Negative Effects of Diurnal Changes in Acidification and Hypoxia on Early-Life Stage Estuarine Fishes
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Seven Treatment Experiments
2.3. Four Treatment Experiments
2.4. Physical and Chemical Water Properties
2.5. Experimental Organisms
2.6. Data & Statistical Analyses
3. Results
3.1. Seven Treatment Diurnal Acidification and Hypoxia Experiments
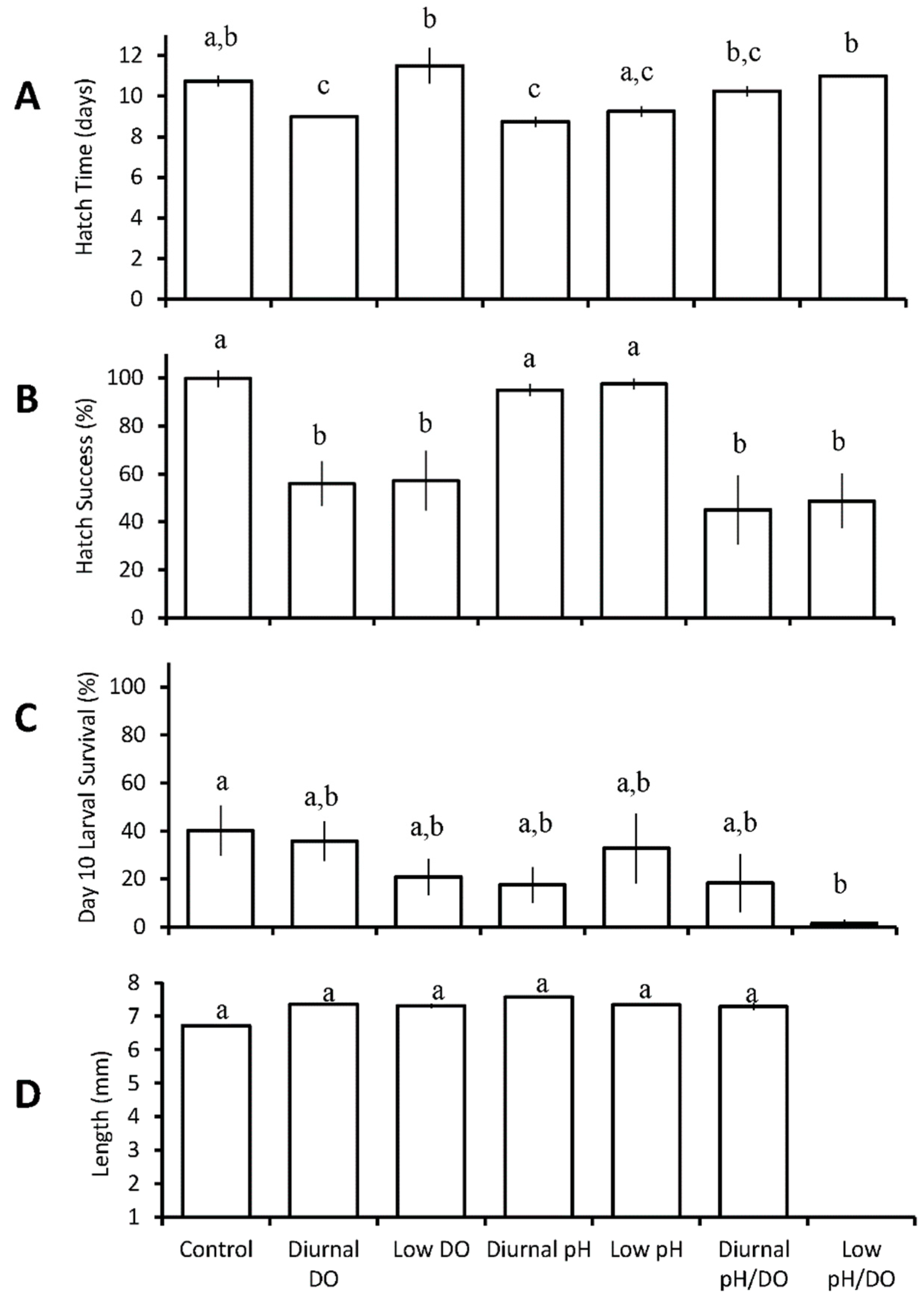
3.1.1. Menidia menidia
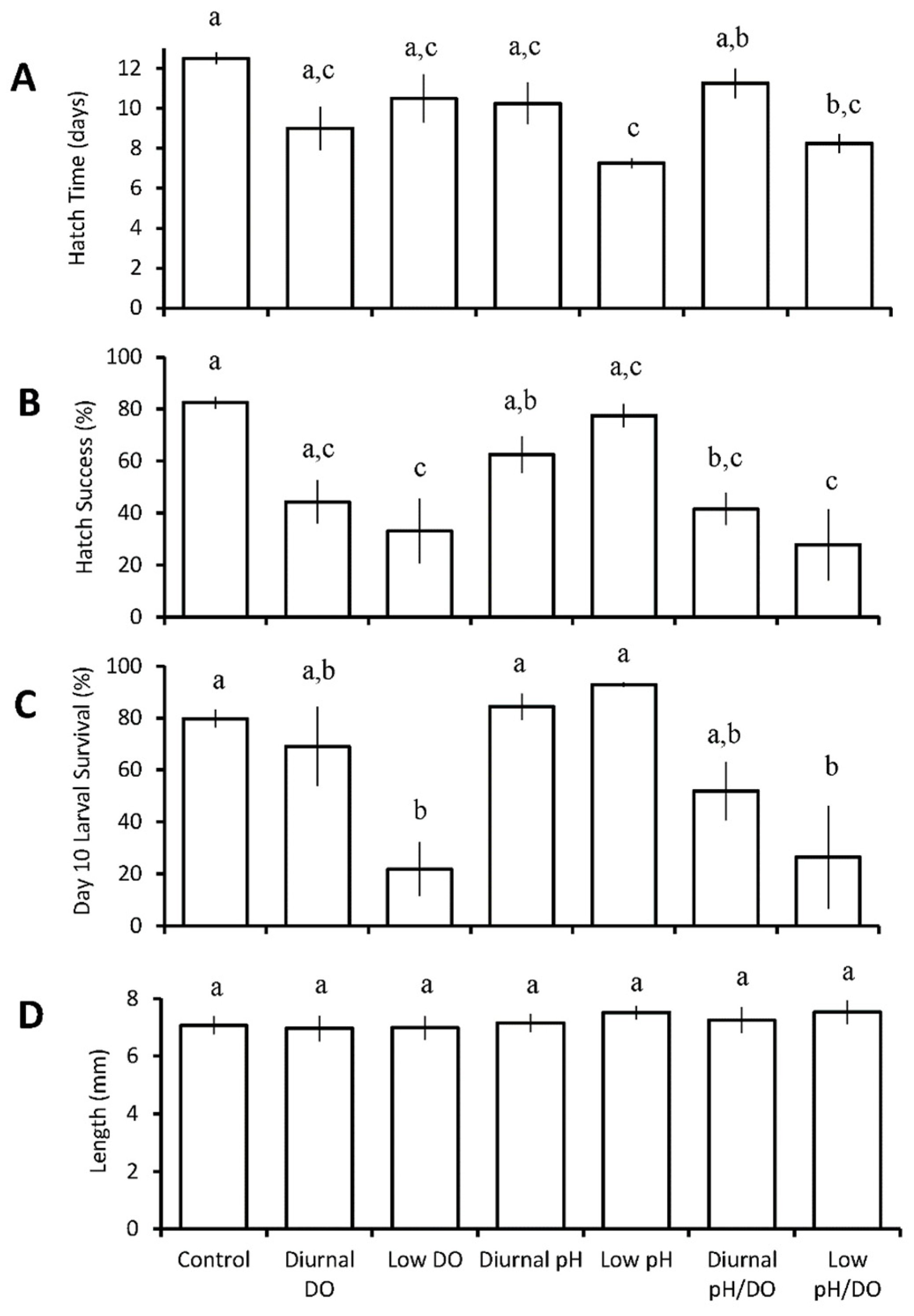
3.1.2. Cyprinodon variegatus
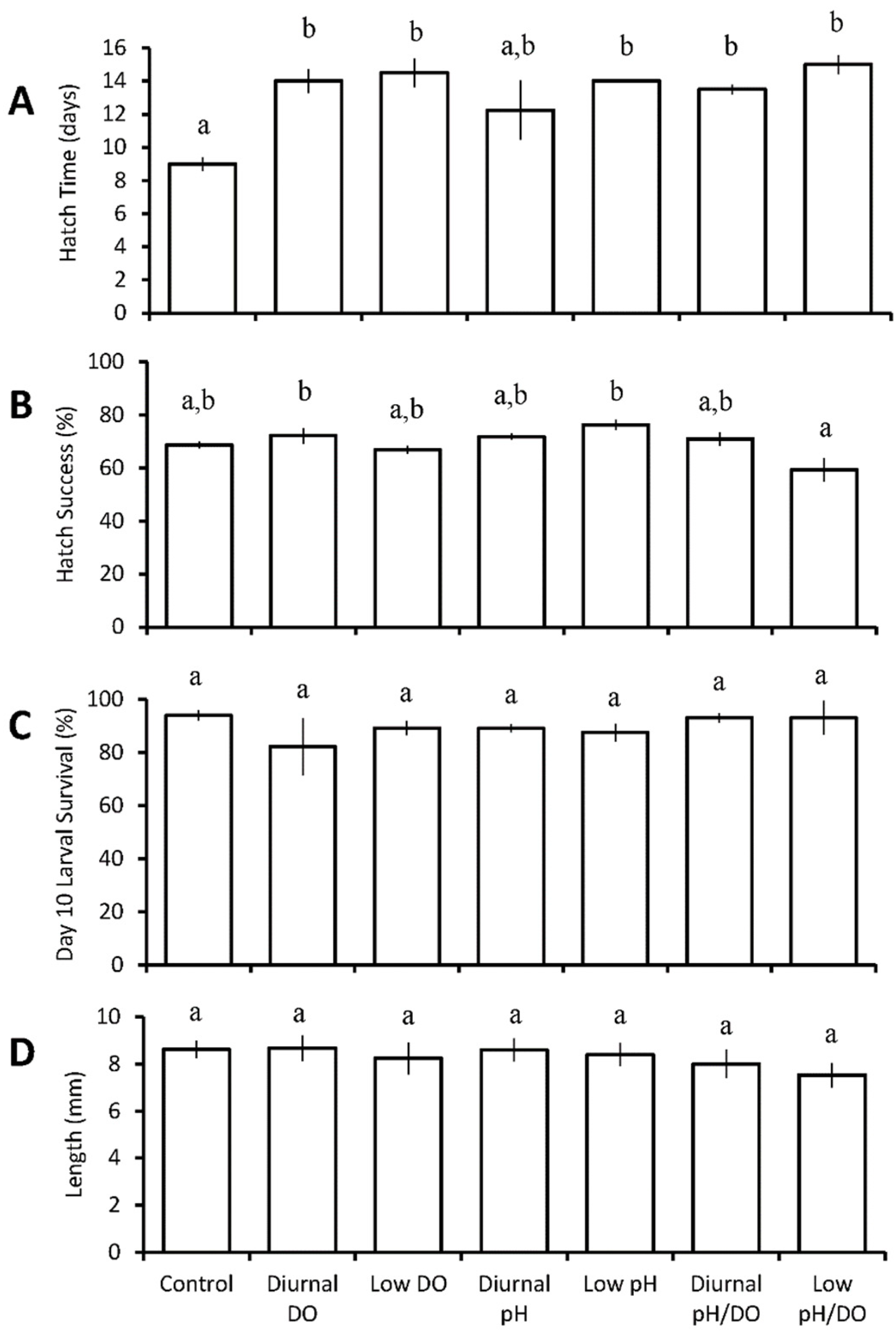
3.1.3. Menidia beryllina
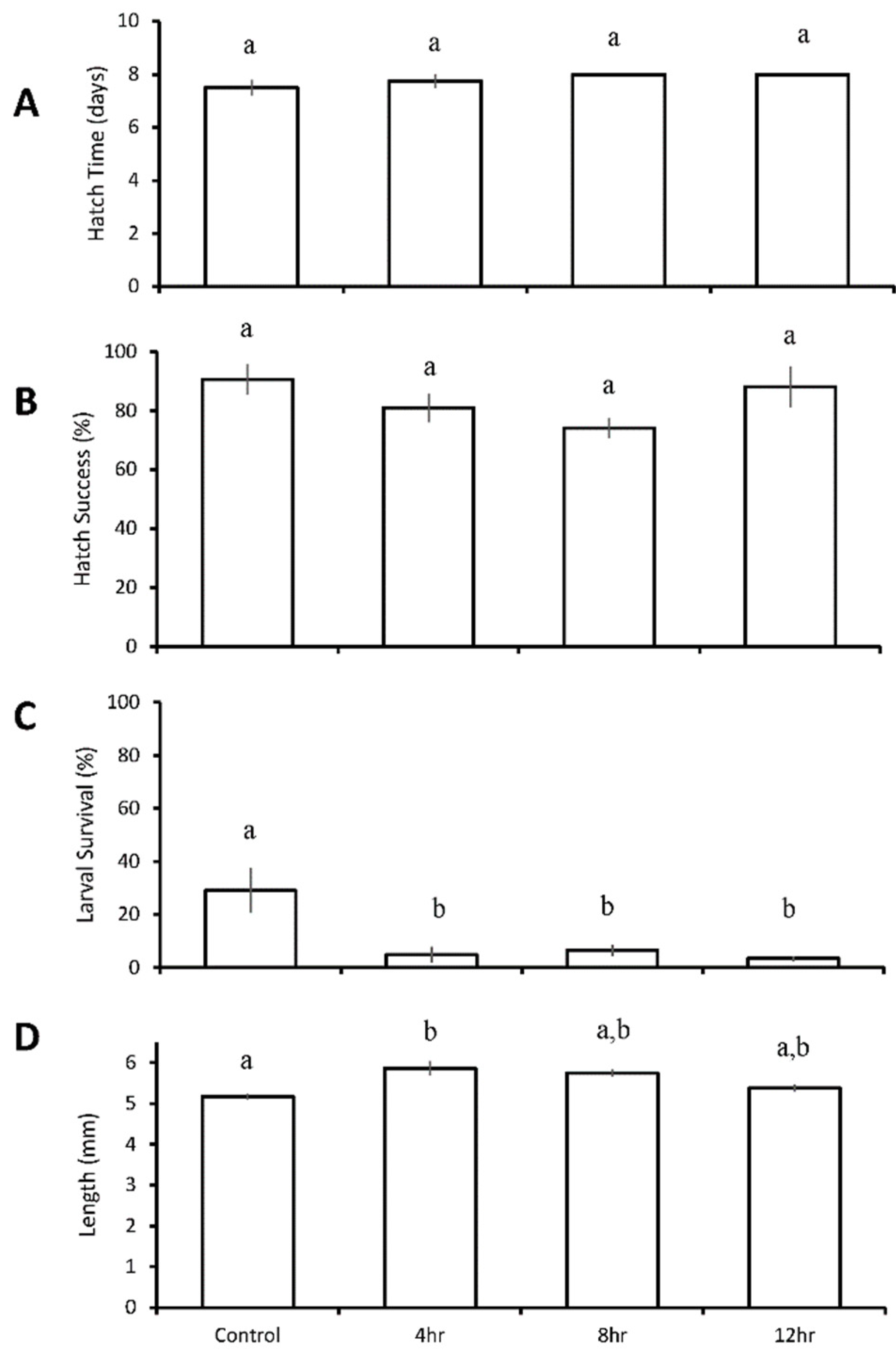
3.2. Four Treatment Diurnal Acidification and Hypoxia Experiments
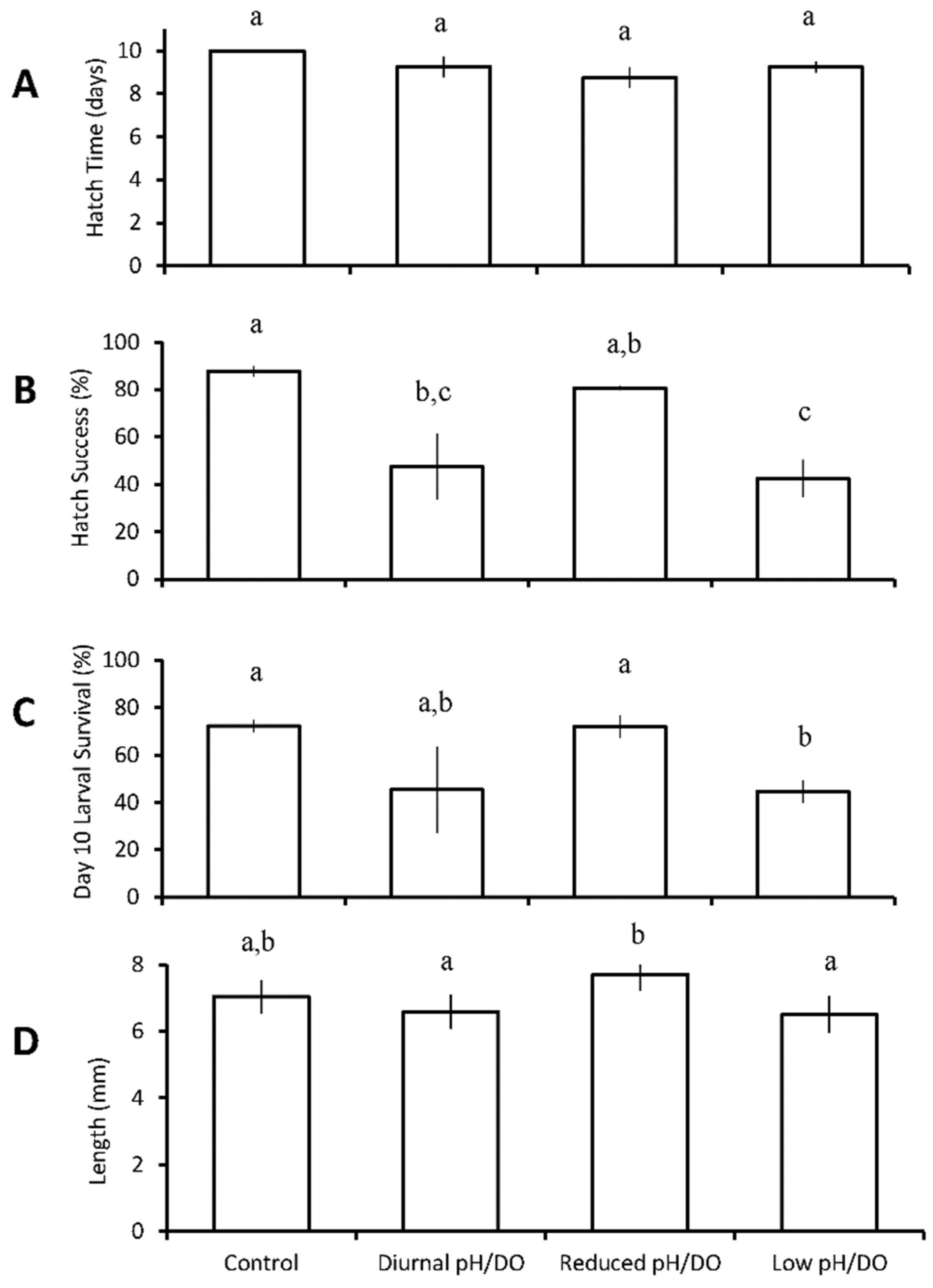
Menidia beryllina
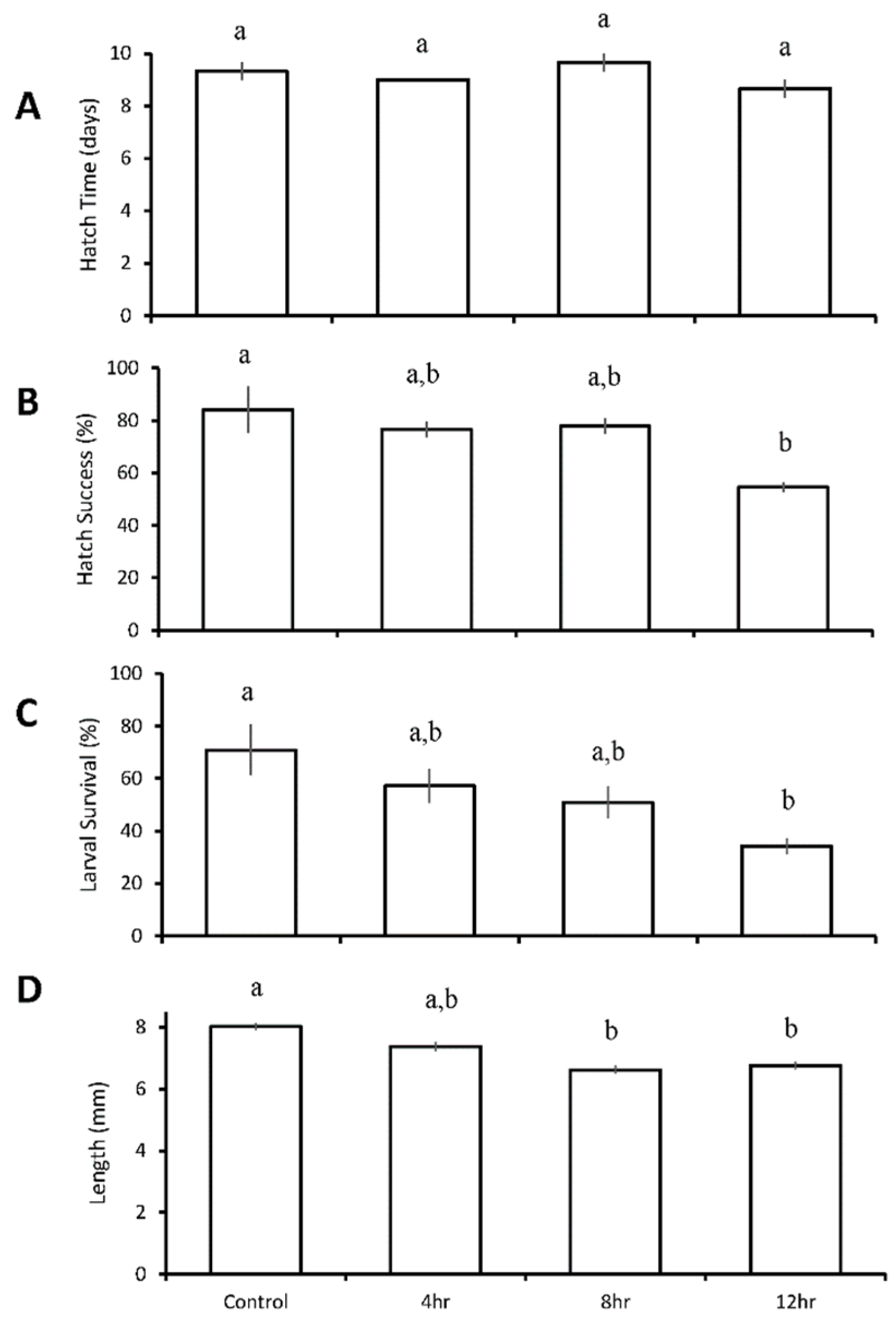
3.3. Diurnal Duration Experiments with Menidia beryllina
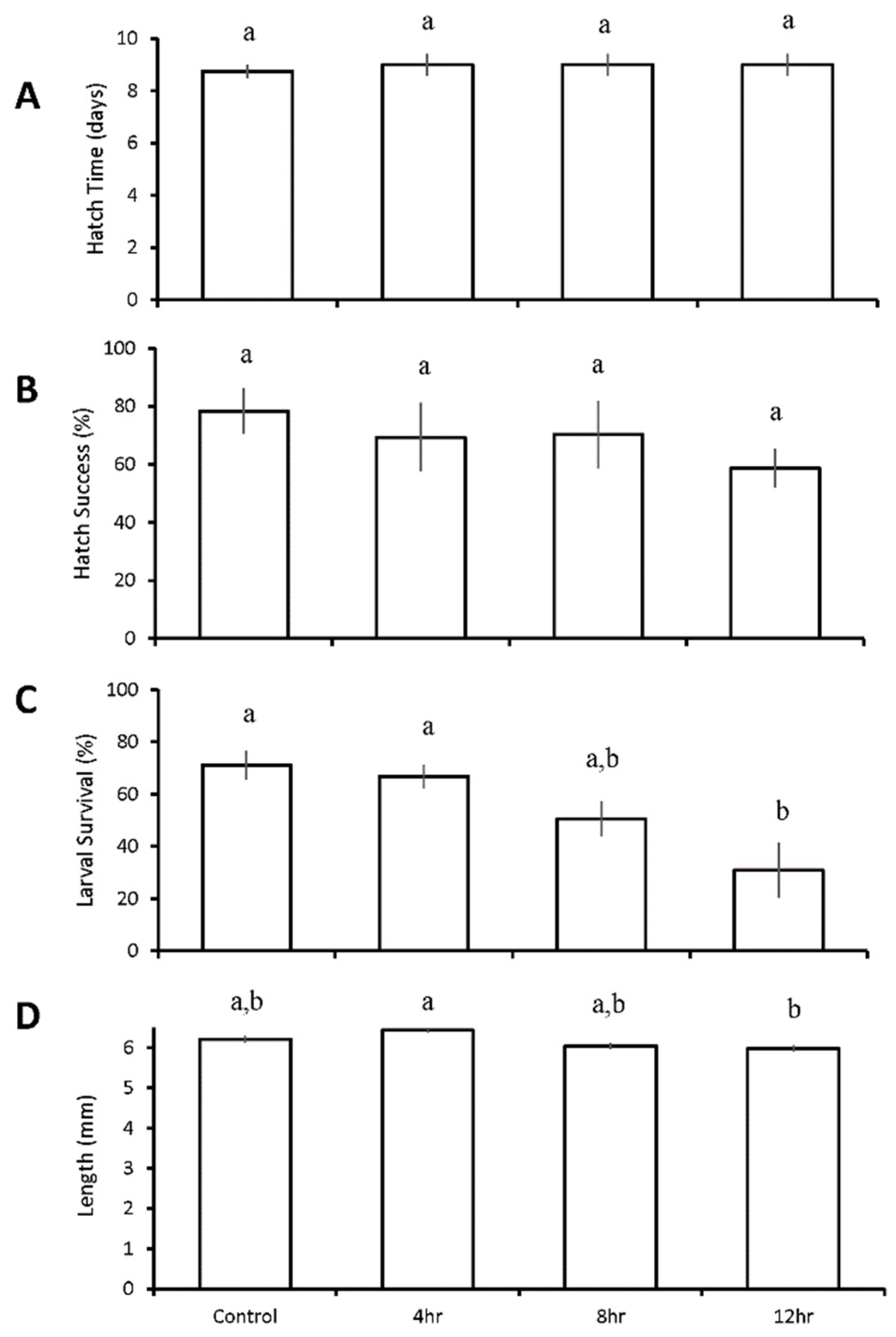
3.4. Diurnal Duration Experiment with Menidia menidia
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Copp, G.H.; Peňáz, M. Ecology of fish spawning and nursery zones in the flood plain, using a new sampling approach. Hydrobiologia 1988, 169, 209–224. [Google Scholar] [CrossRef]
- Hjort, J. Fluctuations in the great fisheries of northern Europe viewed in the light of biological research. Rapports et Proces-Verbaux des Réunions/Conseil Permanent International pour l’Exploration de la Mer 1914, 20, 1–228. Available online: https://core.ac.uk/download/pdf/52047619.pdf (accessed on 29 December 2019).
- Houde, E. Effects of temperature and delayed feeding on growth and survival of larvae of three species of subtropical marine fishes. Mar. Biol. 1974, 26, 271–285. [Google Scholar] [CrossRef]
- Houde, E.D. Emerging from Hjort’s Shadow. J. Northwest Atl. Fish. Sci. 2008, 41, 53–70. [Google Scholar] [CrossRef]
- Doney, S.C.; Ruckelshaus, M.; Duffy, J.E.; Barry, J.P.; Chan, F.; English, C.A.; Galindo, H.M.; Grebmeier, J.M.; Hollowed, A.B.; Knowlton, N.; et al. Climate change impacts on marine ecosystems. Annu. Rev. Mar. Sci. 2012, 4, 11–37. [Google Scholar] [CrossRef]
- Breitburg, D.; Levin, L.A.; Oschlies, A.; Grégoire, M.; Chavez, F.P.; Conley, D.J.; Garçon, V.; Gilbert, D.; Guitérrez, D.; Isensee, K.; et al. Declining oxygen in the global ocean and coastal waters. Science 2018, 359, eaam7240. [Google Scholar] [CrossRef] [PubMed]
- Baumann, H.; Wallace, R.B.; Tagliaferri, T. Large Natural pH, CO2 and O2 Fluctuations in a Temperate Tidal Salt Marsh on Diel, Seasonal, and Interannual Time Scales. Estuar. Coasts 2014, 38, 220–231. [Google Scholar] [CrossRef]
- Portner, H.O.; Peck, M.A. Climate change effects on fishes and fisheries: Towards a cause-and-effect understanding. J. Fish. Biol. 2010, 77, 1745–1779. [Google Scholar] [CrossRef]
- Baumann, H.; Smith, E.M. Quantifying Metabolically Driven pH and Oxygen Fluctuations in US Nearshore Habitats at Diel to Interannual Time Scales. Estuar. Coasts 2017, 41, 1102–1117. [Google Scholar] [CrossRef]
- Wallace, R.B.; Baumann, H.; Grear, J.S.; Aller, R.C.; Gobler, C.J. Coastal ocean acidification: The other eutrophication problem. Estuar. Coast. Shelf. Sci. 2014, 148, 1–13. [Google Scholar] [CrossRef]
- Waldbusser, G.G.; Salisbury, J.E. Ocean acidification in the coastal zone from an organism’s perspective: Multiple system parameters, frequency domains, and habitats. Annu. Rev. Mar. Sci. 2014, 6, 221–247. [Google Scholar] [CrossRef] [PubMed]
- Gobler, C.J.; Baumann, H. Hypoxia and acidification in ocean ecosystems: Coupled dynamics and effects on marine life. Biol. Lett. 2016, 12, 20150976. [Google Scholar] [CrossRef] [PubMed]
- First, P.J. Global Warming of 1.5 °C An IPCC Special Report on the Impacts of Global Warming of 1.5 °C above Pre-Industrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change, Sustainable Development, and Efforts to Eradicate Poverty; World Meterological Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Sunda, W.G.; Cai, W.J. Eutrophication induced CO2-acidification of subsurface coastal waters: Interactive effects of temperature, salinity, and atmospheric PCO2. Environ. Sci. Technol. 2012, 46, 10651–10659. [Google Scholar] [CrossRef] [PubMed]
- Melzner, F.; Thomsen, J.; Koeve, W.; Oschlies, A.; Gutowska, M.A.; Bange, H.W.; Hansen, H.P.; Kortzinger, A. Future ocean acidification will be amplified by hypoxia in coastal habitats. Mar. Biol. 2013, 160, 1875–1888. [Google Scholar] [CrossRef]
- Diaz, R.J.; Rosenberg, R. Marine benthic hypoxia: A review of its ecological effects and the behavioural responses of benthic macrofauna. Oceanogr. Mar. Biol. Annu. Rev. 1995, 33, 245–303. [Google Scholar]
- Vaquer-Sunyer, R.; Duarte, C.M. Thresholds of hypoxia for marine biodiversity. Proc. Natl. Acad. Sci. USA 2008, 105, 15452–15457. [Google Scholar] [CrossRef]
- Frommel, A.Y.; Schubert, A.; Piatkowski, U.; Clemmesen, C. Egg and early larval stages of Baltic cod, Gadus morhua, are robust to high levels of ocean acidification. Mar. Biol. 2012, 160, 1825–1834. [Google Scholar] [CrossRef]
- Hurst, T.P.; Fernandez, E.R.; Mathis, J.T. Effects of ocean acidification on hatch size and larval growth of walleye pollock (Theragra chalcogramma). ICES J. Mar. Sci. 2013, 70, 812–822. [Google Scholar] [CrossRef]
- Baumann, H.; Talmage, S.C.; Gobler, C.J. Reduced early life growth and survival in a fish in direct response to increased carbon dioxide. Nat. Clim. Chang. 2011, 2, 38–41. [Google Scholar] [CrossRef]
- Murray, C.M.; Malvezzi, A.; Gobler, C.J.; Baumann, H. Offspring sensitivity to ocean acidification changes seasonally in a coastal marine fish. Mar. Ecol. Prog. Ser. 2014, 504, 1–11. [Google Scholar] [CrossRef]
- Chambers, R.C.; Candelmo, A.C.; Habeck, E.A.; Poach, M.E.; Wieczorek, D.; Cooper, K.R.; Greenfield, C.E.; Phelan, B.A. Effects of elevated CO2 in the early life stages of summer flounder, Paralichthys dentatus, and potential consequences of ocean acidification. Biogeosciences 2014, 11, 1613–1626. [Google Scholar] [CrossRef]
- Stiasny, M.H.; Mittermayer, F.H.; Sswat, M.; Voss, R.; Jutfelt, F.; Chierici, M.; Puvanendran, V.; Mortensen, A.; Reusch, T.B.H.; Clemmesen, C. Ocean Acidification Effects on Atlantic Cod Larval Survival and Recruitment to the Fished Population. PLoS ONE 2016, 11, e0155448. [Google Scholar] [CrossRef] [PubMed]
- DePasquale, E.; Baumann, H.; Gobler, C.J. Vulnerability of early life stage Northwest Atlantic forage fish to ocean acidification and low oxygen. Mar. Ecol. Prog. Ser. 2015, 523, 145–156. [Google Scholar] [CrossRef]
- Miller, S.H.; Breitburg, D.L.; Burrell, R.B.; Keppel, A.G. Acidification increases sensitivity to hypoxia in important forage fishes. Mar. Ecol. Prog. Ser. 2016, 549, 1–8. [Google Scholar] [CrossRef]
- Wootton, J.T.; Pfister, C.A.; Forester, J.D. Dynamic patterns and ecological impacts of declining ocean pH in a high-resolution multi-year dataset. Proc. Natl. Acad. Sci. USA 2008, 105, 18848–18853. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.; Baumann, H. You Better Repeat It: Complex CO2 × Temperature Effects in Atlantic Silverside Offspring Revealed by Serial Experimentation. Diversity 2018, 10, 69. [Google Scholar] [CrossRef]
- Davidson, M.I.; Targett, T.E.; Grecay, P.A. Evaluating the effects of diel-cycling hypoxia and pH on growth and survival of juvenile summer flounder Paralichthys dentatus. Mar. Ecol. Prog. Ser. 2016, 556, 223–235. [Google Scholar] [CrossRef]
- Dixon, R.L.; Grecay, P.A.; Targett, T.E. Responses of juvenile Atlantic silverside, striped killifish, mummichog, and striped bass to acute hypoxia and acidification: Aquatic surface respiration and survival. J. Exp. Mar. Biol. Ecol. 2017, 493, 20–30. [Google Scholar] [CrossRef]
- Weber, C.I.; Middaugh, D.P.; Hemmer, M.J.; Goodman, L.R. Methods for Measuring the Acute Toxicity of Effluents and Receiving Waters to Freshwater and Marine Organisms; Environmental Monitoring Systems Laboratory, Office of Research and Development, US Environmental Protection Agency: Cincinnati, OH, USA, 1991. [Google Scholar]
- Middaugh, D.P.; Hemmer, M.J. Reproductive ecology of the inland silverside, Menidia beryllina, (Pisces: Atherinidae) from Blackwater Bay, Florida. Copeia 1992, 1, 53–61. [Google Scholar] [CrossRef]
- Middaugh, D.P.; Scott, G.I.; Dean, J.M. Reproductive behavior of the Atlantic silverside, Menidia menidia (Pisces, Atherinidae). Environ. Biol. Fish. 1981, 6, 269–276. [Google Scholar] [CrossRef]
- Chitty, J.D.; Able, K.W. Habitat use, movements and growth of the sheepshead minnow, Cyprinodon variegatus, in a restored salt marsh in Delaware Bay. Bull. N. J. Acad. Sci. 2004, 49, 1–9. [Google Scholar]
- Pikitch, E.K.; Rountos, K.J.; Essington, T.E.; Santora, C.; Pauly, D.; Watson, R.; Sumaila, U.R.; Boersma, P.D.; Boyd, I.L.; Conover, D.O.; et al. The global contribution of forage fish to marine fisheries and ecosystems. Fish Fish. 2014, 15, 43–64. [Google Scholar] [CrossRef]
- Heuer, R.M.; Grosell, M. Physiological impacts of elevated carbon dioxide and ocean acidification on fish. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2014, 307, R1061–R1084. [Google Scholar] [CrossRef] [PubMed]
- Gobler, C.J.; DePasquale, E.L.; Griffith, A.W.; Baumann, H. Hypoxia and acidification have additive and synergistic negative effects on the growth, survival, and metamorphosis of early life stage bivalves. PLoS ONE 2014, 9, e83648. [Google Scholar] [CrossRef]
- Clark, H.R.; Gobler, C.J. Diurnal fluctuations in CO2 and dissolved oxygen concentrations do not provide a refuge from hypoxia and acidification for early-life-stage bivalves. Mar. Ecol. Prog. Ser. 2016, 558, 1–14. [Google Scholar] [CrossRef]
- Millero, F.J. Carbonate constants for estuarine waters. Mar. Freshw. Res. 2010, 61, 139–142. [Google Scholar] [CrossRef]
- Munday, P.L.; Gagliano, M.; Donelson, J.M.; Dixon, D.L.; Thorrold, S.R. Ocean acidification does not affect the early life history development of a tropical marine fish. Mar. Ecol. Prog. Ser. 2011, 423, 211–221. [Google Scholar] [CrossRef]
- Cattano, C.; Claudet, J.; Domenici, P.; Milazzo, M. Living in a high CO2 world: A global meta-analysis shows multiple trait-mediated fish responses to ocean acidification. Ecol. Monog. 2018, 88, 320–335. [Google Scholar] [CrossRef]
- Ekau, W.; Auel, H.; Pörtner, H.O.; Gilbert, D. Impacts of hypoxia on the structure and processes in pelagic communities (zooplankton, macro-invertebrates and fish). Biogeosciences 2010, 7, 1669–1699. [Google Scholar] [CrossRef]
- Bignami, S.; Sponaugle, S.; Hauff, M.; Cowen, R.K. Combined effects of elevated pCO2, temperature, and starvation stress on larvae of a large tropical marine fish. ICES J. Mar. Sci. 2017, 74, 1220–1229. [Google Scholar] [CrossRef]
- Esbaugh, A.J. Physiological implications of ocean acidification for marine fish: Emerging patterns and new insights. J. Comp. Physiol. B 2018, 188, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Rombough, P. The gill of fish larvae. Is it primarily a respiratory or an ionoregulatory structure? J. Fish Biol. 1999, 55, 186–204. [Google Scholar] [CrossRef]
- Ciuhandu, C.; Stevens, E.D.; Wright, P. The effect of oxygen on the growth of Oncorhynchus mykiss embryos with and without a chorion. J. Fish Biol. 2005, 67, 1544–1551. [Google Scholar] [CrossRef]
- Ciuhandu, C.S.; Wright, P.A.; Goldberg, J.I.; Stevens, E.D. Parameters influencing the dissolved oxygen in the boundary layer of rainbow trout (Oncorhynchus mykiss) embryos and larvae. J. Exp. Biol. 2007, 210, 1435–1445. [Google Scholar] [CrossRef] [PubMed]
- Hwang, P.P.; Lee, T.H. New insights into fish ion regulation and mitochondrion-rich cells. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2007, 148, 479–497. [Google Scholar] [CrossRef]
- Oppen-Berntsen, D.O.; Bogsnes, A.; Walther, B.T. The effects of hypoxia, alkalinity and neurochemicals on hatching of Atlantic salmon (Salmo salar) eggs. Aquaculture 1990, 86, 417–430. [Google Scholar] [CrossRef]
- Franke, A.; Clemmesen, C. Effect of ocean acidification on early life stages of Atlantic herring (Clupea harengus L.). Biogeosciences 2011, 8, 3697–3707. [Google Scholar] [CrossRef]
- Miller, G.M.; Watson, S.A.; Donelson, J.M.; McCormick, M.I.; Munday, P.L. Parental environment mediates impacts of increased carbon dioxide on a coral reef fish. Nat. Clim. Chang. 2012, 2, 858–861. [Google Scholar] [CrossRef]
- Green, M.A.; Aller, R.C.; Aller, J.Y. Carbonate dissolution and temporal abundances of foraminifera in Long Island Sound sediments. Limnol. Oceanog. 1993, 38, 331–345. [Google Scholar] [CrossRef]
- Green, M.A.; Waldbusser, G.G.; Reilly, S.L.; Emerson, K.; O’Donnell, S. Death by dissolution: Sediment saturation state as a mortality factor for juvenile bivalves. Limnol. Oceanog. 2009, 54, 1037–1047. [Google Scholar] [CrossRef]
- Mackin, J.E.; Swider, K.T. Organic matter decomposition pathways and oxygen consumption in coastal marine sediments. J. Mar. Res. 1989, 47, 681–716. [Google Scholar] [CrossRef]
- Koenigstein, S.; Mark, F.C.; Gößling-Reisemann, S.; Reuter, H.; Poertner, H.O. Modelling climate change impacts on marine fish populations: Process-based integration of ocean warming, acidification and other environmental drivers. Fish Fish. 2016, 17, 972–1004. [Google Scholar] [CrossRef]
- Tyler, R.M.; Targett, T.E. Juvenile weakfish Cynoscion regalis distribution in relation to diel-cycling dissolved oxygen in an estuarine tributary. Mar. Ecol. Prog. Ser. 2007, 333, 257–269. [Google Scholar] [CrossRef]
- Boeuf, G.; Boujard, D.; Ruyet, J.P.L. Control of the somatic growth in turbot. J. Fish Biol. 1999, 55, 128–147. [Google Scholar] [CrossRef]
- Pichavant, K. Comparative effects of long-term hypoxia on growth, feeding and oxygen consumption in juvenile turbot and European sea bass. J. Fish Biol. 2001, 59, 875–883. [Google Scholar] [CrossRef]
- Miller, T.J.; Crowder, L.B.; Rice, J.A.; Marschall, E.A. Larval Size and Recruitment Mechanisms in Fishes: Toward a Conceptual Framework. Can. J. Fish. Aquat. Sci. 1988, 45, 1657–1670. [Google Scholar] [CrossRef]
- Jarrold, M.D.; Munday, P.L. Diel CO2 cycles and parental effects have similar benefits to growth of a coral reef fish under ocean acidification. Biol. Lett. 2019, 15, 20180724. [Google Scholar] [CrossRef]
| Experiment | Species | Significant Effects | |||
|---|---|---|---|---|---|
| Hatch Time (days) | Hatch Success (%) | Survival (%) | Size (mm) | ||
| Seven treatments: control chronic low DO diurnal low DO chronic low pH diurnal low pH chronic low pH/DO diurnal low pH/DO | M.menidia | diurnal low pH ↓ diurnal low DO ↓ | diurnal and chronic low DO ↓ diurnal and chronic low pH/DO ↓ | chronic low pH/DO ↓ | |
| C. variegatus | chronic low pH ↑ diurnal and chronic low DO ↑ diurnal and chronic low pH/DO ↑ | ||||
| M. beryllina | chronic low pH ↓ chronic low pH/DO ↓ | chronic low DO ↓ diurnal and chronic low pH/DO ↓ | chronic low DO ↓ chronic low pH/DO ↓ | ||
| Four treatments: control chronic low pH/DO diurnal (12 h) low pH/DO chronic reduced pH/DO | M. beryllina | diurnal low pH/DO ↓ chronic low pH/DO ↓ | chronic low pH/DO ↓ | ||
| Four treatment diurnal durations: control 4 h low pH/DO 8 h low pH/DO 12 h low pH/DO | M. beryllina | 12 h ↓ | 12 h ↓ | 8 h ↓ 12 h ↓ | |
| M. beryllina | 12 h ↓ | ||||
| M. menidia | 4 h ↓ 8 h ↓ 12 h ↓ | 4 h ↑ | |||
| Parameter | Continuous | Diurnal pH | Diurnal DO | Diurnal pH/DO | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | Low pH | Low DO | Low pH/DO | Ambient | Low | Mean | Ambient | Low | Mean | Ambient | Low | Mean | |
| pHT daily chem | 7.85 ± 0.05 | 7.20 ± 0.08 | 7.84 ± 0.06 | 7.25 ± 0.07 | 7.64 ± 0.09 | 7.28 ± 0.12 | 7.50 ± 0.21 | 7.84 ± 0.07 | 7.87 ± 0.04 | 7.85 ± 0.06 | 7.68 ± 0.10 | 7.35 ± 0.11 | 7.55 ± 0.19 |
| pCO2 (µatm) | 407 ± 73 | 2255 ± 420 | 406 ± 30 | 1935 ± 124 | 714 ± 390 | 2110 ± 394 | 1412 ± 830 | 416 ± 20 | 468 ± 41 | 442 ± 41 | 443 ± 87 | 1433 ± 622 | 938 ± 670 |
| Ω calcite | 1.66 ± 0.34 | 0.38 ± 0.04 | 1.82 ± 0.26 | 0.49 ± 0.13 | 1.30 ± 0.57 | 0.36 ± 0.08 | 0.83 ± 0.63 | 1.70 ± 0.09 | 1.66 ± 0.26 | 1.68 ± 0.18 | 1.72 ± 0.34 | 0.62 ± 0.22 | 1.17 ± 0.64 |
| Ω aragonite | 1.1 ± 0.26 | 0.3 ± 0.03 | 1.2 ± 0.22 | 0.3 ± 0.07 | 0.9 ± 0.41 | 0.2 ± 0.06 | 0.6 ± 0.44 | 1.1 ± 0.10 | 1.1 ± 0.21 | 1.1 ± 0.15 | 1.2 ± 0.27 | 0.4 ± 0.13 | 0.8 ± 0.44 |
| TDIC (µmol L−1) | 1177 ± 110 | 1329 ± 33 | 1244 ± 82 | 1379 ± 129 | 1253 ± 35 | 1243 ± 39 | 1248 ± 34 | 1217 ± 41 | 1267 ± 87 | 1242 ± 68 | 1244 ± 55 | 1270 ± 135 | 1257 ± 97 |
| CO32− (µmol L−1) | 67.5 ± 14.8 | 15.6 ± 1.8 | 74.2 ± 11.8 | 19.8 ± 4.8 | 53.1 ± 24.1 | 14.8 ± 3.4 | 33.9 ± 25.9 | 69.2 ± 4.8 | 67.6 ± 11.6 | 68.4 ± 8.3 | 69.8 ± 15.0 | 25.0 ± 8.5 | 47.4 ± 26.5 |
| TA (µmol L−1) | 1283 ± 124 | 1287 ± 22 | 1358 ± 95 | 1354 ± 138 | 1322 ± 37 | 1206 ± 54 | 1264 ± 75 | 1324 ± 45 | 1368 ± 100 | 1346 ± 75 | 1350 ± 73 | 1271 ± 110 | 1310 ± 96 |
| Salinity | 28.46 ± 0.0 | 28.46 ± 0.0 | 28.46 ± 0.0 | 28.46 ± 0.0 | 28.46 ± 0.0 | 28.46 ± 0.0 | 28.46 ± 0.0 | 28.46 ± 0.0 | 28.46 ± 0.0 | 28.46 ± 0.0 | 28.46 ± 0.0 | 28.46 ± 0.0 | 28.46 ± 0.0 |
| Dissolved Oxygen (µM) | 6.83 ± 0.28 | 6.77 ± 0.28 | 2.82 ± 0.45 | 2.42 ± 0.64 | 6.69 ± 0.15 | 6.76 ± 0.27 | 6.72 ± 0.21 | 6.63 ± 0.20 | 3.17 ± 1.91 | 5.19 ± 2.11 | 6.63 ± 0.64 | 3.22 ± 1.96 | 5.18 ± 2.18 |
| Temperature (°C) | 23.2 ± 0.4 | 23.2 ± 0.4 | 23.2 ± 0.4 | 23.2 ± 0.4 | 23.2 ± 0.4 | 23.2 ± 0.4 | 23.2 ± 0.4 | 23.2 ± 0.4 | 23.2 ± 0.4 | 23.2 ± 0.4 | 23.2 ± 0.4 | 23.2 ± 0.4 | 23.2 ± 0.4 |
| Parameter | Continuous | Diurnal pH | Diurnal DO | Diurnal pH/DO | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | Low pH | Low DO | Low pH/DO | Ambient | Low | Mean | Ambient | Low | Mean | Ambient | Low | Mean | |
| pHT daily chem | 7.67 ± 0.15 | 7.08 ± 0.12 | 7.74 ± 0.07 | 7.17 ± 0.07 | 7.57 ± 0.19 | 7.27 ± 0.19 | 7.37 ± 0.23 | 7.74 ± 0.05 | 7.71 ± 0.12 | 7.72 ± 0.11 | 7.65 ± 0.13 | 7.37 ± 0.18 | 7.46 ± 0.21 |
| pCO2 (µatm) | 528 ± 96 | 2152 ± 1001 | 519 ± 113 | 1652 ± 658 | 1453 ± 1112 | 1911 ± 899 | 1682 ± 968 | 707 ± 320 | 1046 ± 1040 | 876 ± 735 | 1752 ± 993 | 1413 ± 1762 | 1582 ± 1336 |
| Ω calcite | 1.51 ± 0.24 | 0.68 ± 0.60 | 1.65 ± 0.20 | 0.79 ± 0.44 | 0.85 ± 0.44 | 0.73 ± 0.74 | 0.79 ± 0.56 | 1.33 ± 0.35 | 1.26 ± 0.57 | 1.29 ± 0.44 | 0.62 ± 0.22 | 1.25 ± 0.68 | 0.93 ± 0.58 |
| Ω aragonite | 1.0 ± 0.16 | 0.4 ± 0.39 | 1.1 ± 0.13 | 0.5 ± 0.28 | 0.5 ± 0.28 | 0.5 ± 0.48 | 0.5 ± 0.37 | 0.9 ± 0.23 | 0.8 ± 0.37 | 0.8 ± 0.29 | 0.4 ± 0.14 | 0.8 ± 0.44 | 0.6 ± 0.38 |
| TDIC (µmol L−1) | 1266 ± 196 | 1424 ± 81 | 1301 ± 146 | 1472 ± 65 | 1348 ± 109 | 1338 ± 148 | 1343 ± 120 | 1290 ± 119 | 1317 ± 145 | 1303 ± 124 | 1352 ± 219 | 1355 ± 174 | 1354 ± 183 |
| CO32− (µmol L−1) | 60.7 ± 9.9 | 27.1 ± 24.0 | 66.2 ± 8.0 | 31.7 ± 17.7 | 34.0 ± 17.6 | 29.3 ± 29.6 | 31.7 ± 22.7 | 53.3 ± 13.9 | 50.4 ± 22.9 | 51.9 ± 17.6 | 24.7 ± 8.8 | 50.2 ± 27.5 | 37.5 ± 23.3 |
| TA (µmol L−1) | 1357 ± 201 | 1405 ± 129 | 1400 ± 145 | 1475 ± 51 | 1364 ± 79 | 1331 ± 201 | 1348 ± 143 | 1363 ± 83 | 1374 ± 109 | 1368 ± 90 | 1343 ± 177 | 1400 ± 139 | 1372 ± 150 |
| Salinity | 31.9 ± 2.1 | 31.9 ± 2.1 | 31.9 ± 2.1 | 31.9 ± 2.1 | 31.9 ± 2.1 | 31.9 ± 2.1 | 31.9 ± 2.1 | 31.9 ± 2.1 | 31.9 ± 2.1 | 31.9 ± 2.1 | 31.9 ± 2.1 | 31.9 ± 2.1 | 31.9 ± 2.1 |
| Dissolved Oxygen (µM) | 6.78 ± 0.54 | 6.64 ± 0.61 | 2.74 ± 0.44 | 2.29 ± 0.49 | 6.79 ± 0.68 | 6.51 ± 0.57 | 6.60 ± 0.61 | 6.85 ± 0.60 | 4.35 ± 1.75 | 5.13 ± 1.89 | 7.17 ± 0.40 | 5.04 ± 1.90 | 5.71 ± 1.90 |
| Temperature (°C) | 22.8 ± 1.0 | 22.8 ± 1.0 | 22.8 ± 1.0 | 22.8 ± 1.0 | 22.8 ± 1.0 | 22.8 ± 1.0 | 22.8 ± 1.0 | 22.8 ± 1.0 | 22.8 ± 1.0 | 22.8 ± 1.0 | 22.8 ± 1.0 | 22.8 ± 1.0 | 22.8 ± 1.0 |
| Parameter | Continuous | Diurnal pH | Diurnal DO | Diurnal pH/DO | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | Low pH | Low DO | Low pH/DO | Ambient | Low | Mean | Ambient | Low | Mean | Ambient | Low | Mean | |
| pHT daily chem | 7.78 ± 0.06 | 7.19 ± 0.16 | 7.83 ± 0.05 | 7.27 ± 0.13 | 7.65 ± 0.11 | 7.23 ± 0.02 | 7.62 ± 0.16 | 7.80 ± 0.04 | 7.81 ± 0.02 | 7.80 ± 0.04 | 7.68 ± 0.07 | 7.49 ± 0.03 | 7.66 ± 0.09 |
| pCO2 (µatm) | 313 ± 111 | 2885 ± 462 | 317 ± 130 | 2376 ± 886 | 442 ± 81 | 892 ± 87 | 742 ± 245 | 383 ± 144 | 451 ± 154 | 428 ± 140 | 435 ± 102 | 636 ± 190 | 569 ± 186 |
| Ω calcite | 2.02 ± 0.05 | 0.38 ± 0.06 | 2.08 ± 0.09 | 0.33 ± 0.04 | 2.09 ± 0.37 | 1.17 ± 0.16 | 1.48 ± 0.52 | 2.14 ± 0.51 | 1.84 ± 0.30 | 1.94 ± 0.36 | 1.88 ± 0.14 | 1.27 ± 0.30 | 1.47 ± 0.39 |
| Ω aragonite | 1.3 ± 0.03 | 0.2 ± 0.04 | 1.4 ± 0.06 | 0.2 ± 0.02 | 1.4 ± 0.24 | 0.8 ± 0.10 | 1.0 ± 0.34 | 1.4 ± 0.33 | 1.2 ± 0.20 | 1.3 ± 0.24 | 1.2 ± 0.09 | 0.8 ± 0.19 | 1.0 ± 0.25 |
| TDIC (µmol L−1) | 1140 ± 198 | 1464 ± 38 | 1158 ± 212 | 1255 ± 311 | 1353 ± 19 | 1413 ± 40 | 1393 ± 45 | 1260 ± 93 | 1260 ± 132 | 1260 ± 111 | 1287 ± 94 | 1249 ± 132 | 1262 ± 112 |
| CO32- (µmol L−1) | 82.2 ± 2.1 | 15.4 ± 2.5 | 84.7 ± 4.0 | 13.3 ± 1.5 | 84.9 ± 15.0 | 47.6 ± 6.5 | 60.0 ± 21.0 | 86.7 ± 20.7 | 74.7 ± 12.5 | 78.7 ± 14.7 | 76.3 ± 6.1 | 51.8 ± 12.0 | 59.9 ± 15.9 |
| TA (µmol L−1) | 1284 ± 183 | 1405 ± 25 | 1305 ± 191 | 1208 ± 284 | 1486 ± 9 | 1469 ± 47 | 1474 ± 38 | 1403 ± 49 | 1380 ± 106 | 1388 ± 85 | 1409 ± 76 | 1323 ± 133 | 1352 ± 117 |
| Salinity | 31.7 ± 0.3 | 31.7 ± 0.3 | 31.7 ± 0.3 | 31.7 ± 0.3 | 31.7 ± 0.3 | 31.7 ± 0.3 | 31.7 ± 0.3 | 31.7 ± 0.3 | 31.7 ± 0.3 | 31.7 ± 0.3 | 31.7 ± 0.3 | 31.7 ± 0.3 | 31.7 ± 0.3 |
| Dissolved Oxygen (µM) | 6.91 ± 0.14 | 6.78 ± 0.25 | 2.11 ± 0.33 | 2.44 ± 0.56 | 6.33 ± 1.43 | 6.76 ± 0.02 | 6.37 ± 1.36 | 6.75 ± 0.27 | 2.23 ± 0.53 | 6.34 ± 1.35 | 6.78 ± 0.26 | 2.82 ± 0.64 | 6.40 ± 1.20 |
| Temperature (°C) | 22.8 ± 0.5 | 22.8 ± 0.5 | 22.8 ± 0.5 | 22.8 ± 0.5 | 22.8 ± 0.5 | 22.8 ± 0.5 | 22.8 ± 0.5 | 22.8 ± 0.5 | 22.8 ± 0.5 | 22.8 ± 0.5 | 22.8 ± 0.5 | 22.8 ± 0.5 | 22.8 ± 0.5 |
| Parameter | Continuous | Diurnal pH/DO | ||||
|---|---|---|---|---|---|---|
| Control | Reduced pH/DO | Low pH/DO | Ambient | Low | Mean | |
| pHT daily chem | 7.89 ± 0.07 | 7.43 ± 0.08 | 7.16 ± 0.07 | 7.82 ± 0.05 | 7.32 ± 0.06 | 7.56 ± 0.25 |
| pCO2 (µatm) | 357 ± 57 | 1271 ± 371 | 2368 ± 407 | 422 ± 71 | 818 ± 151 | 620 ± 238 |
| Ω calcite | 2.22 ± 0.54 | 0.88 ± 0.05 | 0.40 ± 0.03 | 2.18 ± 0.87 | 1.03 ± 0.06 | 1.61 ± 0.84 |
| Ω aragonite | 1.5 ± 0.31 | 0.6 ± 0.05 | 0.3 ± 0.01 | 1.5 ± 0.54 | 0.7 ± 0.03 | 1.1 ± 0.54 |
| TDIC (µmol L−1) | 1286 ± 59 | 1480 ± 159 | 1399 ± 130 | 1357 ± 161 | 1303 ± 112 | 1330 ± 132 |
| CO32− (µmol L−1) | 93.9 ± 17.6 | 37.5 ± 4.0 | 17.2 ± 0.5 | 91.8 ± 32.0 | 44.1 ± 2.4 | 68.0 ± 33.0 |
| TA (µmol L−1) | 1437 ± 83 | 1505 ± 143 | 1358 ± 117 | 1498 ± 210 | 1357 ± 106 | 1427 ± 172 |
| Salinity | 31.8 ± 0.6 | 31.8 ± 0.6 | 31.8 ± 0.6 | 31.8 ± 0.6 | 31.8 ± 0.6 | 31.8 ± 0.6 |
| Dissolved Oxygen (µM) | 6.83 ± 0.26 | 4.81 ± 0.22 | 2.47 ± 0.33 | 6.87 ± 0.34 | 2.17 ± 0.52 | 4.44 ± 2.39 |
| Temperature (°C) | 23.2 ± 0.6 | 23.2 ± 0.6 | 23.2 ± 0.6 | 23.2 ± 0.6 | 23.2 ± 0.6 | 23.2 ± 0.6 |
| Parameter | 4 h Diurnal pH/DO | 8 h Diurnal DO/pH | 12 h Diurnal pH/DO | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control | Ambient | Low | Mean | Ambient | Low | Mean | Ambient | Low | Mean | |
| pHT daily chem | 7.94 ± 0.03 | 7.94 ± 0.01 | 7.22 ± 0.09 | 7.95 ± 0.01 | 7.23 ± 0.08 | 7.94 ± 0.00 | 7.23 ± 0.05 | |||
| pCO2 (µatm) | 466 ± 3 | 509 ± 7 | 2782 ± 19 | 1483 ± 1215 | 517 ± 56 | 2901 ± 168 | 1709 ± 1279 | 541 ± 55 | 2895 ± 88 | 1718 ± 1260 |
| Ω calcite | 3.16 ± 0.09 | 2.98 ± 0.18 | 0.69 ± 0.02 | 2.00 ± 1.23 | 2.98 ± 0.10 | 0.67 ± 0.06 | 1.83 ± 1.24 | 2.87 ± 0.10 | 0.67 ± 0.04 | 1.77 ± 1.18 |
| Ω aragonite | 2.04 ± 0.07 | 1.93 ± 0.12 | 0.45 ± 0.01 | 1.29 ± 0.79 | 1.92 ± 0.06 | 0.43 ± 0.04 | 1.18 ± 0.80 | 1.85 ± 0.06 | 0.43 ± 0.03 | 1.14 ± 0.76 |
| TDIC (µmol L−1) | 1789 ± 38 | 1811 ± 58 | 2002 ± 27 | 1893 ± 111 | 1821 ± 71 | 2008 ± 36 | 1915 ± 113 | 1825 ± 66 | 2009 ± 32 | 1917 ± 109 |
| CO32- (µmol L−1) | 127.2 ± 5.5 | 119.9 ± 8.6 | 28.0 ± 1.2 | 80.5 ± 49.5 | 119.6 ± 2.6 | 27.0 ± 2.8 | 73.3 ± 49.5 | 115.3 ± 2.8 | 27.0 ± 2.0 | 71.2 ± 47.2 |
| TA (µmol L−1) | 1968 ± 48 | 1976 ± 71 | 1959 ± 29 | 1969 ± 54 | 1986 ± 66 | 1960 ± 44 | 1973 ± 54 | 1983 ± 61 | 1961 ± 37 | 1972 ± 48 |
| Salinity | 29.6 ± 1.6 | 29.6 ± 1.6 | 29.6 ± 1.6 | 29.6 ± 1.6 | 29.6 ± 1.6 | 29.6 ± 1.6 | 29.6 ± 1.6 | |||
| Dissolved Oxygen (µM) | 7.04 ± 0.58 | 6.90 ± 0.02 | 2.20 ± 0.45 | 6.92 ± 0.02 | 2.04 ± 0.40 | 6.97 ± 0.04 | 2.12 ± 0.60 | |||
| Temperature (°C) | 22.5 ± 0.4 | 22.5 ± 0.4 | 22.5 ± 0.4 | 22.5 ± 0.4 | 22.5 ± 0.4 | 22.5 ± 0.4 | 22.5 ± 0.4 | |||
| Parameter | 4 h Diurnal pH/DO | 8 h Diurnal DO/pH | 12 h Diurnal pH/DO | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control | Ambient | Low | Mean | Ambient | Low | Mean | Ambient | Low | Mean | |
| pHT daily chem | 7.90 ± 0.03 | 7.86 ± 0.01 | 7.21 ± 0.07 | 7.86 ± 0.02 | 7.20 ± 0.07 | 7.86 ± 0.00 | 7.23 ± 0.08 | |||
| pCO2 (µatm) | 506 ± 48 | 538 ± 30 | 2881 ± 79 | 1543 ± 1253 | 521 ± 60 | 2960 ± 98 | 1741 ± 1306 | 543 ± 58 | 2922 ± 125 | 1733 ± 1274 |
| Ω calcite | 2.76 ± 0.37 | 2.60 ± 0.27 | 0.60 ± 0.06 | 1.74 ± 1.09 | 2.71 ± 0.41 | 0.60 ± 0.03 | 1.65 ± 1.16 | 2.62 ± 0.39 | 0.61 ± 0.04 | 1.61 ± 1.10 |
| Ω aragonite | 1.78 ± 0.24 | 1.67 ± 0.18 | 0.39 ± 0.04 | 1.12 ± 0.70 | 1.74 ± 0.27 | 0.39 ± 0.02 | 1.06 ± 0.75 | 1.68 ± 0.25 | 0.39 ± 0.03 | 1.04 ± 0.71 |
| TDIC (µmol L−1) | 1728 ± 33 | 1728 ± 38 | 1910 ± 54 | 1806 ± 105 | 1731 ± 33 | 1931 ± 54 | 1831 ± 115 | 1736 ± 37 | 1929 ± 61 | 1833 ± 113 |
| CO32− (µmol L−1) | 109.8 ± 14.7 | 103.3 ± 10.6 | 23.9 ± 2.4 | 69.3 ± 43.1 | 107.6 ± 16.3 | 23.8 ± 1.2 | 65.7 ± 46.0 | 104.0 ± 15.4 | 24.1 ± 1.7 | 64.1 ± 43.9 |
| TA (µmol L−1) | 1880 ± 55 | 1869 ± 53 | 1856 ± 60 | 1864 ± 52 | 1879 ± 58 | 1875 ± 54 | 1877 ± 52 | 1877 ± 61 | 1875 ± 62 | 1876 ± 57 |
| Salinity | 28.03 ± 0.9 | 28.03 ± 0.9 | 28.03 ± 0.9 | 28.03 ± 0.9 | 28.03 ± 0.9 | 28.03 ± 0.9 | 28.03 ± 0.9 | 28.03 ± 0.9 | 28.03 ± 0.9 | 28.03 ± 0.9 |
| Dissolved Oxygen (µM) | 7.07 ± 0.75 | 7.38 ± 0.01 | 2.64 ± 1.01 | 7.32 ± 0.31 | 2.32 ± 0.83 | 7.56 ± 0.27 | 2.25 ± 0.77 | |||
| Temperature (°C) | 22.8 ± 1.6 | 22.8 ± 1.6 | 22.8 ± 1.6 | 22.8 ± 1.6 | 22.8 ± 1.6 | 22.8 ± 1.6 | 22.8 ± 1.6 | 22.8 ± 1.6 | 22.8 ± 1.6 | 22.8 ± 1.6 |
| Parameter | 4 h Diurnal pH/DO | 8 h Diurnal DO/pH | 12 h Diurnal pH/DO | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control | Ambient | Low | Mean | Ambient | Low | Mean | Ambient | Low | Mean | |
| pHT daily chem | 7.93 ± 0.02 | 7.94 ± 0.01 | 7.27 ± 0.05 | 7.91 ± 0.01 | 7.21 ± 0.04 | 7.91 ± 0.02 | 7.32 ± 0.09 | |||
| pCO2 (µatm) | 526 ± 39 | 523 ± 12 | 3052 ± 164 | 1787 ± 1356 | 528 ± 12 | 2998 ± 31 | 1763 ± 1320 | 544 ± 42 | 3015 ± 129 | 1780 ± 1324 |
| Ω calcite | 2.62 ± 0.21 | 2.64 ± 0.11 | 0.57 ± 0.04 | 1.61 ± 1.11 | 2.61 ± 0.11 | 0.58 ± 0.01 | 1.59 ± 1.09 | 2.57 ± 0.21 | 0.58 ± 0.03 | 1.57 ± 1.08 |
| Ω aragonite | 1.69 ± 0.13 | 1.70 ± 0.07 | 0.37 ± 0.02 | 1.03 ± 0.71 | 1.68 ± 0.07 | 0.37 ± 0.01 | 1.03 ± 0.70 | 1.66 ± 0.13 | 0.37 ± 0.02 | 1.01 ± 0.69 |
| TDIC (µmol L−1) | 1720 ± 15 | 1726 ± 26 | 1923 ± 13 | 1824 ± 107 | 1725 ± 26 | 1910 ± 26 | 1818 ± 102 | 1730 ± 21 | 1917 ± 23 | 1823 ± 102 |
| CO32− (µmol L−1) | 104.4 ± 7.7 | 105.4 ± 3.9 | 22.9 ± 1.4 | 64.1 ± 44.2 | 104.3 ± 4.0 | 23.0 ± 0.3 | 63.6 ± 43.6 | 102.6 ± 8.0 | 23.0 ± 1.1 | 62.8 ± 42.9 |
| TA (µmol L−1) | 1864 ± 26 | 1871 ± 30 | 1863 ± 18 | 1867 ± 24 | 1869 ± 31 | 1852 ± 25 | 1861 ± 27 | 1871 ± 31 | 1858 ± 24 | 1864 ± 27 |
| Salinity | 29.6 ± 0.8 | 29.6 ± 0.8 | 29.6 ± 0.8 | 29.6 ± 0.8 | 29.6 ± 0.8 | 29.6 ± 0.8 | 29.6 ± 0.8 | 29.6 ± 0.8 | 29.6 ± 0.8 | 29.6 ± 0.8 |
| Dissolved Oxygen (µM) | 7.00 ± 0.10 | 7.09 ± 0.03 | 1.95 ± 0.38 | 7.91 ± 0.01 | 2.08 ± 0.40 | 7.25 ± 0.07 | 1.81 ± 0.29 | |||
| Temperature (°C) | 22.4 ± 0.2 | 22.4 ± 0.2 | 22.4 ± 0.2 | 22.4 ± 0.2 | 22.4 ± 0.2 | 22.4 ± 0.2 | 22.4 ± 0.2 | 22.4 ± 0.2 | 22.4 ± 0.2 | 22.4 ± 0.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morrell, B.K.; Gobler, C.J. Negative Effects of Diurnal Changes in Acidification and Hypoxia on Early-Life Stage Estuarine Fishes. Diversity 2020, 12, 25. https://doi.org/10.3390/d12010025
Morrell BK, Gobler CJ. Negative Effects of Diurnal Changes in Acidification and Hypoxia on Early-Life Stage Estuarine Fishes. Diversity. 2020; 12(1):25. https://doi.org/10.3390/d12010025
Chicago/Turabian StyleMorrell, Brooke K., and Christopher J. Gobler. 2020. "Negative Effects of Diurnal Changes in Acidification and Hypoxia on Early-Life Stage Estuarine Fishes" Diversity 12, no. 1: 25. https://doi.org/10.3390/d12010025
APA StyleMorrell, B. K., & Gobler, C. J. (2020). Negative Effects of Diurnal Changes in Acidification and Hypoxia on Early-Life Stage Estuarine Fishes. Diversity, 12(1), 25. https://doi.org/10.3390/d12010025





