Varietal Threat Index for Monitoring Crop Diversity on Farms in Five Agro-Ecological Regions in India
Abstract
:1. Introduction
2. Methods
2.1. Survey Sites
2.2. Focus Group Discussion
2.3. Four-Cell Analysis Method
2.4. Varietal Threat Index
- Few households and on small areas are considered as threatened (TR, Q1);
- Few households but on large areas are considered as near threatened (NT, Q2);
- Many households and on small areas are considered as vulnerable (VU, Q3);
- Many households and on large areas are considered of least concern (LC; Q4); and
- Lost varieties grew in the past 10 years but that are no longer grown (lost, Q5)
3. Results
3.1. FGD Participation by Age and Gender
3.2. Species Richness and Varieties Evenness
3.3. Varietal Threat Index: Threat Status and Trends
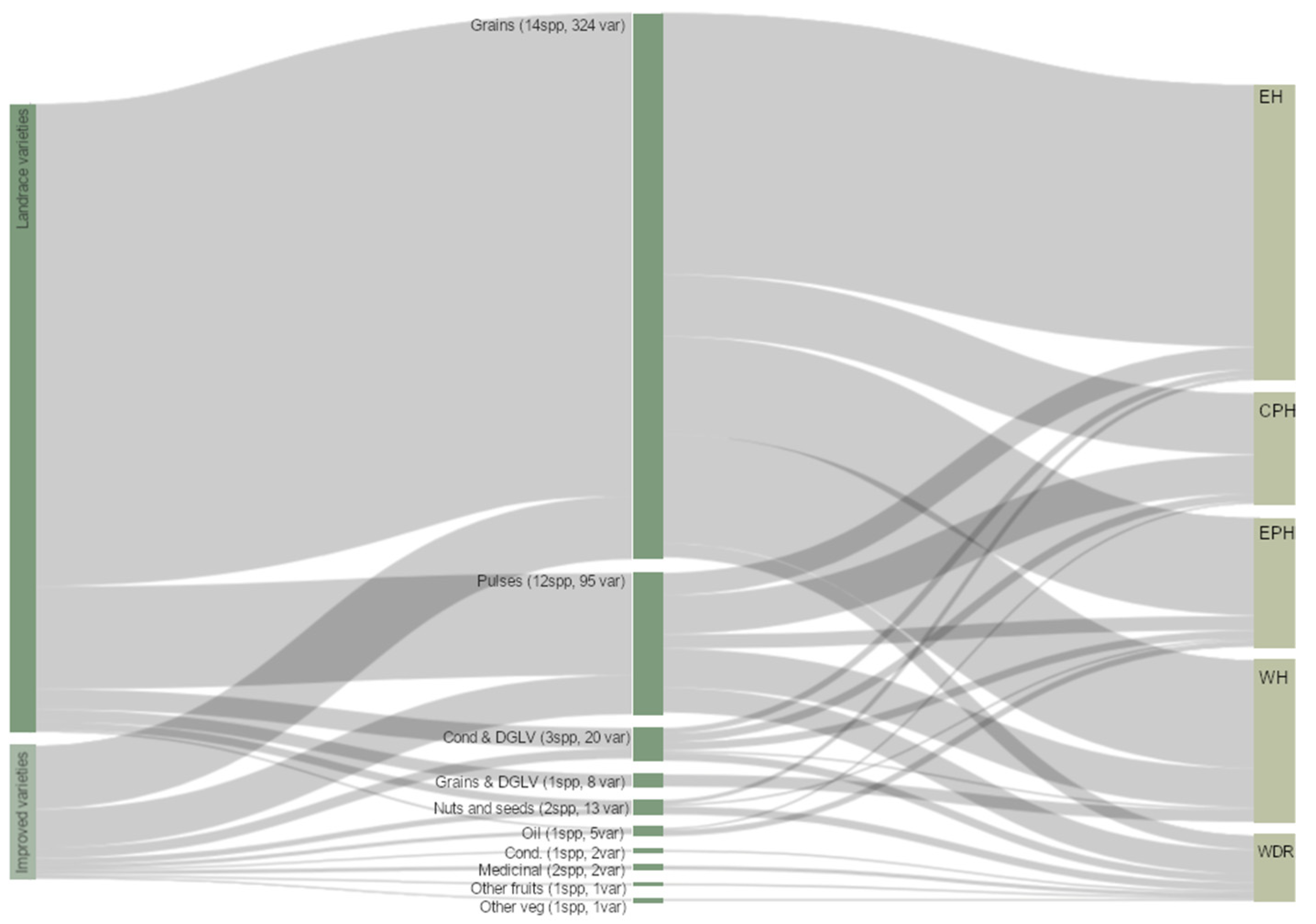
3.4. Crop and Food Groups
4. Discussion
4.1. Varietal Diversity
4.2. Varietal Threat Index
4.3. Monitoring Trends in Varietal Diversity
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De Haan, S.; Burgos, G.; Liria, R.; Rodriguez, F.; Creed-Kanashiro, H.M.; Bonierbale, M. The nutritional contribution of potato varietal diversity in Andean food systems: A case study. Am. J. Potato Res. 2019, 96, 151–163. [Google Scholar] [CrossRef]
- Gotor, E.; Usman, M.A.; Occelli, M.; Fantahun, B.; Fadda, C.; Kidane, Y.G.; Mengistu, D.; Kiros, A.Y.; Mohammed, J.N.; Assefa, M.; et al. Wheat varietal diversification increases Ethiopian smallholders’ food security: Evidence from a participatory development initiative. Sustainability 2021, 13, 1029. [Google Scholar] [CrossRef]
- FAO. The Second Report on the State of the World’s Animal Genetic Resources for Food and Agriculture; Food and Agriculture Organization of the United Nations: Rome, Italy, 2010. [Google Scholar] [CrossRef]
- Bioversity International. Mainstreaming Agrobiodiversity in Sustainable Food Systems: Scientific Foundations for an Agrobiodiversity Index; Bioversity International: Rome, Italy, 2017. [Google Scholar]
- Bisht, I.S.; Rana, J.C.; Yadav, R.; Ahlawat, S.P. Mainstreaming agricultural biodiversity in traditional production landscapes for sustainable development: The Indian Scenario. Sustainability 2020, 12, 10690. [Google Scholar] [CrossRef]
- Gatto, M.; De Haan, S.; Laborte, A.; Bonierbale, M.; Labarta, R.; Hareau, G. Trends in varietal diversity of main staple crops in Asia and Africa and implications for sustainable food systems. Front. Sustain. Food Syst. 2021, 5, 12. [Google Scholar] [CrossRef]
- FAO. Coping with Climate Change: The Roles of Genetic Resources for Food and Agriculture; Food and Agriculture Organization of the United Nations, Ed.; FAO: Rome, Italy, 2015. [Google Scholar]
- Belem, M.; Bazile, D.; Coulibaly, H. Simulating the impacts of climate variability and change on crop varietal diversity in Mali (West-Africa) using agent-based modeling approach. J. Artif. Soc. Soc. Simul. 2018, 21, 8. [Google Scholar] [CrossRef] [Green Version]
- Widawsky, D.; Rozelle, S. Varietal diversity and yield variability in chinese rice production. In Farmers Gene Banks and Crop Breeding: Economic Analyses of Diversity in Wheat Maize and Rice; Smale, M., Ed.; Springer: Dordrecht, The Netherlands, 1998. [Google Scholar] [CrossRef]
- Negri, V.; Maxted, N.; Vetelainen, M. European landraces: On-farm conservation, management and use. In European Landraces: On Farm Conservation, Management and Use; Vetelainen, M., Negri, V., Maxted, N., Eds.; Bioversity International: Rome, Italy, 2009. [Google Scholar]
- Juventia, S.D.; Jones, S.K.; Laporte, M.-A.; Remans, R.; Villani, C.; Estrada-Carmona, N. Text mining national commitments towards agrobiodiversity conservation and use. Sustainability 2020, 12, 715. [Google Scholar] [CrossRef] [Green Version]
- Sthapit, B.R.; Rana, R.B.; Subedi, A.; Gyawali, S.; Bajracharya, J.; Chaudhary, P.; Joshi, B.K.; Sthapit, S.; Joshi, K.D.; Upadhyay, M.P. Participatory four cell analysis (FCA) for local crop diversity. In Good Practices: On-Farm Management of Agricultural Biodiversity in Nepal; NARC, LI-BIRD, IPGRI and IDRC, Nepal: Nagarkot, Nepal, 2006; Volume 260. [Google Scholar]
- FAO. The State of the World’s Biodiversity for Food and Agriculture; Fao Commission on Genetic Resources for Food and Agriculture Assessments: Rome, Italy, 2019. [Google Scholar]
- Dulloo, M.E.; Thormann, I.; Drucker, A.G. 39 What do we have to lose? Monitoring crop genetic diversity. In Enhancing Crop Genepool Use: Capturing Wild Relative and Landrace Diversity for Crop Improvement; Maxted, N., Dulloo, M.E., Lloyd, B.F., Eds.; CABI: Wallingford, UK, 2016; p. 421. [Google Scholar]
- Thormann, I.; Engels, J.M.M. Genetic diversity and erosion—A global perspective. In Genetic Diversity and Erosion in Plants. Sustainable Development and Biodiversity; Ahuja, M., Jain, S., Eds.; Springer: Berlin/Heidelberg, Germany, 2015. [Google Scholar] [CrossRef]
- Bisht, I.S.; Rana, J.C.; Pal Ahlawat, S. The future of smallholder farming in India: Some sustainability considerations. Sustainability 2020, 12, 3751. [Google Scholar] [CrossRef]
- De Haan, S.; Polreich, S.; Rodriguez, F.; Juarez, H.; Ccanto, R.; Alvarez, C.; Otondo, A.; Sainz, H.; Venegas, C.; Kalazich, J. A Long-Term Systematic Monitoring Framework for On-Farm Conserved Potato Landrace Diversity. Enhancing Crop Genepool Use Ca Capturing Wild Relative and Landrace Diversity for Crop Improvement; Maxted, N., Dulloo, M.E., Lloyd, B.F., Eds.; Cabi International: Wallingford, UK, 2016; pp. 289–296. [Google Scholar]
- Raggi, L.; Pacicco, C.L.; Barata, A.M.; Bartha, B.; Heinonen, M.; Maxted, N.; Ralli, P.; Negri, V. Landrace Hotspots Identification in Europe. Farmer’s Pride: Networking, Partnerships and Tools to Enhance in Situ Conservation of European Plant Genetic Resources. 2020. Available online: https://more.bham.ac.uk/farmerspride/wpcontent/uploads/sites/19/2020/10/D1.4_Landrace_hotspots_identification_in_Europe.pdf (accessed on 10 August 2021).
- Jarvis, D.I.; Brown, A.H.D.; Cuong, P.H.; Collado-Panduro, L.; Latournerie-Moreno, L.; Gyawali, S.; Tanto, T.; Sawadogo, M.; Mar, I.; Sadiki, M. A global perspective of the richness and evenness of traditional crop-variety diversity maintained by farming communities. Proc. Natl. Acad. Sci. USA 2008, 105, 5326–5331. [Google Scholar] [CrossRef] [Green Version]
- Dulloo, M.E.; Bissessur, P.; Rana, J. Monitoring plant genetic resources for food and agriculture. In Plant Genetic Resources: A Review of Current Research and Future Needs; Dulloo, M.E., Ed.; Burleigh Dodds Science Publishing: Cambridge, UK, 2021; pp. 55–80. [Google Scholar]
- Padulosi, S.; Bergamini, N.; Lawrence, T. On-Farm Conservation of Neglected and Underutilized Species: Status, Trends and Novel Approaches to Cope with Climate Change: Proceedings of an International Conference, Frankfurt, Germany, 14–16 June 2011; Bioversity International: Rome, Italy, 2012. [Google Scholar]
- FAO. International Targets and Indicators for Biodiversity for Food and Agriculture; Commission on Genetic resources for Food and Agriculture, Thirteenth Regular Session. 2011, CGRFA 13/11/18; FAO: Rome, Italy, 2011. [Google Scholar]
- CBD. AICHI Biodiversity Target. Available online: https://www.cbd.int/sp/targets/ (accessed on 16 June 2021).
- Jones, S.; Estrada-Carmona, N.; Juventia, S.D.; Dulloo, M.; Laporte, M.; Villani, C.; Remans, R. An Agrobiodiversity Index scores show agrobiodiversity is underutilized in national Food System Policy. Nat. Food 2021, 2, 712–723. [Google Scholar] [CrossRef]
- Hoban, S.; Bruford, M.; Jackson, J.D.; Lopes-Fernandes, M.; Heuertz, M.; Hohenlohe, P.A.; Paz-Vinas, I.; Sjögren-Gulve, P.; Segelbacher, G.; Vernesi, C.; et al. Genetic diversity targets and indicators in the CBD post-2020 global biodiversity framework must be improved. Biol. Conserv. 2020, 248, 108654. [Google Scholar] [CrossRef]
- Bruford, M.W.; Davies, N.; Dulloo, M.E.; Faith, D.P.; Walters, M. Monitoring changes in genetic diversity. In The GEO Handbook on Biodiversity Observation Networks; Walters, M., Scholes, R.J., Eds.; Springer International Publishing: Cham, Switzerland, 2017. [Google Scholar] [CrossRef] [Green Version]
- Mondini, L.; Noorani, A.; Pagnotta, M. Assessing plant genetic diversity by molecular tools. Diversity 2009, 1, 19–35. [Google Scholar] [CrossRef] [Green Version]
- Brown, A.H.D.; Hodgkin, T. 2. Measuring, managing, and maintaining crop genetic diversity on farm. In Managing Biodiversity in Agricultural Ecosystems; Jarvis, D.I., Padoch, C., Cooper, H.D., Eds.; Columbia University Press: New York, NY, USA, 2007; pp. 13–33. [Google Scholar]
- Rana, R.B.; Sthapit, B.R.; Garforth, C.; Subedi, A.; Jarvis, D.I. Four-cell analysis as a decision-making tool for conservation of agrobiodiversity on-farm. In On-Farm Conservation of Agricultural Biodiversity in Nepal; Sthapit, B.R., Upadhyay, M.P., Shrestha, P.K., Jarvis, D.I., Eds.; Bioversity International Nepal: Nagarkot, Nepal, 2005; Volume 2, pp. 15–24. [Google Scholar]
- Vernooy, R.; Sthapit, B.; Tjikana, T.T.; Dibiloane, A.; Maluleke, N.; Mukoma, T. Embracing Diversity: Inputs for a Strategy to Support Community Seedbanks in South Africa’s Smallholder Farming Areas: Report of Field Visits to Limpopo and Eastern Cape; Bioversity International: Rome, Italy; Department of Agriculture, Forestry and Fisheries: Pretoria, South Africa, 2013. [Google Scholar]
- Khanal, S.; Dangol, D.R. Assessment of landraces of rice and maize by four cell analysis at Sorabhag, V.D.C. Marang, Nepal. Himal. Biodivers. 2016, 4, 49–52. [Google Scholar]
- Ghimire, Y.N.; Rana, R.B.; Ale, S.; Poudel, I.; Tamang, B.B. Use of agrobiodiversity and crop management practices for climate change adaptation in high hill agriculture of Nepal. J. Agric. Environ. 2017, 18, 6–14. [Google Scholar] [CrossRef]
- Butchart, S.H.M.; Resit Akçakaya, H.; Chanson, J.; Baillie, J.E.M.; Collen, B.; Quader, S.; Turner, W.R.; Amin, R.; Stuart, S.N.; Hilton-Taylor, C. Improvements to the red list index. PLoS ONE 2007, 2, e140. [Google Scholar] [CrossRef]
- IUCN. IUCN Red List Categories and Criteria: Version 3.1; IUCN: Gland, Switzerland, 2012. [Google Scholar]
- Butchart, S.H.M.; Stattersfield, A.J.; Bennun, L.A.; Shutes, S.M.; Akçakaya, H.R.; Baillie, J.E.M.; Stuart, S.N.; Hilton-Taylor, C.; Mace, G.M.; Reid, W.V. Measuring global trends in the status of biodiversity: Red list indices for birds. PLoS Biol. 2004, 2, e383. [Google Scholar] [CrossRef] [Green Version]
- Marler, P.N.; Marler, T.E. An assessment of red list data for the cycadales. Trop. Conserv. Sci. 2015, 8, 1114–1125. [Google Scholar] [CrossRef] [Green Version]
- Lewis, O.T.; Senior, M.J.M. Assessing conservation status and trends for the world’s butterflies: The sampled red list index approach. J. Insect Conserv. 2011, 15, 121–128. [Google Scholar] [CrossRef]
- Hammer, K.; Hammer-Spahillari, M.; Khoshbakht, K. Red lists for cultivated species: Experiences with the IUCN list of threatened plants. In On Farm Conservation of Neglected and Underutilized Species: Status, Trends and Novel Approaches to Cope with Climate Change: Proceedings of an International Conference, Frankfurt, Germany 14–16 June 2011; Padulosi, S., Bergamini, N., Lawrence, T., Eds.; Bioversity International: Rome, Italy, 2012; pp. 127–136. [Google Scholar]
- Davis, U. Minimum Dietary Diversity for Women—A Guide to Measurement; FAO: Rome, Italy, 2016. [Google Scholar]
- Oksanen, J.; Blanchet, F.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Peter, R.; Minchin, P.R.; O’hara, R.B.; Simpson, G.L.; et al. Vegan: Community Ecology Package. R Package Version 2.5-7. Available online: https://CRAN.R-Project.Org/Package=vegan (accessed on 25 August 2021).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.r-project.org/ (accessed on 25 August 2021).
- Patil, I. Visualizations with statistical details: The ‘ggstatsplot’ approach. J. Open Source Softw. 2021, 6, 3167. [Google Scholar] [CrossRef]
- Allaire, J.J.; Gandrud, C.; Russell, K.; Yetman, C.J. NetworkD3: D3 javascript network graphs from R. R Package 2017 Version 0.4, 2014. [Google Scholar]
- Herrero, M.; Thornton, P.K.; Power, B.; Bogard, J.R.; Remans, R.; Fritz, S.; Gerber, J.S.; Nelson, G.; See, L.; Waha, K. Farming and the geography of nutrient production for human use: A transdisciplinary analysis. Lancet Planet. Health 2017, 1, e33–e42. [Google Scholar] [CrossRef] [Green Version]
- Teshome, A.; Torrance, J.K.; Baum, B.; Fahrig, L.; Lambert, J.D.H.; Arnason, J.T. Traditional farmers’ knowledge of sorghum (Sorghum nicolor, [Poaceae]) landrace storability in Ethiopia. Econ. Bot. 1999, 53, 69–78. [Google Scholar] [CrossRef]
- Estrada-Carmona, N.; Attwood, S.; Cole, S.M.; Remans, R.; DeClerck, F. A gendered ecosystem services approach to identify novel and locally-relevant strategies for jointly improving food security, nutrition, and conservation in the barotse floodplain. Int. J. Agric. Sustain. 2020, 18, 351–375. [Google Scholar] [CrossRef]
- Galhena, D.H.; Freed, R.; Maredia, K.M. Home gardens: A promising approach to enhance household food security and wellbeing. Agric. Food Secur. 2013, 2, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Feliciano, D. A review on the contribution of crop diversification to Sustainable Development Goal 1 “No poverty” in different world regions. Sustain. Dev. 2019, 27, 795–808. [Google Scholar] [CrossRef] [Green Version]
- Villamor, G.B.; Van Noordwijk, M.; Djanibekov, U.; Chiong-Javier, M.E.; Catacutan, D. gender differences in land-use decisions: Shaping multifunctional landscapes? Curr. Opin. Environ. Sustain. 2014, 6, 128–133. [Google Scholar] [CrossRef] [Green Version]
- Farnworth, C.R.; Baudron, F.; Andersson, J.A.; Misiko, M.; Badstue, L.; Stirling, C.M. Gender and conservation agriculture in East and Southern Africa: Towards a research agenda. Int. J. Agric. Sustain. 2016, 14, 142–165. [Google Scholar] [CrossRef]
- Nelson, A.R.L.E.; Ravichandran, K.; Antony, U. The impact of the green revolution on indigenous crops of India. J. Ethn. Foods 2019, 6, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Rana, J.C.; Tyagi, R.K.; Sharma, S.K. On-farm conservation–initiatives by NBPGR in the Western Himalayan Region. Indian J. Plant Genet. Resour. 2010, 23, 122–125. [Google Scholar]
- Mandizvo, T.; Odindo, A.O.; Mashilo, J. Citron watermelon potential to improve crop diversification and reduce negative impacts of climate change. Sustainability 2021, 13, 2269. [Google Scholar] [CrossRef]
- Jiménez-Ballesta, R.; García-Navarro, F.J.; García-Giménez, R.; Trujillo-González, J.M.; Iñigo, V.; Asensio, C. Agroecological analysis of cucumber (Cucumis Sativus L.) crops in orchards in a Mediterranean environment. J. Agric. Crop. 2018. [Google Scholar]
- Genesys. Available online: https://www.genesys-pgr.org (accessed on 25 August 2021).

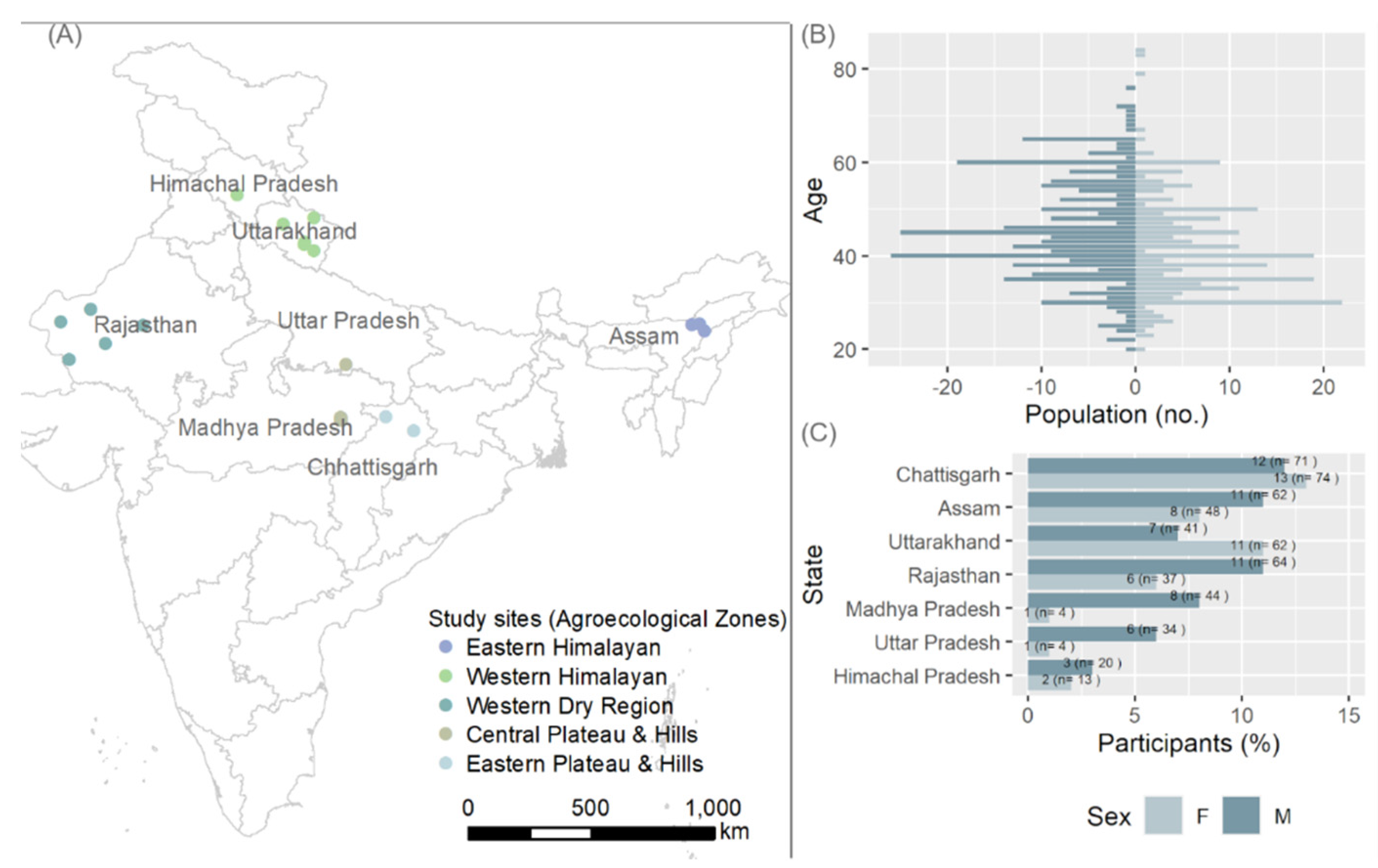
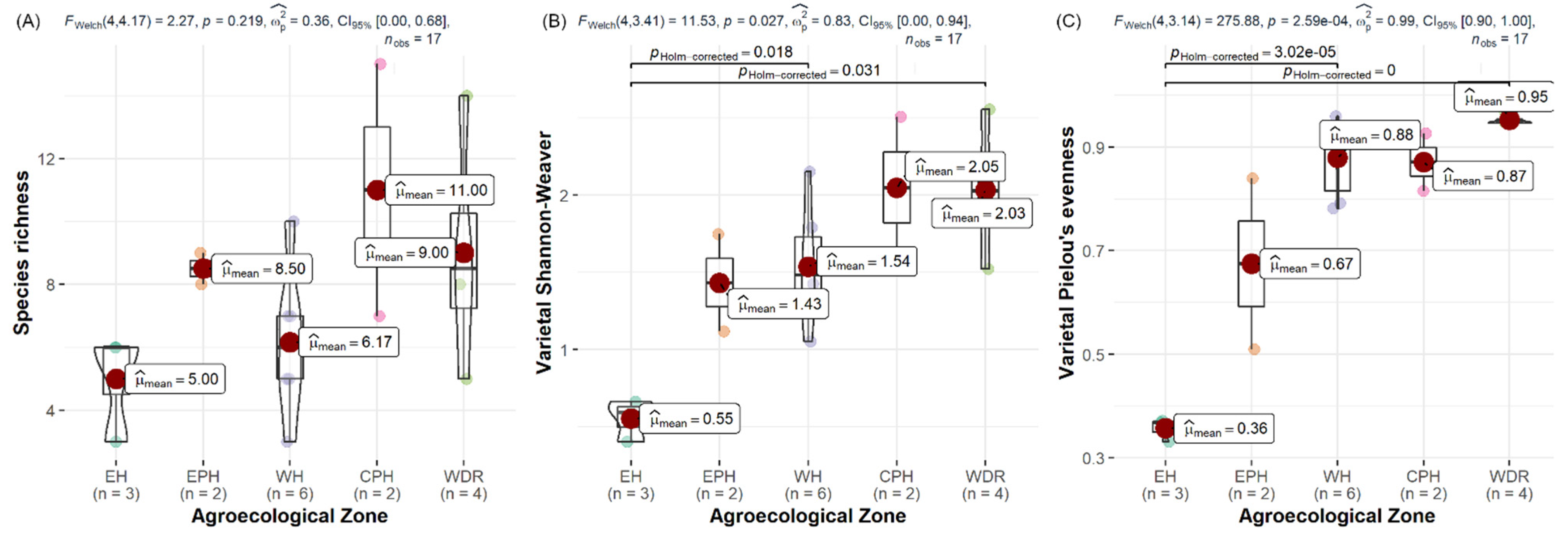
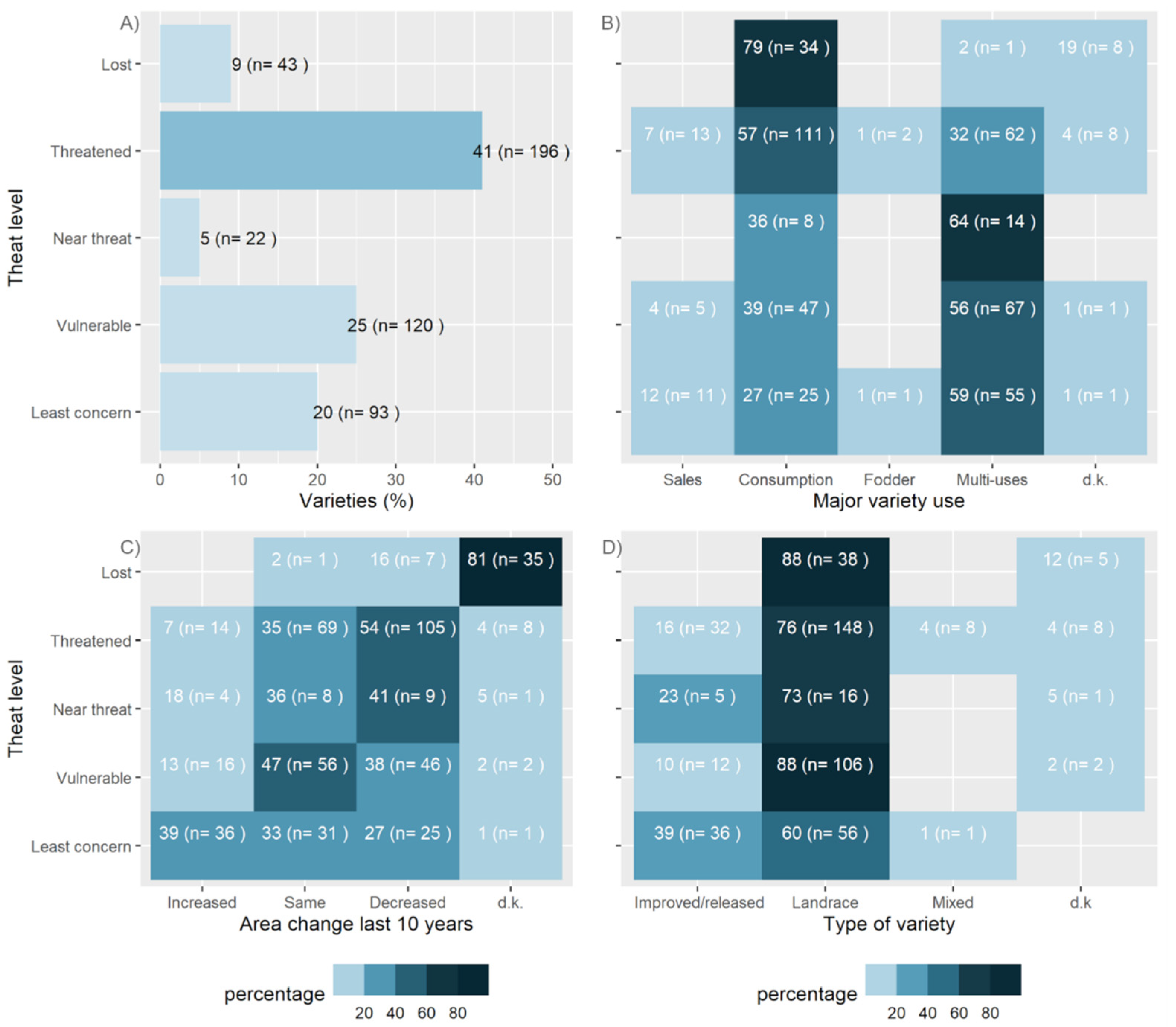
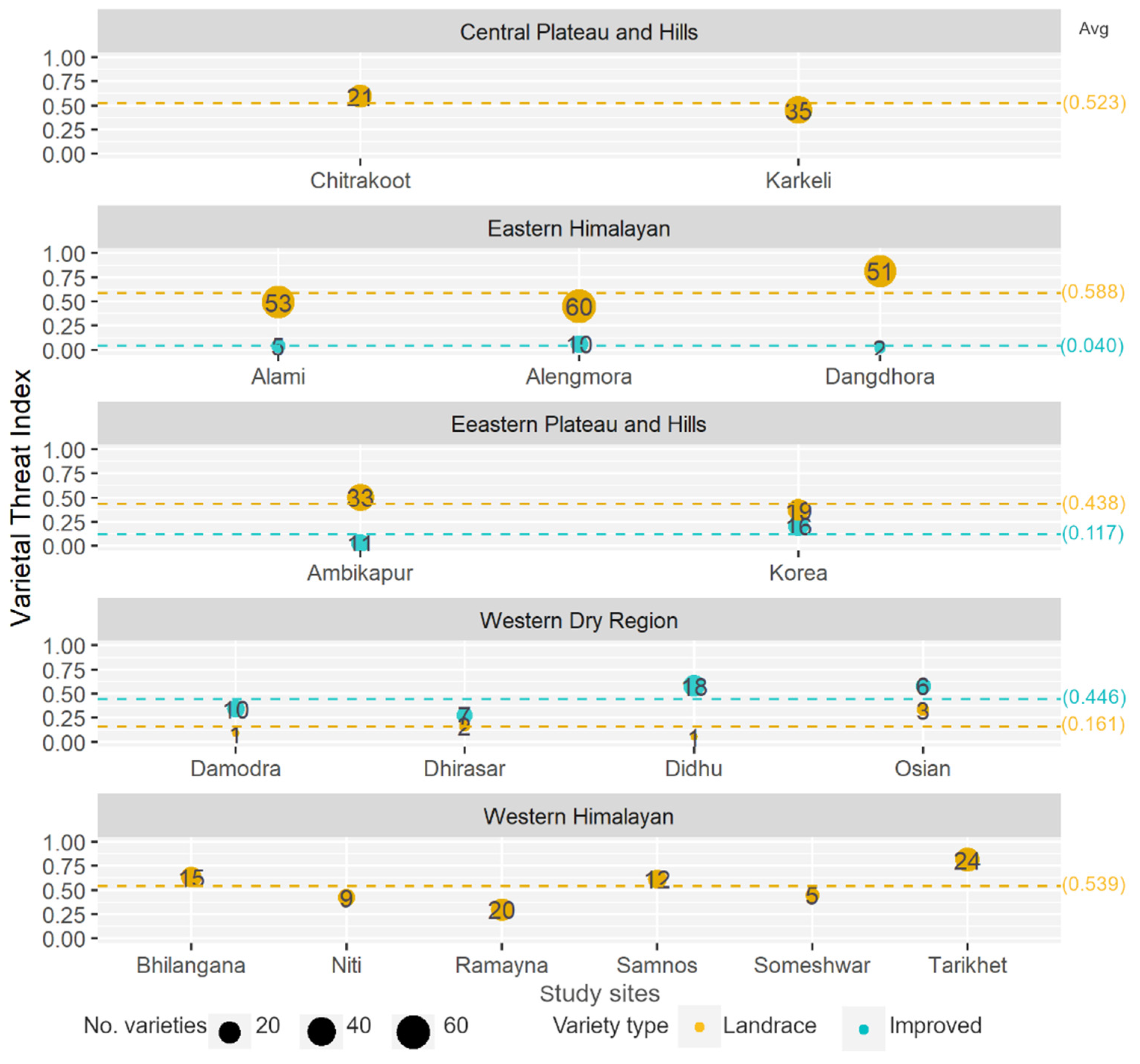
| Agroecological Region/Features | STUDY SITES NAMES (Number of Villages) |
|---|---|
| Eastern Himalaya | |
| Hot, humid, deep, loamy to clayey alluvium-derived soils, available water capacity (AWC) is medium, LGP 240–270 days, and 50–80% of area under forests. High temperatures combined with heavy to very heavy rains. | Dangdhora (7) Alengmora (5) Alami (11) |
| Western Himalaya | |
| Soils shallow, loamy skeletal soils, cold to cool typic-arid, available water capacity (AWC) is low, and length of growing period (LGP) is 60–90 days. The Western Himalayan vegetation is broadleaf and of two types: evergreen and deciduous broadleaved forests. | Bhilangana Valley (5) Niti Valley (8) Tarikhet (8) Someshwar (8) Ramana Nyay (12) Gohar Valley (10) |
| Eastern Plateau and Hills | |
| Moderately to gently sloping basin, hot, moist/dry, subhumid transitional, deep loamy to clayey red and yellow soils, AWC medium, and LGP 150–180 days. Vegetation is tropical moist deciduous, tropical dry deciduous, and subtropical broad-leaved hill forests. | Ambikapur (11) Korea (9) |
| Central Plateau and Hills | |
| Hot, dry, subhumid, deep loamy to clayey mixed red and black soils, AWC medium to high, and LGP 150–180 days. Vegetation of Uttar Pradesh consists mostly of scrub. Forests are generally concentrated in the southern uplands and predominantly consist of tropical dry deciduous types. | Karkeli (17) Chitarkoot (19) |
| Western Dry Region | |
| Hot typic-arid, deep, loamy desert soils (inclusion of saline phase), AWC very low, and LGP 60–90 days. Forest types include dry deciduous forest and thorn forest including scrub jungle. | Dheerasar (6) Didhu (8) Damodara (5) Osian (6) |
| Spread | |||
|---|---|---|---|
| Small Areas (≤10%) | Large Areas (>10%) | ||
| Abundance | Few households (≤ 10%) | Q1-Threatened (TR) (weight:4) | Q2-Near Threatened (NT) (weight:3) |
| Many households (> 10%) | Q3-Vulnerable (VU) (weight:2) | Q4-Least Concern (LC) (weight:0) | |
| Lost varieties | Q5-Varieties which they grew in the past 10 years but were no longer grown (weight:5) | ||
| Crop | Scientific Name of Cultivated Species | Number of Varieties | Percentage |
|---|---|---|---|
| Grain Amaranth | Amaranthus hypochondriacus L. | 8 | 1.7 |
| Groundnut | Arachis hypogaea L. | 2 | 0.4 |
| Indian mustard | Brassica juncea L. | 15 | 3.2 |
| Yellow mustard | Brassica rapa L. | 4 | 0.8 |
| Pigeon pea | Cajanus cajan (Linn.) Huth | 18 | 3.8 |
| Chickpea | Cicer arietinum L. | 7 | 1.5 |
| Bitter cucumber | Citrullus colocynthis (L.) Schrad. | 1 | 0.2 |
| Watermelon | Citrullus lanatus (Thunb.) Matsum. & Nakai | 1 | 0.2 |
| Cumin | Cuminum cyminum L. | 2 | 0.4 |
| Cluster bean | Cyamopsis tetragonoloba (l.) Taub. | 11 | 2.3 |
| Barnyard millet | Echinochloa esculenta (A.Braun) H.Scholz | 1 | 0.2 |
| Finger millet | Eleusine coracana (L.) Gaertn. | 12 | 2.5 |
| Common buckwheat | Fagopyrum esculentum Moench. | 2 | 0.4 |
| Tartary buckwheat | Fagopyrum tataricum (L.) Gaertn. | 2 | 0.4 |
| Soybean | Glycine max (L.) Merr. | 5 | 1.1 |
| Niger | Guizotia abyssinica (L.f.) Cass. | 5 | 1.1 |
| Barley | Hordeum vulgare L. | 1 | 0.2 |
| Lentil | Lens culinaris Medik. | 3 | 0.6 |
| Horse gram | Macrotyloma uniflorum (Lam.) Verdc. | 2 | 0.4 |
| Rice | Oryza sativa L. | 259 | 55.0 |
| Proso millet | Panicum miliaceum Walter | 1 | 0.2 |
| Little millet | Panicum sumatrense Roth | 2 | 0.4 |
| Kodo millet | Paspalum scrobiculatum L. | 1 | 0.2 |
| Pearl millet | Pennisetum glaucum (L.) R. Br. | 9 | 1.9 |
| Common Bean | Phaseolus vulgaris L. | 8 | 1.7 |
| Pea | Pisum sativum L. | 2 | 0.4 |
| Psyllium | Plantago ovata Forssk. | 1 | 0.2 |
| Castor oil | Ricinus communis L. | 1 | 0.2 |
| Sesame | Sesamum indicum L. | 11 | 2.3 |
| Foxtail Millet | Setaria italica (L.) P.Beauv. | 1 | 0.2 |
| Sorghum | Sorghum bicolor (L.) Moench. | 1 | 0.2 |
| Fenugreek | Trigonella foenum-graecum L. | 1 | 0.2 |
| Wheat | Triticum aestivum L. | 23 | 4.9 |
| Moth bean | Vigna aconitifolia (Jacq.) Maréchal | 7 | 1.5 |
| Black gram | Vigna mungo (L.) Hepper | 16 | 3.4 |
| Mung bean | Vigna radiata (L.) R. Wilczek | 15 | 3.2 |
| Rice bean | Vigna umbellate (Thunb.) Ohwi &H.Ohashi | 1 | 0.2 |
| Maize | Zea_mays L. | 9 | 1.9 |
| Total number | 38 | 471 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dulloo, M.E.; Estrada Carmona, N.; Rana, J.C.; Yadav, R.; Grazioli, F. Varietal Threat Index for Monitoring Crop Diversity on Farms in Five Agro-Ecological Regions in India. Diversity 2021, 13, 514. https://doi.org/10.3390/d13110514
Dulloo ME, Estrada Carmona N, Rana JC, Yadav R, Grazioli F. Varietal Threat Index for Monitoring Crop Diversity on Farms in Five Agro-Ecological Regions in India. Diversity. 2021; 13(11):514. https://doi.org/10.3390/d13110514
Chicago/Turabian StyleDulloo, Mohammad Ehsan, Natalia Estrada Carmona, Jai C. Rana, Rashmi Yadav, and Francesca Grazioli. 2021. "Varietal Threat Index for Monitoring Crop Diversity on Farms in Five Agro-Ecological Regions in India" Diversity 13, no. 11: 514. https://doi.org/10.3390/d13110514
APA StyleDulloo, M. E., Estrada Carmona, N., Rana, J. C., Yadav, R., & Grazioli, F. (2021). Varietal Threat Index for Monitoring Crop Diversity on Farms in Five Agro-Ecological Regions in India. Diversity, 13(11), 514. https://doi.org/10.3390/d13110514







