DNA Barcoding of Marine Mollusks Associated with Corallina officinalis Turfs in Southern Istria (Adriatic Sea)
Abstract
1. Introduction
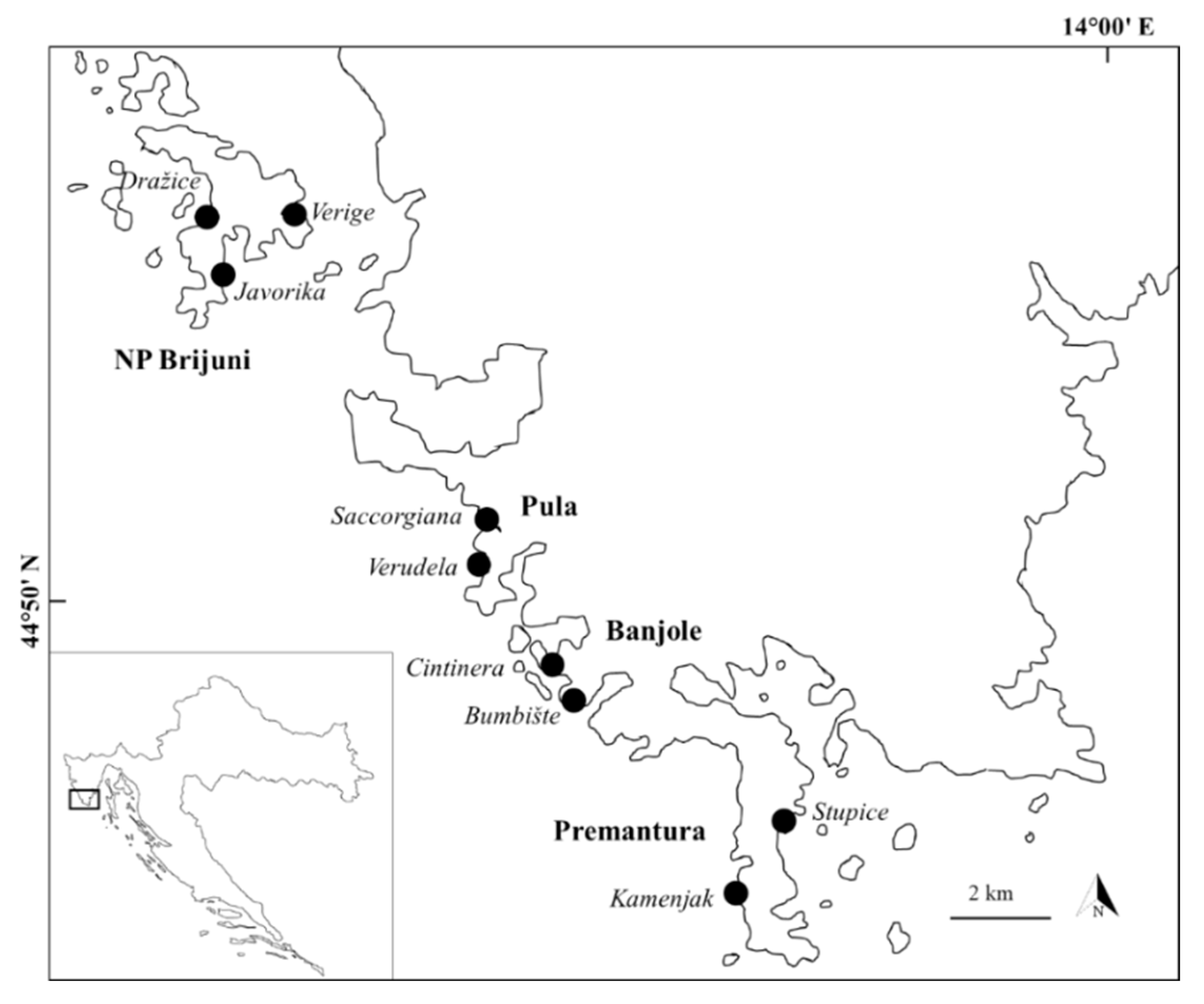
2. Materials and Methods
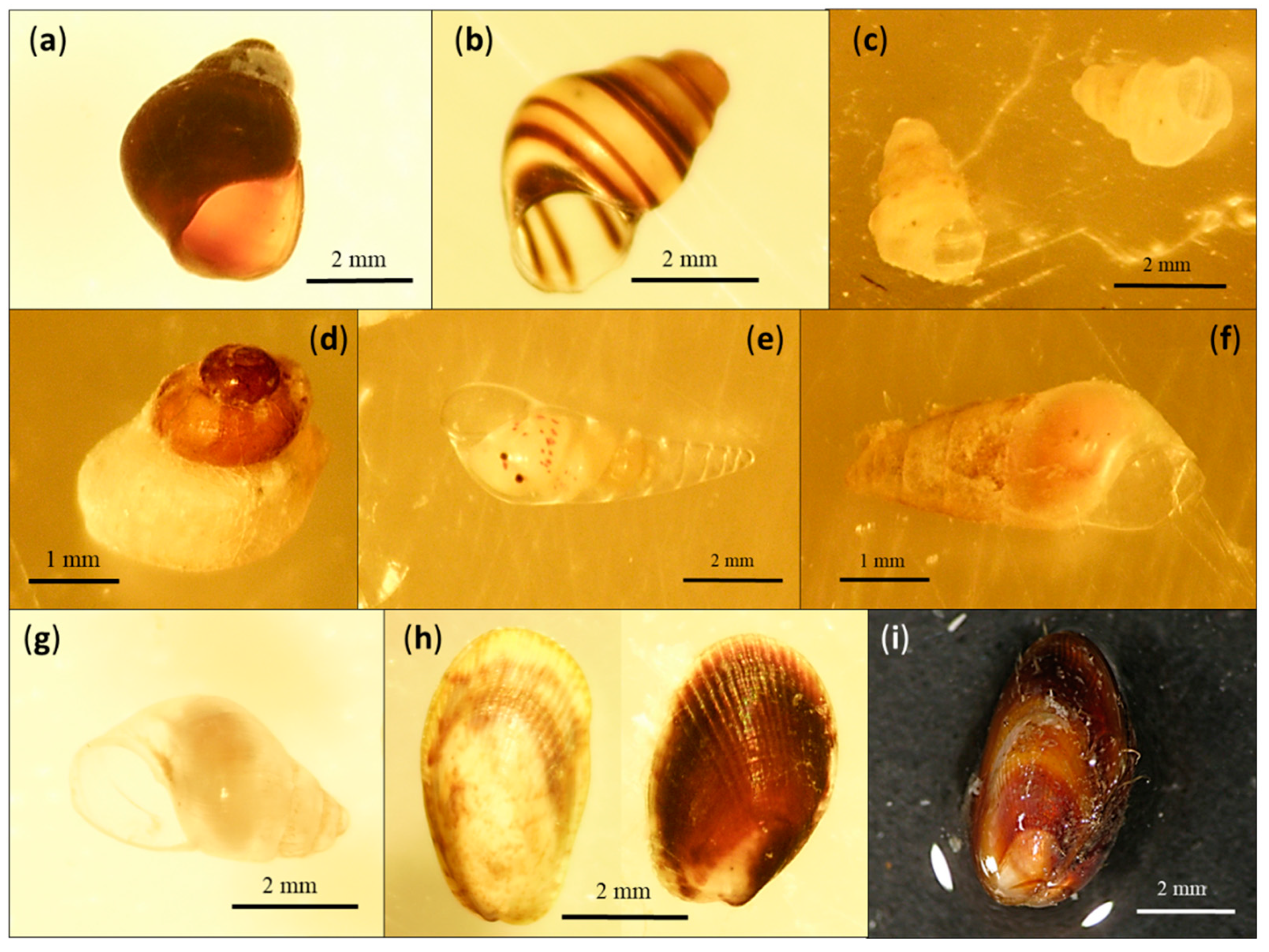
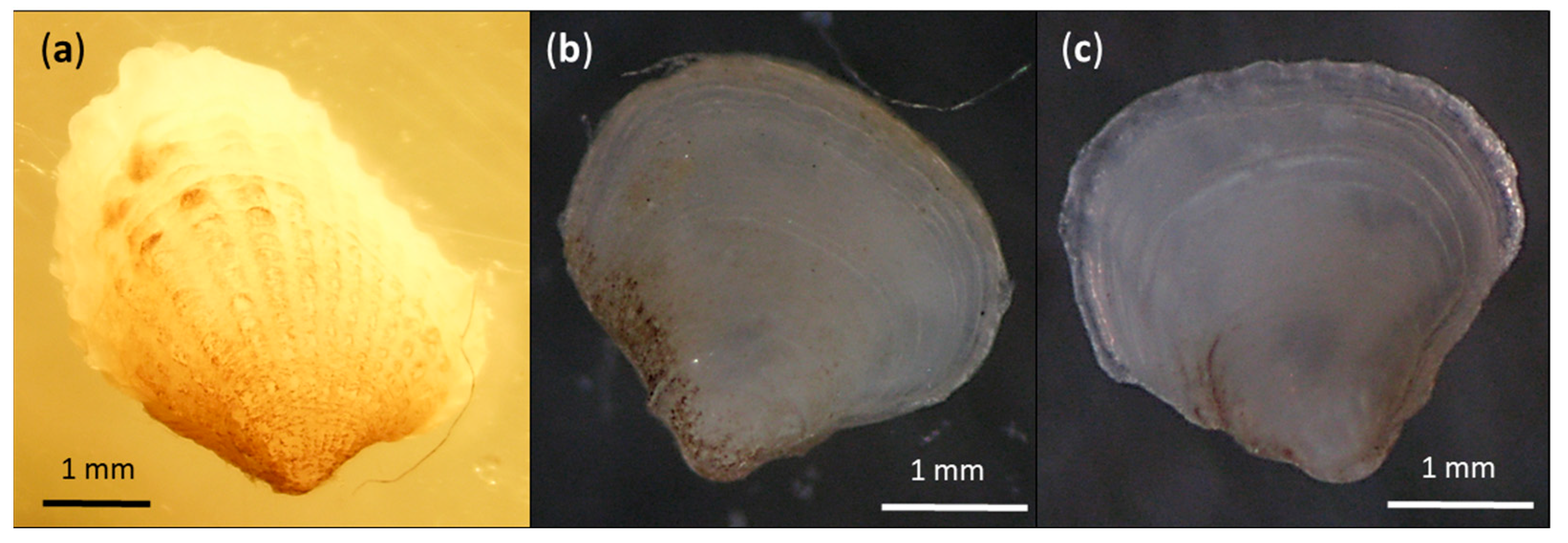
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Williamson, C.J.; Perkins, R.; Voller, M.; Yallop, M.L.; Brodie, J. The Regulation of Coralline Algal Physiology, an in situ Study of Corallina officinalis (Corallinales, Rhodophyta). Biogeosciences 2017, 14, 4485–4498. [Google Scholar] [CrossRef]
- Liuzzi, M.G.; Gappa, J.L. Macrofaunal Assemblages Associated with Coralline Turf: Species Turnover and Changes in Structure at Different Spatial Scales. Mar. Ecol. Prog. Ser. 2008, 363, 147–156. [Google Scholar] [CrossRef]
- Hofmann, L.C.; Yildiz, G.; Hanelt, D.; Bischof, K. Physiological Responses of the Calcifying Rhodophyte, Corallina officinalis (L.), to Future CO2 Levels. Mar. Biol. 2012, 159, 783–792. [Google Scholar] [CrossRef]
- Rendina, F.; Bouchet, P.J.; Appolloni, L.; Russo, G.F.; Sandulli, R.; Kolzenburg, R.; Putra, A.; Ragazzola, F. Physiological Response of the Coralline Alga Corallina officinalis L. to Both Predicted Long-Term Increases in Temperature and Short-Term Heatwave Events. Mar. Environ. Res. 2019, 150, 104764. [Google Scholar] [CrossRef]
- Nikolić, V.; Žuljević, A.; Mangialajo, L.; Antolić, B.; Kušpilić, G.; Ballesteros, E. Cartography of Littoral Rocky-Shore Communities (CARLIT) as a Tool for Ecological Quality Assessment of Coastal Waters in the Eastern Adriatic Sea. Ecol. Indic. 2013, 34, 87–93. [Google Scholar] [CrossRef]
- Iveša, L.; Lyons, D.M.; Devescovi, M. Assessment of the Ecological Status of North-Eastern Adriatic Coastal Waters (Istria, Croatia) Using Macroalgal Assemblages for the European Union Water Framework Directive. Aquat. Conserv. Mar. Freshw. Ecosyst. 2009, 19, 14–23. [Google Scholar] [CrossRef]
- Kelaher, B.P. Changes in Habitat Complexity Negatively Affect Diverse Gastropod Assemblages in Coralline Algal Turf. Oecologia 2003, 135, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Kelaher, B.P.; Castilla, J.C.; Seed, R. Intercontinental Test of Generality for Spatial Patterns among Diverse Molluscan Assemblages in Coralline Algal Turf. Mar. Ecol. Prog. Ser. 2004, 271, 221–231. [Google Scholar] [CrossRef][Green Version]
- Matias, M.G.; Underwood, A.J.; Coleman, R.A. Interactions of Components of Habitats Alter Composition and Variability of Assemblages. J. Anim. Ecol. 2007, 76, 986–994. [Google Scholar] [CrossRef]
- Matias, M.G.; Arenas, F.; Rubal, M.; Pinto, I.S. Macroalgal Composition Determines the Structure of Benthic Assemblages Colonizing Fragmented Habitats. PLoS ONE 2015, 10, e0142289. [Google Scholar] [CrossRef]
- Lavender, J.T.; Dafforn, K.A.; Bishop, M.J.; Johnston, E.L. Small-Scale Habitat Complexity of Artificial Turf Influences the Development of Associated Invertebrate Assemblages. J. Exp. Mar. Biol. Ecol. 2017, 492, 105–112. [Google Scholar] [CrossRef]
- Dommasnes, A. On the Fauna of Corallina officinalis L. in Western Norway. Sarsia 1969, 38, 71–86. [Google Scholar] [CrossRef]
- Akioka, H.; Baba, M.; Masaki, T.; Johansen, H.W. Rocky Shore Turfs Dominated by Corallina (Corallinales, Rhodophyta) in Northern Japan. Phycol. Res. 1999, 47, 199–206. [Google Scholar] [CrossRef]
- Kelaher, B.P.; Chapman, M.G.; Underwood, A.J. Spatial Patterns of Diverse Macrofaunal Assemblages in Coralline Turf and Their Associations with Environmental Variables. J. Mar. Biol. Assoc. UK 2001, 81, 917–930. [Google Scholar] [CrossRef]
- Kelaher, B.P.; Carlos Castilla, J. Habitat Characteristics Influence Macrofaunal Communities in Coralline Turf More than Mesoscale Coastal Upwelling on the Coast of Northern Chile. Estuar. Coast. Shelf Sci. 2005, 63, 155–165. [Google Scholar] [CrossRef]
- Bussell, J.A.; Lucas, I.A.N.; Seed, R. Patterns in the Invertebrate Assemblage Associated with Corallina officinalis in Tide Pools. J. Mar. Biol. Assoc. UK 2007, 87, 383–388. [Google Scholar] [CrossRef]
- Kelaher, B.P.; Castilla, J.C.; Prado, L.; York, P.; Schwindt, E.; Bortolus, A. Spatial Variation in Molluscan Assemblages from Coralline Turfs of Argentinean Patagonia. J. Molluscan Stud. 2007, 73, 139–146. [Google Scholar] [CrossRef]
- Kelaher, B.P.; Castilla, J.C.; Prado, L. Is There Redundancy in Bioengineering for Molluscan Assemblages on the Rocky Shores of Central Chile? Rev. Chil. Hist. Nat. 2007, 80, 173–186. [Google Scholar] [CrossRef]
- Berthelsen, A.K.; Hewitt, J.E.; Taylor, R.B. Biological Traits and Taxonomic Composition of Invertebrate Assemblages Associated with Coralline Turf along an Environmental Gradient. Mar. Ecol. Prog. Ser. 2015, 530, 15–27. [Google Scholar] [CrossRef]
- Berthelsen, A.K.; Hewitt, J.E.; Taylor, R.B. Coralline Turf-Associated Fauna Are Affected More by Spatial Variability than by Host Species Identity. Mar. Biodiv 2015, 45, 689–699. [Google Scholar] [CrossRef]
- Buršić, M.; Iveša, L.; Jaklin, A.; Arko Pijevac, M. A Preliminary Study on the Diversity of Invertebrates Associated with Corallina officinalis Linnaeus in Southern Istrian Peninsula. Acta Adriat. Int. J. Mar. Sci. 2019, 60, 127–135. [Google Scholar] [CrossRef]
- Magill, C.L.; Maggs, C.A.; Johnson, M.P.; O’Connor, N. Sustainable Harvesting of the Ecosystem Engineer Corallina officinalis for Biomaterials. Front. Mar. Sci. 2019, 6. [Google Scholar] [CrossRef]
- Wilson, B.; Hayek, L.-A.C. Calcareous Meiofauna Associated with the Calcareous Alga Corallina officinalis on Bedrock and Boulder-Field Shores of Ceredigion, Wales, UK. J. Mar. Biol. Assoc. UK 2020, 100, 1205–1217. [Google Scholar] [CrossRef]
- Lehmann, T.; Spelda, J.; Melzer, R.; Buršić, M. Pycnogonida (Arthropoda) from Northern Adriatic Corallina officinalis Linnaeus, 1758 Belts. Mediterr. Mar. Sci. 2021, 22, 102–107. [Google Scholar] [CrossRef]
- Hebert, P.D.N.; Cywinska, A.; Ball, S.L.; deWaard, J.R. Biological Identifications through DNA Barcodes. Proc. Biol. Sci. 2003, 270, 313–321. [Google Scholar] [CrossRef]
- Boyer, S.L.; Howe, A.A.; Juergens, N.W.; Hove, M.C. A DNA-Barcoding Approach to Identifying Juvenile Freshwater Mussels (Bivalvia:Unionidae) Recovered from Naturally Infested Fishes. J. N. Am. Benthol. Soc. 2011, 30, 182–194. [Google Scholar] [CrossRef]
- Meyer, R.; Weis, A.; Melzer, R.R. Decapoda of Southern Chile: DNA Barcoding and Integrative Taxonomy with Focus on the Genera Acanthocyclus and Eurypodius. Syst. Biodivers. 2013, 11, 389–404. [Google Scholar] [CrossRef]
- Schander, C.; Willassen, E. What Can Biological Barcoding Do for Marine Biology? Mar. Biol. Res. 2005, 1, 79–83. [Google Scholar] [CrossRef]
- Barco, A.; Raupach, M.J.; Laakmann, S.; Neumann, H.; Knebelsberger, T. Identification of North Sea Molluscs with DNA Barcoding. Mol. Ecol. Resour. 2016, 16, 288–297. [Google Scholar] [CrossRef]
- Trivedi, S.; Aloufi, A.A.; Ansari, A.A.; Ghosh, S.K. Role of DNA Barcoding in Marine Biodiversity Assessment and Conservation: An Update. Saudi J. Biol. Sci. 2016, 23, 161–171. [Google Scholar] [CrossRef]
- Ramirez, J.L.; Rosas-Puchuri, U.; Cañedo, R.M.; Alfaro-Shigueto, J.; Ayon, P.; Zelada-Mázmela, E.; Siccha-Ramirez, R.; Velez-Zuazo, X. DNA Barcoding in the Southeast Pacific Marine Realm: Low Coverage and Geographic Representation despite High Diversity. PLoS ONE 2020, 15, e0244323. [Google Scholar] [CrossRef]
- Yang, C.; Lv, Q.; Zhang, A. Sixteen Years of DNA Barcoding in China: What Has Been Done? What Can Be Done? Front. Ecol. Evol. 2020, 8. [Google Scholar] [CrossRef]
- Tillin, H.M.; Budd, G. Coralline Crust-Dominated Shallow Eulittoral Rockpools; Marine Life Information Network: Biology and Sensitivity Key Information Reviews [on-line]; Marine Biological Association of the United Kingdom: Plymouth, UK, 2016. [Google Scholar]
- Nordsieck, F. Die Europäischen Meeres-Gehäuseschnecken (Prosobranchia): Vom Eismeer bis Kapverden, Mittelmeer und Schwarzes Meer; Gustav Fischer Verlag: Stuttgart, Germany, 1968. [Google Scholar]
- Nordsieck, F. Die Europaischen Meeresmuscheln (Bivalvia): Vom Eismeer bis Kapverden, Mittelmeer und Schwarzes Meer; Gustav Fischer Verlag: Stuttgart, Germany, 1969. [Google Scholar]
- Parenzan, P. Carta D’identità Delle Conchiglie del Mediterraneo. Vol. I. Gasteropodi; Bios Taras: Taranto, Italy, 1970. [Google Scholar]
- Parenzan, P. Carta D’identità Delle Conchiglie del Mediterraneo. Vol. II. Bivalvi, Prima Parte; Bios Taras: Taranto, Italy, 1974. [Google Scholar]
- Sabelli, B.; Giannuzzi-Savelli, R.; Bedulli, D. Catalogo Annotato dei Molluschi Marini del Mediterraneo. Vol. I; Libreria Naturalistica Bolognese: Bologna, Italy, 1990. [Google Scholar]
- Poppe, G.T.; Goto, Y. European Seashells Vol. II (Scaphopoda, Bivalvia, Cephalopoda); Verlag Christa Hemmen: Wiesbaden, Germany, 1993. [Google Scholar]
- Gianuzzi-Savelli, R.; Pusateri, F.; Palmeri, A.; Ebreo, C. Atlante Delle Conchiglie Marine Del Mediterraneo; La Conchiglia: Roma, Italy, 1996. [Google Scholar]
- Gofas, S.; Moreno, D.; Salas, C. (Eds.) Moluscos Marinos de Andalucía. Vol. I; Universidad de Málaga: Málaga, Spain, 2011; ISBN 978-84-9747-356-9. [Google Scholar]
- Gofas, S.; Moreno, D.; Salas, C. (Eds.) Moluscos Marinos de Andalucía. Vol. II; Universidad de Málaga: Málaga, Spain, 2011; ISBN 978-84-9747-356-9. [Google Scholar]
- WoRMS—World Register of Marine Species. Available online: http://www.marinespecies.org/ (accessed on 30 March 2021).
- Folmer, O.; Black, M.; Wr, H.; Lutz, R.; Vrijenhoek, R. DNA Primers for Amplification of Mitochondrial Cytochrome C Oxidase Subunit I from Diverse Metazoan Invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar] [PubMed]
- Geller, J.; Meyer, C.; Parker, M.; Hawk, H. Redesign of PCR Primers for Mitochondrial Cytochrome c Oxidase Subunit I for Marine Invertebrates and Application in All-Taxa Biotic Surveys. Mol. Ecol. Resour. 2013, 13, 851–861. [Google Scholar] [CrossRef]
- Lobo, J.; Costa, P.M.; Teixeira, M.A.L.; Ferreira, M.S.G.; Costa, M.H.; Costa, F.O. Enhanced Primers for Amplification of DNA Barcodes from a Broad Range of Marine Metazoans. BMC Ecol. 2013, 13, 34. [Google Scholar] [CrossRef] [PubMed]
- Duda, M.; Schindelar, J.; Macek, O.; Eschner, A.; Kruckenhauser, L. First Record of Trochulus clandestinus (Hartmann, 1821) in Austria (Gastropoda: Eupulmonata: Hygromiidae). Malacol. Bohemoslov. 2017, 16, 37–43. [Google Scholar]
- Hall, T.A. BioEdit: A User-Friendly Biological Sequence Alignment Editor and Analysis Program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C.; et al. Geneious Basic: An Integrated and Extendable Desktop Software Platform for the Organization and Analysis of Sequence Data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef]
- Madeira, F.; Park, Y.; Lee, J.; Buso, N.; Gur, T.; Madhusoodanan, N.; Basutkar, P.; Tivey, A.R.N.; Potter, S.C.; Finn, R.D.; et al. The EMBL-EBI Search and Sequence Analysis Tools APIs in 2019. Nucleic Acids Res. 2019, 47, W636–W641. [Google Scholar] [CrossRef]
- Ratnasingham, S.; Hebert, P.D.N. BOLD: The Barcode of Life Data System. Mol. Ecol. Notes 2007, 7, 355–364. [Google Scholar] [CrossRef]
- Sayers, E.W.; Beck, J.; Bolton, E.E.; Bourexis, D.; Brister, J.R.; Canese, K.; Comeau, D.C.; Funk, K.; Kim, S.; Klimke, W.; et al. Database Resources of the National Center for Biotechnology Information. Nucleic Acids Res. 2021, 49, D10–D17. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Meyer, C.P.; Paulay, G. DNA Barcoding: Error Rates Based on Comprehensive Sampling. PLoS Biol. 2005, 3, e422. [Google Scholar] [CrossRef] [PubMed]
- Audzijonyte, A.; Krylova, E.M.; Sahling, H.; Vrijenhoek, R.C. Molecular Taxonomy Reveals Broad Trans-Oceanic Distributions and High Species Diversity of Deep-Sea Clams (Bivalvia: Vesicomyidae: Pliocardiinae) in Chemosynthetic Environments. Syst. Biodivers. 2012, 10, 403–415. [Google Scholar] [CrossRef]
- Breure, A.S.H. Caribbean Bulimulus Revisited: Physical Moves and Molecular Traces (Mollusca, Gastropoda, Bulimulidae). PeerJ 2016, 4, e1836. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, H. DNA Barcoding for Species Identification in Deep-Sea Clams (Mollusca: Bivalvia: Vesicomyidae). Mitochondrial DNA Part. A 2018, 29, 1165–1173. [Google Scholar] [CrossRef] [PubMed]
- Ratnasingham, S.; Hebert, P.D.N. A DNA-Based Registry for All Animal Species: The Barcode Index Number (BIN) System. PLoS ONE 2013, 8, e66213. [Google Scholar] [CrossRef]
- Zhang, J.; Kapli, P.; Pavlidis, P.; Stamatakis, A. A General Species Delimitation Method with Applications to Phylogenetic Placements. Bioinformatics 2013, 29, 2869–2876. [Google Scholar] [CrossRef]
- Puillandre, N.; Brouillet, S.; Achaz, G. ASAP: Assemble Species by Automatic Partitioning. Mol. Ecol. Resour. 2021, 21, 609–620. [Google Scholar] [CrossRef]
- Puillandre, N.; Lambert, A.; Brouillet, S.; Achaz, G. ABGD, Automatic Barcode Gap Discovery for Primary Species Delimitation: Abgd, automatic barcode gap discovery. Mol. Ecol. 2012, 21, 1864–1877. [Google Scholar] [CrossRef]
- Katoh, K.; Rozewicki, J.; Yamada, K.D. MAFFT Online Service: Multiple Sequence Alignment, Interactive Sequence Choice and Visualization. Brief. Bioinform. 2019, 20, 1160–1166. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Dufayard, J.-F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New Algorithms and Methods to Estimate Maximum-Likelihood Phylogenies: Assessing the Performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Lefort, V.; Longueville, J.-E.; Gascuel, O. SMS: Smart Model Selection in PhyML. Mol. Biol. Evol. 2017, 34, 2422–2424. [Google Scholar] [CrossRef] [PubMed]
- Radulovici, A.E.; Archambault, P.; Dufresne, F. DNA Barcodes for Marine Biodiversity: Moving Fast Forward? Diversity 2010, 2, 450–472. [Google Scholar] [CrossRef]
- Layton, K.K.S.; Martel, A.L.; Hebert, P.D. Patterns of DNA Barcode Variation in Canadian Marine Molluscs. PLoS ONE 2014, 9, e95003. [Google Scholar] [CrossRef] [PubMed]
- Jaksch, K.; Eschner, A.; Rintelen, T.V.; Haring, E. DNA Analysis of Molluscs from a Museum Wet Collection: A Comparison of Different Extraction Methods. BMC Res. Notes 2016, 9, 348. [Google Scholar] [CrossRef]
- Knebelsberger, T.; Stöger, I. DNA Extraction, Preservation, and Amplification. Methods Mol. Biol 2012, 858, 311–338. [Google Scholar] [CrossRef]
- Galindo, L.A.; Puillandre, N.; Strong, E.E.; Bouchet, P. Using Microwaves to Prepare Gastropods for DNA Barcoding. Mol. Ecol. Resour. 2014, 14, 700–705. [Google Scholar] [CrossRef]
- Hebert, P.D.N.; Penton, E.H.; Burns, J.M.; Janzen, D.H.; Hallwachs, W. Ten Species in One: DNA Barcoding Reveals Cryptic Species in the Neotropical Skipper Butterfly Astraptes Fulgerator. Proc. Natl. Acad. Sci. USA 2004, 101, 14812–14817. [Google Scholar] [CrossRef]
- Sweeney, B.W.; Battle, J.M.; Jackson, J.K.; Dapkey, T. Can DNA Barcodes of Stream Macroinvertebrates Improve Descriptions of Community Structure and Water Quality? J. N. Am. Benthol. Soc. 2011, 30, 195–216. [Google Scholar] [CrossRef]
- Hebert, P.D.N.; Stoeckle, M.Y.; Zemlak, T.S.; Francis, C.M. Identification of Birds through DNA Barcodes. PLoS Biol. 2004, 2, e312. [Google Scholar] [CrossRef] [PubMed]
- Meier, R.; Zhang, G.; Ali, F. The Use of Mean Instead of Smallest Interspecific Distances Exaggerates the Size of the “Barcoding Gap” and Leads to Misidentification. Syst. Biol. 2008, 57, 809–813. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Li, Q.; Kong, L.; Yu, H.; Zheng, X.; Yu, R.; Dai, L.; Sun, Y.; Chen, J.; Liu, J.; et al. DNA Barcoding Reveal Patterns of Species Diversity among Northwestern Pacific Molluscs. Sci. Rep. 2016, 6, 33367. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Affenzeller, S.; Haar, N.; Steiner, G. Revision of the Genus Complex Gibbula: An Integrative Approach to Delineating the Eastern Mediterranean Genera Gibbula Risso, 1826, Steromphala Gray, 1847, and Phorcus Risso, 1826 Using DNA-Barcoding and Geometric Morphometrics (Vetigastropoda, Trochoidea). Org. Divers. Evol. 2017, 17, 789–812. [Google Scholar] [CrossRef]
- Barco, A.; Aissaoui, C.; Houart, R.; Bonomolo, G.; Crocetta, F.; Oliverio, M. Revision of the Ocinebrina aciculata Species Complex (Mollusca: Gastropoda: Muricidae) in the Northeastern Atlantic Ocean and Mediterranean Sea. J. Molluscan Stud. 2018, 84, 19–29. [Google Scholar] [CrossRef]
- Barroso, C.X.; de Freitas, J.E.P.; Matthews-Cascon, H.; Bezerra, L.E.A.; Lotufo, T.M.d.C. Molecular Evidences Confirm the Taxonomic Separation of Two Sympatric Congeneric Species (Mollusca, Gastropoda, Neritidae, Neritina). ZooKeys 2020, 904, 117–130. [Google Scholar] [CrossRef]
- Ran, K.; Li, Q.; Qi, L.; Li, W.; Kong, L. DNA Barcoding for Identification of Marine Gastropod Species from Hainan Island, China. Fish. Res. 2020, 225, 105504. [Google Scholar] [CrossRef]
- Fujisawa, T.; Barraclough, T.G. Delimiting Species Using Single-Locus Data and the Generalized Mixed Yule Coalescent Approach: A Revised Method and Evaluation on Simulated Data Sets. Syst. Biol. 2013, 62, 707–724. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Rannala, B.; Yang, Z. Bayesian Species Delimitation Can Be Robust to Guide-Tree Inference Errors. Syst. Biol. 2014, 63, 993–1004. [Google Scholar] [CrossRef]
- Dellicour, S.; Flot, J.-F. Delimiting Species-Poor Data Sets Using Single Molecular Markers: A Study of Barcode Gaps, Haplowebs and GMYC. Syst. Biol. 2015, 64, 900–908. [Google Scholar] [CrossRef]
- Yang, Z. The BPP Program for Species Tree Estimation and Species Delimitation. Curr. Zool. 2015, 61, 854–865. [Google Scholar] [CrossRef]
- Mallo, D.; De Oliveira Martins, L.; Posada, D. SimPhy: Phylogenomic Simulation of Gene, Locus, and Species Trees. Syst. Biol. 2016, 65, 334–344. [Google Scholar] [CrossRef]
- Jackson, N.D.; Carstens, B.C.; Morales, A.E.; O’Meara, B.C. Species Delimitation with Gene Flow. Syst. Biol. 2017, 66, 799–812. [Google Scholar] [CrossRef]
- Luo, A.; Ling, C.; Ho, S.Y.W.; Zhu, C.-D. Comparison of Methods for Molecular Species Delimitation Across a Range of Speciation Scenarios. Syst. Biol. 2018, 67, 830–846. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Li, Q.; Kong, L.; Zheng, X. DNA Barcoding and Phylogenetic Analysis of Pectinidae (Mollusca: Bivalvia) Based on Mitochondrial COI and 16S RRNA Genes. Mol. Biol. Rep. 2011, 38, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Barco, A.; Evans, J.; Schembri, P.J.; Taviani, M.; Oliverio, M. Testing the Applicability of DNA Barcoding for Mediterranean Species of Top-Shells (Gastropoda, Trochidae, Gibbula s.l.). Mar. Biol. Res. 2013, 9, 785–793. [Google Scholar] [CrossRef]
- Furfaro, G.; Modica, M.V.; Oliverio, M.; Mariottini, P. A DNA-Barcoding Approach to the Phenotypic Diversity of Mediterranean Species of Felimare Ev. Marcus & Er. Marcus, 1967 (Mollusca: Gastropoda), with a Preliminary Phylogenetic Analysis. Ital. J. Zool. 2016, 83, 195–207. [Google Scholar] [CrossRef]
- Pejovic, I.; Ardura, A.; Miralles, L.; Arias, A.; Borrell, Y.J.; Garcia-Vazquez, E. DNA Barcoding for Assessment of Exotic Molluscs Associated with Maritime Ports in Northern Iberia. Mar. Biol. Res. 2016, 12, 168–176. [Google Scholar] [CrossRef]
- Giribet, G.; Wheeler, W. On Bivalve Phylogeny: A High-Level Analysis of the Bivalvia (Mollusca) Based on Combined Morphology and DNA Sequence Data. Invertebr. Biol. 2002, 121, 271–324. [Google Scholar] [CrossRef]
- Borrell, Y.J.; Miralles, L.; Huu, H.D.; Mohammed-Geba, K.; Garcia-Vazquez, E. DNA in a Bottle—Rapid Metabarcoding Survey for Early Alerts of Invasive Species in Ports. PLoS ONE 2017, 12, e0183347. [Google Scholar] [CrossRef] [PubMed]
- Taylor, H.R.; Harris, W.E. An Emergent Science on the Brink of Irrelevance: A Review of the Past 8 Years of DNA Barcoding. Mol. Ecol. Resour. 2012, 12, 377–388. [Google Scholar] [CrossRef]
- Bonsdorff, E. Effects of Experimental Oil Exposure on the Fauna Associated with Corallina officinalis L. in Intertidal Rock Pools. Sarsia 1983, 68, 149–155. [Google Scholar] [CrossRef]
- Grahame, J.; Hanna, F.S. Factors Affecting the Distribution of the Epiphytic Fauna of Corallina officinalls (L.) on an Exposed Rocky Shore. Ophelia 1989, 30, 113–129. [Google Scholar] [CrossRef]
- Olabarria, C.; Chapman, M.G. Comparison of Patterns of Spatial Variation of Microgastropods between 2 Contrasting Intertidal Habitats. Mar. Ecol. Prog. Ser. Mar. Ecol. Progr Ser. 2001, 220, 201–211. [Google Scholar] [CrossRef]
| Morphological Determination (Family) | BOLD ID/BLAST Hit (Family; % Similarity) | BIN-RESL (AD/DNN %) | bPTP | ABGD | ASAP | SPECIES Submitted to BOLD (Family)/BOLD Process ID |
|---|---|---|---|---|---|---|
| GASTROPODA Bittium reticulatum (Cerithiidae) | Bittium reticulatum (Cerithiidae; 95.59) | BOLD:ACV9171 (1.77/2.52) | B | C | B | Bittium reticulatum (Cerithiidae)/CROMI001-19 |
| Eatonina sp. juv. (Cingulopsidae) | Sassia bassi (Cymatiidae; 83.28) Amphissa reticulata (Columbellidae; 83.26) | BOLD:AEA4980 * (N/A/16.69) | n.a. | n.a. | n.a. | ** Eatoninasp. (Cingulopsidae)/ CROMI010-19 |
| Eatonina cossurae (Cingulopsidae) | Crepidula convexa (Calyptraeidae; 82.07) Fluminicola sp. (Hydrobiidae; 82.19) | BOLD:AEA4781 * (N/A/17.98) | n.a. | n.a. | n.a. | ** Eatonina cossurae (Cingulopsidae)/ CROMI057-19 |
| Vitreolina antiflexa (Eulimidae) | cf. Crinolamia sp. (Eulimidae; 80.27) | BOLD:AEA6078 * (N/A/16.53) | A | A | A | ** Vitreolina antiflexa (Eulimidae)/ CROMI044-19 |
| cf. Episcomitra cornicula juv. (Mitridae) | Episcomitra cornicula (Mitridae; 98.48) | BOLD:ACT4873 (1.8/7.01) | C | C | C | Episcomitra cornicula (Mitridae)/ CROMI043-19 |
| Muricopsis cristata juv. (Muricidae) | Ocenebra cf. edwardsii (Muricidae; 100.00) | BOLD:ACF7365 (0.19/4.98) | D | D | D | Ocenebracf. edwardsii (Muricidae)/ CROMI059-19 |
| Patella cf. caerulea juv. (Patellidae) | Patella caerulea (Patellidae; 99.56) | BOLD:AAC1245 (0.32/4.74) | C | C | C | Patella caerulea (Patellidae)/ CROMI046-19 |
| Megastomia winfriedi juv. (Pyramidellidae) | Odostomia (Pyramidellidae; 83.64) Boonea cincta (Pyramidellidae; 83.09) | BOLD:AEA7694 * (0.81/15.12) | A | A | A | ** Megastomia winfriedi (Pyramidellidae)/CROMI008-19 |
| Megastomia winfriedi juv. (Pyramidellidae) | Odostomia (Pyramidellidae; 82.17) | BOLD:AEA7694 * (0.81/15.12) | A | A | A | ** Megastomia winfriedi (Pyramidellidae)/CROMI061-19 |
| Odostomia plicata (Pyramidellidae) | (Pyramidellidae; 73.12) | BOLD:AEA6873 * (N/A/25.65) | A | A | A | ** Odostomia plicata (Pyramidellidae)/CROMI048-19 |
| Rissoella sp. juv. (Rissoellidae) | Rissoella japonica (Rissoellidae; 82.01) | BOLD:AEA8121 * (1.99/17.55) | A | A | A | Rissoella sp. (Rissoellidae)/ CROMI009-19 |
| Rissoella sp. juv. (Rissoellidae) | Rissoella japonica (Rissoellidae; 82.36) | BOLD:AEA8121 * (1.99/17.55) | A | A | A | Rissoella sp. (Rissoellidae)/ CROMI034-19 |
| cf. Alvania carinata juv. (Rissoidae) | Cerithium adustum (Cerithiidae; 84.87) Cerithium nodulosum (Cerithiidae; 83.94) | BOLD:AEA4597 * (N/A/15.57) | A | A | A | **Alvaniacf. carinata (Rissoidae)/CROMI041-19 |
| cf. Alvania discors juv. (Rissoidae) | Alvania angioyi (Rissoidae; 86.70) Cingula trifasciata (Rissoidae; 86.35) | BOLD:AEA4635 * (N/A/13.48) | A | A | A | Alvania sp. (Rissoidae)/ CROMI045-19 |
| Crisilla maculata (Rissoidae) | Rissoella japonica (Rissoellidae; 83.94) | BOLD:AEA8121 * (1.99/17.55) | A | A | A | Rissoella sp. (Rissoellidae)/ CROMI053-19 |
| cf. Crisilla maculata (Rissoidae) | Setia turriculata (Rissoidae; 88.06) | BOLD:AEA7055 * (0.32/12.34) | A | A | A | **Crisillacf. maculata (Rissoidae)/ CROMI038-19 |
| cf. Crisilla maculata (Rissoidae) | Setia turriculata (Rissoidae; 87.89) | BOLD:AEA7055 * (0.32/12.34) | A | A | A | **Crisillacf. maculata (Rissoidae)/ CROMI039-19 |
| Rissoidae indet. juv. (Rissoidae) | Rissoella japonica (Rissoellidae; 83.20) | BOLD:AEA8121 * (1.99/17.55) | A | A | A | Rissoella sp. (Rissoellidae)/ CROMI060-19 |
| cf. Gibbula ardens juv. (Trochidae) | Gibbula turbinoides (Trochidae; 96.47) | BOLD:AEA5856 * (0.16/3.69) | B | D | D | Gibbula cf. turbinoides (Trochidae)/ CROMI042-19 |
| cf. Gibbula ardens juv. (Trochidae) | Steromphala adriatica (Trochidae; 99.68) | BOLD:ACA1591 # (0.68/6.41) | E | E | E | Steromphala adriatica (Trochidae)/ CROMI040-19 |
| Gibbula turbinoides juv. (Trochidae) | Gibbula turbinoides (Trochidae; 96.31) | BOLD:AEA5856 * (0.16/3.69) | B | D | D | Gibbula cf. turbinoides (Trochidae)/ CROMI047-19 |
| Sinezona cingulata juv. (Scissurellidae) | Cochlostoma erika (Megalomastomatidae; 80.12) Bellamya robertsoni (Viviparidae; 80.00) | BOLD:AEA3659 * (N/A/19.26) | A | A | A | Gastropoda CROMI007-19 |
| Phorcus turbinatus juv. (Trochidae) | Phorcus turbinatus (Trochidae; 100.00) | BOLD:ACB7685 (0.7/10.57) | C | C | C | Phorcus turbinatus (Trochidae)/ CROMI002-19 |
| Phorcus turbinatus juv. (Trochidae) | Phorcus turbinatus (Trochidae; 100.00) | BOLD:ACB7685 (0.7/10.57) | C | C | C | Phorcus turbinatus (Trochidae)/ CROMI058-19 |
| BIVALVIA Arca tetragona juv. (Arcidae) | Striarca lactea (Noetiidae; 98.62) Arca tetragona (Arcidae; 80.19) | BOLD:ADK1293 (1.14/4.29) | D | D | D | Striarca lactea (Noetiidae)/ CROMI011-19 |
| Carditidae indet. juv. (Carditidae) | Cardita calyculata (Carditidae; 95.07) | BOLD:AEA7765 * (0.75/5.07) | B | C | C | Cardita calyculata (Carditidae)/ CROMI017-19 |
| Carditidae indet. juv. (Carditidae) | Cardita calyculata (Carditidae; 95.07) | BOLD:AEA7765 * (0.75/5.07) | B | C | C | Cardita calyculata (Carditidae)/ CROMI018-19 |
| Carditidae indet. juv. (Carditidae) | Cardita calyculata (Carditidae; 95.07) | BOLD:AEA7765 * (0.75/5.07) | B | C | C | Cardita calyculata (Carditidae)/ CROMI019-19 |
| Carditidae indet. juv. (Carditidae) | Cardita calyculata (Carditidae; 95.07) | BOLD:AEA7765 * (0.75/5.07) | B | C | C | Cardita calyculata (Carditidae)/ CROMI020-19 |
| Carditidae indet. juv. (Carditidae) | Cardita calyculata (Carditidae; 95.22) | BOLD:AEA7765 * (0.75/5.07) | B | C | C | Cardita calyculata (Carditidae)/ CROMI027-19 |
| Carditidae indet. juv. (Carditidae) | Cardita calyculata (Carditidae; 95.38) | BOLD:AEA7765 * (0.75/5.07) | B | C | C | Cardita calyculata (Carditidae)/ CROMI035-19 |
| Lasaea adansoni juv. (Lasaeidae) | Lasaea rubra (Lasaeidae; 94.83) | BOLD:AEA6973 * (N/A/5.45) | B | E | E | Lasaea cf. rubra (Lasaeidae)/ CROMI006-19 |
| Lucinella divaricata juv. (Lucinidae) | Lucinoma annulata (Lucinidae; 76.11) | BOLD:AEA7064 * (N/A/23.76) | A | A | A | Lucinella sp. (Lucinidae)/ CROMI016-19 |
| Gregariella semigranata juv. (Mytilidae) | Mytilus edulis (Mytilidae; 80.59) Mytilus galloprovincialis (Mytilidae; 79.56) | BOLD:AEA7588 * (N/A/21.06) | A | A | A | ** Gregariella semigranata (Mytilidae)/ CROMI062-19 |
| Lithophaga litophaga juv. (Mytilidae) | Lithophaga litophaga (Mytilidae; 99.82) | BOLD:AAX6397 (0.18/30.32) | C | C | C | Lithophaga litophaga (Mytilidae)/ CROMI013-19 |
| Musculus costulatus juv. (Mytilidae) | Crenella faba (Mytilidae; 74.96) Musculus niger, M. discolor (Mytilidae; 73.00) | BOLD:AEA4861 * (0/13.53) | B | B | B | ** Musculus cf. costulatus (Mytilidae)/ CROMI003-19 |
| Musculus costulatus juv. (Mytilidae) | Musculus costulatus (Mytilidae; 86.36) | BOLD:AEA4861 * (0/13.53) | B | B | B | ** Musculuscf. costulatus (Mytilidae)/ CROMI005-19 |
| Musculus sp. juv. (Mytilidae) | Musculus niger, M. discolor (Mytilidae; 74.00) | BOLD:AEA6766 * (N/A/4.62) | A | A | A | Musculus sp. (Mytilidae)/ CROMI015-19 |
| Mytilaster minimus juv. (Mytilidae) | Mytilus galloprovincialis (Mytilidae; 100.00) | BOLD:AAA2184 # (1.8/4.39) | D, E | D, E | D, E | Mytilus galloprovincialis (Mytilidae)/ CROMI004-19 |
| Mytilaster sp. juv. (Mytilidae) | Mytilus galloprovincialis (Mytilidae; 99.17) | BOLD:AAA2184 # (1.8/4.39) | D, E | D, E | D, E | Mytilus galloprovincialis (Mytilidae)/ CROMI052-19 |
| Mytilus galloprovincialis juv. (Mytilidae) | Mytilus galloprovincialis (Mytilidae; 100.00) | BOLD: AAA2184 # (1.8/4.39) | D, E | D, E | D, E | Mytilus galloprovincialis (Mytilidae)/ CROMI056-19 |
| Ostrea sp. juv. (Chamidae) | Chama gryphoides (Chamidae; 98.85) | BOLD:AAW6880 (0.16/22.17) | C | C | C | Chama gryphoides (Chamidae)/ CROMI014-19 |
| Veneridae indet. 1 juv. (Veneridae) | Austrovenus stutchburyi (Veneridae; 82.98) | BOLD:AEA5297 * (1.09/18.26) | A | A | A | Veneridae CROMI012-19 |
| Veneridae indet. 1 juv. (Veneridae) | Austrovenus stutchburyi (Veneridae; 82.86) | BOLD:AEA5297 * (1.09/18.26) | A | A | A | Veneridae CROMI029-19 |
| Veneridae indet. 1 juv. (Veneridae) | Austrovenus stutchburyi (Veneridae; 82.83) | BOLD:AEA5297 * (1.09/18.26) | A | A | A | Veneridae CROMI030-19 |
| Veneridae indet. 2 juv. (Veneridae) | Austrovenus stutchburyi (Veneridae; 82.74) | BOLD:AEA5058 * (0.44/17.18) | A | A | A | Veneridae CROMI021-19 |
| Veneridae indet. 2 juv. (Veneridae) | Austrovenus stutchburyi (Veneridae; 83.74) | BOLD:AEA5058 * (0.44/17.18) | A | A | A | Veneridae CROMI022-19 |
| Veneridae indet. 2 juv. (Veneridae) | Austrovenus stutchburyi (Veneridae; 83.50) | BOLD:AEA5058 * (0.44/17.18) | A | A | A | Veneridae CROMI023-19 |
| Veneridae indet. 2 juv. (Veneridae) | Austrovenus stutchburyi (Veneridae; 83.89) | BOLD:AEA5058 * (0.44/17.18) | A | A | A | Veneridae CROMI024-19 |
| Veneridae indet. 2 juv. (Veneridae) | Austrovenus stutchburyi (Veneridae; 83.89) | BOLD:AEA5058 * (0.44/17.18) | A | A | A | Veneridae CROMI025-19 |
| Veneridae indet. 2 juv. (Veneridae) | Austrovenus stutchburyi (Veneridae; 83.74) | BOLD:AEA5058 * (0.44/17.18) | A | A | A | Veneridae CROMI026-19 |
| Veneridae indet. 3 juv. (Veneridae) | Kurtiella bidentata (Lasaeidae; 85.34) | BOLD:AEA5056 * (N/A/15.14) | A | A | A | Veneridae CROMI028-19 |
| Veneridae indet. 4 juv. (Veneridae) | Kellia suborbicularis (Lasaeidae; 80.26) Petricola rugosa (Veneridae; 79.51) | BOLD:AEA5057 * (N/A/18.97) | A | A | A | Veneridae CROMI055-19 |
| POLYPLACOPHORA Acanthochitona fascicularis juv. (Acanthochitonidae) | Acanthochitona sp. (Acanthochitonidae; 88.77) | BOLD:AEA6823 * (N/A/12.1) | A | A | A | Acanthochitona fascicularis (Acanthochitonidae)/ CROMI037-19 |
| GASTROPODA | c,eOdostomia plicata | aIrus irus |
| c,eAlvania cf. carinata | cPatella caerulea | b,eLasaea cf. rubra |
| dAlvania sp. 1 | c,ePhorcus turbinatus | aLima lima |
| aAlvania sp. 2 | aPusilllina philippi | c,eLithophaga lithophaga |
| a,eAmmonicera fischeriana | a,eRissoa splendida | d,eLucinella sp. |
| cBittium reticulatum | d,eRissoella sp. | cMusculus cf. costulatus |
| a,eCrisilla beniamina | a,eSetia sp. | d,eMusculus sp. |
| aCrisilla innominata | d Gastropoda indet. | cMytilus galloprovincialis |
| a,eCrisilla iunoniae | bSteromphala adriatica | b,eStriarca lactea |
| c,eCrisilla cf. maculata | cVitreolina antiflexa | d Veneridae indet. 1 |
| cEatonina cossurae | d Veneridae indet. 2 | |
| d,eEatonina sp. | BIVALVIA | d Veneridae indet. 3 |
| c,eEpiscomitra cornicula | bCardita calyculata | d Veneridae indet. 4 |
| c,eGibbula cf. turbinoides | b,eChama gryphoides | a Veneridae indet. 5 |
| a,e cf. Gibbula ardens | aFlexopecten glaber | |
| cMegastomia winfriedi | c,eGregariella semigranata | POLYPLACOPHORA |
| b Ocenebra cf. edwardsii | a,eHiatella rugosa | cAcanthochitona fascicularis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buršić, M.; Iveša, L.; Jaklin, A.; Arko Pijevac, M.; Kučinić, M.; Štifanić, M.; Neal, L.; Bruvo Mađarić, B. DNA Barcoding of Marine Mollusks Associated with Corallina officinalis Turfs in Southern Istria (Adriatic Sea). Diversity 2021, 13, 196. https://doi.org/10.3390/d13050196
Buršić M, Iveša L, Jaklin A, Arko Pijevac M, Kučinić M, Štifanić M, Neal L, Bruvo Mađarić B. DNA Barcoding of Marine Mollusks Associated with Corallina officinalis Turfs in Southern Istria (Adriatic Sea). Diversity. 2021; 13(5):196. https://doi.org/10.3390/d13050196
Chicago/Turabian StyleBuršić, Moira, Ljiljana Iveša, Andrej Jaklin, Milvana Arko Pijevac, Mladen Kučinić, Mauro Štifanić, Lucija Neal, and Branka Bruvo Mađarić. 2021. "DNA Barcoding of Marine Mollusks Associated with Corallina officinalis Turfs in Southern Istria (Adriatic Sea)" Diversity 13, no. 5: 196. https://doi.org/10.3390/d13050196
APA StyleBuršić, M., Iveša, L., Jaklin, A., Arko Pijevac, M., Kučinić, M., Štifanić, M., Neal, L., & Bruvo Mađarić, B. (2021). DNA Barcoding of Marine Mollusks Associated with Corallina officinalis Turfs in Southern Istria (Adriatic Sea). Diversity, 13(5), 196. https://doi.org/10.3390/d13050196







