Urban Sprawl, Food Subsidies and Power Lines: An Ecological Trap for Large Frugivorous Bats in Sri Lanka?
Abstract
1. Introduction
2. Materials and Methods
3. Results
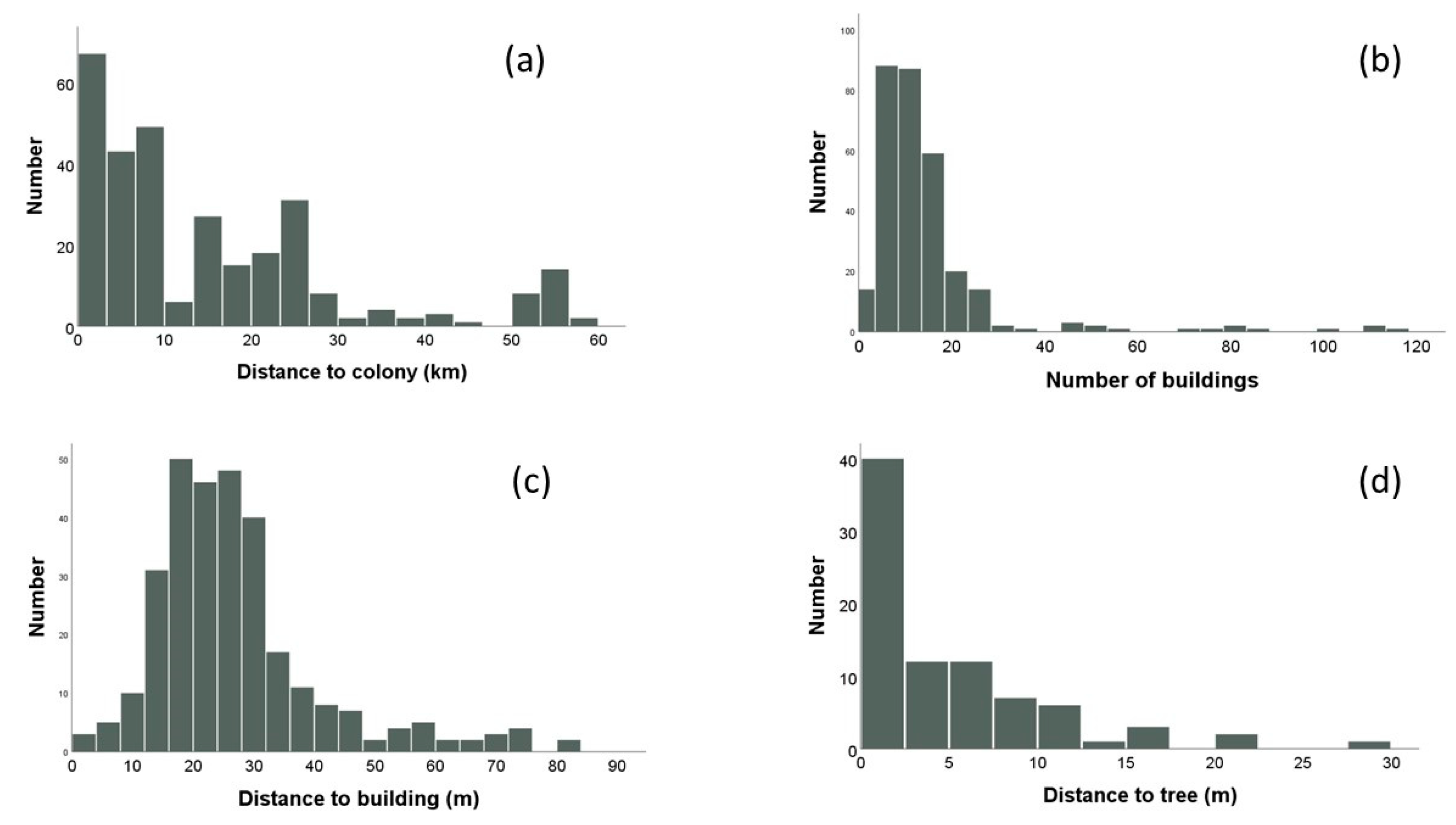
3.1. Location of Electrocuted Flying Foxes
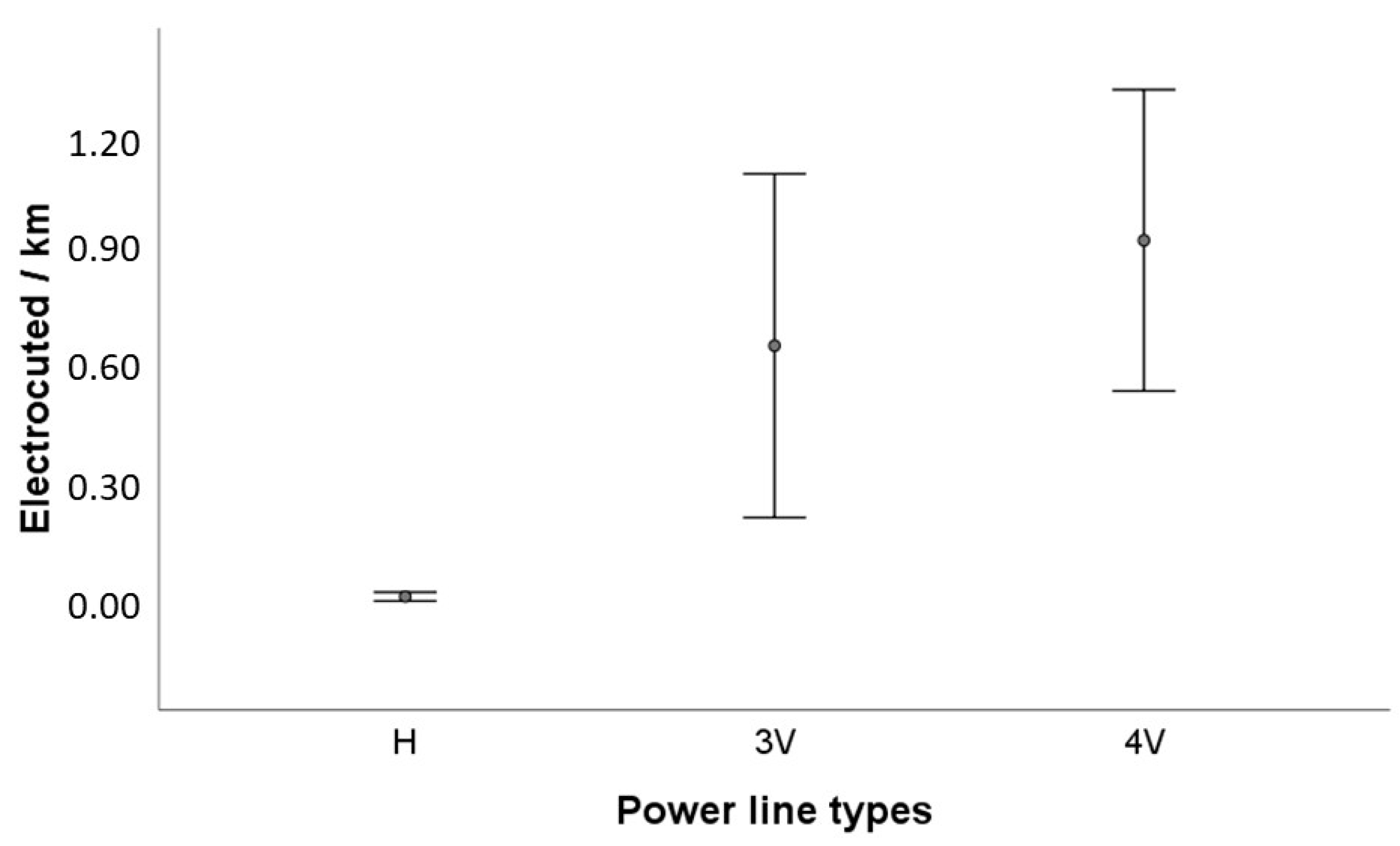
3.2. Risk of Electrocution
4. Discussion
4.1. Urbanization as an Ecological Trap for Flying Foxes
4.2. Conservation Implications: Population Decline and the Loss of Ecological Functions
4.3. Mitigation Options and Further Research
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Frick, W.F.; Kingston, T.; Flanders, J. A Review of the major threats and challenges to global bat conservation. Ann. N. Y. Acad. Sci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Mickleburgh, S.P.; Hutson, A.M.; Racey, P.A. A review of the global conservation status of bats. Oryx 2002, 36, 18–34. [Google Scholar] [CrossRef]
- Voigt, C.C.; Kingston, T. Bats in the Anthropocene: Conservation of Bats in a Changing World; Springer Science+ Business Media: New York, NY, USA, 2016. [Google Scholar]
- Rydell, J.; Eklöf, J.; Sánchez-Navarro, S. Age of enlightenment: Long-term effects of outdoor aesthetic lights on bats in churches. Roy. Soc. Open Sci. 2017, 4, 161077. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Brito, D.; Carrete, M.; Ibáñez, C.; Juste, J.; Tella, J.L. Nest-site competition and killing by invasive parakeets cause the decline of a threatened bat population. Roy. Soc. Open Sci. 2018, 5, 172477. [Google Scholar] [CrossRef]
- Cryan, P.M.; Gorresen, P.M.; Hein, C.D.; Schirmacher, M.R.; Diehl, R.H.; Huso, M.M.; Hayman, D.T.; Fricker, P.D.; Bonaccorso, F.J.; Johnson, D.H.; et al. Behavior of bats at wind turbines. Proc. Nat. Acad. Sci. USA 2014, 111, 15126–15131. [Google Scholar] [CrossRef]
- Foo, C.F.; Bennett, V.J.; Hale, A.M.; Korstian, J.M.; Schildt, A.J.; Williams, D.A. Increasing evidence that bats actively forage at wind turbines. PeerJ 2017, 5, e3985. [Google Scholar] [CrossRef]
- Hayes, M.A.; Hooton, L.A.; Gilland, K.L.; Grandgent, C.; Smith, R.L.; Lindsay, S.R.; Collins, J.D.; Schumacher, S.M.; Rabie, P.A.; Gruver, J.C.; et al. A smart curtailment approach for reducing bat fatalities and curtailment time at wind energy facilities. Ecol. Appl. 2019, 29, e01881. [Google Scholar] [CrossRef]
- O’shea, T.J.; Cryan, P.M.; Hayman, D.T.; Plowright, R.K.; Streicker, D.G. Multiple mortality events in bats: A global review. Mam. Rev. 2016, 46, 175–190. [Google Scholar] [CrossRef]
- Manville, A.M. Impacts to birds and bats due to collisions and electrocutions from some tall structures in the United States: Wires, towers, turbines, and solar arrays—state of the art in addressing the problems. In Problematic Wildlife; Springer: Cham, Switzerland, 2016; pp. 415–442. [Google Scholar]
- Sinha, Y.P. Contribution to the Knowledge of Bats (Mammalia: Chiroptera) of North East Hills, India; Occ. Paper No. 174; Zoological Survey of India: Patna, India, 1999; pp. 1–52. [Google Scholar]
- Aziz, M.A.; Ali, A.H.M.; Hasan, M.K.; Tonchangya, P.K.; Sarker, A.; Atiquzzaman, K.M.; Dutta, S.; Makayching; Rahman, K.M.Z. Some notes on three species of bats of Jahangirnagar University, Bangladesh. Zoo. J. 2007, 22, 2729–2731. [Google Scholar]
- Molur, S.; Molur, P.; Ravichandran, B. Electrocuted Flying foxes in Madikeri, Coorge. Bat Net News. 2007, 8, 44. [Google Scholar]
- Ali, A. Population trend and conservation status of Indian flying fox Pteropus giganteus Brunnich, 1782 (Chiroptera: Pteropodidae) in Western Assam. Ecoscan 2010, 4, 311–312. [Google Scholar]
- Rajeshkumar, S.; Raghunathan, C.; Krishnamoorthy, V. Observation on Electrocution of Flying Fox (Pteropus giganteus) In Andaman Islands and their conservation strategies. J. Andaman Sci. Assoc. 2013, 3718, 213–215. [Google Scholar]
- Bhandarkar, S.V.; Paliwal, G.T. A record of giant roost of Pteropus giganteus (brunnich, 1782) in Baghnadi district Rajnandgaon Chhattisgarh. Adv. Inn. Res. 2017, 4, 33–36. [Google Scholar]
- Oo, K.S.; Win, H.L.; Bates, P.J.; Pearch, M. The ecology and distribution of Flying Foxes (Chiroptera: Pteropodidae: Pteropus) in Tanintharyi Region, Myanmar with a first mainland record of Pteropus hypomelanus geminorum from Myeik. J. Threat. Taxa 2017, 9, 10528–10537. [Google Scholar] [CrossRef]
- Chouhan, R.; Shrivastava, S. Observation on electrocution of Indian flying fox (Pteropus giganteus) in Ramganjmandi, Kota,(Rajasthan) and their conservation strategies. Int. J. Res. Engin. Sci. Manag. 2019, 2, 648–649. [Google Scholar]
- Kundu, S.; Kumar, V.; Tyagi, K.; Rath, S.; Pakrashi, A.; Saren, P.C.; Laishram, K.; Chandra, K. Mitochondrial DNA identified bat species in northeast India: Electrocution mortality and biodiversity loss. Mitoc. DNA Part. B 2019, 4, 2454–2458. [Google Scholar] [CrossRef]
- Tidemann, C.R. The biology and management of the grey-headed flying-fox, Pteropus poliocephalus. Acta Chiropt. 1999, 1, 151–164. [Google Scholar]
- Tidemann, C.R.; Nelson, J.E. Life expectancy, causes of death and movements of the grey-headed flying-fox (Pteropus poliocephalus) inferred from banding. Acta Chirop. 2011, 13, 419–429. [Google Scholar] [CrossRef]
- Krystufek, B. On the Indian flying foxes (Pteropus giganteus) colony in Paradeniya Botanical gardens, Sri Lanka. Hystrix 2009, 20, 29–35. [Google Scholar]
- Wijeyeratne, G.D.S. Mammals of Sri Lanka; Bloomsbury Publishing PLC, New Holland: London, UK, 2016; pp. 19–20. [Google Scholar]
- Molur, S.; Srinivasulu, C.; Bates, P.; Francis, C. Pteropus giganteus. The IUCN Red List of Threatened Species 2008:e.T18725A8511108. Available online: http://dx.doi.org/10.2305/IUCN.UK.2008.RLTS.T18725A8511108.en (accessed on 21 December 2019).
- Adriaens, T.; Sutton-Croft, M.; Owen, K.; Brosens, D.; van Valkenburg, J.; Kilbey, D.; Groom, Q.; Ehmig, C.; Thürkow, F.; Van Hende, P.; et al. Trying to engage the crowd in recording invasive alien species in Europe: Experiences from two smartphone applications in northwest Europe. Manag. Biol. Invas. 2015, 6, 215–225. [Google Scholar] [CrossRef]
- Vercayie, D.; Herremans, M. Citizen science and smartphones take roadkill monitoring to the next level. Nat. Conserv. 2015, 11, 29–40. [Google Scholar] [CrossRef]
- Mishra, R.; Dookia, S.; Bhattacharya, P. Avenue Plantations as Biodiversity Havens: A Case Study of Population Status of the Indian Flying Fox, Pteropus giganteus Brunnich, 1782 and Implications for Its Conservation in the Urban Megacity, Delhi, India. Proc. Zool. Soc. 2019, 1–10. [Google Scholar] [CrossRef]
- Chaturvedi, S.K.; Singh, S. Status and distribution of Indian flying fox (Pteropus giganteus) in district Chitrakoot, Uttar Pradesh, India. Int. J. Zool. Stu. 2018, 3, 126–128. [Google Scholar]
- Russo, D.; Ancillotto, L. Sensitivity of bats to urbanization: A review. Mam. Biol. 2015, 80, 205–212. [Google Scholar] [CrossRef]
- Rebolo-Ifrán, N.; Tella, J.L.; Carrete, M. Urban conservation hotspots: Predation release allows the grassland-specialist burrowing owl to perform better in the city. Sci. Rep. 2017, 7, 3527. [Google Scholar] [CrossRef]
- Luna, A.; Romero-Vidal, P.; Hiraldo, F.; Tella, J.L. Cities may save some threatened species but not their ecological functions. PeerJ 2018, 6, e4908. [Google Scholar] [CrossRef]
- Oro, D.; Genovart, M.; Tavecchia, G.; Fowler, M.S.; Martínez-Abraín, A. Ecological and evolutionary implications of food subsidies from humans. Ecol. Lett. 2013, 16, 1501–1514. [Google Scholar] [CrossRef]
- Electricity sector in Sri Lanka. Available online: www.en.wikipedia.org/wiki/Electricity_sector_in_Sri_Lanka (accessed on 26 December 2019).
- Sri Lanka. Available online: www.en.wikipedia.org/wiki/Sri_Lanka (accessed on 26 December 2019).
- McIlwee, A.P.; Martin, L. On the intrinsic capacity for increase of Australian flying-foxes (Pteropus spp., Megachiroptera). Aust. Zool. 2002, 32, 76–100. [Google Scholar] [CrossRef]
- Meyer, C.F.; Aguiar, L.M.; Aguirre, L.F.; Baumgarten, J.; Clarke, F.M.; Cosson, J.F.; Villegas, S.E.; Fahr, J.; Faria, D.; Furey, N.; et al. Long-term monitoring of tropical bats for anthropogenic impact assessment: Gauging the statistical power to detect population change. Biol. Conserv. 2010, 143, 2797–2807. [Google Scholar] [CrossRef]
- Hodgkison, R.; Balding, S.T.; Zubald, A.; Kunz, T.H. Fruit bats (Chiroptera: Pteropodidae) as seed dispersers and pollinators in a lowland Malaysian rain forest. Biotropica 2003, 35, 491–502. [Google Scholar] [CrossRef]
- Corlett, R.T. Frugivory and seed dispersal by vertebrates in tropical and subtropical Asia: An update. Glob. Ecol. Conserv. 2017, 11, 1–22. [Google Scholar] [CrossRef]
- Lewanzik, D.; Voigt, C.C. Artificial light puts ecosystem services of frugivorous bats at risk. J. Appl. Ecol. 2014, 51, 388–394. [Google Scholar] [CrossRef]
- Florens, F.B.; Baider, C.; Marday, V.; Martin, G.M.; Zmanay, Z.; Oleksy, R.; Krivek, G.; Vincenot, C.E.; Strasberg, D.; Kingston, T. Disproportionately large ecological role of a recently mass-culled flying fox in native forests of an oceanic island. J. Nat. Conserv. 2017, 40, 85–93. [Google Scholar] [CrossRef]
- Valiente-Banuet, A.; Aizen, M.A.; Alcántara, J.M.; Arroyo, J.; Cocucci, A.; Galetti, M.; García, M.B.; García, D.; Gómez, J.M.; Jordano, P.; et al. Beyond species loss: The extinction of ecological interactions in a changing world. Funct. Ecol. 2015, 29, 299–307. [Google Scholar] [CrossRef]
- Akçakaya, H.R.; Rodrigues, A.S.; Keith, D.A.; Milner-Gulland, E.J.; Sanderson, E.W.; Hedges, S.; Mallon, D.P.; Grace, M.K.; Long, B.; Meijaard, E.; et al. Assessing ecological function in the context of species recovery. Conserv. Biol. 2019. [Google Scholar] [CrossRef] [PubMed]
- MacGregor, K.A.; Lemaître, J. The management utility of large-scale environmental drivers of bat mortality at wind energy facilities: The effects of facility size, elevation and geographic location. Glob. Ecol. Conserv. 2019, 6, e00871. [Google Scholar] [CrossRef]
- Allison, T.D. Bats and Wind Energy: Impacts, Mitigation, and Tradeoffs; American Wind Wildlife Institute: Washington, DC, USA, 2018. [Google Scholar]
- Smallwood, K.S.; Bell, D.A. Relating Bat Passage Rates to Wind Turbine Fatalities. Diversity 2020, 12, 84. [Google Scholar] [CrossRef]
- Hoffmaster, E.; Vonk, J.; Mies, R. Education to action: Improving public perception of bats. Animals 2016, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Borda-de-Água, L.; Ascensão, F.; Sapage, M.; Barrientos, R.; Pereira, H.M. On the identification of mortality hotspots in linear infrastructures. Basic Appl. Ecol. 2019, 34, 25–35. [Google Scholar] [CrossRef]
- Vincenot, C.E.; Koyama, L.; Russo, D. Near threatened? First report of unsuspected human-driven decline factors in the Ryukyu flying fox (Pteropus dasymallus) in Japan. Mam. Biol. 2015, 80, 273–277. [Google Scholar] [CrossRef][Green Version]
- Kibuule, M.; Pomeroy, D. Avian mortality rates on a power line near Kampala, Uganda. Scopus 2015, 34, 52–55. [Google Scholar]
- The IUCN Red List of Threatened Species. Version 2019-3. Available online: https://www.iucnredlist.org (accessed on 28 December 2019).


| Tree Species | N | % |
|---|---|---|
| Mangifera indica | 51 | 41.80 |
| Musa x paradisiaca | 26 | 21.31 |
| Psidium guajava | 10 | 8.20 |
| Artocarpus heterophyllus | 9 | 7.38 |
| Ficus religiosa | 9 | 7.38 |
| Terminalia catappa | 6 | 4.92 |
| Carica papaya | 4 | 3.28 |
| Unidentified species | 2 | 1.64 |
| Limonia acidissima | 1 | 0.82 |
| Annona muricata | 1 | 0.82 |
| Annona reticulata | 1 | 0.82 |
| Areca catechu | 1 | 0.82 |
| Chrysophyllum monopyrenum | 1 | 0.82 |
| Estimate | s.e. | Wald χ2 | d.f. | p | |
|---|---|---|---|---|---|
| Intercept | 0.865 | 0.1253 | 47.682 | 1 | 0.000 |
| Power line: H | −4.332 | 0.5809 | 55.595 | 1 | 0.000 |
| Power line: 3V | −1.477 | 0.3091 | 22.827 | 1 | 0.000 |
| Power line length | 0.080 | 0.0100 | 63.455 | 1 | 0.000 |
| Habitat: agricultural | −0.525 | 0.2496 | 4.416 | 1 | 0.036 |
| Habitat: suburban | −0.930 | 0.1491 | 38.943 | 1 | 0.000 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tella, J.L.; Hernández-Brito, D.; Blanco, G.; Hiraldo, F. Urban Sprawl, Food Subsidies and Power Lines: An Ecological Trap for Large Frugivorous Bats in Sri Lanka? Diversity 2020, 12, 94. https://doi.org/10.3390/d12030094
Tella JL, Hernández-Brito D, Blanco G, Hiraldo F. Urban Sprawl, Food Subsidies and Power Lines: An Ecological Trap for Large Frugivorous Bats in Sri Lanka? Diversity. 2020; 12(3):94. https://doi.org/10.3390/d12030094
Chicago/Turabian StyleTella, José L., Dailos Hernández-Brito, Guillermo Blanco, and Fernando Hiraldo. 2020. "Urban Sprawl, Food Subsidies and Power Lines: An Ecological Trap for Large Frugivorous Bats in Sri Lanka?" Diversity 12, no. 3: 94. https://doi.org/10.3390/d12030094
APA StyleTella, J. L., Hernández-Brito, D., Blanco, G., & Hiraldo, F. (2020). Urban Sprawl, Food Subsidies and Power Lines: An Ecological Trap for Large Frugivorous Bats in Sri Lanka? Diversity, 12(3), 94. https://doi.org/10.3390/d12030094







