Abstract
This study assessed the reproductive success of a temperate dioecious shrub, Canada buffaloberry, Shepherdia canadensis (L.) Nutt., in central Alberta, Canada, by examining the effects of spatial patterns and overstory canopy on flower and fruit production. S. canadensis is more abundant and productive (more fruit) at forest edges and in forest gaps, suggesting a dependence on higher light conditions than is typical of late-seral forests. We used path analysis to demonstrate that flower and fruit production exhibited density-dependent effects at a scale of 50 m2 around focal female plants. Fruit production was positively affected by male intraspecific density (pollen supply) and negatively affected by female intraspecific density (pollen competition), but not correlated with overall intraspecific density. The effects of sex-differentiated density are partly due to pollinator responses to male plant density. Flower production was positively affected by overall intraspecific density. A pollen supplementation trial doubled fruit production relative to a control, demonstrating that local male density (pollen availability) and pollinator activity can limit fruit production in S. canadensis. Canopy cover was negatively related to both flower and total fruit production, with approximately one-third (34%) of the total effect of canopy on fruit production due to the effect of canopy on flower production. The commonly observed negative association between canopy cover and fruit production in buffaloberry, therefore, is partly a result of the reduction first in flower number and second in fruit set. This study clarifies the mechanisms associated with the often-noted observation, but not previously assessed at the level of individuals, that reproductive output in S. canadensis is density dependent, limited by canopy cover through reductions in both flowering and fruit set, and pollinator limited. These findings hold implications for managing animal species that depend on the fruit of S. canadensis and suggest future directions for research on dioecious and actinorhizal species.
1. Introduction
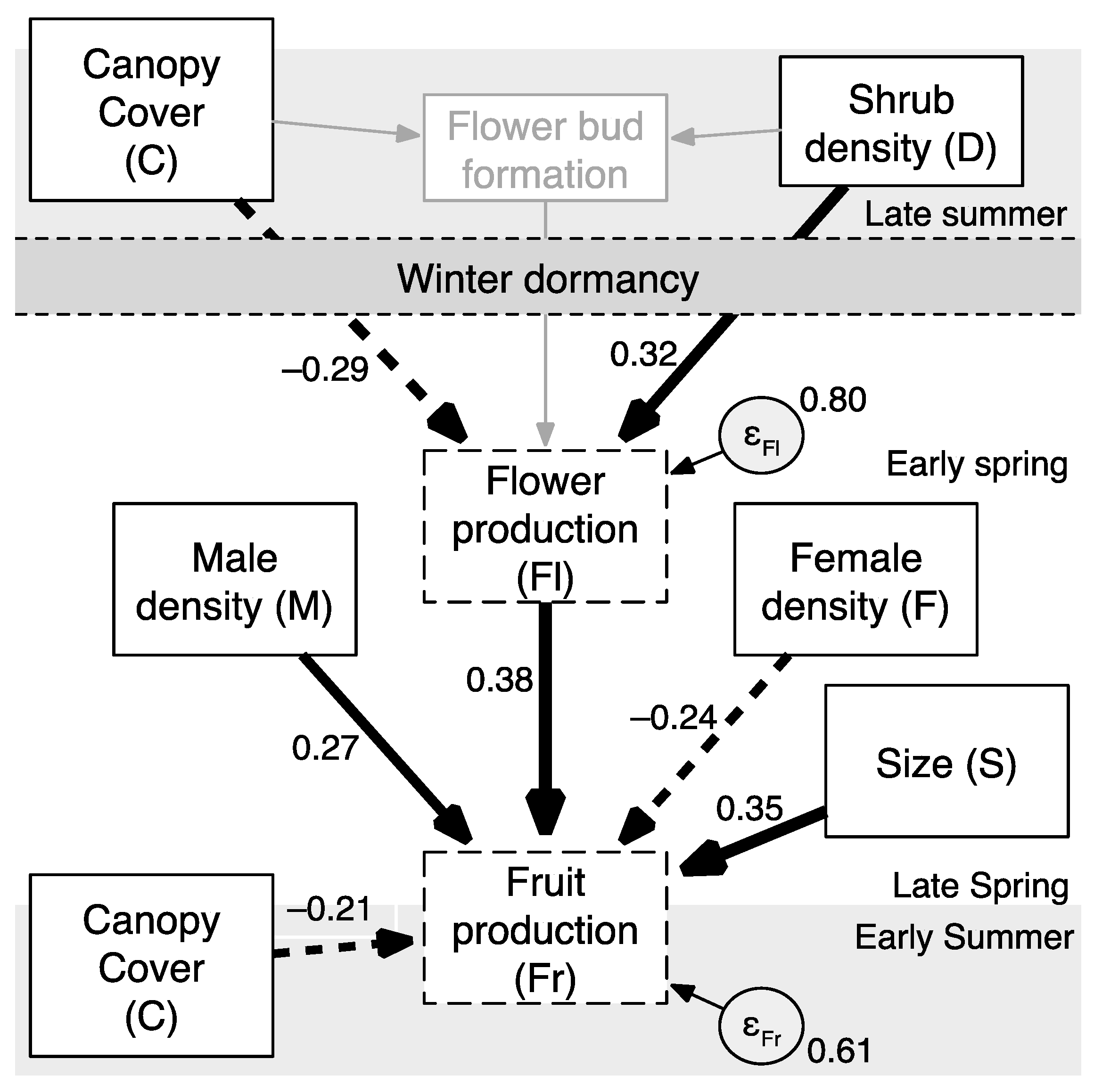
Patterns of animal and plant diversity in temperate regions show concentrations of biodiversity in forests and forest edges [1,2]. An important understory and edge species of North American forests is Canada buffaloberry, or soopolallie, Shepherdia canadensis (L.) Nutt. (Elaeagnaceae). Buffaloberry is a deciduous and dioecious shrub, widespread over the forested and mountainous regions of North America [3,4]. The shrub is an important food resource for mammals and birds that feed on the hundreds and often thousands of small (<1 cm long) fruits produced by individual female shrubs [4,5,6] (Figure 1).

Figure 1.
The small and densely packed red fruit of Shepherdia canadensis (Photo by S. E. Nielsen, 31 July 2009).
Previous studies have shown that fruit production in buffaloberry is affected by light and pollen availability [7,8], as is the case for other angiosperms [9,10,11]. High overstory canopy cover reduces the availability of light to understory plants, typically resulting in lower fruit and seed production and understory biomass, particularly under dense canopy [10,12,13]. Optimal light requirements for plant species vary [14,15], but canopy closure limits woody plant biomass [15,16] and understory diversity [1] across forest types. Buffaloberry occurs less frequently and produces less fruit as canopy cover increases [8,17,18], as in other dioecious shrubs [19]. Logging, fires, and other disturbances that decrease canopy cover and reduce competition with canopy trees for light favor growth and higher rates of reproductive success for buffaloberry and other understory species [16,18,20]. Buffaloberry may be particularly sensitive to light conditions since it is an actinorhizal nitrogen-fixing plant, most genera of which are shade-intolerant [21].
The phenology of buffaloberry is consistent with shade-intolerant plants. It forms flower buds in the late summer of the year prior to fruit production and is dormant through winter, with flower emergence occurring shortly after snowmelt and before canopy leaf emergence [22,23]. Fruiting occurs from late June to September depending on summer temperatures [22,24]. Flower bud formation and fruit maturation occurs under a fully developed canopy, while flowering occurs before canopy leaf emergence in spring. The plant may match available resources at the end of the growing season as it forms flower primordia and also in the spring during flowering and fruit development as is common in temperate species such as the dioecious shrub Rhamnus alpinus [25]. As resource conditions at the time of anthesis and fruit development are imperceptible to the plant at the time of flower bud formation, the bet-hedging hypothesis [11,25], where plants are assumed to form flower buds in accordance with potential and ideal resource conditions, may partly explain lower buffaloberry fruit production under low light conditions. Light availability in other species has been correlated with flower number [19,26], thereby limiting fruit and seed formation by setting a limit on the number of fruits that can potentially mature [11]. Since light availability is correlated with air temperature and air humidity [27,28], measures of light availability may be a proxy for conditions that may also affect pollinator activity [13,29,30].
As with light availability, the quantity of available pollen often affects reproductive output [9,31]. While dioecy prevents geitonogamy, it also makes pollen receipt and fruit production dependent on male densities surrounding female plants [32,33,34]. A higher density of males surrounding a female plant will increase the local quantity of pollen while increasing the probability that pollinators will transfer pollen to stigmas [7,30,35]. However, the degree to which male density affects fruit production is connected to the type of pollinator associated with particular plant species. Dioecious species in temperate regions often undergo anthesis in early spring [19,36,37]. Pollination in early spring is often facilitated by fly pollination since flies can be active in cooler temperatures [38,39]. Flies active in early spring are often generalists, consuming pollen indiscriminately from flowers of different species [40]. Generalist pollinators are unlikely to carry pollen between flowers of the same species over moderate to long distances since they forage among many food sources and are likely to displace pollen from a particular species before they reach a distant conspecific plant [7,40]. While specialized pollinators can be more effective than generalists over moderate to long distances as they focus on particular species at different times of the day and season [41], many specialized pollinators also avoid female flowers because of the lack of a pollen reward [42,43,44]. Dioecious and monoecious species that are pollinated by generalist insects are, as a result, often dependent on conspecific density over short distances [7,40], while dimorphic plant species that rely on pollination over moderate distances tend to be pollinated abiotically [45].
Buffaloberry is pollinated primarily by syrphid flies, and to a lesser extent, other fly species [7,40]. Its pollination syndrome is that of the generalist fly pollinator [39], with small (~5 mm in diameter), open, bowl-shaped, green-yellow flowers that produce little nectar [39,40]. This association with generalist flies entails that the distances over which pollinators transfer pollen from male to female plants are relatively small, in a 2–6 m radius surrounding female buffaloberry individuals [7,46], a short pollinator foraging distance also observed in other dioecious plants that depend on generalist flies [30]. Given the short foraging distances of generalist flies, proximate male densities increase the supply of pollen available to female flowers, while female densities increase competition for pollen [46]. While high male density and low female density increase the supply of pollen available for pollination in dioecious plants pollinated by generalist flies, higher plant densities may attract pollinators by increasing reward patch sizes, making isolated female plants further prone to pollen limitation as a result of reduced pollinator attraction [11,25,47]. Female dioecious plants pollinated by generalist insects, therefore, should demonstrate pollen limitation when male density surrounding the female is low, due to the ineffectual transfer of pollen over large distances by generalist pollinators. Dioecious plants pollinated by generalist insects may thus be expected to be among the majority of angiosperms where fruit and seed production is likely to be limited by pollen transfer [31]. Pollen supplementation trials [11,48,49] may, therefore, be expected to demonstrate pollen limitation in dioecious species pollinated by generalist insects.
Previous studies of buffaloberry have not simultaneously assessed the combined effects of light and pollen availability at the level of individual plants on flower and fruit production. This study assesses factors influencing fruit production in buffaloberry at the level of individual shrubs. The study specifically asks: (1) To what degree is flower and fruit production in female buffaloberry individuals affected by light availability and how much of this effect is due to the effects of canopy cover on flower production as a mediating variable? (2) Do the effects of spatial pollen availability—pollen supply and competition near female plants—explain additional variation in buffaloberry fruit production? (3) Is there evidence of pollen limitation in buffaloberry, as a case of a dioecious plant pollinated by generalist insects?
2. Materials and Methods
2.1. Study Site
The study was carried out from April to July 2012 and in May of 2014 in Terwillegar Park (53.479946° N, 113.607827° W), a 186 hectare multipurpose recreation site in southwest Edmonton, Alberta, Canada (455.7 mm mean annual precipitation (76% rain, 24% snow), 14.5 °C mean growing season (May–September) temperature [50]). The park consists of natural vegetation with minimal development. Historical disturbances included small-scale agriculture (first half of the 20th century) and gravel extraction until 1986 [51]. Vegetation consists of semi-open to closed forests of trembling aspen (Populus tremuloides) and balsam poplar (Populus balsamifera) representative of parkland forest in the northern Central Parkland Natural Subregion [52,53]. The number of buffaloberry individuals in the park is in the 1000s, with mean density of 10.4 shrubs per 100 m2 [46].
2.2. Flower and Fruit Production
During the early spring of 2012, we randomly selected 60 focal female buffaloberry individuals distributed among 5 sites in the park. The maximum distance between sites was ~530 m. Each shrub was located at least 11.28 m (radius of a 400 m2 plot) from other focal female plants. All plants were located within or bordering the semi-open to closed deciduous aspen-poplar forest (with an average of 67% canopy cover) since the shrub is largely absent in forest patches with the presence of evergreen conifers. In May, we counted flowers on four branch segments per shrub (~30 cm each, = 32 cm, SD = 6 cm), with each of the four branch segments in one cardinal direction. We sub-sampled individual shrubs because of high flower production (often > 5000 flowers/plant) and to avoid damaging the flowers between flowering and fruit production. The measure for flower production used is, therefore, an estimate of average flowers per cm of branch length. In July, we counted the fruit produced on the previously marked branch segments to estimate the number of fruit per centimeter of branch length. We also counted all fruit produced by each focal shrub (total fruit production per shrub). To estimate canopy cover above marked plants, we calculated the average of four concave spherical densiometer measurements [54,55] during the fruit counting period in mid-summer when canopy leaf cover was greatest. Densiometer measurements were taken above each plant, which averaged 1.32 m in height (Table 1). To assess the effects of shrub density around focal female plants, we measured the surrounding density of both male and female buffaloberry individuals at a 50 m2 scale (3.99 m radius) using circular plots. Distances of all shrubs to the focal female shrub were measured with a Haglöf DME 201 Cruiser (Långsele, Sweden). Plant height and width measurements were taken to estimate plant size, but average height (m) of major stems was determined to be the most effective proxy for plant size and is used in the path model.

Table 1.
Environmental, demographic, and reproductive variables for Shepherdia canadensis individuals in Terwillegar Park (Edmonton, Alberta, Canada).
2.3. Pollen Supplementation
Pollen limitation was assessed by a pollen supplementation trial completed in May of 2014. This trial assessed fruit set on 15 female shrubs randomly selected from the area, but not from shrubs used for assessing the relationship between flower and fruit production as a function of light availability, shrub size, and surrounding male and female density (above section). A sub-sampling method was used, with two branches on the north and two branches on the south side of the plant selected and flowers counted on 30 cm branch segments, measured from the tip of the branch. One branch in each direction was hand pollinated, while the other was left as a control (n = 60 branches). The branches were marked for resampling. Treatment branches were pollinated with flowers from neighboring male plants (~15 male flowers) to emulate the pollen quality of the proximate environment. Between hand pollination and fruit maturation, a supplemented and control branch of one plant were broken by an unknown factor, leading to a reduction of two branches in the analysis. Fruit set was calculated as the percentage of flowers that became fruit on each branch.
2.4. Statistical Analysis
To evaluate the results of the supplemental pollination trial, we used a mixed-effect model, with individual plant included as a random effect and treatment type (control or supplemented) as the fixed effect, using the “mixed” function of Stata (release 13) [56]. For the analysis of factors affecting flower and fruit production, path models were used to estimate (1) the influence of canopy cover (C) and overall buffaloberry density (D) on flower production (Fl) of the focal females, and (2) the influence of canopy cover (C), male (M) and female (F) buffaloberry density, focal plant size (S), and focal plant flower production (Fl) on the fruit production (Fr) of focal female shrubs (Equations (1)–(3)). Flower production was, therefore, a mediation variable between light availability and fruit production (Figure 2). Preliminary analyses demonstrated that variables did not vary systematically across site and site was not used in subsequent analyses. We used Stata (release 13) and its sembuilder function [57] to construct and run the path model (Dataset S1; File S1). The untransformed variables were not multivariate normal (Mardia mSkewness (P < 0.001), Mardia mKurtosis (P = 0.04), and the Doornik-Hansen (P < 0.001) tests rejected the null hypothesis of multivariate normality). Variables were, therefore, normalized using Royston’s V’ (Royston 1991) (Appendix A, Tables S1–S3). For the transformed variables, Mardia mSkewness (P = 0.40), Mardia mKurtosis (P = 0.29), and Doornik–Hansen (P = 0.15) tests provided support for multivariate normality. Since transformed variables were used in the model, the results are reported in standardized coefficients. In the model, the flower number variable was converted to flower density (number of flowers per centimeter of branch segment) to remove bias associated with small differences in branch segment length.
Fl = βFlCC + βFlDD,
Fr = βFrFlFl + βFrCC + βFrFF + βFrMM + βFrSS,
Fr = (βFlCC)(βFrFl) + (βFlDD)(βFrFl) + βFrCC + βFrFF + βFrMM + βFrSS,
Fr = (−0.29C)(0.38) + (0.32D)(0.38) − 0.21C − 0.24F + 0.27M + 0.35S,
Fr = −0.11C + 0.12D − 0.21C − 0.24F + 0.27M + 0.35S,
Fr = (−0.29C)(0.38) + (0.32D)(0.38) − 0.21C − 0.24F + 0.27M + 0.35S,
Fr = −0.11C + 0.12D − 0.21C − 0.24F + 0.27M + 0.35S,
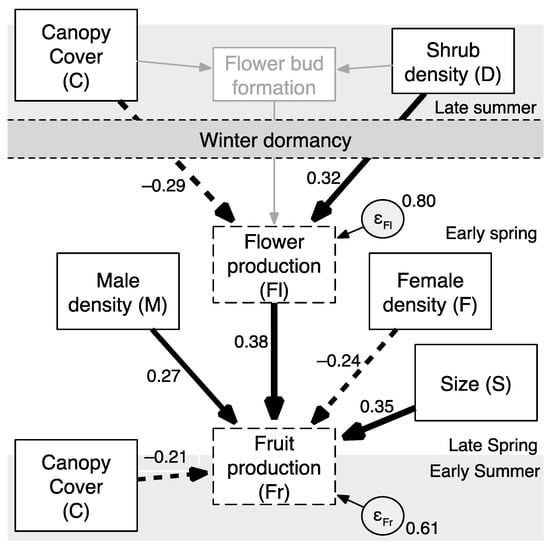
Figure 2.
Model describing the effect of light availability (% canopy cover), plant (male, female, and total) density, shrub size (m), and flower production (#/cm of branch) on total fruit production of Shepherdia canadensis in Central Alberta, Canada. Figures beside each path indicate direct effects reported as standardized coefficients. Dashed paths indicate negative coefficients. Indirect effects are not displayed (see Table 2). Dashed rectangles indicate endogenous variables. “Density (F)” and “Density (M)” represent density of female and male plants surrounding the focal female shrub, respectively. “Density (D)” indicates the aggregate (total) male and female density surrounding the focal female shrub. Grey paths and rectangle surrounding flower bud formation indicate theorized phenological paths [22], while all black arrows and rectangles indicate measured variables and estimated path weights.
3. Results
3.1. Flower and Fruit Production
Environmental, demographic, and reproductive variables are summarized in Table 1. The path model explained 20% of the variation in flower production among shrubs (R2 = 0.20) and 39% of the variation in fruit production (R2 = 0.39) (Figure 2; File S1). It had good overall fit (χ2 = 2.001, df = 4, P = 0.736), with a root mean squared error of approximation below 0.001 and a comparative fit index of 1.000 indicating support for the model structure.
As expected, larger shrubs produced more total fruit, and flower production (flowers per cm of branch length; flower density) was positively related to total fruit production (Table 2). Canopy cover was inversely related to flower and fruit production. The total effect of canopy cover on fruit production (βC(total)Fr) was composed of the direct effect of canopy cover on fruit production (66% of the total effect; βFrC/βC(total)Fr) and the indirect effect of canopy cover on fruit production through flower production (34% of the total effect; βFrFlC/βC(total)Fr). That is, canopy cover indirectly reduced fruit production by reducing flower production, and this indirect effect accounted for approximately one-third of the reduction in fruit production due to canopy cover.

Table 2.
Direct and indirect path relationships (hypotheses) and their coefficients (β) from a path model relating flower and fruit production for a population of Shepherdia canadensis in Edmonton, Alberta, Canada 1.
Total buffaloberry density did not directly affect fruit production (P = 0.824) but did directly affect flower production (βFlD); as density of the shrubs increased, flower production per unit of branch length increased demonstrating a positive density-dependent effect (Table 2). Total density and male density were not correlated (r < 0.3). Density had a positive indirect effect on fruit production through the effect of total density on flower production since density increased flower production and greater flower production was associated with higher fruit production (βFrFlD). The estimated standardized model represents a positive effect of overall intraspecific density, male density, and plant size, and a negative effect of canopy cover and female density. The derivation of Equation (4) from Equation (3) demonstrates how the coefficients in the model (Figure 2) are summarized as total effects.
Fr = 0.12D − 0.32C − 0.24F + 0.27M + 0.35S,
A second model with fruit density (fruit number per centimeter of branch) as the response variable was also estimated using the same set of explanatory variables (Appendix B; File S2; Table S4). All coefficients in this fruit density model were of the same direction and significance as in the model reported in Table 2 other than the relationship between size and the response variable (fruit density), which was non-significant (P = 0.707).
3.2. Pollination Environment and Supplementation
Pollen limitation was assessed through the effects of sex-differentiated density dependence of fruit production in 2012 and the pollen supplementation trial in 2014. In the 2012 study, density of males and females influenced fruit set (Table 2). Greater male (pollen donor) density surrounding focal female shrubs increased fruit production, while female (pollen competitor) density decreased fruit production. In the 2014 study, the linear mixed model with individual plant as a random factor had good overall fit (χ2 = 7.21, df = 1, P < 0.01) and the likelihood-ratio test comparing the model with a one-level ordinary linear regression was significant (χ2 = 9.13, df = 1, P < 0.01). The model demonstrated that hand pollination increased fruit set. Control flowers had a fruit set of 11.4%, while supplemental pollination more than doubled fruit set at 23.3% (Equation (5); n branches = 58, n shrubs = 15).
Fruit Set = 11.4 + 11.9 × Supplemented
4. Discussion
Canopy cover reduces fruit production in buffaloberry by reducing flower production and directly limiting the ability of individual shrubs to mature fruit. Male density in a 3.99 m radius surrounding female shrubs positively affected fruit production, while greater female density in this radius negatively affected fruit production. Increased pollen supply (male density) and decreased pollen competition (female density), therefore, increased fruit production among individual fruiting shrubs. Buffaloberry flower production also demonstrated a density-dependent effect—as total density of shrubs increased, flower production also increased. The pollen supplementation experiment demonstrated that buffaloberry fruit production was limited by pollen availability.
4.1. Plant Size
As is commonly demonstrated in buffaloberry and other plant species [7,37,58], we found shrub size increased total fruit production in buffaloberry. This may be a result of increased photosynthetic carbon gain due to higher leaf surface area with increasing shrub size [59]. Shrub size, however, did not influence fruit density (Appendix B), suggesting that larger plants did not attract pollinators as a result of greater patch reward size. This association in buffaloberry is in contrast to other flowering species where the reward patch size of individual plants or inflorescences increases seed and/or fruit set as a result of increased pollinator attraction [60,61]. The lack of relationship between plant size and fruit set in buffaloberry would be expected for plants pollinated by flies, which tend to visit few flowers per plant irrespective of plant size [39] including in the case of buffaloberry’s pollinators [40]. As we discuss below, a more significant factor for this species is the male and female density within a 50 m2 area surrounding female plants.
4.2. Positive Effect of Light Availability on Flower and Fruit Production
Light availability commonly limits plant fitness by influencing the rate of photosynthetic carbon gain [59,62] and the activity of pollinators [13,29]. Ultimately, however, life history determines whether increased light availability will affect fruit production [63,64]. Due to interspecific competition and particular niche adaptation, some dioecious shrubs occur more frequently in conditions of low to moderate light availability in forests [65]. In cases where low or moderate light availability increases plant abundance, the niche may include other conditions that reduce fitness of competing plants [66]. Likewise, biomass and photosynthetic rate may not simply increase with greater light, but rather peak at an intermediate irradiance level as a result of a specific niche adapted to low or moderate light availability [67]. Light availability can also interact with temperature, with some shrubs demonstrating significant differences in photosynthetic plasticity in response to light conditions at certain temperature ranges [68].
Despite the effects of niche and other interacting environmental conditions, in dioecious and other fruiting shrubs, light availability has been frequently linked to increased flower number [58,62] and increased fruit production [13,19,37,58,62]. Buffaloberry has shown a consistent association between light availability and flower and fruit production [8,17], demonstrating the centrality of light for fitness of the shrub. Similar to other actinorhizal plants that depend on light availability to drive nitrogen fixation [21,69], buffaloberry is shade-intolerant, and thus depends on forest disturbances [8,18,70]. Buffaloberry should not, however, simply be considered a pioneer species, as the dichotomous pioneer–climax framework is better conceptualized as a continuum [20,63]. The shrub does, however, display traits favoring disturbances that lead to canopy gaps, crown-replacing fires, and tree canopies with higher rates of understory irradiance [17,18], placing it more toward the pioneer side of life history responses to disturbance. Our results, demonstrating both reduced flower and fruit production as direct effects of higher canopy cover, support this classification.
Because of the effects of canopy cover on buffaloberry fruit production, selective thinning of overstory trees, or other prescribed disturbances, has been recommended for increasing fruit production in other dioecious shrubs [37]. For example, selective harvest of trees could be a management tool for improving brown bear habitat in northwestern North America where buffaloberry is a significant food source for bears [71,72,73]. Interactions between light availability and other elements of buffaloberry’s niche in different environments may further affect how buffaloberry fruit production responds to environmental conditions. Slope and aspect (direction that a slope faces) are secondary to light availability in affecting buffaloberry fruit production [18], but interactions between light availability and temperature, precipitation, and soil conditions should be considered further to improve site-level management recommendations.
4.3. Intraspecific Density and Flower Production
Increased intraspecific density often negatively influences fecundity, including in dioecious plants [63,74]. Our results indicate that increasing buffaloberry density, as noted above, is related to fruit production in sex-specific ways. Overall buffaloberry density, however, while not positively related to fruit production, did increase flower production. Our explanations of this pattern cannot be definitive given the study design, but our results suggest directions for future inquiry. Buffaloberry is an actinorhizal nitrogen-fixing shrub [21,75], forming associations with Frankia bacteria and mycorrhizae that facilitate nitrogen fixation in root nodules [76,77]. Since actinorhizal plants increase Frankia abundance in the soil [78,79,80], infectivity of the soil [81,82], and the prevalence of symbiotic mycorrhizae [77,83], often in host-specific ways [81,82], increasing density of buffaloberry may lead to more nodulation of new roots and thus more resources available for reproduction. That overall buffaloberry density would increase flower number but not fruit number could be related to the seasonal carbon budget of the shrub. In late summer, when buffaloberry sets flower primordia for the following year, the plant has completed fruiting and has been photosynthetically active for several months [22]. Since nitrogen fixation by Frankia in root nodules requires a steady supply of sucrose for the operation of nitrogenase [69,84], late summer may be when the plant has a greater ability to drive nitrogen fixation and this could explain why we see the effect on flower production. This study highlights the need to better understand how the carbon budget at different phenological stages interacts with soil conditions and actinorhizal plant density. Future research on buffaloberry and other fruiting actinorhizal species should examine how fruit production is related to Frankia abundance, infectivity, and nitrogen-fixing capacity.
4.4. Pollination Environment and Supplementation
Male density is not uniformly associated with greater fruit production in dioecious plants [85], although greater male density commonly results in higher pollination success and fruit production [32,33,34]. The most common pollinators of buffaloberry are generalist (primarily syrphid) flies that forage over short distances [7,40], consistent with our results and others demonstrating a positive association between male density and buffaloberry fruit production over a distance of 2–6 m from the focal plant [7,46]. Within the 3.99-m radius (50 m2 area) of our focal female plants, greater female density reduces fruit production, which may be the result of generalist pollinators distributing pollen among more female plants as female density increases. Pollination of buffaloberry, therefore, may not only be spatially restricted by pollen supply (as demonstrated by [7]), but also by pollen competition. Intraspecific pollen competition between females of other dioecious plants may also be expected, and further research on pollen limitation for individual dioecious and gynodioecious female plants could assess both pollen supply and pollen competition as a function of sex-biased density.
Inadequate pollination has been estimated to affect reproductive success in over half of angiosperms [31,86,87]. We found pollen supplementation led to a marked increase (doubling) in buffaloberry fruit set. As a dioecious species, buffaloberry is dependent on pollen transfer between individuals, and pollen limitation may be expected to be more common across dioecious species as a result of this obligate outcrossing [88]. The dependence upon insect pollination, combined with short foraging distances of generalist fly pollinators, can lead to expectations of higher pollen limitation since the plants must depend on pollen transfer from relatively stochastic and density-dependent pollen vectors [7]. Buffaloberry flowers soon after snowmelt in cooler temperatures and pollen limitation may also be expected in early-blooming and alpine plant species as a result of lower overall pollinator activity in cooler temperatures [10,39,89]. The association between cool temperatures and reduced pollinator activity has led to the cautionary observation that if climate change leads to variations in spring-flowering phenology in early-flowering species, plants may flower at times of restricted pollinator activity, leading to low fruit set and reduction of seedling recruitment [10,90].
5. Conclusions
While an open canopy may facilitate buffaloberry fruit production in a variety of conditions [18,70], this study demonstrates that overstory canopy cover and local buffaloberry shrub density are both consequential in determining fruit and flower production of the shrub. Owing to its actinorhizal nitrogen-fixing capacity and relative shade-intolerance—fruit production in buffaloberry has been shown to drop significantly when canopy cover is above 50% [8]—buffaloberry can be characterized as an early-successional species. As an early-flowering dioecious shrub, pollinated by generalist flies [7,40], buffaloberry is susceptible to pollen limitation as a result of conspecific sex-biased density dependence. Buffaloberry presence and fruit production, and brown bear feeding on buffaloberry fruit, has been associated with open forests such as immature evergreen stands and deciduous stands [8,17,18,70]. Our study lends support to recommendations of increasing the extent of young, regenerating forests with minimal scarification of soils [8] to increase the availability of buffaloberry fruit and animal species that depend on them.
Supplementary Materials
The following are available online at https://www.mdpi.com/1424-2818/12/1/37/s1. Dataset S1: Shepherdia canadensis data collected in 2012 (sheet 1), 2014 (sheet 2), and the 2012 data formatted for analysis (sheet 3). File S1: Stata structural equation (sem) model file for Shepherdia canadensis total fruit production. File S2: Stata structural equation (sem) model file for Shepherdia canadensis fruit density. Table S1: Summary of transformations performed on variables in the analysis done to conform to normality assumptions in the path models. Table S2: Variable transformations for each parameter assessed in the path models for Shepherdia canadensis fruit production showing tests conducted for each variable and the corresponding Shapiro–Francia W’, Royston V’, z, and p value. Table S3: Meaning of normality statistics. Table S4: Direct and indirect path relationships (hypotheses) and their coefficients (β) from a path model relating flower and fruit per cm of branch for a population of Shepherdia canadensis in Edmonton, Alberta, Canada.
Author Contributions
Conceptualization, T.J.B. and S.E.N.; data curation, T.J.B.; formal analysis, T.J.B. and S.E.N.; funding acquisition, S.E.N.; investigation, T.J.B. and S.E.N.; methodology, T.J.B. and S.E.N.; project administration, T.J.B. and S.E.N.; resources, S.E.N.; software, T.J.B.; validation, T.J.B. and S.E.N.; visualization, T.J.B. and S.E.N.; writing—original draft preparation, T.J.B.; writing—review and editing, T.J.B. and S.E.N. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by a Natural Sciences and Engineering Research Council of Canada Discovery Grant, #RGPIN-2014-04842.
Acknowledgments
The authors acknowledge R. Touchie, N. Stewart, K. Johnson, and A. Braid for field assistance.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Appendix A
Variable Normality
The sem function in Stata requires adherence to normality assumptions [57]. Fitting these assumptions was desirable due to the results available in sem, but not in gsem [57]. Royston derived V’ from the Shapiro–Francia W’ statistic [91], which enables the quantification of normality [91,92,93,94,95]. V’ was used as our measure of normality. Large values of V’ indicate non-normality; smaller values indicate greater normality (see Tables S1–S3). As the model used transformed variable coefficients, we reported standardized results.
Appendix B
The results of an additional path model using fruit density (fruit per cm of branch) as the response variable are included in the supplementary material (Table S4). In this model, shrub size did not affect fruit density. The path coefficients for the endogenous flower production variable in this model are identical to those included in the main body of the paper (see Table 2). For the endogenous fruit density variable, canopy cover was inversely related to fruit density. Male buffaloberry density was positively related to fruit density, while female buffaloberry density was inversely related to fruit density.
The path model explained 20% of the variation in flower production among shrubs (R2 = 0.20) and 46% of the variation in fruit production (R2 = 0.46). It had moderate overall fit (χ2 = 4.937, df = 4, P = 0.294), with a root mean squared error of approximation of 0.062 and a comparative fit index of 0.979 further indicating moderate fit. Lower model fit was primarily due to the inclusion of the plant size variable, which was non-significant (P = 0.707), but we include plant size in this model because the lack of a relationship between size and fruit density has implications for the relationship between reward patch size and pollinator attraction (see Section 4.1 of the discussion).
References
- Bartels, S.F.; Chen, H.Y.H. Is understory plant species diversity driven by resource quantity or resource heterogeneity? Ecology 2010, 91, 1931–1938. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Chen, H.Y.H.; Thomas, S.C.; Shahi, C. Linking resource availability and heterogeneity to understorey species diversity through succession in boreal forest of Canada. J. Ecol. 2018, 106, 1266–1276. [Google Scholar] [CrossRef]
- USDA. Shepherdia Canadensis (L.) Nutt.; United States Department of Agriculture Natural Resources Conservation Service: Washington, DC, USA, 2019.
- Walkup, C.J. Shepherdia canadensis. In Fire Effects Information System; United Sates Department of Agriculture, Forest Service, Fire Sciences Laboratory: Washington, DC, USA, 1991. [Google Scholar]
- Denny, C.K.; Stenhouse, G.B.; Nielsen, S.E. Scales of selection and perception: Landscape heterogeneity of an important food resource influences habitat use by a large omnivore. Wildl. Biol. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Krebs, C.J.; Cowcill, K.; Boonstra, R.; Kenney, A.J. Do changes in berry crops drive population fluctuations in small rodents in the southwestern Yukon? J. Mammal. 2010, 91, 500–509. [Google Scholar] [CrossRef]
- Lin, S.-Y.; Nol, E.; Dorken, M.E. Spatial dynamics of pollination in dioecious Shepherdia canadensis (Elaeagnaceae). Plant Ecol. 2015, 216, 1213–1223. [Google Scholar] [CrossRef]
- Nielsen, S.E.; Munro, R.H.M.; Bainbridge, E.L.; Stenhouse, G.B.; Boyce, M.S. Grizzly bears and forestry II: Distribution of grizzly bear foods in clearcuts of west-central Alberta, Canada. For. Ecol. Manag. 2004, 199, 67–82. [Google Scholar] [CrossRef]
- Ashman, T.-L.; Knight, T.M.; Steets, J.A.; Amarasekare, P.; Burd, M.; Campbell, D.R.; Dudash, M.R.; Johnston, M.O.; Mazer, S.J.; Mitchell, R.J.; et al. Pollen limitation of plant reproduction: Ecological and evolutionary causes and consequences. Ecology 2004, 85, 2408–2421. [Google Scholar] [CrossRef]
- Kudo, G.; Ida, T.Y.; Tani, T. Linkages between phenology, pollination, photosynthesis, and reproduction in deciduous forest understory plants. Ecology 2008, 89, 321–332. [Google Scholar] [CrossRef]
- Stephenson, A.G. Flower and fruit abortion: Proximate causes and ultimate functions. Annu. Rev. Ecol. Syst. 1981, 12, 253–279. [Google Scholar] [CrossRef]
- Alaback, P.B. Dynamics of understory biomass in sitka spruce-western hemlock forests of southeast Alaska. Ecology 1982, 63, 1932–1948. [Google Scholar] [CrossRef]
- Eckerter, T.; Buse, J.; Förschler, M.; Pufal, G. Additive positive effects of canopy openness on European bilberry (Vaccinium myrtillus) fruit quantity and quality. For. Ecol. Manag. 2019, 433, 122–130. [Google Scholar] [CrossRef]
- Barbier, S.; Gosselin, F.; Balandier, P. Influence of tree species on understory vegetation diversity and mechanisms involved—A critical review for temperate and boreal forests. For. Ecol. Manag. 2008, 254, 1–15. [Google Scholar] [CrossRef]
- Kumar, P.; Chen, H.Y.H.; Searle, E.B.; Shahi, C. Dynamics of understorey biomass, production and turnover associated with long-term overstorey succession in boreal forest of Canada. For. Ecol. Manag. 2018, 427, 152–161. [Google Scholar] [CrossRef]
- Burnham, K.M.; Lee, T.D. Canopy gaps facilitate establishment, growth, and reproduction of invasive Frangula alnus in a Tsuga canadensis dominated forest. Biol. Invasions 2010, 12, 1509–1520. [Google Scholar] [CrossRef]
- Denny, C.; Nielsen, S. Spatial heterogeneity of the forest canopy scales with the heterogeneity of an understory shrub based on fractal analysis. Forests 2017, 8, 146. [Google Scholar] [CrossRef]
- Hamer, D. Buffaloberry [Shepherdia canadensis (L.) Nutt.] fruit production in fire-successional bear feeding sites. J. Range Manag. 1996, 49, 520–529. [Google Scholar] [CrossRef]
- Antos, J.A.; Allen, G.A. Patterns of reproductive effort in male and female shrubs of Oemleria cerasiformis: A 6-year study. J. Ecol. 1999, 87, 77–84. [Google Scholar] [CrossRef]
- Abe, S.; Motai, H.; Tanaka, H.; Shibata, M.; Kominami, Y.; Nakashizuka, T. Population maintenance of the short-lived shrub Sambucus in a deciduous forest. Ecology 2008, 89, 1155–1167. [Google Scholar] [CrossRef]
- Wheeler, C.T.; Akkermans, D.L.; Berry, A.M. Frankia and actinorhizal plants: A historical perspective. In Nitrogen-Fixing Actinorhizal Symbioses; Pawlowski, K., Newton, W.E., Eds.; Nitrogen Fixation: Origins, Applications, and Research Progress; Springer: Dordrecht, The Netherlands, 2008; Volume 6, pp. 1–24. [Google Scholar]
- Hayes, P.A.; Steeves, T.A.; Neal, B.R. An architectural analysis of Shepherdia canadensis and Shepherdia argentea: Patterns of shoot development. Can. J. Bot. 1989, 67, 1870–1877. [Google Scholar] [CrossRef]
- Krebs, C.J.; Boonstra, R.; Cowcill, K.; Kenney, A.J. Climatic determinants of berry crops in the boreal forest of the southwestern Yukon. Botany 2009, 87, 401–408. [Google Scholar] [CrossRef]
- Thilenius, F.J.; Evans, K.E.; Garrett, C.E. Shepherdia, buffaloberry. In Seeds of Woody Plants in the United States; Schopmeyer, C.S., Ed.; U.S. Forest Service, Department of Agriculture: Washington, DC, USA, 1974; pp. 771–773. [Google Scholar]
- Bañuelos, M.-J.; Obeso, J.-R. How is fruit production regulated in the dioecious fleshy-fruited shrub Rhamnus alpinus? Basic Appl. Ecol. 2005, 6, 249–259. [Google Scholar] [CrossRef]
- Timmerman-Erskine, M.; Boyd, R.S. Reproductive biology of the endangered plant Clematis socialis (Ranunculaceae). J. Torrey Bot. Soc. 1999, 126, 107–116. [Google Scholar] [CrossRef]
- Barkman, J.J. Canopies and microclimate of tree species mixtures. In The Ecology of Mixed Species Stands of Trees; Blackwell Scientific Publications: Oxford, UK, 1992; pp. 181–188. [Google Scholar]
- Sharpe, F. The biologically significant attributes of forest canopies to small birds. Northwest Sci. 1996, 70, 86–93. [Google Scholar]
- Boulanger-Lapointe, N.; Järvinen, A.; Partanen, R.; Herrmann, T.M. Climate and herbivore influence on Vaccinium myrtillus over the last 40 years in northwest Lapland, Finland. Ecosphere 2017, 8, e01654. [Google Scholar] [CrossRef]
- Westerbergh, A.; Saura, A. Gene flow and pollinator behaviour in Silene dioica populations. Oikos 1994, 71, 215. [Google Scholar] [CrossRef]
- Knight, T.M.; Steets, J.A.; Vamosi, J.C.; Mazer, S.J.; Burd, M.; Campbell, D.R.; Dudash, M.R.; Johnston, M.O.; Mitchell, R.J.; Ashman, T.-L. Pollen limitation of plant reproduction: Pattern and process. Annu. Rev. Ecol. Evol. Syst. 2005, 36, 467–497. [Google Scholar] [CrossRef]
- Carlsson-Graner, U.; Elmqvist, T.; Agren, J.; Gardfjell, H.; Ingvarsson, P. Floral sex ratios, disease and seed set in dioecious Silene dioica. J. Ecol. 1998, 86, 79–91. [Google Scholar] [CrossRef]
- House, S.M. Pollination success in a population of dioecious rain forest trees. Oecologia 1993, 96, 555–561. [Google Scholar] [CrossRef]
- Steven, J.C.; Waller, D.M. Isolation affects reproductive success in low-density but not high-density populations of two wind-pollinated Thalictrum species. Plant Ecol. 2007, 190, 131–141. [Google Scholar] [CrossRef]
- Hargreaves, A.L.; Harder, L.D.; Johnson, S.D. Consumptive emasculation: The ecological and evolutionary consequences of pollen theft. Biol. Rev. 2009, 84, 259–276. [Google Scholar] [CrossRef]
- Peeters, L.; Totland, Ø. Wind to insect pollination ratios and floral traits in five alpine Salix species. Can. J. Bot. 1999, 77, 556–563. [Google Scholar]
- Schulz, K.; Zasada, J.; Nauertz, E. Annual, local, and individual variation in the inflorescence and fruit production of eastern leatherwood (Dirca palustris L. Thymelaeaceae). J. Torrey Bot. Soc. 2004, 131, 292–304. [Google Scholar] [CrossRef]
- Bierzychudek, P. Pollinators increase the cost of sex by avoiding female flowers. Ecology 1987, 68, 444–447. [Google Scholar] [CrossRef]
- Willmer, P. Pollination and Floral Ecology; Princeton University Press: Princeton, NJ, USA; Woodstock, UK, 2011. [Google Scholar]
- Borkent, C.J.; Harder, L.D. Flies (Diptera) as pollinators of two dioecious plants: Behaviour and implications for plant mating. Can. Entomol. 2007, 139, 235–246. [Google Scholar] [CrossRef]
- Kevan, P.G. Insect pollination of high arctic flowers. J. Ecol. 1972, 60, 831–847. [Google Scholar] [CrossRef]
- Ashman, T.-L.; Stanton, M. Seasonal variation in pollination dynamics of sexually dimorphic Sidalcea oregana ssp. spicata (Malvaceae). Ecology 1991, 72, 993–1003. [Google Scholar] [CrossRef]
- Charlesworth, D. Why are unisexual flowers associated with wind pollination and unspecialized pollinators? Am. Nat. 1993, 141, 481–490. [Google Scholar] [CrossRef]
- Eckhart, V.M. The effects of floral display on pollinator visitation vary among populations of Phacelia linearis (Hydrophyllaceae). Evol. Ecol. 1991, 5, 370–384. [Google Scholar] [CrossRef]
- Renner, S.S.; Ricklefs, R.E. Dioecy and its correlates in the flowering plants. Am. J. Bot. 1995, 82, 596–606. [Google Scholar] [CrossRef]
- Johnson, K.M.; Nielsen, S.E. Demographic effects on fruit set in the dioecious shrub Canada buffaloberry (Shepherdia canadensis). PeerJ 2014, 2, e526. [Google Scholar] [CrossRef]
- Ghazoul, J. Floral diversity and the facilitation of pollination. J. Ecol. 2006, 94, 295–304. [Google Scholar] [CrossRef]
- Haig, D.; Westoby, M. On limits to seed production. Am. Nat. 1988, 131, 757–759. [Google Scholar] [CrossRef]
- Knight, T.M.; Steets, J.A.; Ashman, T.-L. A quantitative synthesis of pollen supplementation experiments highlights the contribution of resource reallocation to estimates of pollen limitation. Am. J. Bot. 2006, 93, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Government of Canada. Canadian Climate Normals 1981–2010 Station Data; Government of Canada: Ottawa, ON, Canada, 2014.
- City of Edmonton. Terwillegar Park Concept Plan Study: Opportunities and Constraints Analysis; City of Edmonton: Edmonton, AB, Canada, 2007. [Google Scholar]
- Alberta Environmental Protection. The Parkland Natural Region of Alberta; Government of Alberta: Edmonton, AB, Canada, 1997.
- Downing, D.J.; Pettapiece, W.W. Natural Regions and Subregions of Alberta; Government of Alberta, Natural Regions Committee: Edmonton, AB, Canada, 2006.
- Fiala, A.C.S.; Garman, S.L.; Gray, A.N. Comparison of five canopy cover estimation techniques in the western Oregon Cascades. For. Ecol. Manag. 2006, 232, 188–197. [Google Scholar] [CrossRef]
- Lemmon, P.E. A spherical densiometer for estimating forest overstory density. For. Sci. 1956, 2, 314–320. [Google Scholar]
- StataCorp. Stata Multilevel Mixed-Effects Reference Manual (Release 13); Stata Press: College Station, TX, USA, 2013. [Google Scholar]
- StataCorp. Structural Equation Modeling Reference Manual (Release 13); Stata Press: College Station, TX, USA, 2013. [Google Scholar]
- Osunkoya, O.O. Population structure and breeding biology in relation to conservation in the dioecious Gardenia actinocarpa (Rubiaceae): A rare shrub of North Queensland rainforest. Biol. Conserv. 1999, 88, 347–359. [Google Scholar] [CrossRef]
- Kozlowski, T.T.; Pallardy, S.G. Growth Control in Woody Plants; Academic Press: Toronto, ON, USA, 1997; ISBN 978-0-12-424210-4. [Google Scholar]
- Glaettli, M.; Barrett, S.C.H. Pollinator responses to variation in floral display and flower size in dioecious Sagittaria latifolia (Alismataceae). New Phytol. 2008, 179, 1193–1201. [Google Scholar]
- Matsuhisa, S.; Ushimaru, A. Sexual dimorphism in floral longevity and flowering synchrony in relation to pollination and mating success in three dioecious Ilex species. Am. J. Bot. 2015, 102, 1187–1197. [Google Scholar] [CrossRef]
- Niesenbaum, R.A. Light or pollen: Seasonal limitations on female reproductive success in the understory shrub Lindera benzoin. J. Ecol. 1993, 81, 315–323. [Google Scholar] [CrossRef]
- Alvarez-Buylla, E.R.; Martinez-Ramos, M. Demography and allometry of Cecropia obtusifolia, a neotropical pioneer tree: An evaluation of the climax-pioneer paradigm for tropical rain forests. J. Ecol. 1992, 80, 275–290. [Google Scholar] [CrossRef]
- Amadeu, L.S.N.; Sampaio, M.B.; dos Santos, F.A.M. Influence of light and plant size on the reproduction and growth of small palm tree species: Comparing two methods for measuring canopy openness. Am. J. Bot. 2016, 103, 1678–1686. [Google Scholar] [CrossRef] [PubMed]
- Paudel, S.; Battaglia, L.L. The role of light, soil and human factors on the probability of occurrence of an invasive and three native plant species in coastal transitions of coastal Mississippi, USA. J. Plant Ecol. 2015, 8, 491–500. [Google Scholar] [CrossRef]
- Sultan, S.E.; Wilczek, A.M.; Hann, S.D.; Brosi, B.J. Contrasting ecological breadth of co-occurring annual Polygonum species. J. Ecol. 1998, 86, 363–383. [Google Scholar] [CrossRef]
- Hudson, J.E.; Levia, D.F.; Hudson, S.A.; Bais, H.P.; Legates, D.R. Phenoseasonal subcanopy light dynamics and the effects of light on the physiological ecology of a common understory shrub, Lindera benzoin. PLoS ONE 2017, 12, e0185894. [Google Scholar] [CrossRef] [PubMed]
- De la Bandera, M.d.C.; Traveset, A.; Valladares, F.; Gulías, J. Gender, season and habitat: Patterns of variation in photosynthetic activity, growth and fecundity in Thymelaea velutina. Acta Oecol. Montrouge 2008, 34, 294–302. [Google Scholar] [CrossRef][Green Version]
- Valdéz, M. Frankia ecology. In Nitrogen-Fixing Actinorhizal Symbioses; Pawlowski, K., Newton, W.E., Eds.; Springer: Dordrecht, The Netherlands, 2008. [Google Scholar]
- Hamer, D.; Herrero, S. Wildfire’s influence on grizzly bear feeding ecology in Banff National Park, Alberta. Int. Conf. Bear Res. Manag. 1987, 7, 179–186. [Google Scholar] [CrossRef]
- McLellan, B.N. Implications of a high-energy and low-protein diet on the body composition, fitness, and competitive abilities of black (Ursus americanus) and grizzly (Ursus arctos) bears. Can. J. Zool. 2011, 89, 546–558. [Google Scholar] [CrossRef]
- McLellan, B.N.; Hovey, F.W. The diet of grizzly bears in the Flathead River drainage of southeastern British Columbia. Can. J. Zool. 1995, 73, 704–712. [Google Scholar] [CrossRef]
- Munro, R.H.M.; Nielsen, S.E.; Price, M.H.; Stenhouse, G.B.; Boyce, M.S. Seasonal and diel patterns of grizzly bear diet and activity in west-central Alberta. J. Mammal. 2006, 87, 1112–1121. [Google Scholar] [CrossRef]
- Antonovics, J.; Levin, D.A. The ecological and genetic consequences of density-dependent regulation in plants. Annu. Rev. Ecol. Syst. 1980, 11, 411–452. [Google Scholar] [CrossRef]
- Huguet, V.; Batzli, J.M.; Zimpfer, J.F.; Gourbière, F.; Dawson, J.O.; Fernandez, M.P. Nodular symbionts of Shepherdia, Alnus, and Myrica from a sand dune ecosystem: Trends in occurrence of soilborne Frankia genotypes. Can. J. Bot. 2004, 82, 691–699. [Google Scholar] [CrossRef]
- Batzli, J.M.; Zimpfer, J.F.; Huguet, V.; Smyth, C.A.; Fernandez, M.; Dawson, J.O. Distribution and abundance of infective, soilborne Frankia and host symbionts Shepherdia, Alnus, and Myrica in a sand dune ecosystem. Can. J. Bot. 2004, 82, 700–709. [Google Scholar] [CrossRef]
- Visser, S.; Danielson, R.M.; Parkinson, D. Field performance of Elaeagnus commutata and Shepherdia canadensis (Elaeagnaceae) inoculated with soil containing Frankia and vesicular–arbuscular mycorrhizal fungi. Can. J. Bot. 1991, 69, 1321–1328. [Google Scholar] [CrossRef]
- Burleigh, S.H.; Dawson, J.O. Occurrence of Myrica-nodulating Frankia in Hawaiian volcanic soils. Plant Soil. 1994, 164, 283–289. [Google Scholar] [CrossRef]
- Huss-Danell, K. Actinorhizal symbioses and their N2 fixation. New Phytol. 1997, 136, 375–405. [Google Scholar] [CrossRef]
- Zitzer, S.F.; Dawson, J.O. Soil properties and actinorhizal vegetation influence nodulation of Alnus glutinosa and Elaeagnus angustifolia by Frankia. Plant Soil. 1992, 140, 197–204. [Google Scholar] [CrossRef]
- Nickel, A.; Pelz, O.; Hahn, D.; Saurer, M.; Siegwolf, R.; Zeyer, J. Effect of inoculation and leaf litter amendment on establishment of nodule-forming Frankia populations in soil. Appl. Environ. Microbiol. 2001, 67, 2603–2609. [Google Scholar] [CrossRef]
- Zimpfer, J.F.; Kennedy, G.J.; Smyth, C.A.; Hamelin, J.; Navarro, E.; Dawson, J.O. Localization of Casuarina -infective Frankia near Casuarina cunninghamiana trees in Jamaica. Can. J. Bot. 1999, 77, 1248–1256. [Google Scholar]
- Barea, J.M.; Azcon-Aguilar, C. Mycorrhizas and their significance in nodulating nitrogen-fixing plants. Adv. Agron. 1983, 36, 1–54. [Google Scholar]
- McClure, P.R.; Coker, G.T.; Schubert, K.R. Carbon dioxide fixation in roots and nodules of Alnus glutinosa: I: Role of phosphoenolpyruvate carboxylase and carbamyl phosphate synthetase in dark CO2 fixation, citrulline synthesis, and N2 fixation. Plant Physiol. 1983, 71, 652–657. [Google Scholar] [CrossRef]
- House, S.M. Population density and fruit set in three dioecious tree species in Australian tropical rain forest. J. Ecol. 1992, 80, 57–69. [Google Scholar] [CrossRef]
- Dauber, J.; Biesmeijer, J.C.; Gabriel, D.; Kunin, W.E.; Lamborn, E.; Meyer, B.; Nielsen, A.; Potts, S.G.; Roberts, S.P.M.; Sõber, V.; et al. Effects of patch size and density on flower visitation and seed set of wild plants: A pan-European approach. J. Ecol. 2010, 98, 188–196. [Google Scholar] [CrossRef]
- Labouche, A.-M.; Richards, S.A.; Pannell, J.R. Effects of pollination intensity on offspring number and quality in a wind-pollinated herb. J. Ecol. 2017, 105, 197–208. [Google Scholar] [CrossRef]
- Crossman, A.; Charlesworth, D. Breakdown of dioecy: Models where males acquire cosexual functions. Evolution 2014, 68, 426–440. [Google Scholar] [CrossRef]
- McCabe, L.M.; Colella, E.; Chesshire, P.; Smith, D.; Cobb, N.S. The transition from bee-to-fly dominated communities with increasing elevation and greater forest canopy cover. PLoS ONE 2019, 14, e0217198. [Google Scholar] [CrossRef]
- Miller-Rushing, A.J.; Primack, R.B. Global warming and flowering times in Thoreau’s concord: A community perspective. Ecology 2008, 89, 332–341. [Google Scholar] [CrossRef]
- Shapiro, S.S.; Francia, R.S. An approximate analysis of variance test for normality. J. Am. Stat. Assoc. 1972, 67, 215–216. [Google Scholar] [CrossRef]
- Gould, W.; Rogers, W. sg3.4: Summary of tests of normality. Stata Tech. Bull. 1991, 1, 20–23. [Google Scholar]
- Gould, W. sg11.1: Quantile regression with bootstrapped standard errors. Stata Tech. Bull. 1992, 2, 19–21. [Google Scholar]
- StataCorp Swilk—Shapiro–Wilk and Shapiro–Francia Tests for Normality. Available online: http://www.stata.com/manuals13/rswilk.pdf (accessed on 17 January 2020).
- Royston, P. Estimating departure from normality. Stat. Med. 1991, 10, 1283–1293. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).