A Review of Human-Elephant Ecological Relations in the Malay Peninsula: Adaptations for Coexistence
Abstract
1. Introduction
2. Methodology
2.1. Study Site
2.2. Study Species
2.3. Literature Review
2.4. Field Observations
2.5. Analysis
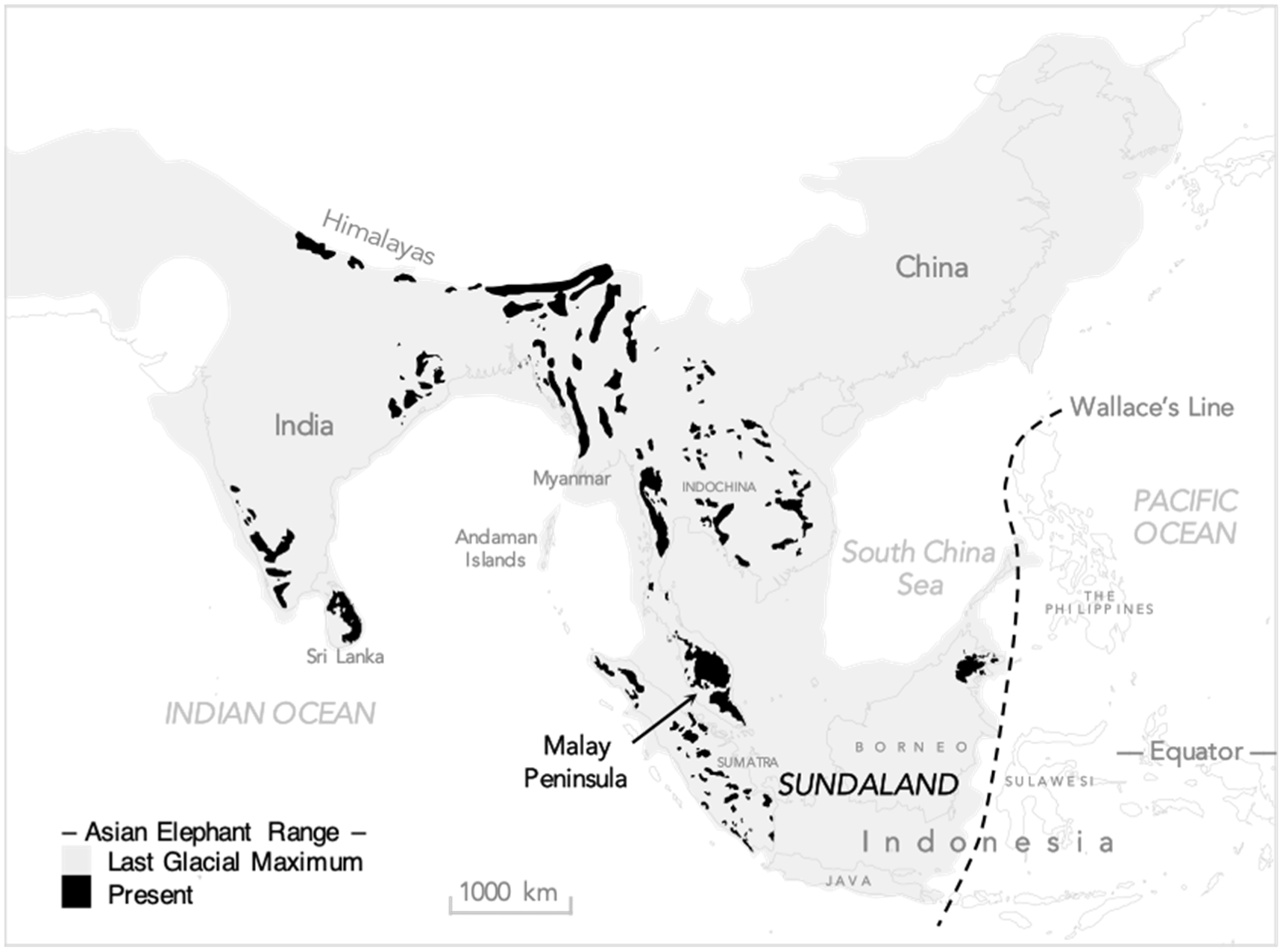
3. Overview of the Human–Elephant Ecological Overlap
4. Spatio-Temporal Niche Overall and Partitioning: The Elephant and the Person in the Room
4.1. Eurytopic Sympatry: From Coast to Mountain Crest
4.2. Shared Pathways: Elephant Forest Trails
4.3. Facultative Arborealism
4.4. Temporal Niche Partitioning: Diurnal & Nocturnal Activity Patterns
5. Trophic Niche: One Bite at a Time
5.1. Finding Food in the Rainforest
5.2. Dessert in the Green Desert: Carbohydrates from Palms
5.3. A Proboscidean Perspective on the Wild Yam Problem
5.4. Honey Hunters
5.5. Fruit Gardeners
5.6. Swidden Farming: Elephants in the Fallow Field
5.7. Predation: Calories from Animal Protein
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Shoshani, J.; Tassy, P. Advances in Proboscidean Taxonomy & Classification, Anatomy & Physiology, and Ecology & Behavior. Quat. Int. 2005, 126, 5–20. [Google Scholar]
- Palkopoulou, E.; Lipson, M.; Mallick, S.; Nielsen, S.; Rohland, N.; Baleka, S.; Karpinski, E.; Ivancevic, A.M.; To, T.-H.; Kortschak, R.D. A Comprehensive Genomic History of Extinct and Living Elephants. Proc. Natl. Acad. Sci. USA 2018, 115, E2566–E2574. [Google Scholar] [CrossRef]
- Agam, A.; Barkai, R. Elephant and Mammoth Hunting during the Paleolithic: A Review of the Relevant Archaeological, Ethnographic and Ethno-Historical Records. Quaternary 2018, 1, 3–31. [Google Scholar] [CrossRef]
- Ben-Dor, M.; Gopher, A.; Hershkovitz, I.; Barkai, R. Man the Fat Hunter: The Demise of Homo erectus and the Emergence of a New Hominin Lineage in the Middle Pleistocene (ca. 400 Kyr) Levant. PLoS ONE 2011, 6, e28689. [Google Scholar] [CrossRef]
- Smith, B.D.; Zeder, M.A. The Onset of the Anthropocene. Anthropocene 2013, 4, 8–13. [Google Scholar] [CrossRef]
- Malhi, Y.; Doughty, C.E.; Galetti, M.; Smith, F.A.; Svenning, J.-C.; Terborgh, J.W. Megafauna and Ecosystem Function from the Pleistocene to the Anthropocene. Proc. Natl. Acad. Sci. USA 2016, 113, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Brittain, S.; Bata, M.N.; Ornellas, P.D.; Milner-Gulland, E.J.; Rowcliffe, M. Combining Local Knowledge and Occupancy Analysis for a Rapid Assessment of the Forest Elephant Loxodonta cyclotis in Cameroon’s Timber Production Forests. Oryx 2020, 54, 90–100. [Google Scholar] [CrossRef]
- Choudhury, A.; Lahiri Choudhury, D.K.; Desai, A.; Duckworth, J.W.; Easa, P.S.; Johnsingh, A.J.T.; Fernando, P.; Hedges, S.; Gunawardena, M.; Kurt, F.; et al. Elephas maximus. The IUCN Red List of Threatened Species 2008. Available online: https://doi.org/10.2305/IUCN.UK.2008.RLTS.T7140A12828813.en (accessed on 31 October 2020).
- Chase, M.J.; Schlossberg, S.; Griffin, C.R.; Bouché, P.J.C.; Djene, S.W.; Elkan, P.W.; Ferreira, S.; Grossman, F.; Kohi, E.M.; Landen, K.; et al. Continent-Wide Survey Reveals Massive Decline in African Savannah Elephants. PeerJ 2016, 4, e2354. [Google Scholar] [CrossRef]
- Blake, S.; Hedges, S. Sinking the Flagship: The Case of Forest Elephants in Asia and Africa. Conserv. Biol. 2004, 18, 1191–1202. [Google Scholar] [CrossRef]
- Lima-Ribeiro, M.S.; Nogués-Bravo, D.; Terribile, L.C.; Batra, P.; Diniz-Filho, J.A.F. Climate and Humans Set the Place and Time of Proboscidean Extinction in Late Quaternary of South America. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2013, 392, 546–556. [Google Scholar] [CrossRef]
- Stuart, A.J. The Extinction of Woolly Mammoth (Mammuthus primigenius) and Straight-Tusked Elephant (Palaeoloxodon antiquus) in Europe. Quat. Int. 2005, 126–128, 171–177. [Google Scholar] [CrossRef]
- Lobban, R.A., Jr.; De Liedekerke, V. Elephants in Ancient Egypt and Nubia. Anthrozoös 2000, 13, 232–244. [Google Scholar] [CrossRef]
- Elvin, M. The Retreat of the Elephants: An Environmental History of China, Illustrated ed.; Yale University Press: New Haven, CT, USA, 2001; ISBN 978-0-300-11993-0. [Google Scholar]
- Parker, I.S.C.; Graham, A.D. Men, Elephants and Competition. Symp. Zool. Soc. Lond. 1989, 61, 241–252. [Google Scholar]
- Barnes, R.F.W. The Conflict between Humans and Elephants in the Central African Forests. Mammal Rev. 1996, 26, 67–80. [Google Scholar] [CrossRef]
- Sukumar, R. A Brief Review of the Status, Distribution and Biology of Wild Asian Elephants, Elephas maximus. Int. Zoo Yearb. 2006, 40, 1–8. [Google Scholar] [CrossRef]
- Laden, G.T. Ethnoarchaeology and Land Use Ecology of the Efe (Pygmies) of the Ituri Rain Forest, Zaire. PhD Thesis, Harvard University, Cambridge, MA, USA, 1992. [Google Scholar]
- Endicott, K. Malaysia’s Original People: Past, Present and Future of the Orang Asli; NUS Press: Singapore, 2016; ISBN 978-9971-69-861-4. [Google Scholar]
- Whitmore, T.C. Tropical Rain Forests of the Far East; Clarendon Press: Oxford, UK, 1975. [Google Scholar]
- Kealhofer, L. Looking into the Gap: Land Use and the Tropical Forests of Southern Thailand. Asian Perspect. 2003, 42, 72–95. [Google Scholar] [CrossRef]
- Ray, N.; Adams, J. A GIS-Based Vegetation Map of the World at the Last Glacial Maximum (25,000-15,000 BP). Internet Archaeol. 2001, 11, 2. [Google Scholar] [CrossRef]
- Haynes, G. Mammoths, Mastodonts, and Elephants: Biology, Behavior and the Fossil Record; Cambridge University Press: Cambridge, UK, 1993; ISBN 978-0-521-45691-3. [Google Scholar]
- Cerling, T.E.; Harris, J.M.; Leakey, M.G. Browsing and Grazing in Elephants: The Isotope Record of Modern and Fossil Proboscideans. Oecologia 1999, 120, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Todd, N.E. New Phylogenetic Analysis of the Family Elephantidae Based on Cranial-Dental Morphology. Anat. Rec. Adv. Integr. Anat. Evol. Biol. 2010, 293, 74–90. [Google Scholar] [CrossRef] [PubMed]
- Puspaningrum, M.R.; van den Bergh, G.D.; Chivas, A.R.; Setiabudi, E.; Kurniawan, I. Isotopic Reconstruction of Proboscidean Habitats and Diets on Java since the Early Pleistocene: Implications for Adaptation and Extinction. Quat. Sci. Rev. 2020, 228, 106007. [Google Scholar] [CrossRef]
- Vidya, T.N.C.; Sukumar, R.; Melnick, D.J. Range-Wide MtDNA Phylogeography Yields Insights into the Origins of Asian Elephants. Proc. R. Soc. B Biol. Sci. 2009, 278, 798. [Google Scholar] [CrossRef]
- van der Made, J. The Evolution of the Elephants and Their Relatives in the Context of Changing Climate and Geography. In Elefantentreich—Eine Fossilwelt in Europa; Verlag Beier & Beran: Langenweißbach, Germany, 2010; pp. 340–360. ISBN 978-3-939414-48-3. [Google Scholar]
- Wen, H.; Jian, Y.; He, Y.; Gao, Y. Initial research on wild elephants in China during the historical period. In The Change of the Plant and Animal in China During Different Historical Period; Wen, R., Wen, H., Eds.; Chongqing Press: Chongqing, China, 1995; pp. 185–201. [Google Scholar]
- Détroit, F.; Mijares, A.S.; Corny, J.; Daver, G.; Zanolli, C.; Dizon, E.; Robles, E.; Grün, R.; Piper, P.J. A New Species of Homo from the Late Pleistocene of the Philippines. Nature 2019, 568, 181–186. [Google Scholar] [CrossRef] [PubMed]
- van den Bergh, G.D.; de Vos, J.; Sondaar, P.Y. The Late Quaternary Palaeogeography of Mammal Evolution in the Indonesian Archipelago. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2001, 171, 385–408. [Google Scholar] [CrossRef]
- Louys, J.; Curnoe, D.; Tong, H. Characteristics of Pleistocene Megafauna Extinctions in Southeast Asia. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2007, 243, 152–173. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Y.; Janis, C.M.; Goodall, R.H.; Purnell, M.A. An Examination of Feeding Ecology in Pleistocene Proboscideans from Southern China (Sinomastodon, Stegodon, Elephas), by Means of Dental Microwear Texture Analysis. Quat. Int. 2017, 445, 60–70. [Google Scholar] [CrossRef]
- Ma, J.; Wang, Y.; Jin, C.; Hu, Y.; Bocherens, H. Ecological Flexibility and Differential Survival of Pleistocene Stegodon orientalis and Elephas maximus in Mainland Southeast Asia Revealed by Stable Isotope (C, O) Analysis. Quat. Sci. Rev. 2019, 212, 33–44. [Google Scholar] [CrossRef]
- Olivier, R.C. Ecology and behavior of living elephants: Bases for assumptions concerning the extinct woolly mammoths. In Paleoecol. of Beringia; Academic Press: New York, NY, USA, 1982; pp. 291–305. [Google Scholar]
- Olivier, R. Distribution and Status of the Asian Elephant. Oryx 1978, 14, 379–424. [Google Scholar] [CrossRef]
- Terborgh, J.; Davenport, L.C.; Ong, L.; Campos-Arceiz, A. Foraging Impacts of Asian Megafauna on Tropical Rain Forest Structure and Biodiversity. Biotropica 2018, 50, 84–89. [Google Scholar] [CrossRef]
- Olivier, R. On the Ecology of the Asian Elephant, Elephas maximus Linn, with Particular Reference to Malaya and Sri Lanka. Ph.D. Thesis, University of Cambridge, Cambridge, UK, 1978. [Google Scholar]
- Roberts, P.; Louys, J.; Zech, J.; Shipton, C.; Kealy, S.; Carro, S.S.; Hawkins, S.; Boulanger, C.; Marzo, S.; Fiedler, B. Isotopic Evidence for Initial Coastal Colonization and Subsequent Diversification in the Human Occupation of Wallacea. Nat. Commun. 2020, 11, 1–11. [Google Scholar] [CrossRef]
- Louys, J.; Roberts, P. Environmental Drivers of Megafauna and Hominin Extinction in Southeast Asia. Nature 2020, 586, 402–406. [Google Scholar] [CrossRef]
- Rambo, A.T. Human Ecology of the Orang Asli: A Review of Research on the Environmental Relations of the Aborigines of Peninsular Malaysia. Fed. Mus. J. 1979, 24, 41–74. [Google Scholar]
- Lim, T. Malaysia: Illegalities in Forest Clearance for Large-Scale Commercial Plantations; Forest Trends Association: Washington, DC, USA, 2013. [Google Scholar]
- Hannigan, T. Beyond Control: Orientalist Tensions and the History of the “Upas Tree” Myth. J. Commonw. Lit. 2020, 55, 173–189. [Google Scholar] [CrossRef]
- Noor, F.A. The Discursive Construction of Southeast Asia in 19th Century Colonial-Capitalist Discourse; Amsterdam University Press: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Rappaport, R.A. Pigs for the Ancestors: Ritual in the Ecology of a New Guinea People; Waveland Press: Long Grove, USA, 2000. [Google Scholar]
- Reich, D. Who We Are and How We Got Here: Ancient DNA and the New Science of the Human Past; Oxford University Press: Oxford, UK, 2018. [Google Scholar]
- Lev, M.; Barkai, R. Elephants Are People, People Are Elephants: Human–Proboscideans Similarities as a Case for Cross Cultural Animal Humanization in Recent and Paleolithic Times. Quat. Int. 2016, 406, 239–245. [Google Scholar] [CrossRef]
- Endicott, K. The Hunting Methods of the Batek Negritos of Malaysia. Canberra Anthropol. 1979, 2, 7–22. [Google Scholar] [CrossRef]
- Rambo, A.T. Why Are the Semang? Ecology and Ethnogenesis of Aboriginal Groups in Peninsular Malaysia. In Ethnic Diversity and the Control of Natural Resources in Southeast Asia; Rambo, A.T., Gillogly, K., Hutterer, K.L., Eds.; University of Michigan Press: Ann Arbor, MI, USA, 1988; pp. 19–35. [Google Scholar]
- Eisenberg, J.F.; Seidensticker, J. Ungulates in Southern Asia: A Consideration of Biomass Estimates for Selected Habitats. Biol. Conserv. 1976, 10, 293–308. [Google Scholar] [CrossRef]
- Evans, I.H.N. The Negritos of Malaya; University Press: Cambridge, UK, 1937. [Google Scholar]
- Lye, T.-P. Changing Pathways: Forest Degradation and the Batek of Pahang, Malaysia; Lexington Books: Lanham, MD, USA, 2004. [Google Scholar]
- Endicott, K.; Bellwood, P. The Possibility of Independent Foraging in the Rain Forest of Peninsular Malaysia. Hum. Ecol. 1991, 19, 151–185. [Google Scholar] [CrossRef]
- Torre, J.A.; Lechner, A.M.; Wong, E.P.; Magintan, D.; Saaban, S.; Campos-Arceiz, A. Using Elephant Movements to Assess Landscape Connectivity under Peninsular Malaysia’s Central Forest Spine Land Use Policy. Conserv. Sci. Pract. 2019, 1. [Google Scholar] [CrossRef]
- Evans, L.J.; Asner, G.P.; Goossens, B. Protected Area Management Priorities Crucial for the Future of Bornean Elephants. Biol. Conserv. 2018, 221, 365–373. [Google Scholar] [CrossRef]
- Symington, C.F. Foresters’ Manual of Dipterocarps; Ashton, P.S., Appanah, S., Revs Barlow, H.S., Eds.; Forest Research Institute Malaysia: Kuala Lumpur, Malaysia, 2004. [Google Scholar]
- Owen-Smith, N. Pleistocene Extinctions: The Pivotal Role of Megaherbivores. Paleobiology 1987, 13, 351–362. [Google Scholar] [CrossRef]
- Haynes, G. Mammoth Landscapes: Good Country for Hunter-Gatherers. Quat. Int. 2006, 142–143, 20–29. [Google Scholar] [CrossRef]
- Wadey, J.; Beyer, H.L.; Saaban, S.; Othman, N.; Leimgruber, P.; Campos-Arceiz, A. Why Did the Elephant Cross the Road? The Complex Response of Wild Elephants to a Major Road in Peninsular Malaysia. Biol. Conserv. 2018, 218, 91–98. [Google Scholar] [CrossRef]
- Wall, J.; Douglas-Hamilton, I.; Vollrath, F. Elephants Avoid Costly Mountaineering. Curr. Biol. 2006, 16, R527–R529. [Google Scholar] [CrossRef]
- Blake, S.; Inkamba-Nkulu, C. Fruit, Minerals, and Forest Elephant Trails: Do All Roads Lead to Rome? Biotropica 2004, 36, 392–401. [Google Scholar] [CrossRef]
- Kingdon, J. Mammalian evolution in Africa. In Mammals of Africa; Kingdon, J., Happold, D., Butynski, T., Hoffmann, M., Happold, M., Kalina, J., Eds.; Bloomsbury: London, UK, 2013; Volume 1, pp. 75–100. ISBN 978-1-4081-8996-2. [Google Scholar]
- Keil, P.G. Elephant-Human Dandi: How Humans and Elephants Move through the Fringes of Forest and Village. Confl. Negot. Coexistence Rethink. Hum.-Elephant Relat. South Asia 2016, 242–271. [Google Scholar] [CrossRef]
- Lim, T. Human-Elephant Relations in Peninsular Malaysia. Ph.D. Thesis, University of Nottingham, Nottingham, UK, 2019. [Google Scholar]
- Kromann-Clausen, A. How Mineral Deposits Impact Behaviour of Megafauna and Shape the Structure of a Malaysian Forest. Master’s Thesis, University of Copenhagen, København, Denmark, 2015. [Google Scholar]
- Campos-Arceiz, A.; Blake, S. Megagardeners of the Forest–the Role of Elephants in Seed Dispersal. Acta Oecologica 2011, 37, 542–553. [Google Scholar] [CrossRef]
- Ong, L.; McConkey, K.; Solana-Mena, A.; Campos-Arceiz, A. Elephant Frugivory and Wild Boar Seed Predation of Irvingia malayana, a Large-Fruited Tree, in a Rainforest of Peninsular Malaysia. Raffles Bull. Zool. 2019, 67, 160170. [Google Scholar] [CrossRef]
- Remis, M.J. The Gorilla Paradox. In Primate Locomotion: Recent Advances; Strasser, E., Fleagle, J.G., Rosenberger, A.L., McHenry, H.M., Eds.; Springer: Boston, MA, USA, 1998; pp. 95–106. ISBN 978-1-4899-0092-0. [Google Scholar]
- Goodall, J.M. Nest Building Behavior in the Free Ranging Chimpanzee. Ann. N. Y. Acad. Sci. 1962, 102, 455–467. [Google Scholar] [CrossRef]
- Prasetyo, D.; Ancrenaz, M.; Morrogh-Bernard, H.C.; Atmoko, S.S.U.; Wich, S.A.; van Schaik, C.P. Nest Building in Orangutans; Oxford University Press: Oxford, UK; ISBN 978-0-19-170756-8.
- Noble, A.G. Traditional Buildings: A Global Survey of Structural Forms and Cultural Functions; I.B. Tauris: London, UK, 2007; ISBN 978-1-84511-305-6. [Google Scholar]
- Baker, S.W. The Rifle and the Hound in Ceylon; Longman: London, UK, 1854. [Google Scholar]
- Santiapillai, C. Mitigation of Human-Elephant Conflicts in Sri Lanka. Gajah 1996, 15, 1–8. [Google Scholar]
- Henderson, P.; Mornement, A. Treehouses; Frances Lincoln: London, UK, 2005; ISBN 978-0-7112-2437-7. [Google Scholar]
- Kroeber, A.L. Peoples of the Philippines; American Museum Press: New York, NY, USA, 1919. [Google Scholar]
- Skeat, W.W. Pagan Races of the Malay Peninsula; Macmillan: London, UK, 1906; Volume 2. [Google Scholar]
- Cameron, J. Our Tropical Possessions in Malayan India: Being a Descriptive Account of Singapore, Penang, Province Wellesley, and Malacca; Their Peoples, Products, Commerce, and Government; Smith, Elder & Co.: London, UK, 1865. [Google Scholar]
- Kelsall, H.J. Account of a Trip up the Pahang, Tembeling, and Tahan Rivers, and an Attempt to Reach Gunong Tahan. J. Straits Br. R. Asiat. Soc. 1894, 25, 33–56. [Google Scholar]
- Hornaday, W.T. Account of a Naturalist’s Visit to the Territory of Selangor. J. Straits Br. R. Asiat. Soc. 1879, 3, 124–131. [Google Scholar]
- Dunn, F.L. Rain-Forest Collectors and Traders: A Study of Resource Utilization in Modern and Ancient Malaya; Malaysian Branch of the Royal Asiatic Society: Kuala Lumpur, Malaysia, 1975. [Google Scholar]
- Roseman, M. Singers of the Landscape: Song, History, and Property Rights in the Malaysian Rain Forest. Am. Anthropol. 1998, 100, 106–121. [Google Scholar] [CrossRef]
- Manickam, S.K. Taming the Wild: Aborigines and Racial Knowledge in Colonial Malaya; NUS Press: Singapore, 2015. [Google Scholar]
- Schebesta, P. Among the Forest Dwarfs of Malaya; Hutchinson: London, UK, 1929. [Google Scholar]
- Dentan, R.K. The Semai: A Nonviolent People of Malaya; Holt, Rinehart, and Winston: New York, NY, USA, 1968. [Google Scholar]
- Larramendi, A. Shoulder Height, Body Mass, and Shape of Proboscideans. Acta Palaeontol. Pol. 2015, 61, 537–574. [Google Scholar] [CrossRef]
- Hii, N. Asian Elephants’ Social Structure and Mineral Lick Usage in a Malaysian Rainforest Using Camera Traps. Master’s Thesis, University of Nottingham, Selangor, Malaysia, 2017. [Google Scholar]
- Campos-Arceiz, A.; Takatsuki, S.; Ekanayaka, S.K.K.; Hasegawa, T. The Human-Elephant Conflict in Southeastern Sri Lanka: Type of Damage, Seasonal Patterns, and Sexual Differences in the Raiding Behavior of Elephants. Gajah 2009, 31, 5–14. [Google Scholar]
- Wilson, S.; Davies, T.E.; Hazarika, N.; Zimmermann, A. Understanding Spatial and Temporal Patterns of Human–Elephant Conflict in Assam, India. Oryx 2015, 49, 140–149. [Google Scholar] [CrossRef]
- Robarchek, C.A.; Robarchek, C.J. A comparative study of Waorani and Semai. In Aggression and Peace in Humans and Other Primates; Silverberg, J., Patrick Gray, J., Eds.; Oxford University Press: Oxford, UK, 1992; pp. 189–213. [Google Scholar]
- Headland, T.N. The Wild Yam Question: How Well Could Independent Hunter-Gatherers Live in a Tropical Rain Forest Ecosystem? Hum. Ecol. 1987, 15, 463–491. [Google Scholar] [CrossRef]
- Bailey, R.C.; Headland, T.N. The Tropical Rain Forest: Is It a Productive Environment for Human Foragers? Hum. Ecol. 1991, 19, 261–285. [Google Scholar] [CrossRef]
- Headland, T.N. Could ‘Pure’ Hunter-Gatherers Live in a Rain Forest? Available online: https://scholars.sil.org/thomas_n_headland/controversies/wild_yam (accessed on 27 October 2020).
- Hart, T.B.; Hart, J.A. The Ecological Basis of Hunter-Gatherer Subsistence in African Rain Forests: The Mbuti of Eastern Zaire. Hum. Ecol. 1986, 14, 29–55. [Google Scholar] [CrossRef]
- Dierenfeld, E.S. Nutrition. In Biology, Medicine, and Surgery of Elephants; Fowler, M., Mikota, S.K., Eds.; Blackwell Publishing: Ames, IA, USA, 2008; pp. 57–65. ISBN 978-0-470-34411-8. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; Institute of Medicine of the National Academies: Washington, DC, USA, 2002; ISBN 978-0-309-08525-0. [Google Scholar]
- Dentan, R.K. Potential Food Sources for Foragers in Malaysian Rainforest: Sago, Yams and Lots of Little Things. Bijdr. Tot Taal-Land-En Volkenkd. J. Humanit. Soc. Sci. Southeast Asia 1991, 147, 420–444. [Google Scholar] [CrossRef]
- Brosius, J.P. Foraging in Tropical Rain Forests: The Case of the Penan of Sarawak, East Malaysia (Borneo). Human Ecol. 1991, 19, 123–150. [Google Scholar] [CrossRef]
- Kitanishi, K. Variability in the Subsistence Activities and Distribution of Food among Different Aged Males of the Aka Hunter-Gatheres in Northeastern Congo. Afr. Study Monogr. 1996, 17, 35–57. [Google Scholar] [CrossRef]
- Endicott, K.M.; Endicott, K.L. The Headman Was a Woman: The Gender Egalitarian Batek of Malaysia; Waveland: Long Grove, IL, USA, 2008. [Google Scholar]
- Stanton, W.R. Perspective on, and Future Prospects for, the Sago Industry. Sago Palm 1993, 1, 2–7. [Google Scholar]
- Phillipps, Q. Phillipps’ Field Guide to the Mammals of Borneo and Their Ecology: Sabah, Sarawak, Brunei, and Kalimantan; Princeton University Press: Princeton, NJ, USA, 2016. [Google Scholar]
- Dounias, E. The Management of Wild Yam Tubers by the Baka Pygmies in Southern Cameroon. Afr. Study Monogr. Suppl. Issue 2001, 26, 135–156. [Google Scholar] [CrossRef]
- Turner, I.M. A Catalogue of the Vascular Plants of Malaya. Gardens’ Bul. 1994, 47, 19980602199. [Google Scholar]
- Burkill, I.H. A Dictionary of the Economic Products of the Malay Peninsula. Ministry of Agriculture: Kuala Lumpur, Malaysia, 1935 (2002 Reprint). Available online: https://books.google.com.my/books?id=a4AKAQAAIAAJ (accessed on 13 January 2021).
- English, M.; Ancrenaz, M.; Gillespie, G.; Goossens, B.; Nathan, S.; Linklater, W. Foraging Site Recursion by Forest Elephants Elephas maximus borneensis. Curr. Zool. 2014, 60, 551–559. [Google Scholar] [CrossRef]
- Yamamoto-Ebina, S.; Saaban, S.; Campos-Arceiz, A.; Takatsuki, S. Food Habits of Asian Elephants Elephas maximus in a Rainforest of Northern Peninsular Malaysia. Mammal Study 2016, 41, 155–161. [Google Scholar] [CrossRef]
- Kuchikura, Y. Wild Yams in the Tropical Rain Forest: Abundance and Dependence among the Semaq Beri in Peninsular Malaysia. Man Cult. Ocean. 1993, 9, 102. [Google Scholar]
- Barton, H.; Denham, T. Prehistoric Vegeculture and Social Life in Island Southeast Asia and Melanesia. In Why cultivate? Anthropological and Archaeological Approaches to Foraging-Farming Transitions in Southeast Asia; Barker, G., Janowski, M., Eds.; McDonald Institute for Anthropological Research: Cambridge, UK, 2011; pp. 17–25. [Google Scholar]
- Yasuoka, H. Concentrated Distribution of Wild Yam Patches: Historical Ecology and the Subsistence of African Rainforest Hunter-Gatherers. Hum. Ecol. 2009, 37, 577–587. [Google Scholar] [CrossRef]
- Knight, J. Half-man, half-elephant: Shapeshifting among the Baka. In Natural Enemies: People-wildlife Conflicts in Anthropological Perspective; Routledge: Abingdon, UK, 2000; pp. 50–77. ISBN 978-0-415-22440-6. [Google Scholar]
- Sukumar, R. The Living Elephants: Evolutionary Ecology, Behavior, and Conservation; Oxford University Press: New York, NY, USA, 2003; ISBN 978-0-19-510778-4. [Google Scholar]
- Suba, R.B.; Beveridge, N.G.P.; Kustiawan, W.; De Snoo, G.R.; De Iongh, H.H. Foraging Ecology and Diet of Bornean Elephants (Elephas maximus borneensis) in the Sebuku Forest Area, North Kalimantan Province of Indonesia: Do the Choices Matter? Integr. Zool. 2018, 13, 219–223. [Google Scholar] [CrossRef]
- Kurt, F.; Hartl, G.B.; Tiedemann, R. Tuskless Bulls in Asian Elephant Elephas maximus. History and Poulation Genetics of a Man-Made Phenomenon. Acta Theriol. 1995, 40, 125–144. [Google Scholar] [CrossRef]
- Zaya, D.N.; Howe, H.F. The Anomalous Kentucky Coffeetree: Megafaunal Fruit Sinking to Extinction? Oecologia 2009, 161, 221–226. [Google Scholar] [CrossRef]
- Wilson, M.J. Clay Mineralogical and Related Characteristics of Geophagic Materials. J. Chem. Ecol. 2003, 29, 1525–1547. [Google Scholar] [CrossRef]
- Lundquist, C.A.; Varnedoe Jr, W.W. Salt Ingestion Caves. Int. J. Speleol. 2006, 35, 2. [Google Scholar] [CrossRef]
- Elyana, F.N.; Al-Mekhlafi, H.M.; Ithoi, I.; Abdulsalam, A.M.; Dawaki, S.; Nasr, N.A.; Atroosh, W.M.; Abd-Basher, M.H.; Al-Areeqi, M.A.; Sady, H. A Tale of Two Communities: Intestinal Polyparasitism among Orang Asli and Malay Communities in Rural Terengganu, Malaysia. Parasit. Vectors 2016, 9, 398. [Google Scholar] [CrossRef] [PubMed]
- Nadkarni, N.M. Diversity of Species and Interactions in the Upper Tree Canopy of Forest Ecosystems. Am. Zool. 1994, 34, 70–78. [Google Scholar] [CrossRef]
- Latinis, D.K. The Development of Subsistence System Models for Island Southeast Asia and Near Oceania: The Nature and Role of Arboriculture and Arboreal-Based Economies. World Archaeol. 2000, 32, 41–67. [Google Scholar] [CrossRef]
- Kraft, T.S.; Venkataraman, V.V.; Dominy, N.J. A Natural History of Human Tree Climbing. J. Hum. Evol. 2014, 71, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Seeley, T.D.; Seeley, R.H.; Akratanakul, P. Colony Defense Strategies of the Honeybees in Thailand. Ecol. Monogr. 1982, 52, 43–63. [Google Scholar] [CrossRef]
- Bulbeck, F.D. The Guar Kepah human remains. In The Perak Man and Other Prehistoric Skeletons of Malaysia; Penerbit Universiti Sains Malaysia: Penang, Malaysia, 2005; pp. 383–423. ISBN 978-983-3391-12-7. [Google Scholar]
- Estienne, V.; Boesch, C. Underground Honey Extraction by Chimpanzees, Honey Badgers and Forest Elephants in Loango National Park, Gabon. Folia Primatol. 2015, 86, 276–277. [Google Scholar]
- King, L.; Pardo, M.; Weerathunga, S.; Kumara, T.V.; Jayasena, N.; Soltis, J.; de Silva, S. Wild Sri Lankan Elephants Retreat from the Sound of Disturbed Asian Honey Bees. Curr. Biol. 2018, 28, R64–R65. [Google Scholar] [CrossRef] [PubMed]
- Ridley, H.N. On the Dispersal of Seeds by Mammals. J. Straits Br. R. Asiat. Soc. 1894, 25, 11–32. [Google Scholar]
- Maisels, F.; Blake, S.; Turkalo, A. Wild Forest Elephants Shake down Fruit and Leaves from Trees. Pachyderm 2002, 33, 88–90. [Google Scholar]
- Rutten, M. Over Olifantshoopen [On Elephant Dung]. Trop. Nat. 1939, 28, 19. [Google Scholar]
- Campos-Arceiz, A.; Traeholt, C.; Jaffar, R.; Santamaria, L.; Corlett, R.T. Asian Tapirs Are No Elephants When It Comes To Seed Dispersal. Biotropica 2012, 44, 220–227. [Google Scholar] [CrossRef]
- Plotnik, J.M.; Shaw, R.C.; Brubaker, D.L.; Tiller, L.N.; Clayton, N.S. Thinking with Their Trunks: Elephants Use Smell but Not Sound to Locate Food and Exclude Nonrewarding Alternatives. Anim. Behav. 2014, 88, 91–98. [Google Scholar] [CrossRef]
- Corlett, R.T. How to Be a Frugivore (in a Changing World). Acta Oecologica 2011, 37, 674–681. [Google Scholar] [CrossRef]
- Corner, E.J.H. The Durian Theory or the Origin of the Modern Tree on JSTOR. Ann. Bot. 1949, 13, 367–414. [Google Scholar] [CrossRef]
- Mayer, C. Trapping Wild Animals in Malay Jungles; Duffield: New York, NY, USA, 1922. [Google Scholar]
- Majid, A.; Kruspe, N. Hunter-Gatherer Olfaction Is Special. Curr. Biol. 2018, 28, 409–413. [Google Scholar] [CrossRef]
- Terrill, A.; Burenhult, N. Orientation as a Strategy of Spatial Reference. Stud. Lang. Int. J. Spons. Found. Found. Lang. 2008, 32, 93–136. [Google Scholar] [CrossRef]
- Moore, J.H.; Sittimongkol, S.; Campos-Arceiz, A.; Sumpah, T.; Eichhorn, M.P. Fruit Gardens Enhance Mammal Diversity and Biomass in a Southeast Asian Rainforest. Biol. Conserv. 2016, 194, 132–138. [Google Scholar] [CrossRef]
- Lim, T.; Loke, V.; Mena, A.; Pura, P.; Angah, R.; Tan, A.; Campos-Arceiz, A. Mapping the Distribution of People, Elephants, and Human-Elephant Conflict in Temengor Forest Complex, Peninsular Malaysia. Malay. Nat. J. 2017, 2017, 31–49. [Google Scholar]
- Wharton, C.H.; Komarek, E.V. Man, Fire and Wild Cattle in Southeast Asia. Proc. An. Tall Timbers Fire Ecol. Conf. 1968, 8, 107–167. [Google Scholar]
- Sukumar, R. The Asian Elephant: Ecology and Management; Cambridge University Press: Cambridge, UK, 1992. [Google Scholar]
- Lorimer, J. Elephants as Companion Species: The Lively Biogeographies of Asian Elephant Conservation in Sri Lanka. Trans. Inst. Br. Geogr. 2010, 35, 491–506. [Google Scholar] [CrossRef]
- Fernando, P.; Wikramanayake, E.; Weerakoon, D.; Jayasinghe, L.K.A.; Gunawardene, M.; Janaka, H.K. Perceptions and Patterns of Human–Elephant Conflict in Old and New Settlements in Sri Lanka: Insights for Mitigation and Management. Biodivers. Conserv. 2005, 14, 2465–2481. [Google Scholar] [CrossRef]
- Speth, J.D.; Spielmann, K.A. Energy Source, Protein Metabolism, and Hunter-Gatherer Subsistence Strategies. J. Anthropol. Archaeol. 1983, 2, 1–31. [Google Scholar] [CrossRef]
- Speth, J.D. The Paleoanthropology and Archaeology of Big-Game Hunting: Protein, Fat, or Politics? Springer: New York, NY, USA, 2010; pp. 149–161. [Google Scholar]
- Brown, C.; Warburton, K. Differences in Timidity and Escape Responses between Predator-Naive and Predator-Sympatric Rainbowfish Populations. Ethology 1999, 105, 491–502. [Google Scholar] [CrossRef]
- Hubback, T.B. The Malay Elephant. J. Bombay Nat. Hist. Soc. 1941, 42, 483–509. [Google Scholar]
- Tshen, L.T. Quaternary Elephas Fossils from Peninsular Malaysia: Historical Overview and New Material. Raffles Bull. Zool. 2013, 139–153. [Google Scholar]
- Muhammad, R.F.; Tshen, L.T.; Ibrahim, N.; Azmi Abdul Razak, M.; Mohd Razif, F.; Kem, Z.; Boon Tat, C. First discovery of Stegodon (Proboscidea) in Malaysia. War. Geol. 2020, 46, 196–198. [Google Scholar] [CrossRef]
- Loke, V.P.W.; Lim, T.; Campos-Arceiz, A. Hunting Practices of the Jahai Indigenous Community in Northern Peninsular Malaysia. Glob. Ecol. Conserv. 2020, 21, e00815. [Google Scholar] [CrossRef]
- Rambo, A.T. Bows, Blowpipes and Blunderbusses: Ecological Implications of Weapons Change among the Malaysian Negritos. Malays. Nat. J. 1978, 22, 209–216. [Google Scholar]
- Pfeffer, P. Fauna of humid tropical Asia. In Natural Resources of Tropical Asia; UNESCO: Paris, France, 1974; pp. 295–306. [Google Scholar]
- Stearman, A.M. Making a Living in the Tropical Forest: Yuqui Foragers in the Bolivian Amazon. Hum. Ecol. 1991, 19, 245–260. [Google Scholar] [CrossRef]
- Watson, P.J. Archaeology, Anthropology, and the Culture Concept. Am. Anthropol. 1995, 97, 683–694. [Google Scholar] [CrossRef]
- Gosselain, O.P. To Hell with Ethnoarchaeology! Archaeol. Dialogues 2016, 23, 215–228. [Google Scholar] [CrossRef]
- Barth, F. Ethnic Groups and Boundaries: The Social Organization of Culture Difference. In Results of a Symposium Held at the University of Bergen, 23rd to 26th February 1967; Universitetsforlage: Bergen, Norway, 1969. [Google Scholar]
- Roseman, M. Healing Sounds from the Malaysian Rainforest: Temiar Music and Medicine; University of California Press: Berkeley, CA, USA, 1991. [Google Scholar]
- Laundré, J.W.; Hernández, L.; Ripple, W.J. The Landscape of Fear: Ecological Implications of Being Afraid. Open Ecol. J. 2010, 3, 1–7. [Google Scholar] [CrossRef]
- Singh, J. Materialities of the Non-Human Animal and the Species Matrix of Postcolonial Remains in Selected Contemporary Writing. Interventions 2018, 20, 666–681. [Google Scholar] [CrossRef]
- Naveh, D.; Bird-David, N. How Persons Become Things: Economic and Epistemological Changes among N Ayaka Hunter-Gatherers. J. R. Anthropol. Inst. 2014, 20, 74–92. [Google Scholar] [CrossRef]
- Mumby, H.S.; Plotnik, J.M. Taking the Elephants’ Perspective: Remembering Elephant Behavior, Cognition and Ecology in Human-Elephant Conflict Mitigation. Front. Ecol. Evol. 2018, 6, 122. [Google Scholar] [CrossRef]
- Torre, J.A.d.l.; Wong, E.P.; Lechner, A.M.; Zulaikha, N.; Zawawi, A.; Abdul-Patah, P.; Saaban, S.; Goossens, B.; Campos-Arceiz, A. There Will Be Conflict—Agricultural Landscapes Are Prime, Rather than Marginal, Habitats for Asian Elephants. Anim. Conserv. 2020. [Google Scholar] [CrossRef]



| Activity Pattern (Section 4.4) | ||
|---|---|---|
| Habitat (Section 4.1) |  Day Day |  Night Night |
 Arboreal (Section 4.3) Arboreal (Section 4.3) |  |  |
 Terrestrial (Section 4.2) Terrestrial (Section 4.2) |  |  |
| Food Source (Section 5 of This Paper) |  Elephants |  Humans |  Tigers |
|---|---|---|---|
| Plants | |||
 Fruit (e.g., durian: Section 5.5) Fruit (e.g., durian: Section 5.5) | ⚫ | ⚫ | ● |
 Leaves, stems (palms: Section 5.2, grass: Section 5.6) Leaves, stems (palms: Section 5.2, grass: Section 5.6) | ⚫ | ● | ○ |
 Roots (tubers: Section 5.3) Roots (tubers: Section 5.3) | ● | ⚫ | ○ |
| Animals (Section 5.7) | |||
 In trees (birds, squirrels, monkeys, honey (Section 5.4)) In trees (birds, squirrels, monkeys, honey (Section 5.4)) | ○ | ⚫ | ● |
 On the ground (pigs, deer, bovids) On the ground (pigs, deer, bovids) | ○ | ● | ⚫ |
 In holes*; ‘lots of little things’ (grubs, snails, fish) In holes*; ‘lots of little things’ (grubs, snails, fish) | ○ | ⚫ | ● |
| Group | Height Above Ground | Rationale Given | Period & Ref. |
|---|---|---|---|
| Korowai, New Guinea | 35 m | slavers, “cannibals” | 20th century [74] |
| Illongot, Luzon | 18 m | head-hunters | 20th century [75] |
| Temuan, Selangor | 9–12 m | elephants | ca. 1900 [76] |
| Aboriginal Malays | 9 m | not given | 19th century [77] |
| Semai, Pahang | 4.6–6 m | not given | 19th century [78] |
| Semelai, Pahang | 4 m | tigers, elephants | 1980 (R. Gianno pers. comm., 24 July 2021) |
| Temuan), Selangor | 3.7 m | elephants | 19th century [79,80] |
| Temiar, Perak/Kelantan | 3 m | tigers, slavers, elephants | 20th century [64,81] |
| Jakun, Johor | 1.5–2.7 m* | elephants, tigers | ca. 1900 [76,82] |
| Menraq (in cliffs and “large” trees) | elephants, tigers | ca. 1900 [51,76,83] | |
| Semai (in the “sturdiest longhouse”) | elephants, slavers | ca. 1960 [84] | |
| Energy | Protein | |
|---|---|---|
| Elephant (1) | 290.3 MJ (2) | 2400 g (6.7 g per kg BW) |
| Human (3) | 10.9 MJ | 46 g (0.7 g per kg BW) |
| Food source | Gross MJ (1) | Nett MJ (2) | Protein | Ref. | Section |
|---|---|---|---|---|---|
 Elephant (3605 kg whole animal) (3) Elephant (3605 kg whole animal) (3) | 9361 | 8392 | 283,738 g | [4] | Section 5.7 |
 Sago (90 kg starch from 1 tree) Sago (90 kg starch from 1 tree) | 1314 | 1149 | 180 g | [96,97] | Section 5.2 |
 Pig (50 kg meat from 1 animal) (4) Pig (50 kg meat from 1 animal) (4) | 310 | 246 | 8195 g | [97] | Section 5.7 |
 Yam (14.4 kg @ 0.8 kg per hr) Yam (14.4 kg @ 0.8 kg per hr) | 71 | 31 | 220 g | [53] | Section 5.3 |
 Honey (4.2 kg) Honey (4.2 kg) | 53 | 15 | 13 g | [98,99] | Section 5.4 |
 Arboreal game (7 kg) Arboreal game (7 kg) | 51 | 13 | 2170 g | [53] | Section 5.7 |
 Durian (27 kg fruit; 5.4 kg pulp) (5) Durian (27 kg fruit; 5.4 kg pulp) (5) | 33 | -3 | 79 g | – | Section 5.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lim, T.; Campos-Arceiz, A. A Review of Human-Elephant Ecological Relations in the Malay Peninsula: Adaptations for Coexistence. Diversity 2022, 14, 36. https://doi.org/10.3390/d14010036
Lim T, Campos-Arceiz A. A Review of Human-Elephant Ecological Relations in the Malay Peninsula: Adaptations for Coexistence. Diversity. 2022; 14(1):36. https://doi.org/10.3390/d14010036
Chicago/Turabian StyleLim, Teckwyn, and Ahimsa Campos-Arceiz. 2022. "A Review of Human-Elephant Ecological Relations in the Malay Peninsula: Adaptations for Coexistence" Diversity 14, no. 1: 36. https://doi.org/10.3390/d14010036
APA StyleLim, T., & Campos-Arceiz, A. (2022). A Review of Human-Elephant Ecological Relations in the Malay Peninsula: Adaptations for Coexistence. Diversity, 14(1), 36. https://doi.org/10.3390/d14010036






