Abstract
The concept of generic diversity expresses the ‘diversification’ of species into genera in a community. Since niche overlap is assumed to be higher in congeneric species, competition should increase generic diversity. On the other hand, generic diversity might be lower in highly selective environments, where only species with similar adaptations can survive. We used the distribution of tenebrionid beetles in Central Italy to investigate how generic diversity varies with elevation from sea level to 2400 m altitude. Generic diversity of geophilous tenebrionids decreased sharply with elevation, whereas the generic diversity of xylophilous tenebrionids showed similarly high values across the gradient. These results suggest that geophilous species are more sensitive to variation in environmental factors, and that the advantages of close relationships (similar adaptations to harsh conditions) are greater than the possible drawbacks (competition). This is consistent with the fact that geophilous tenebrionids are mostly generalist detritivores, and hence weakly affected by competition for resources. By contrast, xylophilous species are more protected from harsh/selective conditions, but more limited by competition for microhabitats and food. Our results support the environmental filtering hypothesis for the species composition of tenebrionid beetles along an elevational gradient.
1. Introduction
Elevational variations in biodiversity are among the most investigated patterns in biogeography [1,2]. Many environmental characteristics (including climate, land use, soil composition, and geological settings), as well as species richness, beta-diversity, functional diversity, biogeographical composition, and several aspects of community structure, vary markedly with altitude, which makes elevational gradients ideal to investigate patterns of variation in biodiversity in response to environmental factors [3,4,5,6,7,8,9,10,11,12,13,14,15,16].
The concept of generic diversity has been introduced to express the ‘diversification’ of species into genera [17], as the inverse of the probability that two species selected at random from a community belong to the same genus. Despite its simplicity, this measure has important ecological implications. In general, it is assumed that morphological distance between species is associated with ecological similarity [18,19,20,21]. As congeneric species are expected to be morphologically more similar than non-congeners, they should also have more overlapping niches than species belonging to different genera. This concept has been originally formulated by Elton, who concluded that there is “a strong tendency for the species of any genus to be … unable to coexist permanently on the same area of the same habitat” [22]. This idea represented a major step in the history of ecology, leading to the development of a variety of approaches to investigate the possible role of competition from patterns of species co-occurrence [23,24,25,26,27,28,29,30]. Although competition may occur between non-congeners, and congeners may not significantly compete, in general, taxonomically closely related species are also ecologically similar, and hence subject to higher competition [31,32,33,34,35,36,37]. If competition is stronger between congeneric species, species belonging to different genera will co-occur in the same habitats more often than congeneric species, thus increasing generic diversity (because species are less concentrated into genera). On the other hand, in virtue of their morphological similarity, congeneric species are also expected to share similar adaptations [21]. Thus, especially in highly selective environments, generic diversity might be high because only species with similar adaptations (and hence probably belonging to the same genera) can survive [38]. Therefore, a low generic diversity indicates that the advantages of a close relationship, as expressed by belonging to the same genus, might largely compensate for any associated drawbacks. The balance between the two contrasting selective forces exerted by competition and habitat requirements might lead generic diversity to increase with resource limitations, and to decrease with environmental severity. Although some papers investigated variations in generic diversity among beetle communities [34,39,40], to the best of our knowledge, no research has attempted to study how generic diversity can be influenced by elevation.
In this paper, we used the distribution of tenebrionid beetles in Latium (Central Italy) to investigate how generic diversity varies with elevation along a gradient from sea level to 2400 m altitude and in relation with species lifestyle. Tenebrionid beetles are a family of beetles, which exhibit an extraordinarily wide range of ecological diversity, occurring from deserts to boreal forests. Most species are saprophagous, feeding on humus, leaf litter, decaying wood, windblown detritus, carrion, and dung. However, some tenebrionids are predators or semipredators, and many others feed on fungi and lichens. The vast majority of tenebrionids can be divided into two groups according to the adult main lifestyle: (1) those that occur in the soil (geophilous species), and (2) those associated with wood (xylophilous species) [41].
Elevational gradients reflect increasing stressful conditions [3,4,7,42,43,44], which are particularly severe at high altitudes, because of low temperatures, high radiation, strong winds, low soil nutrients, unstable substrates, and short plant growing seasons [5,45,46]. As a result, we expect that only few tenebrionid species will be able to survive at high altitudes, resulting in very low generic diversity because of the higher probability of belonging to the same genera. By contrast, at low altitudes, thanks to the presence of more favorable conditions, even species with very different morphological characteristics, and hence which are non-congeneric, may coexist, leading to higher values of generic diversity. Thus, we expect an overall pattern of decreasing generic diversity with elevation, and a different response between geophilous and xylophilous species. Competition is expected to be generally not important in geophilous species, since those are mainly omnivores and can easily find resting places under stones, but it might relevant to xylophilous species, which depend on plants. Thus, we expect that generic diversity will be relatively high for xylophilous beetles, even at high elevation, if they compete for the same plant species as resting places or as food sources (dead wood, and/or associated fungi or lichens).
2. Materials and Methods
2.1. Study Area
Latium is an Italian region extending for 17,200 km2 in the central part of the Italian peninsula. Most of the area is occupied by flat and hilly landscapes. However, the region also includes several mountain chains that can exceed 2000 m elevation. Coasts are mainly represented by sandy shores, whereas a large alluvial plain, corresponding to the course of the river Tevere, characterizes the central part of the region. The city of Rome (which extends for 360 km2 and hosts about 3 million inhabitants) is located in this alluvial plain. Most of the region presents a typical Mediterranean climate, with, however, obvious elevational variations. Whereas temperatures are high (9–10 °C in winter and 24–25 °C in summer) and annual rainfall scarce (700 mm) along the coasts, mountain areas are characterized by low temperatures (below 0 °C in winter) and abundant rainfall (1200 mm) [47].
2.2. Data Sources
We used the dataset assembled by Fattorini [12,48], which included 3,561 tenebrionid records, plus 526 new records, spanning from 1860 to 2015. Data were obtained from the examination of museum and private collections, publications, and unpublished lists for a total of 27,476 specimens. Overall, they form a random sample not affected by biases due to collector preferences for certain biotopes, sites, or species. Possible elevational shifts due to climate change occurring from 1860 to 2015 did not extend far enough to influence the overall patterns [12,49].
Sample sites were georeferenced (latitude and longitude decimal degrees) using digital topographic maps. Thanks to the high density of place names in study area, true collecting places were likely very close to the position of locality reported on the maps [50]. Elevation was taken as recorded by the collector (when available) or retrieved from topographic maps.
In total, we considered the vertical distribution of 84 native species and subspecies of Tenebrionidae. Recent morphological [51,52,53,54] and molecular [55,56,57] analyses showed that some tenebrionid populations traditionally classified as subspecies are evolutionarily significant units, usually demanding a species status. Thus, we considered both species and subspecies as terminal taxonomic units of equal rank. The term “species” will be used for simplicity. We omitted from the analysis the genus Lagria and the subfamily Alleculinae (formerly considered a separate family), because the ecology of many of these beetles is completely different from that of all other tenebrionids (many alleculines are flower-visiting insects) and their biology and distribution in the study area are poorly known. We also excluded synanthropic species associated with human food, and which became cosmopolitan or subcosmopolitan, and alien species introduced into Italy.
For each species, we established the main lifestyle of adults, coded as geophilous (i.e., beetles that occur in the soil, usually taking shelter under stones, or in the sand under debris) vs. xylophilous (e.g., beetles that occur in rotten wood or on living trees, usually into subcortical spaces) using information reported in Aliquò et al. [58] and personal observations of the first author.
Taxonomy follows Löbl and Smetana [59]. We recognize that the interpretation of genus boundaries can vary among taxonomists (e.g., some genera can be considered subgenera), and the monophyly of most tenebrionid genera remains untested. However, the vast majority of species here investigated have been unambiguously classified to the same genera by different researchers, and so genus boundaries are fairly stable.
2.3. Data Analysis
We divided the whole elevational gradient into 24 belts of 100 m (0–100, 101–200, 201–300 m, etc.). As commonly done in biodiversity studies on elevational gradients, we considered species as continuously distributed along the gradient from the lowest to the highest recorded belt, assuming possible gaps due to sampling incompleteness [60,61,62].
To express the actual distribution over genera of the species present in each elevational band (i.e., the generic diversity, GD), we used the method proposed by Williams [17] and based on Simpson’s index [63]. The total number of possible ways in which two species can be taken at random from any collection of N species, classified into genera, each with ni species, respectively, is N(N−1)/2, whereas the total number of possible congeneric pairs is ni(ni−1)/2. Hence, the chance that any two species picked independently and at random from the community will belong to the same genus is Σni(ni−1)/N(N−1), by which N(N−1)/Σni(ni−1) can be used as a measure of the “generic diversity” (GD) of N. GD is therefore a number that will increase with the number of genera over which the N species are distributed. Simpson’s index is an unbiased estimate of diversity when the sample size varies, provided no samples of size 0 or 1 are included. We calculated GD across elevational belts for all tenebrionids and for the two main ecological groups (geophilous vs. xylophilous).
To model elevational patterns for number of genera, number of species, and GD, we tested various functions (linear, semilogarithmic, power, polynomial, and exponential) [12,64] and selected the best fit model according to the goodness-of-fit (R2) and Akaike Information Criterion (AIC). Curvilinear models were linearized by using log10-transformations. We used Analysis of Covariance (ANCOVA) to compare regression slopes. Since the species–elevation relationship may be an indirect effect of decreasing available area due to the basically conical shape of mountains [65], we also used here the procedure recommended by McCain [66] to control for variation in belt area. We first calculated the amount of land surface in each 100 m elevational belt using a digital elevational model. Then, assuming a power function S = cAz for the species area–relationship (where S is the number of species, A is area), we calculated the constant c (c = S/Az), which is a measure of species density (i.e., number of species per area unit) and regressed c-values against elevation. The same approach was used for the genus–elevation relationship. In both cases, for the z-value we used the canonical value of Preston (0.25) [66,67].
Errors refer to standard errors and significance was set at α = 0.05. Analyses were performed using R version 3.2.0 [68].
3. Results
Both the total number of species (species richness) and the total number of genera decreased with elevation according to a semilogarithmic function (Table 1, Figure 1a). The two curves showed significantly different slopes (ANCOVA: F1,44 = 30.63, p < 0.0000001), indicating a faster decline in species richness than in the number of genera. The same pattern was found for the geophilous species (Table 1, Figure 1b). Species richness declined with elevation faster than the number of genera here too (ANCOVA: F1,44 = 46.68, p < 0.0000001). In the case of the xylophilous species, both species richness and number of genera declined linearly with elevation (Table 1, Figure 1c). However, also in this case species richness declined with elevation faster than the number of genera (ANCOVA: F1,44 = 14.44, p = 0.0004). Use of number of species or number of genera per area unit (c-values) produced analogous results (see Supplementary Materials, Table S2). The slope of the curve for the number of species was higher than that for the number of genera (all species: ANCOVA: F1,44 = 12.85, p = 0.0008; the geophilous species: ANCOVA: F1,44 = 8.845, p = 0.0048, and xylophilous species: ANCOVA: F1,44 = 6.701., p = 0.0130).

Table 1.
Results of OLS regression models for the number of tenebrionid species (S) and genera (G), and generic diversity (GD) as function of elevation (E). Errors refer to Standard Errors. R2 = goodness-of-fit, t = Student’s t value.
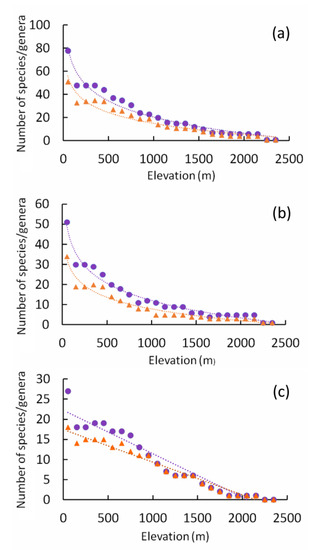
Figure 1.
Relationships of number of tenebrionid species (purple circles) and genera (orange triangles) with elevation (in meters) calculated for all species (a), geophilous species (b), and xylophilous species (c). Dotted lines are regression lines. Regression models were semilog functions for panels (a,b), and linear functions for panel (c). Regression equations and goodness-of fit values are given in Table 1.
Overall GD declined linearly with elevation (Table 1, Figure 2a). When geophilous and xylophilous species were analyzed separately, we found that geophilous species followed a semilogarithmic model (Table 1, Figure 2b), whereas no trend was apparent for the xylophilous species. The slope of the GD equation for geophilous species did not differ significantly from that of geophilous species richness (ANCOVA: F1,40 = 0.45, p = 0.506), but was significantly higher than that observed for the number of geophilous genera (ANCOVA: F1,40 = 12.89, p = 0.0009).
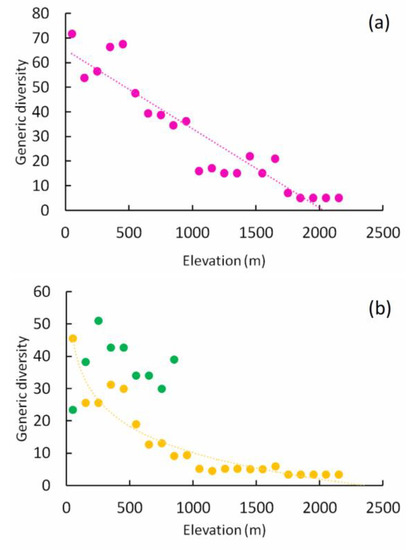
Figure 2.
Relationships between generic diversity and elevation (in meters) for the total number of species (panel a, pink circles) and for geophilous (yellow circles) and xylophilous (green circles) species separately (panel b). Dotted lines are regression lines (linear function in panel a; semilog function in panel b). Regression equations and goodness-of fit values are given in Table 1.
4. Discussion
Overall, we found that both the number of species and the number of genera declined sharply with elevation. This pattern is consistent with a monotonic decrease in species richness with elevation in response to variation in abiotic and biotic factors observed for various animal groups and in a variety of contexts [1,69,70,71].
In general, elevational patterns in species richness tend to follow one the following trends [9]: (1) monotonic decreasing richness with increasing elevation (as in our analyses); (2) plateaus in richness across low elevations, then decreasing, with or (3) without a mid-elevation peak; and (4) a hump-shaped pattern with a mid-elevational peak. The monotonic pattern was the second most common in Rahbek’s reviews [72,73] (about 20% of studies analyzed). A monotonic decrease, however, can follow different models. The best fit model, in our case, was a semilog function, which indicates that the number of species declines very sharply from lowland to medium altitudes, and then continues to decline, but at a decreasing rate. Patterns of species richness and number of genera found in our study are similar to the decline in mammal species richness with increasing elevation in Europe [74], but with a substantial difference. In the case of mammals, the decrease of species richness with elevation was uniformly smooth from above 400 m up to about 3000 m, but there was a lower number of species at 0–150 m than at 200–250 m. This peak has been interpreted as a consequence of a depressive effect on species richness at very low elevations due to massive habitat alteration (such as extensive agriculture and urbanization) [74]. In our case, the highest number of species was found in the 0–100 belt. This can be explained by two not mutually exclusive reasons. First, tenebrionid beetles include several groups adapted to arid conditions and associated with coastal environments, such as sand dunes [39,49,58]. Thus, tenebrionid are speciose also in lowland areas were mammals are poorly diversified. Second, tenebrionids can be less negatively affected by landscape changes than mammals. Although animal response to habitat loss and fragmentation is species-specific, mammals seem to be more sensitive that other groups [75], and the tenebrionids of Latium include many species that can survive profound landscape alteration, including urbanization. Although urbanization also negatively affects tenebrionids [76,77], these animals form rich communities even in small green areas within the largest city (Rome) present in the study area [78,79].
The pattern found for the number of genera was similar to that modelled for species richness, but characterized by a lower slope, which indicates that the number of species decreased with increasing elevation faster than the number of genera. This suggests that the factors depressing tenebrionid diversity along the altitude tend to prune more severely the number of species than the number of genera, which means that species tend to be more concentrated in fewer genera at higher altitudes as a result of a filtering process. This is clearly shown by the pattern of generic diversity. We found that, in general, generic diversity decreased with elevation, thus paralleling the decrease in species richness. While the monotonic decrease in species richness with elevation is a widely recognized and well investigated pattern, our study is the first research showing a monotonic decrease for generic diversity. This decline of generic diversity with increasing elevation suggests that environmental conditions become progressively less favorable to most species; most genera are progressively eliminated, and the remaining species tend to belong to few genera that include the few species adapted to cope with increasing challenging conditions.
Many studies have shown that natural communities are not only assembled through neutral processes (e.g., dispersal and stochastic events), but are also driven by environmental conditions [80,81,82,83]. A widely accepted ecological hypothesis suggests that such environmental drivers act as hierarchical ‘filters’ constraining the assemblage of communities, i.e., progressively selecting species [84,85,86]. Our results support the environmental filtering hypothesis for the species composition of tenebrionid beetles along an elevational gradient, indicating that the harsh conditions of high altitude environments allow the persistence of species belonging to few genera adapted to cope with high temperatures and lack of water. An analysis based on climatic data gathered from 29 meteorological stations distributed along this elevational gradient revealed strong negative correlations between elevation and the following climatic measures: annual maximum, minimum and average temperatures, and Mitrakos’ summer drought stress and year drought stress [12]. These decreases in temperatures and aridity with altitude may act as an important factor driving species impoverishment by filtering thermophilic and xerophilic species. On the other hand, Mitrakos’ winter cold stress and year cold stress increased with altitude [12], which also supports the possible role of factors associated with elevation (such as temperature, humidity, or radiation) as filtering factors for thermo–xerophilic species.
In general, low values of generic diversity may be a consequence of the preference of related species for similar habitats. It is expected for the habitats preferred by congeneric species to be more similar than those occupied by species of different genera; leading species that are ecologically closely related to frequently coexist in the same habitats, if interspecific interferences are not more important than possible intraspecific interferences [34,87]. Most insect communities seem to be not influenced by competition [87,88], and there is some evidence that, consistent with this general pattern, tenebrionid guilds are not regulated by competitive interactions [89,90,91], although the presence of different life strategies in lichen-feeding Helopini has been interpreted as a mechanism possibly reducing competition among species inhabiting the same tree or stone [92]. Additionally, even when insect interspecific competition has been reported, severe effects, such as competitive exclusion, were not demonstrated [87,93].
If tenebrionids are subdivided into two main ecological groups (geophilous and xylophilous) two distinct patterns emerge. Geophilous tenebrionids conform to the general pattern of decreasing generic diversity, whereas xylophilous species tend to maintain high values of generic diversity across the gradient. These results suggest that geophilous species are more sensitive to variation in environmental factors, and the advantages of close relationship (similar adaptations to harsh conditions) are on average greater than the drawbacks (competition). This is consistent with the fact that geophilous tenebrionids are mostly generalist detritivores, and hence competition for food is expected to be low [94]. By contrast, xylophilous species, being associated with vegetation, may be less directly exposed to harsh conditions, but may be more subject to competition to find appropriate microhabitats and food. Xylophilous species are typically saproxylic species and competition has been frequently invoked to explain segregation patterns in saproxylic beetles, although the relative importance of competition in structuring saproxylic communities and how competition intensity varies are open questions [95].
Interestingly, above 1000 m, which typically marks the transition from the sclerophyll evergreen vegetation and heliophilic broadleaves dominated by the oak (Quercus spp.) to the sciaphilous broadleaves dominated by the beech Fagus sylvatica L. [96,97,98], generic diversity of xylophilous tenebrionids could not be calculated, because each genus was represented by only one species (e.g., Helops caeruleus, Corticeus unicolor, Bolitophagus reticulatus), which suggests a combination of competition and very harsh conditions. There is a long lasting debate whether changes in vegetational structure or composition along elevational gradients are abrupt, allowing the recognition of distinct vegetational belts, or gradual, making it impossible to identify clear separations between belts [2]. Our results support the notion that the boundary between oakwoods and beechwods at around 1000 m is probably an abrupt transition, representing a strong discontinuity for the generic diversity of xylophilous tenebrionids.
We are aware that the use of genera can be problematic because of possible discrepancies in their delimitation by different taxonomists. For example, with reference to the fauna studied in this research, Colpotus is regarded as a true genus by most taxonomists, but it has been recently considered as a subgenus of Pedinus [99]. However, for the vast majority of the species considered in this paper, assignment to genera is not disputed, which makes our results robust. Recent phylogenetic reconstructions based on molecular data tend to support traditional tenebrionid taxonomy, at least for the major clades [100,101], but genus delimitations are still based on morphological similarities. The degree of morphological distance accepted by taxonomists to assign species to different genera varies among groups. For example, some tenebrionid genera are very heterogeneous, including a lot of species that exhibit a great morphological diversity (see, for example, the genus Anomalipus [102], which includes some 60 species and subspecies), whereas other groups, such as mammal taxa, typically include fewer and morphologically more similar species (for example, the entire family of Mustelidae includes some 65 species assigned to 23 genera [103]). The different criteria used for delimiting genera in different taxa may make it difficult cross-taxon comparisons in generic diversity patterns.
We are aware that our approach does not allow a direct test for the role of competition, mainly because there is no guarantee that species present in the same belt are really syntopic, i.e., that they co-occur in the same sites within the belt (and, in general, this is unlikely for some species). Thus, it would be interesting in the future to study within-belt co-occurrence patterns. However, our comparative analyses provide interesting insights into the role of species ecology into the possible relative effects of competition and adaptation.
Supplementary Materials
The following are available online at https://www.mdpi.com/1424-2818/12/2/47/s1: Table S1. Tenebrionid species distribution across the elevational gradient in Latium (Central Italy). Table S2. Results obtained using area-corrected values of number of species and number of genera.
Author Contributions
Conceptualization, C.M., L.D.B., and S.F.; methodology, G.S. and S.F.; formal analysis, C.M., G.S., and S.F.; investigation, L.P., M.B., and S.F.; data curation, C.M.; writing—original draft preparation, C.M., G.S., L.D.B., L.P., M.B., and S.F.; writing—review and editing, C.M., G.S., L.D.B., and S.F.; visualization, L.D.B.; supervision, L.P., M.B., and S.F. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
We are grateful to the five anonymous reviewers for their comments on a previous version of this paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Peters, M.; Hemp, A.; Appelhans, T.; Behler, C.; Classen, A.; Detsch, F.; Ensslin, A.; Ferger, S.W.; Frederiksen, S.B.; Gebert, F.; et al. Predictors of elevational biodiversity gradients change from single taxa to the multi-taxa community level. Nat. Commun. 2016, 7, 13736. [Google Scholar] [CrossRef] [PubMed]
- Fattorini, S.; Di Biase, L.; Chiarucci, A. Recognizing and interpreting vegetational belts: New wine in the old bottles of a von Humboldt’s legacy. J. Biogeogr. 2019, 46, 1643–1651. [Google Scholar] [CrossRef]
- Callaway, R.M.; Brooker, R.W.; Choler, P.; Kikvidze, Z.; Lortie, C.J.; Michalet, R.; Paolini, L.; Pugnaire, F.I.; Newingham, B.; Aschehoug, E.T.; et al. Positive interactions among alpine plants increase with stress. Nature 2002, 417, 844–848. [Google Scholar] [CrossRef] [PubMed]
- Kikvidze, Z.; Pugnaire, F.I.; Brooker, R.W.; Choler, P.; Lortie, C.J.; Michalet, R.; Callaway, R.M. Linking patterns and processes in alpine plant communities: A global study. Ecology 2005, 86, 1395–1400. [Google Scholar] [CrossRef]
- Körner, C. The use of “altitude” in ecological research. Trends Ecol. Evol. 2007, 22, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Körner, C. Alpine Treelines – Functional Ecology of the Global High Elevation Tree Limits, 1st ed.; Springer: Basel, Switzerland, 2012; p. 220. [Google Scholar]
- Le Roux, P.C.; McGeoch, M.A. Interaction intensity and importance along two stress gradients: Adding shape to the stress–gradient hypothesis. Oecologia 2010, 162, 733–745. [Google Scholar] [CrossRef] [PubMed]
- Körner, C. Alpine Plant Life: Functional Plant Ecology of High Mountain Ecosystems, 2nd ed.; Springer-Verlag: Heidelberg, Germany, 1999; p. 349. [Google Scholar]
- McCain, C.M.; Grytnes, J.A. Elevational gradients in species richness. In Encyclopedia of LifeSciences (ELS); John Wiley & Sons: Chichester, UK, 2010; pp. 1–10. [Google Scholar]
- Hoiss, B.; Krauss, J.; Potts, S.G.; Roberts, S.; Steffan–Dewenter, I. Altitude acts as an environmental filter on phylogenetic composition, traits and diversity in bee communities. Proc. R. Soc. Lond. 2012, 279, 4447–4456. [Google Scholar] [CrossRef]
- Sanders, N.J. Elevational gradients in ant species richness: Area, geometry, and Rapoport’s rule. Ecography 2002, 25, 25–32. [Google Scholar] [CrossRef]
- Fattorini, S. Disentangling the effects of available area, mid-domain constraints, and species environmental tolerance on the altitudinal distribution of tenebrionid beetles in a Mediterranean area. Biodivers. Conserv. 2014, 23, 2545–2560. [Google Scholar] [CrossRef]
- Luo, Y.; Liu, J.; Tan, S.; Cadotte, M.W.; Xu, K.; Gao, L.; Li, D. Trait variation and functional diversity maintenance of understory herbaceous species coexisting along an elevational gradient in Yulong Mountain, Southwest China. Plant Divers. 2016, 38, 303–311. [Google Scholar] [CrossRef]
- Sanders, N.J.; Rahbek, C. The patterns and causes of elevational diversity gradients. Ecography 2012, 35, 1–3. [Google Scholar] [CrossRef]
- Camacho, L.; Avilés, L. Decreasing predator density and activity explain declining predation of insect prey along elevational gradients. Am. Nat. 2019, 194, 334–343. [Google Scholar] [CrossRef]
- Lazarina, M.; Charalampopoulos, A.; Psaralexi, M.; Krigas, N.; Michailidou, D.E.; Kallimanis, A.S.; Sgardelis, S.P. Diversity patterns of different life forms of plants along an elevational gradient in Crete, Greece. Diversity 2019, 11, 200. [Google Scholar] [CrossRef]
- Williams, C.B. Intra-generic competition as illustrated by Moreau’s records of east african bird communities. J. Anim. Ecol. 1951, 20, 246–253. [Google Scholar] [CrossRef]
- Ricklefs, R.E.; Cox, G.W. Morphological similarity and ecological overlap among passerine birds on St. Kitts, British West Indies. Oikos 1977, 29, 60–66. [Google Scholar] [CrossRef]
- Douglas, M.E.; Matthews, W.J. Does morphology predict ecology? Hypothesis testing within a freshwater stream fish assemblage. Oikos 1992, 65, 213–224. [Google Scholar] [CrossRef]
- Melville, J.; Harmon, L.J.; Losos, J.B. Intercontinental community convergence of ecology and morphology in desert lizards. Proc. R. Soc. Lond. 2005, 273, 557–563. [Google Scholar] [CrossRef]
- Valiente-Banuet, A.; Verdú, M. Temporal shifts from facilitation to competition occur between closely related taxa. J. Ecol. 2008, 96, 489–494. [Google Scholar] [CrossRef]
- Elton, C.S. Competition and the structure of ecological communities. J. Anim. Ecol. 1946, 15, 54–68. [Google Scholar] [CrossRef]
- Simberloff, S. The taxonomic diversity of island biotas. Evolution 1970, 24, 23–47. [Google Scholar] [CrossRef]
- Diamond, J.L. Niche shifts and the rediscovery of interspecific competition. Am. Sci. 1978, 66, 322–331. [Google Scholar]
- Connor, E.F.; Simberloff, S. Interspecific competition and species co-occurrence patterns on islands: Null models and the evaluation of evidence. Oikos 1983, 41, 455–465. [Google Scholar] [CrossRef]
- Weiher, E.; Keddy, P. Ecological Assembly Rules: Perspectives, Advances, Retreats; Cambridge University Press: Cambridge, UK, 1999; p. 430. [Google Scholar]
- Gotelli, N.J. Null model analysis of species co-occurrence patterns. Ecology 2000, 81, 2606–2621. [Google Scholar] [CrossRef]
- Gotelli, N.J.; McCabe, D.J. Species co-occurrence: A meta-analysis of J.M. Diamond’s assembly rules model. Ecology 2002, 83, 2091–2096. [Google Scholar] [CrossRef]
- Sanderson, J.G.; Pimm, S.L. Patterns in Nature. The Analysis of Species Co-occurrences; University of Chicago Press: Chicago, IL, USA, 2015; p. 206. [Google Scholar]
- Ulrich, W.; Jabot, F.; Gotelli, N.J. Competitive interactions change the pattern of species co-occurrences under neutral dispersal. Oikos 2017, 126, 91–100. [Google Scholar] [CrossRef]
- Miller, R.S. Pattern and process in competition. Adv. Ecol. Res. 1967, 4, 1–74. [Google Scholar]
- Jenssen, T.A. Shift in the structural habitat of Anolis opalinus due to congeneric competition. Ecology 1973, 54, 863–869. [Google Scholar] [CrossRef]
- McCluskey, E.S. Generic diversity in phase of rhythm in Formicine ants. Psyche 1973, 80, 295–304. [Google Scholar] [CrossRef]
- Den Boer, P.J. Exclusion or coexistence and the taxonomic or ecological relationship between species. Neth. J. Zool 1980, 30, 278–306. [Google Scholar] [CrossRef]
- Moulton, M.P.; Pimm, S.L. The extent of competition in shaping an introduced avifauna. In Community Ecology; Diamond, J., Case, T.J., Eds.; Harper & Row: New York, NY, USA, 1986; pp. 80–97. [Google Scholar]
- Kaplan, I.; Denno, R.F. Interspecific interactions in phytophagous insects revisited: A quantitative assessment of competition theory. Ecol. Lett. 2007, 10, 977–994. [Google Scholar] [CrossRef]
- Burns, J.H.; Strauss, S.Y. More closely related species are more ecologically similar in an experimental test. Proc. Natl. Acad. Sci.USA 2011, 108, 5302–5307. [Google Scholar] [CrossRef] [PubMed]
- Keddy, P.A. Competition, 2nd ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001; p. 552. [Google Scholar]
- de los Santos, A. Generic diversity patterns in the Tenebrionid beetle communities (Col. Tenebrionidae). Ecologia Mediterranea 1994, 20, 125–136. [Google Scholar]
- Gómez-González, L.A.; de los Santos, A.; Alonso, C. Carabid generic diversity and inter-island similarity in the Macaronesian Region. In Natural History and Applied Ecology of Carabid Beetles; Brandmayr, P., Lövei, G.L., Brandmayr, T., Zetto Casale, A., Vigna Taglianti, A., Eds.; Pensoft: Sofia, Bulgaria, 2000; pp. 53–60. [Google Scholar]
- Lawrence, J.F.; Spilman, T.J. Tenebrionidae. In Immature Insects; Stehr, F.W., Ed.; Kendall Hunt Publishing: Dubuque, ID, USA, 1991; Volume 2, pp. 520–524. [Google Scholar]
- Cavieres, L.A.; Arroyo, M.T.K.; Molina-Montenegro, M.; Torres, C.; Peñaloza, A. Nurse effect of Bolax gummifera (Apiaceae) cushion plants in the alpine vegetation of the Chilean Patagonian Andes. J. Veg. Sci. 2002, 13, 547–554. [Google Scholar] [CrossRef]
- Kikvidze, Z.; Michalet, R.; Brooker, R.W.; Cavieres, L.A.; Lortie, C.J.; Pugnaire, F.I.; Callaway, R.M. Climatic drivers of plant–plant interactions and diversity in alpine communities. Alp. Bot. 2011, 121, 63–70. [Google Scholar] [CrossRef]
- Antonsson, A.; Björk, R.G.; Molau, U. Nurse plant effect of the cushion plant Silene acaulis (L.) Jacq. in an alpine environment in the subarctic Scandes, Sweden. Plant Ecol. Divers. 2009, 2, 17–25. [Google Scholar] [CrossRef]
- Körner, C. Why are there global gradients in species richness? Mountains might hold the answer. Trends Ecol. Evol. 2000, 15, 513–514. [Google Scholar] [CrossRef]
- Li, X.J.; Li, X.; Zhang, X.; Wu, J.; Shen, Z.; Zhang, Y.; Xu, X.; Fan, T.; Zhao, Y.; Yan, W. Root biomass distribution in alpine ecosystems of the northern Tibetan Plateau. Environ. Earth Sci. 2011, 64, 1911–1919. [Google Scholar] [CrossRef]
- Salvati, L.; Perini, L.; Bajocco, S.; Sabbi, A. Climate aridity and land use change: A regional-scale analysis. Geogr. Res. 2012, 50, 193–203. [Google Scholar] [CrossRef]
- Fattorini, S. Regional insect inventories require long time, extensive spatial sampling and good will. PLoS ONE 2013, 8, e62118. [Google Scholar] [CrossRef]
- Fattorini, S.; Salvati, L. Tenebrionid beetles as proxy indicators of climate aridity in a Mediterranean area. Ecol. Indic. 2014, 38, 256–261. [Google Scholar] [CrossRef]
- Ruffo, S.; Stoch, F. Checklist and Distribution of the Italian Fauna; Museo di Storia Naturale di Verona: Verona, Italy, 2006; p. 304. [Google Scholar]
- Trichas, A. The genus Dendarus Latreille, 1829 (Coleoptera, Tenebrionidae: Dendarini) in Greece (A systematic account of the genus with description of a new species and four new systematic combinations). In Advances in Arachnology and Developmental Biology; Makarov, S.E., Dimitrijević, R.N., Eds.; SASA, Belgrade and UNESCO MAB Serbia: Belgrade, Serbia, 2008; pp. 417–462. [Google Scholar]
- Ferrer, J. Contribución al conocimiento de los Asinini iberobaleares. Segunda nota. Las Alphasida (Glabrasida) del grupo Tricostatae Escalera 1922 (Coleoptera, Tenebrionidae, Pimeliinae). Boln. SEA 2008, 43, 61–73. [Google Scholar]
- Condamine, F.L.; Soldati, L.; Rasplus, J.Y.; Kergoat, G.J. New insights on systematics and phylogenetics of Mediterranean Blaps species (Coleoptera: Tenebrionidae: Blaptini), assessed through morphology and dense taxon sampling. Syst. Entomol. 2011, 36, 340–361. [Google Scholar] [CrossRef]
- Ferrer, J. Contribución al conocimiento del Género Phylan Dejean, 1821, y descripción de una specie nueva del género Heliopates Dejean, 1834 (Coleoptera, Tenebrionidae, Pedinini). Boln. SEA 2011, 49, 75–82. [Google Scholar]
- Pons, J.; Bruvo, B.; Petitpierre, E.; Plohl, M.; Ugarkovic, D.; Juan, C. Complex structural features of satellite DNA sequences in the genus Pimelia (Coleoptera: Tenebrionidae): Random differential amplification from a common ‘satellite DNA library’. Heredity 2004, 9, 418–427. [Google Scholar] [CrossRef]
- Soldati, F.; Soldati, L. Species delimitation using morphological and molecular tools in the Asida (Polasida) jurinei Solier, 1836 species complex. Preliminary results. (Coleoptera: Tenebrionidae: Tentyrinae). Cahiers Scientifiques Muséum Lyon 2006, 10, 111–116. [Google Scholar]
- Stroscio, S.; Baviera, C.; Frati, F.; Lo Paro, G.; Nardi, F. Deep genetic divergence in the darkling beetle Pimelia rugulosa (Coleoptera, Tenebrionidae) reflects Plio-Pleistocenic paleogeographic history of Sicily. J Zool. Syst. Evol. Res. 2011, 49, 196–203. [Google Scholar] [CrossRef]
- Aliquò, V.; Rastelli, M.; Rastelli, S.; Soldati, F. Coleotteri Tenebrionidi d’Italia; CD-ROM Museo Civico di Storia Naturale di Carmagnola: Carmagnola, Italy, 2006. [Google Scholar]
- Löbl, I.; Smetana, A. Catalogue of Palaearctic Coleoptera. Vol. 5. Tenebrionoidea; Apollo Books: Stenstrup, UK, 2008; p. 670. [Google Scholar]
- Grytnes, J.A.; Vetaas, O.R. Species richness and altitude: A comparison between null models and interpolated plant species richness along the Himalayan altitudinal gradient, Nepal. Am. Nat. 2002, 159, 294–304. [Google Scholar] [CrossRef]
- Mena, J.L.; Vázquez-Domínguez, E. Species turnover on elevational gradients in small rodents. Glob. Ecol. Biogeogr. 2005, 14, 539–547. [Google Scholar] [CrossRef]
- Rowe, R. Environmental and geometric drivers of small mammal diversity along elevational gradients in Utah. Ecography 2009, 32, 411–422. [Google Scholar] [CrossRef]
- Simpson, E.H. Measurement of Diversity. Nature 1949, 63, 688. [Google Scholar] [CrossRef]
- Fontanilla, A.M.; Nakamura, A.; Xu, Z.; Cao, M.; Kitching, R.L.; Tang, Y.; Burwell, C.J. Taxonomic and Functional Ant Diversity Along tropical, Subtropical, and Subalpine Elevational Transects in Southwest China. Insects 2019, 10, 128. [Google Scholar] [CrossRef] [PubMed]
- McCain, C.M. Global analysis of reptile elevational diversity. Glob. Ecol. Biogeogr. 2010, 19, 541–553. [Google Scholar] [CrossRef]
- McCain, C.M. Area and mammalian elevational diversity. Ecology 2007, 88, 76–86. [Google Scholar] [CrossRef]
- Preston, F.W. The canonical distribution of commonness and rarity. Part I. Ecology 1962, 43, 185–215. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2015. [Google Scholar]
- Stevens, G.C. The elevational gradient in altitudinal range: An extension of Rapoport’s latitudinal rule to altitude. Am. Nat. 1992, 140, 893–911. [Google Scholar] [CrossRef]
- Kaspari, M.; O’Donnell, S.; Kercher, J.R. Energy, density, and constraints to species richness: Ant assemblages along a productivity gradient. Am. Nat. 2000, 155, 280–293. [Google Scholar] [CrossRef]
- Lomolino, M.V.; Riddle, B.R.; Whittaker, R.J.; Brown, J.H. Biogeography, 4th ed.; Sinauer Associates: Sunderland, MA, USA, 2010; p. 878. [Google Scholar]
- Rahbek, C. The elevational gradient of species richness: A uniform pattern? Ecography 1995, 18, 200–205. [Google Scholar] [CrossRef]
- Rahbek, C. The role of spatial scale and the perception of large-scale species-richness patterns. Ecol. Lett. 2005, 8, 224–239. [Google Scholar] [CrossRef]
- Amori, G.; Boitani, L.; Milana, G.; Maiorano, L.; Luiselli, L. Endemism and diversity in European montane mammals: Macro-ecological patterns. Biol. J. Linn. Soc. 2019, 128, 225–237. [Google Scholar] [CrossRef]
- Prugh, L.R.; Hodges, K.E.; Sinclair, R.E.; Brashares, J.S. Effect of habitat area and isolation on fragmented animal populations. Proc. Natl. Acad. Sci. USA 2008, 105, 20770–20775. [Google Scholar] [CrossRef]
- Fattorini, S. Insect rarity, extinction and conservation in urban Rome (Italy): A 120-year-long study of tenebrionid beetles. Insect Conserv. Divers. 2011, 4, 307–315. [Google Scholar] [CrossRef]
- Fattorini, S. Insect extinction by urbanization: A long term study in Rome. Biol. Conserv. 2011, 144, 370–375. [Google Scholar] [CrossRef]
- Fattorini, S. Urban biodiversity hotspots are not related to the structure of green spaces: A case study of tenebrionid beetles from Rome, Italy. Urban Ecosyst. 2014, 17, 1033–1045. [Google Scholar] [CrossRef]
- Fattorini, S.; Galassi, D.M.P. Role of urban green spaces for saproxylic beetle conservation: A case study of tenebrionids in Rome, Italy. J. Insect. Conserv. 2016, 20, 737–745. [Google Scholar] [CrossRef]
- Petchey, O.L.; Evans, K.L.; Fishburn, I.S.; Gaston, K.J. Low functional diversity and no redundancy in British avian assemblages. J. Anim. Ecol. 2007, 76, 977–985. [Google Scholar] [CrossRef]
- Villéger, S.; Miranda, J.R.; Hernández, D.F.; Mouillot, D. Contrasting changes in taxonomic vs. functional diversity of tropical fish communities after habitat degradation. Ecol. Appl. 2010, 20, 1512–1522. [Google Scholar] [CrossRef]
- Mason, N.W.; de Bello, F.; Doležal, J.; Lepš, J. Niche overlap reveals the effects of competition, disturbance and contrasting assembly processes in experimental grassland communities. J. Ecol. 2011, 99, 788–796. [Google Scholar] [CrossRef]
- Funk, J.L.; Larson, J.E.; Ames, G.M.; Butterfield, B.J.; Cavender-Bares, J.; Firn, J.; Laughlin, D.C.; Sutton-Grier, A.E.; Williams, L.; Wright, J. Revisiting the Holy Grail: Using plant functional traits to understand ecological processes. Biol. Rev. 2017, 92, 1156–1173. [Google Scholar] [CrossRef]
- Emerson, B.C.; Gillespie, R.G. Phylogenetic analysis of community assembly and structure over space and time. Trends Ecol. Evol. 2008, 23, 619–630. [Google Scholar] [CrossRef]
- Kraft, N.J.B.; Adler, P.B.; Godoy, O.; James, E.C.; Fuller, S.; Levine, J.M. Community assembly, coexistence and the environmental filtering metaphor. Funct. Ecol. 2015, 29, 592–599. [Google Scholar] [CrossRef]
- Cadotte, M.W.; Tucker, C.M. Should environmental filtering be abandoned? Trends Trends. Ecol. Evol. 2017, 32, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Price, P.W.; Denno, R.F.; Eubanks, M.D.; Finke, D.L.; Kaplan, I. Insect Ecology: Behavior, Populations and Communities; Cambridge University Press: Cambridge, UK, 2011; p. 82. [Google Scholar]
- Shorrocks, B.; Rosewell, J.; Edwards, K.; Atkinson, W. Interspecific competition is not a major organizing force in many insect communities. Nature 1984, 310, 310–312. [Google Scholar] [CrossRef]
- Wise, D.H. A Removal Experiment with Darkling Beetles: Lack of evidence for interspecific competition. Ecology 1981, 62, 727–738. [Google Scholar] [CrossRef]
- Carpaneto, G.M.; Fattorini, S. Spatial and seasonal organisation of a darkling beetle (Coleoptera: Tenebrionidae) community inhabiting a Mediterranean coastal dune system. Ital. J. Zool. 2001, 68, 207–214. [Google Scholar] [CrossRef]
- Fattorini, S.; Bergamaschi, D.; Mantoni, C.; Acosta, A.T.R.; Di Giulio, A. Niche partitioning in tenebrionid species (Coleoptera: Tenebrionidae) inhabiting Mediterranean coastal dunes. Eur. J. Entomol. 2016, 113, 462–468. [Google Scholar] [CrossRef]
- Nabozhenko, M.V.; Keskin, B.; Nabozhenko, S.V. Life forms and strategies of lichen-feeding darkling beetles (Coleoptera, Tenebrionidae: Helopini). Entomol. Rev. 2017, 97, 735–746. [Google Scholar] [CrossRef]
- Niemelä, J. Interspecific competition in ground-beetle assemblages (Carabidae): What have we learned? Oikos 1993, 66, 325–335. [Google Scholar] [CrossRef]
- Fattorini, S. Ecology and conservation of tenebrionid beetles in Mediterranean coastal areas. In Insect Ecology and Conservation; Fattorini, S., Ed.; Research Signpost: Trivandrum, Kerala, 2008; pp. 165–297. [Google Scholar]
- Brin, A.; Bouget, C. Biotic interactions between saproxylic insect species. In Saproxylic Insects: Diversity, Ecology and Conservation (Zoological Monographs 1); Ulyshen, M.D., Ed.; Springer: Cham, Switzerlnad, 2018; pp. 471–514. [Google Scholar]
- Giacomini, V.; Fenaroli, L. Conosci l’Italia. La flora; Touring Club Italiano: Milano, Italy, 1958; p. 276. [Google Scholar]
- Blasi, C.; Stanisci, A.; Filesi, L.; Milanese, A.; Perinelli, E.; Riggio, L. Syndinamics of lowland Quercus frainetto & Q. cerris forests in Lazio (central Italy). Fitosociologia 2002, 39, 23–43. [Google Scholar]
- Piovesan, G.; Biondi, F.; Bernabei, M.; di Filippo, A.; Schirone, B. Spatial and altitudinal bioclimatic zones of the Italian Peninsula identified from a beech (Fagus sylvatica L.) tree-ring network. Acta Oecol. 2005, 27, 197–210. [Google Scholar] [CrossRef]
- Kaminski, M.J.; Iwan, D. Revision of the Subtribe Pedinina (Tenebrionidae: Pedinini). Ann. Zool. 2017, 67, 585–607. [Google Scholar] [CrossRef]
- Kamiński, M.J.; Kanda, K.; Raś, M.; Smith, A.D. Pythiopina, an enigmatic subtribe of darkling beetles (Coleoptera: Tenebrionidae: Pedinini): Taxonomic revision, microtomography, ecological niche models and phylogenetic position. Syst. Entomol. 2018, 43, 147–165. [Google Scholar] [CrossRef]
- Fattorini, S.; Mantoni, C.; Audisio, P.; Biondi, M. Taxonomic variation in levels of endemism: A case study of Italian tenebrionid beetles. Insect. Conserv. Divers. 2019, 12, 351–361. [Google Scholar] [CrossRef]
- Endroedy-Younga, S. Revision of the Genus Anomalipus Latreille, 1846 (Coleoptera: Tenebrionidae: Platynotini); Transvaal Museum Monograph No. 6; Transvaal Museum: Pretoria, South Africa, 1988; p. 129. [Google Scholar]
- Nowak, R.M. Walker’s Mammals of the World, 5th ed.; Johns Hopkins University Press: Baltimore, MD, USA, 1991; p. 1614. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).