The Current Status of Indigenous Ovine Genetic Resources in Southern Africa and Future Sustainable Utilisation to Improve Livelihoods
Abstract
1. Introduction
2. Ovine Genetic Resources in SADC
2.1. Genetic Diversity of Ovine Genetic Resources in SADC
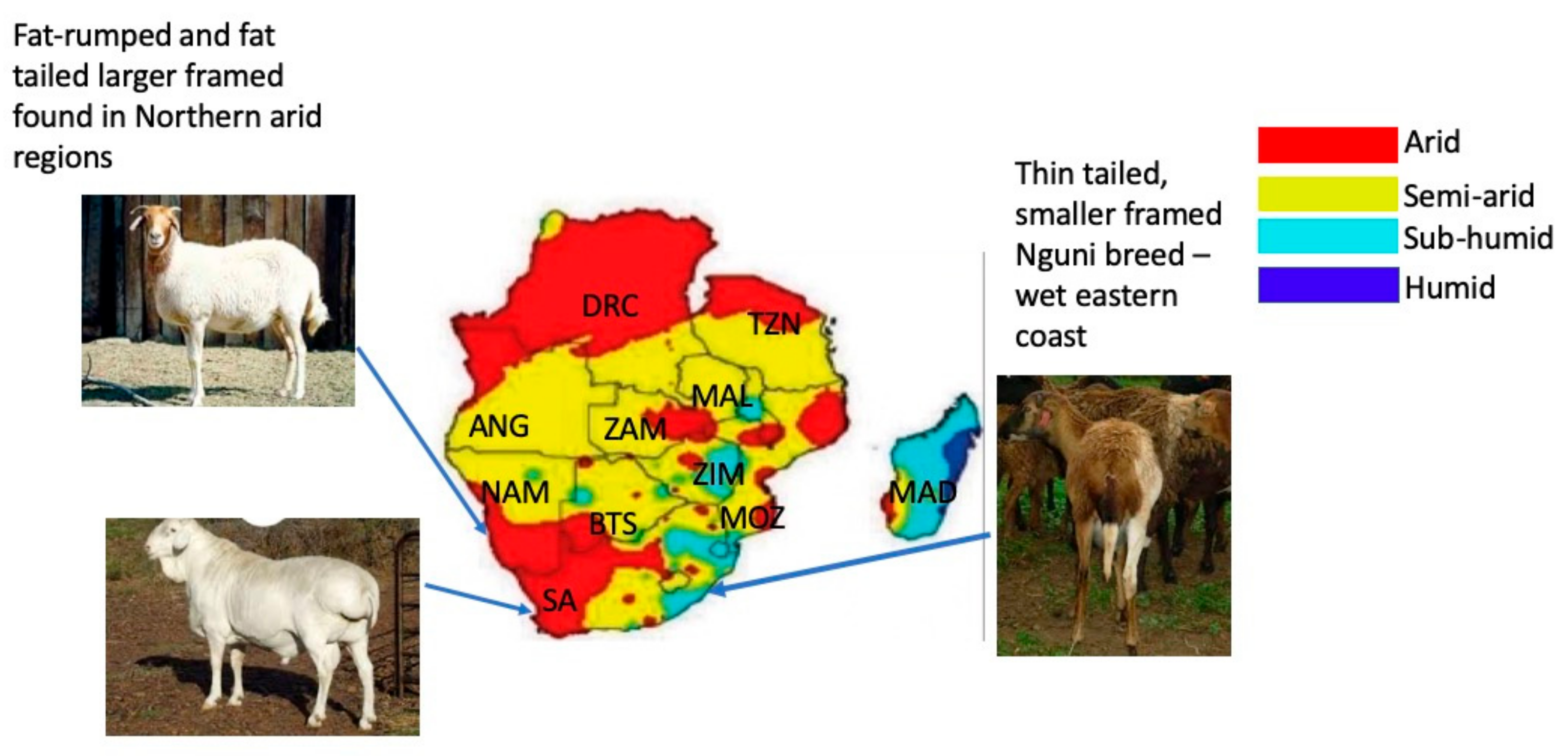
2.2. Phenotypic Characterisation and Traits Important for Selection
3. Sustainable Utilisation of Ovine Genetic Resources
3.1. Implementation of Structured Breeding Programs
3.2. Conservation of Genetic Resources
3.3. Marketing
3.4. Feed Resources
3.5. Health and Diseases
3.6. Gender and Age Issues
4. Possible Ideas for Innovations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Van Marle-Kőster, E.; Visser, C.; Makgahlela, M.; Cloete, S.W.P. Genomic technologies for food security: A review of challenges and opportunities in Southern Africa. Food Res. Int. 2015, 76, 971–979. [Google Scholar] [CrossRef]
- Department of Agriculture, Forestry and Fisheries. Abstract of Agricultural Statistics; Department of Agriculture, Forestry and Fisheries: Pretoria, South Africa, 2018. [Google Scholar]
- African Union—Interafrican Bureau for Animal Resources (AU-IBAR). Strategic Plan 2010–2014; African Union: Nairobi, Kenya, 2009. [Google Scholar]
- Schoeman, S.J.; Cloete, S.W.P.; Olivier, J.J. Returns on investment in sheep and goat breeding in South Africa. Livest. Sci. 2010, 130, 70–82. [Google Scholar] [CrossRef]
- Bijlsma, R.; Loeschcke, V. Genetic erosion impedes adaptive responses to stressful environments. Evol. Appl. 2012, 5, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.M. The Damara in the context of Southern Africa fat-tailed sheep breeds. Trop. Anim. Health Prod. 2011, 43, 1427–1441. [Google Scholar] [CrossRef]
- Lord, J.S.; Hargrove, J.W.; Torr, S.J.; Vale, G.A. Climate change and African trypanosomiasis vector populations in Zimbabwe’s Zambezi valley: A mathematical modelling study. PLoS Med. 2018, 15, e1002675. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.A. Emerging parasite diseases of sheep. Vet. Parasitol. 2012, 189, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Martin, V.; Chevalier, V.; Ceccato, P.; Anyamba, A.; De Simone, L.; Lubroth, J.; de La Rocque, S.; Domenech, J. The impact of climate change on the epidemiology and control of Rift valley fever. Rev. Sci. Technol. Off. Int. Epiz. 2008, 27, 413–426. [Google Scholar] [CrossRef]
- Marai, I.F.M.; El-Darawany, A.A.; Fadiel, A.; Abdel-Hafez, M.A.M. Physiological traits as affected by heat stress in sheep—A review. Small Rumin. Res. 2007, 71, 1–12. [Google Scholar] [CrossRef]
- Goddard, M. Fitness traits in animal breeding programs. In Adaptation and Fitness in Farm Animal Populations; van der Werf, J.H.J., Graser, H.-U., Frankham, R., Gondro, C., Eds.; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Knap, P.W. Breeding robust pigs. Aust. J. Exp. Agric. 2005, 45, 763–773. [Google Scholar] [CrossRef]
- FAO-DAD-IS. Available online: www.fao.org/dad-is/en (accessed on 20 November 2019).
- Soma, P.; Kotze, A.; Grobler, J.P.; Van Wyk, J.B. South African sheep breeds: Population genetic structure and conservation implications. Small Rumin. Res. 2012, 103, 112–119. [Google Scholar] [CrossRef]
- Sandenbergh, L.; Cloete, S.W.P.; Olivier, J.J. Assessing the occurrence of hybridisation in endangered indigenous sheep. In Proceedings of the 11th World Congress on Genetics Applied to Livestock Production, Auckland, New Zealand, 11–16 February 2018. [Google Scholar]
- Sandenbergh, L.; Cloete, S.W.P.; Roodt-Wilding, R.; Snyman, M.A.; Van der Merwe, A.E. Evaluation of the OvineSNP50 chip for use in four South African sheep breeds. S. Afr. J. Anim. Sci. 2016, 46, 89–93. [Google Scholar] [CrossRef]
- Greyvenstein, O.F.C.; Reich, C.M.; Van Marle-Koster, E.; Riley, E.D.G.; Hayes, B.J. Polyceraty (multi-horns) in Damara sheep maps to ovine chromosome 2. Anim. Genet. 2016, 47, 263–266. [Google Scholar] [CrossRef] [PubMed]
- Wilson, T.R. Small Ruminant Production and Small Ruminant Genetic Resources in Tropical Africa; FAO: North Devon, UK, 1991. [Google Scholar]
- Kunene, N.W. Characterisation of Indigenous Zulu (Nguni) Sheep for Utilisation Improvement and Conservation. Ph.D. Thesis, University of Kwa-Zulu Natal, Kwa-Zulu Natal, South Africa, 2010. [Google Scholar]
- Selepe, M.M.; Ceccobelli, S.; Lasagna, E.; Kunene, N.W. Genetic structure of South African Nguni (Zulu) sheep populations reveals admixture with exotic breeds. PLoS ONE 2018, 13, e0196276. [Google Scholar] [CrossRef] [PubMed]
- Molotsi, A.H.; Taylor, J.F.; Cloete, S.W.P.; Muchadeyi, F.; Decker, J.E.; Whitacre, J.K.; Sandenbergh, L.; Dzama, K. Genetic diversity and population structure of South African smallholder farmer sheep breeds determined using the OvineSNP50 beadchip. Trop. Anim. Health Prod. 2017, 49, 1771–1777. [Google Scholar] [CrossRef] [PubMed]
- Cloete, S.W.P.; Olivier, J.J.; Sandenbergh, L.; Snyman, M.A. The adaption of the South Africa sheep industry to new trends in animal breeding and genetics: A review. S. Afr. J. Anim. Sci. 2014, 44, 308–321. [Google Scholar] [CrossRef]
- Norberg, E.; Sørensen, A.C. Inbreeding trend and inbreeding depression in the Danish 1518 populations of Texel, Shropshire, and Oxford Down. J. Anim. Sci. 2007, 85, 299–304. [Google Scholar] [CrossRef]
- Kunene, N.W.; Bezuidenhout, C.C.; Nsahlai, I.V. Genetic and phenotypic diversity in Zulu sheep populations: Implications for exploitation and conservation. Small Rumin. Res. 2009, 84, 100–107. [Google Scholar] [CrossRef]
- Portes, J.V.; Somavilla, A.L.; Grion, A.L.; Dias, L.T.; Teixeira, R.A. Genetic parameters for post-weaning visual scores and reproductive traits in Suffolk sheep. Span. J. Agric. Res. 2018, 16, e04SC01. [Google Scholar] [CrossRef]
- Lemos, M.V.A.; Chiaia, H.L.J.; Berton, M.P.; Feitosa, F.L.B.; Aboujaoude, C.; Venturini, G.C.; Oliveira, L.G.; Albuquerque, H.N.; Baldi, F. Reaction norms for the study of genotype environment interaction for growth and indicator traits of sexual precocity in Nellore cattle. Genet. Mol. Res. 2015, 14, 7151–7162. [Google Scholar] [CrossRef]
- Snyman, M.A.; Buys, T.; Jonker, M.L. Production and reproduction performance of Namaqua Afrikaner sheep. In Proceedings of the 6th Global Conference on the Conservation of Domestic Animal Genetic Resources, Magalies Park, South Africa, 9–13 October 2005. [Google Scholar]
- Cloete, S.W.P.; Cloete, J.J.E.; Scholtz, A.J. Genetic parameters for tick count and udder health in commercial and indigenous ewes in South Africa. Vet. Parasitol. 2016, 230, 33–42. [Google Scholar] [CrossRef]
- Burger, A.; Hoffman, L.C.; Cloete, J.J.E.; Muller, M.; Cloete, S.W.P. Carcass composition of Namaqua Afrikaner, Dorper and SA Mutton Merino ram lambs reared under extensive conditions. S. Afr. J. Anim. Sci. 2013, 43 (Suppl. 1), S27–S32. [Google Scholar] [CrossRef]
- Kilminster, T.F.; Greef, J.C. A note on the reproductive performance of Damara, Dorper and Merino sheep under optimum management and nutrition for Merino ewes in the eastern wheatbelt of Western Australia. Trop. Anim. Health Prod. 2011, 43, 1459–1464. [Google Scholar] [CrossRef] [PubMed]
- Van Harten, S.; Kilminster, T.; Scanlon, T.; Milton, J.; Oldham, C.; Greeff, J.; Almeida, A.M. Fatty acid composition of the ovine longissimus dorsi muscle: Effect of feed restriction in threebreeds of different origin. J. Sci. Food Agric. 2016, 96, 1777–1782. [Google Scholar] [CrossRef] [PubMed]
- Miller, B.A.; Chapwanya, A.; Kilminster, T.; Scanlon, T.; Milton, J.; Osorio, H.; Oldham, C.; Greeff, J.; Bergfelt, D.R.; Campos, A.M.O.; et al. The ovine hepatic mitochondrial proteome: Understanding seasonal weight loss tolerance in two distinct breeds. PLoS ONE 2019, 14, e0212580. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, K.; Abba, Y.; Ramli, N.S.B.; Marimuthu, M.; Omar, M.A.; Abdullah, F.F.J.; Sadiq, M.A.; Tijjani, A.; Chung, E.L.T.; Lila, M.A.M. The use of FAMACHA in estimation of gastrointestinal nematodes and total worm burden in Damara and Barbados Blackbelly cross sheep. Trop. Anim. Health Prod. 2016, 48, 1013–1020. [Google Scholar] [CrossRef]
- Van Harten, S.; Brito, R.; Almeida, A.M.; Scanlon, T.; Kilminster, T.; Milton, J.; Greeff, J.; Oldham, C.; Cardoso, L.A. Gene expression of regulatory enzymes involved in the intermediate metabolism of sheep subjected to feed restriction. Animal 2013, 7, 439–445. [Google Scholar] [CrossRef]
- Palma, M.; Scanlon, T.; Kilminster, T.; Milton, J.; Oldham, C.; Greeff, J.; Matzapetakis, M.; Almeida, A.M. The hepatic and skeletal muscle ovine metabolomes as affected by weightloss: A study in three sheep breeds using NMR-metabolomics. Sci. Rep. 2016, 6, 39120. [Google Scholar] [CrossRef]
- Matika, O.; Van Wyk, J.B.; Erasmus, G.J.; Baker, R.L. Phenotypic and genetic relationships between lamb and ewe traits for the Sabi sheep of Zimbabwe. S. Afr. J. Anim. Sci. 2001, 31, 215–222. [Google Scholar] [CrossRef]
- Rocha, A.; McKinnon, D.; Wilson, R.T. Comparative performance of Landim and Blackhead Persian sheep in Mozambique. Small Rum. Res. 1990, 3, 527–538. [Google Scholar] [CrossRef]
- Molotsi, A.H.; Dube, B.; Oosting, S.; Marandure, T.; Mapiye, C.; Cloete, S.W.P.; Dzama, K. Genetic traits of relevance to sustainability of smallholder sheep farming systems in South Africa. Sustainability 2017, 9, 1225. [Google Scholar] [CrossRef]
- Nigussie, H.; Legesse, Y.; Pal, S.K. Domestic Animal Diversity Information System (DAD-IS): Its contribution for Animal Genetic Resources (AnGR) management. In Proceedings of the 19th Annual Conference of Ethiopian Society of Animal Production (ESAP), Addis Ababa, Ethiopia, 15–17 December 2011; pp. 17–21. [Google Scholar]
- Haile, A.; Gizaw, S.; Getachew, T.; Rischkowsky, B. Challenges in small ruminant breeding programs and resulting investment priorities in Ethiopia. In Proceedings of the World Congress on Genetics Applied to Livestock Production, Auckland, New Zealand, 11–16 February 2018. [Google Scholar]
- Gizaw, S.; Komen, H.; Windig, J.J.; Hanotte, O.; Van Arendonk, J.A.M. Conservation priorities for Ethiopian sheep breeds combining threat status, breed merits and contributions to genetic diversity. Genet. Sel. Evol. 2008, 40, 433–447. [Google Scholar] [PubMed]
- Qwabe, S.O.; Van Marle-Köster, E.; Visser, C. Genetic diversity and population structure of the endangered Namaqua Afrikaner sheep. Trop. Anim. Health Prod. 2013, 45, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Mavule, B.S.; Sarti, F.M.; Lasagna, E.; Kunene, N.W. Morphological differentiation amongst Zulu sheep populations in KwaZulu-Natal, South Africa, as revealed by multivariate analysis. Small Rumin. Res. 2016, 140, 50–56. [Google Scholar] [CrossRef]
| Breeds | SADC Country | Exotic/Indigenous/Synthethic | Risk Level | Population Numbers Minimum (Updated between 1990–2018) | Population Numbers Maximum (Updated between 1990–2018) |
|---|---|---|---|---|---|
| Africaner | South Africa | Indigenous | Unknown | Unknown | Unknown |
| Afrino | South Africa | Synthetic (South African Mutton Merino 50%, Blinkhaar Afrikaner 25%, Merino 25%) | Unknown | 1000 | 3141 |
| Angola long-legged | Angola | Indigenous | Unknown | Unknown | Unknown |
| Angola maned | Angola | Indigenous | Unknown | Unknown | Unknown |
| Bahu | Democratic republic of Congo | Indigenous | Unknown | Unknown | Unknown |
| Baluba | Democratic republic of Congo | Indigenous | Unknown | Unknown | Unknown |
| Basotho sheep | Lesotho | Indigenous | Unknown | Unknown | Unknown |
| Bezuidenhout | South Africa | Synthetic/Indigenous (derived from Ronderib Afrikaner and wooled Persian in 1917) | Unknown | 20 | 100 |
| Blackheaded Persian | Angola, South Africa, Zimbabwe, Botswana, Namibia, Mozambique, Tanzania, Mauritius | Indigenous (transboundary breed) | Not at risk | 12,100 | Unknown |
| Blinkhaar Ronderib Afrikaner | South Africa, Namibia | Indigenous | Unknown | 1000 | Unknown |
| Boesmanlander | South Africa | Developed breed | Unknown | Unknown | Unknown |
| Damara | South Africa, Botswana, Namibia, | Indigenous (transboundary breed) | Not at risk | 6100 | 20,114 |
| Dormer | South Africa | Synthetic (transboundary breed) | Not at risk | 1000 | 35,707 |
| Dorper | South Africa, Zimbabwe, Malawi, Botswana, Namibia, Angola, Swaziland and Mauritius | Synthetic/Indigenous (transboundary) | Not at risk | 606,100 | 1304,810 |
| Dohne Merino | South Africa, Namibia | Synthetic (transboundary) | Not at risk | 1000 | 316,454 |
| Hottentot | South Africa | Indigenous | Extinct | 0 | 0 |
| Karakul | South Africa, Botswana, Namibia, Mozambique, Angola, | Exotic (transboundary breed) | Not at risk | 160,120 | 240,785 |
| Meatmaster | South Africa, Namibia | Synthetic | At risk | 3000 | 15,927 |
| Man’gati | Tanzania | Indigenous | Not at risk | 5000 | 10,000 |
| Merino | South Africa, Malawi, Lesotho, Angola | Exotic | Unknown | 15,000,000 | Unknown |
| Mondombes | Angola | Indigenous | Unknown | Unknown | Unknown |
| Merino Landsheep | South Africa | Not at risk | 1000 | 25,294 | |
| Mouton indigène moyen à courte patte | Democratic republic of Congo | Indigenous | Unknown | Unknown | Unknown |
| Mouton indigène à longue patte | Democratic republic of Congo | Indigenous | Unknown | Unknown | Unknown |
| Namaqua Afrikaner | South Africa | Indigenous | Unknown | 100 | 1000 |
| Nguni | South Africa, Malawi, Mozambique, Swaziland | Indigenous | Unknown | 109,800 | Unknown |
| Pedi | South Africa | Indigenous | At risk | 120 | 342 |
| Ronderib Afrikaner | South Africa | Indigenous | Unknown | 10,000 | Unknown |
| Ronderib Merino | South Africa | indigenous | Unknown | Unknown | Unknown |
| South African Mutton Merino | South Africa, Zimbabwe, Namibia | Developed in SA (transboundary breed) | Not at risk | 1000 | 258,647 |
| Steekhaar | South Africa | Indigenous | Unknown | 100 | Unknown |
| Van Rooy | South Africa, Zimbabwe, Namibia | Synthetic—(Ronderib Africander and Rambouillet) (transboundary) | At risk | 1000 | 1081 |
| Vandor | South Africa | Synthetic—(Rambouillet and Van Rooy) | Unknown | 100 | Unknown |
| Walrich Vleismerino | South Africa | Developed in SA | Extinct | 0 | 0 |
| White Dorper | South Africa | Synthetic | Unknown | Unknown | Unknown |
| White wooled Mountain | South Africa | Developed in SA | Extinct | 0 | 0 |
| Wooled Persian | South Africa | Origin from Arabia | Extinct | 0 | 0 |
| Sabi | Zimbabwe | Indigenous (transboundary) | At risk locally | 50 | 300 |
| Tswana | Zimbabwe, Botswana | Indigenous (transboundary) | Not at risk | 250,000 in Zim | Unknown |
| Wiltiper | Zimbabwe | Synthetic (Wiltshire horn and blackhead Persian) | Unknown | 12,000 | Unknown |
| Primitif | Comoros | Indigenous | Unknown | Unknown | Unknown |
| Gellaper | Namibia | Indigenous | At risk | 100 | 2000 |
| Veldmaster | Namibia | Indigenous | At risk | 2000 | 6000 |
| Landim | Mozambique | Indigenous | Unknown | 150,000 | Unknown |
| East African Blackheaded | Tanzania | Indigenous | Not at risk | 800,000 | 1979,952 |
| Gogo Tanzania long-tailed | Tanzania | Indigenous | Not at risk | 3000,000 | 5182,627 |
| Kasulu | Tanzania | Indigenous | Not at risk | 200,000 | 300,000 |
| Maasai | Tanzania | Indigenous | Not at risk | 600,000 | 1500,000 |
| Rodrigaise | Mauritius | Indigenous | Unknown | 780 | Unknown |
| Sukuma | Tanzania | Indigenous (transboundary) | Not at risk | 1000,000 | 1500,000 |
| Tanzania Long-tailed | Tanzania | Indigenous (transboundary) | Not at risk | 2000 | 3000 |
| Urambo | Tanzania | Indigenous | Not at risk | 100,000 | 200,000 |
| Zunu | Angola | Indigenous | Unknown | Unknown | Unknown |
| What | How | Who | When |
|---|---|---|---|
| Community-based breeding programme | Two nucleus flocks to be established
Two systems will be implemented and run parallel, namely (i) conservation and (ii) terminal crossbreeding. The conservation system will be done to maintain the indigenous breeds, and the terminal will be for marketing the crossbreds with improved weight. |
|
|
| Nutrition for animals |
|
| Five years |
| Land availability | Land will be made available for
|
| Two years |
| Health and diseases |
|
| Five years for farmers to be self-reliant |
| Gender and age issues |
|
| Five to ten years |
| Market access |
|
| Five years to build market facilities and to ensure contracts for farmers |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molotsi, A.H.; Dube, B.; Cloete, S.W.P. The Current Status of Indigenous Ovine Genetic Resources in Southern Africa and Future Sustainable Utilisation to Improve Livelihoods. Diversity 2020, 12, 14. https://doi.org/10.3390/d12010014
Molotsi AH, Dube B, Cloete SWP. The Current Status of Indigenous Ovine Genetic Resources in Southern Africa and Future Sustainable Utilisation to Improve Livelihoods. Diversity. 2020; 12(1):14. https://doi.org/10.3390/d12010014
Chicago/Turabian StyleMolotsi, Annelin Henriehetta, Bekezela Dube, and Schalk Willem Petrus Cloete. 2020. "The Current Status of Indigenous Ovine Genetic Resources in Southern Africa and Future Sustainable Utilisation to Improve Livelihoods" Diversity 12, no. 1: 14. https://doi.org/10.3390/d12010014
APA StyleMolotsi, A. H., Dube, B., & Cloete, S. W. P. (2020). The Current Status of Indigenous Ovine Genetic Resources in Southern Africa and Future Sustainable Utilisation to Improve Livelihoods. Diversity, 12(1), 14. https://doi.org/10.3390/d12010014





