Brachyuran Crabs (Decapoda) Associated with Rhodolith Beds: Spatio-Temporal Variability at Gran Canaria Island
Abstract
1. Introduction
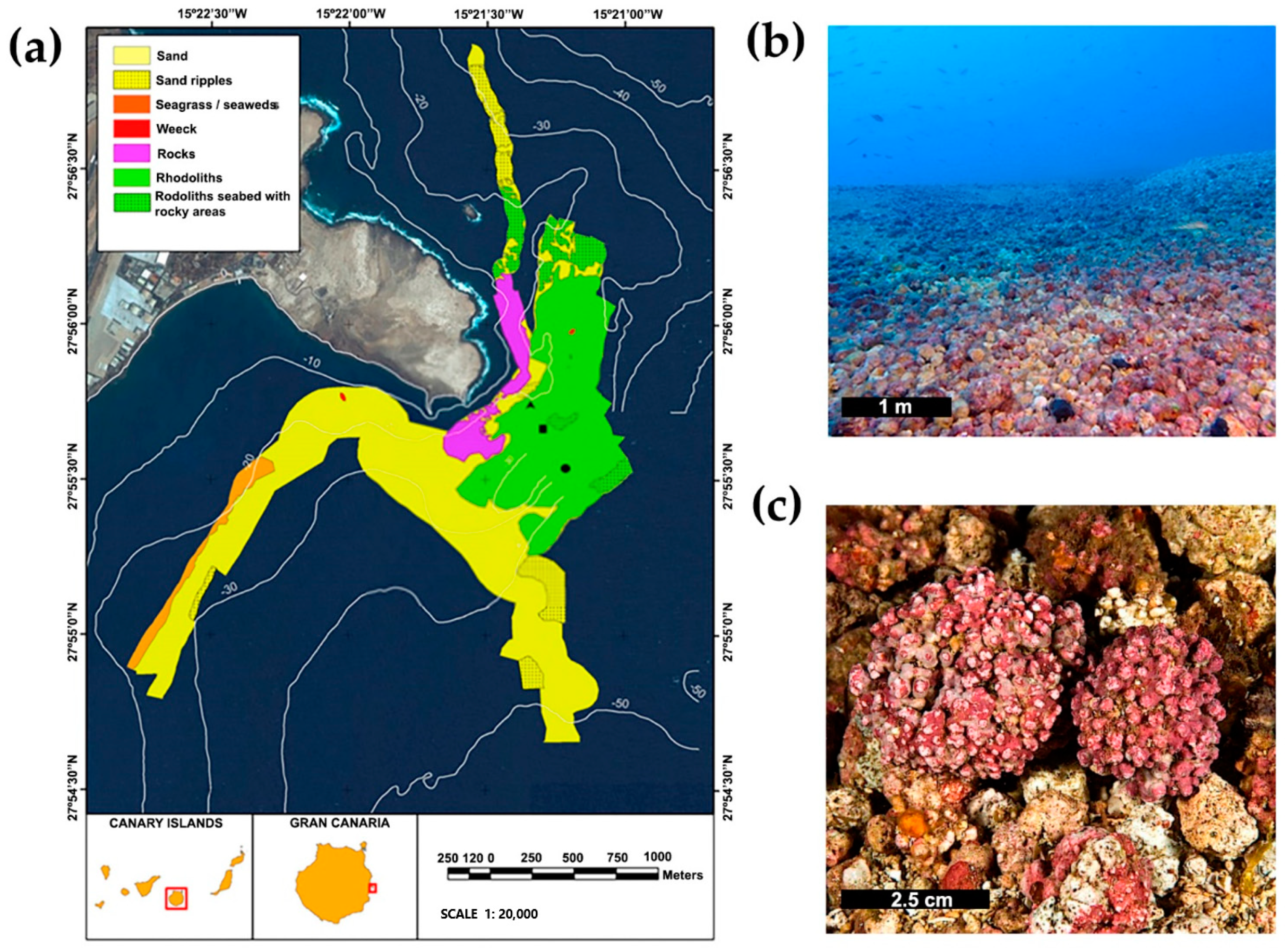
2. Materials and Methods
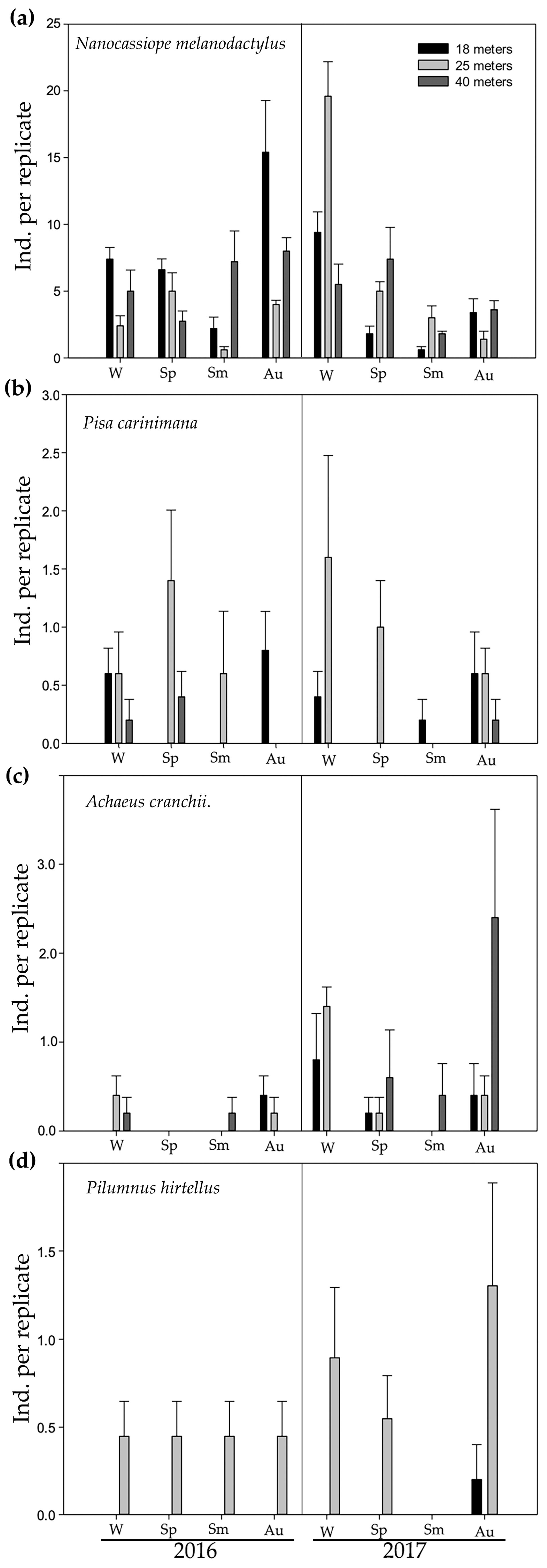
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Riosmena-Rodríguez, R.; Nelson, W.; Aguirre, J. Rhodolith/maërl Beds: A Global Perspective; Springer International Publishing: Basel, Switzerland, 2017. [Google Scholar]
- Ginsburg, R.N.; Bosellini, A. Form and internal structure of recent algal nodules (Rhodolites) from Bermuda. J. Geol. 1971, 79, 669–682. [Google Scholar] [CrossRef]
- Steller, D.L.; Riosmena-Rodríguez, R.; Foster, M.S.; Roberts, C.A. Rhodolith bed diversity in the Gulf of California: The importance of rhodolith structure and consequences of disturbance. Aquat. Conserv. Mar. Freshw. Ecosyst. 2003, 13, S5–S20. [Google Scholar] [CrossRef]
- Sciberras, M.; Rizzo, M.; Mifsud, J.R.; Camilleri, K.; Borg, J.A.; Lanfranco, E.; Schembri, P.J. Habitat structure and biological characteristics of a maerl bed off the northeastern coast of the Maltese Islands (central Mediterranean). Mar. Biodivers. 2009, 39, 251–264. [Google Scholar] [CrossRef]
- Steller, D.L.; Cáceres-Martínez, C. Coralline algal rhodoliths enhance larval settlement and early growth of the pacific calico scallop Argopecten ventricosus. Mar. Ecol. Prog. Ser. 2009, 396, 49–60. [Google Scholar] [CrossRef]
- Basso, D.; Babbini, L.; Kaleb, S.; Bracchi, V.A.; Falace, A. Monitoring deep Mediterranean rhodolith beds. Aquat. Conserv. Mar. Freshw. Ecosyst. 2016, 26, 549–561. [Google Scholar] [CrossRef]
- Otero-Ferrer, F.; Mannarà, E.; Cosme, M.; Falace, A.; Montiel-Nelson, J.A.; Espino, F.; Tuya, F. Early-faunal colonization patterns of discrete habitat units: A case study with rhodolith-associated vagile macrofauna. Estuar. Coast. Shelf Sci. 2019, 218, 9–22. [Google Scholar] [CrossRef]
- Foster, M.S. Rhodoliths: Between rocks and soft places. J. Phycol. 2001, 37, 659–667. [Google Scholar] [CrossRef]
- Nelson, W.A.; New Zealand Ministry for Primary Industries. Rhodolith Beds in Northern New Zealand: Characterisation of Associated Biodiversity and Vulnerability to Environmental Stressors. 2012. Available online: http://fs.fish.govt.nz/Doc/23064/AEBR_99.pdf.ashx (accessed on 2 February 2020).
- Hernandez-Kantun, J.J.; Hall-Spencer, J.M.; Grall, J.; Adey, W.; Rindi, F.; Maggs, C.A.; Peña, V. North Atlantic Rhodolith Beds. In Coastal Research Library; Springer: Cham, Switzerland, 2017; Volume 15. [Google Scholar]
- Rebelo, C.; Johnson, M.E.; Quartau, R.; Rasser, M.W.; Melo, C.S.; Neto, A.I.; Ávila, S.P. Modern rhodoliths from the insular shelf of Pico in the Azores (Northeast Atlantic Ocean). Estuar. Coast. Shelf Sci. 2018, 210, 7–17. [Google Scholar] [CrossRef]
- Riera, R.; Delgado, J.D.; Rodríguez, M.; Monterroso, Ó.; Ramos, E. Macrofaunal communities of threatened subtidal maërl seabeds on Tenerife (Canary Islands, north-east Atlantic Ocean) in summer. Acta Oceanol. Sin. 2012, 31, 98–105. [Google Scholar] [CrossRef]
- Hinojosa-Arango, G.; Riosmena-Rodriguez, R. Influence of Rhodolith-Forming Species and Growth-Form on Associated Fauna of Rhodolith Beds in the Central-West Gulf of California, Mexico. Mar. Ecol. 2014, 25, 109–127. [Google Scholar] [CrossRef]
- Kamenos, N.A.; Moore, P.G.; Hall-Spencer, J.M. Maerl grounds provide both refuge and high growth potential for juvenile queen scallops (Aequipecten opercularis L.). J. Exp. Mar. Biol. Ecol. 2004, 313, 241–254. [Google Scholar] [CrossRef]
- Amado-Filho, G.M.; Maneveldt, G.; Manso, R.C.C.; Marins-Rosa, B.V.; Pacheco, M.R.; Guimaraes, S.M. Estructura de los mantos de rodolitos de 4 a 55 metros de profundidad en la costa sur del estado de Espírito Santo, Brazil. Cienc. Mar. 2007, 33, 399–410. [Google Scholar] [CrossRef]
- De Grave, S. The Influence of Sedimentary Heterogeneity on Within Maerl Bed Differences in Infaunal Crustacean Community. Estuar. Coast. Shelf Sci. 1999, 49, 153–163. [Google Scholar] [CrossRef]
- Veras, P.C.; Pierozzi, I.; Lino, J.B.; Amado-Filho, G.M.; de Senna, A.R.; Santos, C.S.G.; Pereira-Filho, G.H. Drivers of biodiversity associated with rhodolith beds from euphotic and mesophotic zones: Insights for management and conservation. Perspect. Ecol. Conserv. 2020, in press. [Google Scholar] [CrossRef]
- Bouzon, J.L.; Freire, A.S. The Brachyura and Anomura fauna (Decapoda; Crustacea) in the Arvoredo Marine Biological Reserve on the southern brazilian coast. Braz. J. Biol. 2007, 67, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Borg, J.A.; Howege, H.M.; Lanfranco, E.; Micallef, S.; Mifsud, C.; Schembri, P.J. The macrobenthic species of the infralittoral to circalittoral transition zone off the northeastern coast of Malta (Central Mediterranean). Xjenza 1998, 3, 16–24. [Google Scholar]
- Koettker, A.G.; Lopes, R.M. Meroplankton spatial structure and variability on Abrolhos Bank and adjacent areas, with emphasis on brachyuran larvae. Cont. Shelf Res. 2013, 70, 97–108. [Google Scholar] [CrossRef]
- Herrera-Martínez, P.; Álvarez-Hernández, S.; del Carmen Méndez-Trejo, M.; Riosmena-Rodríguez, R. Invertebrates: Structure of the Community and Biodiversity Associated to Rhodolith-Sponge Complex at Magdalena Bay. South Baja California. In Invertebrates: Classification, Evolution and Biodiversity; Nova Science Publishers: New York, NY, USA, 2013; pp. 107–130. [Google Scholar]
- Abele, L.G. Species diversity of decapod crustaceans in marine habitats. Ecology 1974, 55, 156–161. [Google Scholar] [CrossRef]
- Hines, A.H. Larval patterns in the life histories of brachyuran crabs (Crustacea, Decapoda, Brachyura). Bull. Mar. Sci. 1986, 39, 444–466. [Google Scholar]
- González, J.A. Brachyuran crabs (Crustacea: Decapoda) from the Canary Islands (eastern Atlantic): Checklist, zoogeographic considerations and conservation. Sci. Mar. 2016, 80, 89–102. [Google Scholar] [CrossRef]
- González, J.A.; Triay-Portella, R.; Martins, A.; Lopes, E. Checklist of brachyuran crabs (Crustacea: Decapoda) from the Cape Verde Islands, with a biogeographic comparison with the Canary Islands (Eastern Atlantic). Cah. Biol. Mar. 2017, 58, 137–151. [Google Scholar] [CrossRef]
- Otero-Ferrer, F.; Cosme, M.; Tuya, F.; Espino, F.; Haroun, R. Effect of depth and seasonality on the functioning of rhodolith seabeds. Estuar. Coast. Shelf Sci. 2020, 106579. [Google Scholar] [CrossRef]
- Haroun, R.J.; Gil-Rodríguez, M.C.; de Castro, J.D.; Prud’Homme Van Reine, W.F. A checklist of the marine plants from the Canary Islands (central eastern Atlantic Ocean). Bot. Mar. 2002, 45, 139–169. [Google Scholar] [CrossRef]
- Pardo, C.; Lopez, L.; Peña, V.; Hernández-Kantún, J.; Le Gall, L.; Bárbara, I.; Barreiro, R. A multilocus species delimitation reveals a striking number of species of coralline algae forming maerl in the OSPAR maritime area. PLoS ONE 2014, 9, e104073. [Google Scholar] [CrossRef] [PubMed]
- Monod, T. Hippidea et Brachyura ouest-africains. Ifan 1956, 45, 1–674. [Google Scholar]
- Zariquiey, A.R. Crustáceos decápodos ibéricos. Investig. Pesq. 1968, 32, 1–510. [Google Scholar]
- Manning, R.B.; Holthuis, L.B. West African Brachyura Crabs (Crustacea: Decapoda); Smithsonian Institution Press: Washington, DC, USA, 1981. [Google Scholar]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest package: Tests in linear mixed effects models. J. Stat. Softw. 2017, 82. [Google Scholar] [CrossRef]
- Harrison, X.A.; Donaldson, L.; Correa-Cano, M.E.; Evans, J.; Fisher, D.N.; Goodwin, C.E.D.; Robinson, B.S.; Hodgson, D.J.; Inger, R. A brief introduction to mixed effects modelling and multi-model inference in ecology. PeerJ 2018, 6, e4794. [Google Scholar] [CrossRef]
- Abele, L.G. Comparative habitat diversity and faunal relationships between the Pacific and Caribbean Panamanian decapod Crustacea: A preliminary report with remarks on the crustacean fauna of Panama. Bull. Biol. Soc. Wash. 1972, 2, 125–138. [Google Scholar]
- Spivak, E.D.; Arévalo, E.; Cuesta, J.A.; González-Gordillo, J.I. Population structure and reproductive biology of the stone crab Xantho poressa (Crustacea: Decapoda: Xanthidae) in the ‘Corrales de Rota’ (south-western Spain), a human-modified intertidal fishing area. J. Mar. Biol. Assoc. U. K. 2010, 90, 323–334. [Google Scholar] [CrossRef][Green Version]
- Connell, S.D. Assembly and maintenance of subtidal habitat heterogeneity: Synergistic effects of light penetration and sedimentation. Mar. Ecol. Prog. Ser. 2005, 289, 53–61. [Google Scholar] [CrossRef]
- Pascelli, C.; Riul, P.; Riosmena-Rodríguez, R.; Scherner, F.; Nunes, M.; Hall-Spencer, J.M.; de Oliveira, E.C.; Horta, P. Seasonal and depth-driven changes in rhodolith bed structure and associated macroalgae off Arvoredo island (southeastern Brazil). Aquat. Bot. 2013, 111, 62–65. [Google Scholar] [CrossRef]
- McConnico, L.A.; Carmona, G.H.; Morales, J.S.M.; Rodríguez, R.R. Temporal variation in seaweed and invertebrate assemblages in shallow rhodolith beds of Baja California Sur, México. Aquat. Bot. 2017, 139, 37–47. [Google Scholar] [CrossRef]
- Thomsen, M.S.; Wernberg, T.; Altieri, A.; Tuya, F.; Gulbransen, D.; McGlathery, K.J.; Holmer, M.; Silliman, B.R. Habitat cascades: The conceptual context and global relevance of facilitation cascades via habitat formation and modification. Integr. Comp. Biol. 2010, 50, 158–175. [Google Scholar] [CrossRef]
- Navarro-Mayoral, S.; Fernandez-Gonzalez, V.; Otero-Ferrer, F.; Tuya, F. Spatio-temporal variability of amphipod assemblages associated with rhodolith seabeds. Mar. Freshw. Res. 2020, 71, 1–8. [Google Scholar] [CrossRef]
- Arnott, G.; Elwood, R.W. Assessment of fighting ability in animal contests. Anim. Behav. 2009, 77, 991–1004. [Google Scholar] [CrossRef]
- Steller, D.L.; Foster, M.S. Environmental factors influencing distribution and morphology of rhodoliths in Bahía Concepción, BCS, México. J. Exp. Mar. Biol. Ecol. 1995, 194, 201–212. [Google Scholar] [CrossRef]
- Riul, P.; Lacouth, P.; Pagliosa, P.R.; Christoffersen, M.L.; Horta, P.A. Rhodolith beds at the easternmost extreme of South America: Community structure of an endangered environment. Aquat. Bot. 2009, 90, 315–320. [Google Scholar] [CrossRef]
- Amado-Filho, G.M.; Maneveldt, G.; Pereira-Filho, G.; Manso, R.; Bahia, R.; Barros-Barreto, M.; Guimaraes, S.M. Seaweed diversity associated with a Brazilian tropical rhodolith bed. Cienc. Mar. 2010, 36, 371–391. [Google Scholar] [CrossRef]
- Weber-Van Bosse, A.; Foslie, M. The corallinaceae of the Siboga Expedition. Leid. Bull. Rep. 1904, 61, 1–110. [Google Scholar]
- Turner, H.V.; Wolcott, D.L.; Wolcott, T.G.; Hines, A.H. Post-mating behavior, intramolt growth, and onset of migration to Chesapeake Bay spawning grounds by adult female blue crabs, Callinectes sapidus Rathbun. J. Exp. Mar. Biol. Ecol. 2003, 295, 107–130. [Google Scholar] [CrossRef]
- Aguilar, R.; Hines, A.H.; Wolcott, T.G.; Wolcott, D.L.; Kramer, M.A.; Lipcius, R.N. The timing and route of movement and migration of post-copulatory female blue crabs, Callinectes sapidus Rathbun, from the upper Chesapeake Bay. J. Exp. Mar. Biol. Ecol. 2005, 319, 117–128. [Google Scholar] [CrossRef]
- Fransen, C.H.J.M. Preliminary Report on Crustacea Collected in the Eastern Part of the North Atlantic during the CANCAP and Mauritania Expeditions of the Former Rijksmuseum van Natuurlijke Historie, Leiden; Nationaal Naturhistorisch Museum: Leiden, The Netherlands, 1991. [Google Scholar]
- Sinclair, M.E. Agonistic behavior of the stone crab, Menippe mercenaria (Say). Anim. Behav. 1977, 25, 193–207. [Google Scholar] [CrossRef]
- Silliman, B.R.; Layman, C.A.; Geyer, K.; Zieman, J.C. Predation by the black-clawed mud crab, Panopeus herbstii, in mid-Atlantic salt marshes: Further evidence for top-down control of marsh grass production. Estuaries 2004, 27, 188–196. [Google Scholar] [CrossRef]
- Brown, K.M.; Keenan, S.F.; Banks, P.D. Dominance hierarchies in xanthid crabs: Roles in resource-holding potential and field distributions. Mar. Ecol. Prog. Ser. 2005, 291, 189–196. [Google Scholar] [CrossRef]
- Thatje, S.; Hall, S. The effect of temperature on the evolution of per offspring investment in a globally distributed family of marine invertebrates (Crustacea: Decapoda: Lithodidae). Mar. Biol. 2016, 163, 48. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mateo-Ramírez, Á.; García Raso, J.E. Temporal changes in the structure of the crustacean decapod assemblages associated with Cymodocea nodosa meadows from the Alboran Sea (Western Mediterranean Sea). Mar. Ecol. 2012, 33, 302–316. [Google Scholar] [CrossRef]
- Mateo-Ramírez, Á.; Urra, J.; Marina, P.; Rueda, J.L.; García Raso, J.E. Crustacean decapod assemblages associated with fragmented Posidonia oceanica meadows in the Alboran Sea (Western Mediterranean Sea): Composition, temporal dynamics and influence of meadow structure. Mar. Ecol. 2016, 37, 344–358. [Google Scholar] [CrossRef]



| Brachyuran Species | Dominance (%) | Abundance |
|---|---|---|
| Nanocassiope melanodactylus (A. Milne-Edwards, 1867) | 83.03 | 635 |
| Pisa carinimana Miers, 1879 | 5.74 | 44 |
| Achaeus cranchii Leach, 1817 | 5.09 | 39 |
| Pilumnus hirtellus (Linnaeus, 1761) | 1.83 | 14 |
| Acanthonyx lunulatus (Risso, 1816) | 1.70 | 13 |
| Micropisa ovata Stimpson, 1858 | 0.91 | 7 |
| Paractaea sp. | 0.78 | 6 |
| Eurynome sp. | 0.39 | 3 |
| Ebalia sp. | 0.26 | 2 |
| Thalamita poissonii (Audouin, 1826) | 0.26 | 2 |
| Estimate | Std. Error | z Value | Pr(> |z|) | |
|---|---|---|---|---|
| (Intercept) | 0.0001 | 0.2928 | 0.000 | 1.0000 |
| Depth [25 m] | 0.8755 | 0.3470 | 2.523 | 0.0116 * |
| Depth [40 m] | 0.4700 | 0.3888 | 1.209 | 0.2267 |
| Season [Spring] | 0.6419 | 0.3620 | 1.773 | 0.0762 |
| Season [Summer] | 0.0953 | 0.4116 | 0.232 | 0.8169 |
| Season [Autumn] | 0.7419 | 0.3688 | 2.012 | 0.0443 * |
| Depth [25 m]: Season [Spring] | −0.5241 | 0.4480 | −1.170 | 0.2421 |
| Depth [40 m]: Season [Spring] | −0.9295 | 0.5256 | −1.769 | 0.0770 |
| Depth [25 m]: Season [Summer] | −0.9708 | 0.5388 | −1.802 | 0.0716 |
| Depth [40 m]: Season [Summer] | −0.1598 | 0.5508 | −0.290 | 0.7717 |
| Depth [25 m]: Season [Autumn] | −0.5878 | 0.4526 | −1.299 | 0.1941 |
| Depth [40 m]: Season [Autumn] | −0.5700 | 0.5090 | −1.120 | 0.2628 |
| Estimate | Std. Error | z Value | Pr(>|z|) | |
|---|---|---|---|---|
| (Intercept) | 1.4351 | 0.2571 | 5.583 | 0.0001 *** |
| Depth [25 m] | −0.1268 | 0.3679 | −0.344 | 0.7305 |
| Depth [40 m] | 0.1335 | 0.3594 | 0.372 | 0.7102 |
| Season [Spring] | 0.6931 | 0.3468 | 1.999 | 0.0456 * |
| Season [Summer] | −1.0986 | 0.4240 | −2.591 | 0.0096 ** |
| Season [Autumn] | 0.8056 | 0.3449 | 2.336 | 0.0195 * |
| Depth [25 m]: Season [Spring] | 0.3964 | 0.4908 | 0.808 | 0.4193 |
| Depth [40 m]: Season [Spring] | −0.7142 | 0.4969 | −1.437 | 0.1506 |
| Depth [25 m]: Season [Summer] | 0.3781 | 0.5890 | 0.642 | 0.5209 |
| Depth [40 m]: Season [Summer] | 1.0341 | 0.5544 | 1.865 | 0.0622 |
| Depth [25 m]: Season [Autumn] | −1.1207 | 0.5173 | −2.166 | 0.0303 * |
| Depth [40 m]: Season [Autumn] | −0.6164 | 0.4915 | −1.254 | 0.2098 |
| Estimate | Std. Error | z Value | Pr(> |z|) | |
|---|---|---|---|---|
| (Intercept) | 4.1866 | 0.1836 | 22.8010 | 0.0001 *** |
| Depth (25 m) | 0.2578 | 0.1890 | 1.3640 | 0.1753 |
| Depth (40 m) | −0.4960 | 0.1892 | −2.6220 | 0.0010 ** |
| Season (Spring) | 0.1497 | 0.2121 | 0.7060 | 0.4816 |
| Season (Summer) | 0.3480 | 0.2240 | 1.5540 | 0.1231 |
| Season (Autumn) | −0.7900 | 0.2102 | −3.7570 | 0.0003 *** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Latorre, C.; Triay-Portella, R.; Cosme, M.; Tuya, F.; Otero-Ferrer, F. Brachyuran Crabs (Decapoda) Associated with Rhodolith Beds: Spatio-Temporal Variability at Gran Canaria Island. Diversity 2020, 12, 223. https://doi.org/10.3390/d12060223
Sánchez-Latorre C, Triay-Portella R, Cosme M, Tuya F, Otero-Ferrer F. Brachyuran Crabs (Decapoda) Associated with Rhodolith Beds: Spatio-Temporal Variability at Gran Canaria Island. Diversity. 2020; 12(6):223. https://doi.org/10.3390/d12060223
Chicago/Turabian StyleSánchez-Latorre, Clara, Raül Triay-Portella, Marcial Cosme, Fernando Tuya, and Francisco Otero-Ferrer. 2020. "Brachyuran Crabs (Decapoda) Associated with Rhodolith Beds: Spatio-Temporal Variability at Gran Canaria Island" Diversity 12, no. 6: 223. https://doi.org/10.3390/d12060223
APA StyleSánchez-Latorre, C., Triay-Portella, R., Cosme, M., Tuya, F., & Otero-Ferrer, F. (2020). Brachyuran Crabs (Decapoda) Associated with Rhodolith Beds: Spatio-Temporal Variability at Gran Canaria Island. Diversity, 12(6), 223. https://doi.org/10.3390/d12060223






