Stable Isotope Analyses of Multiple Tissues of Great Shearwaters (Ardenna Gravis) Reveals Long-Term Dietary Stability, Short-Term Changes in Diet, and Can be Used as a Tool to Monitor Food Webs
Abstract
1. Introduction
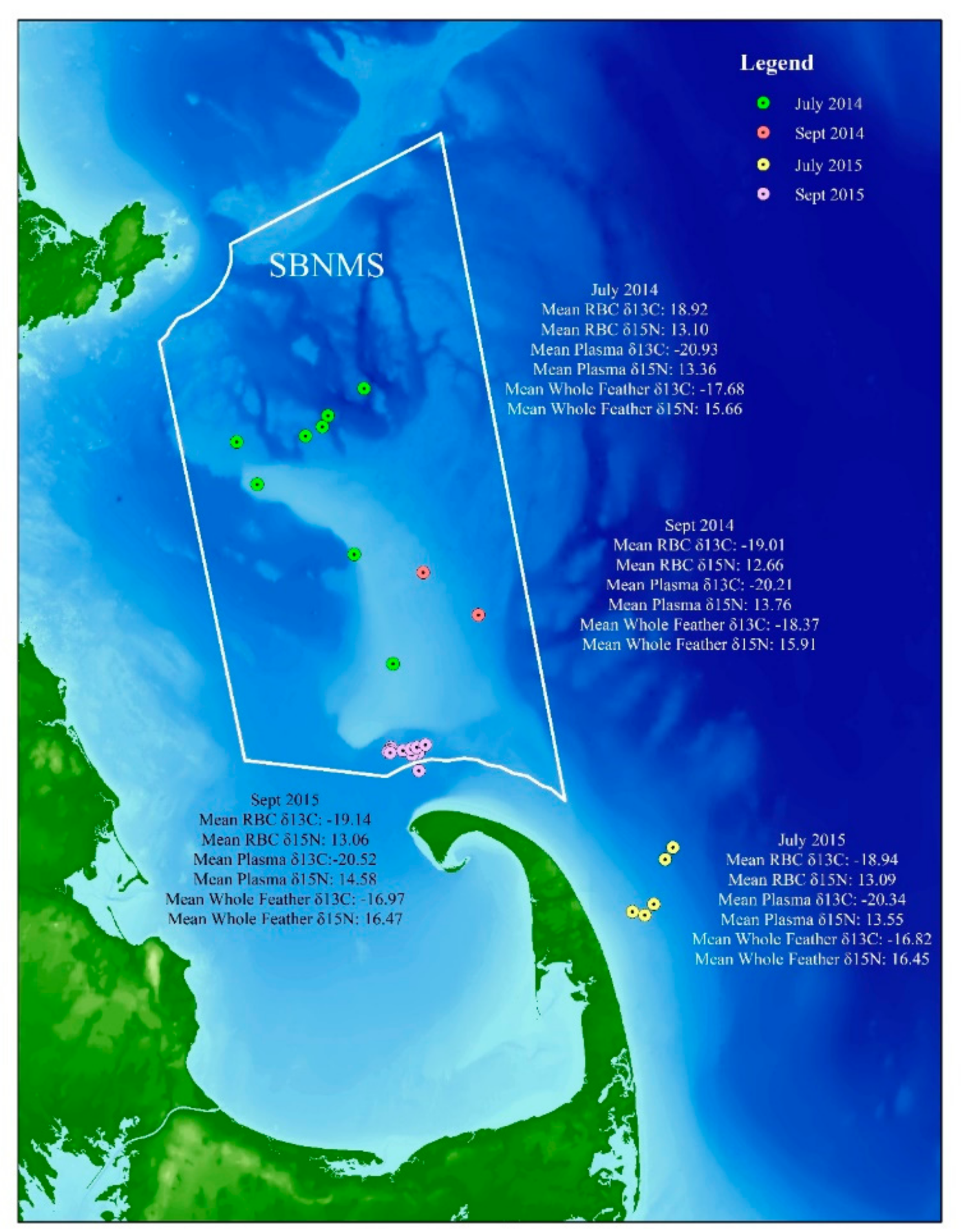
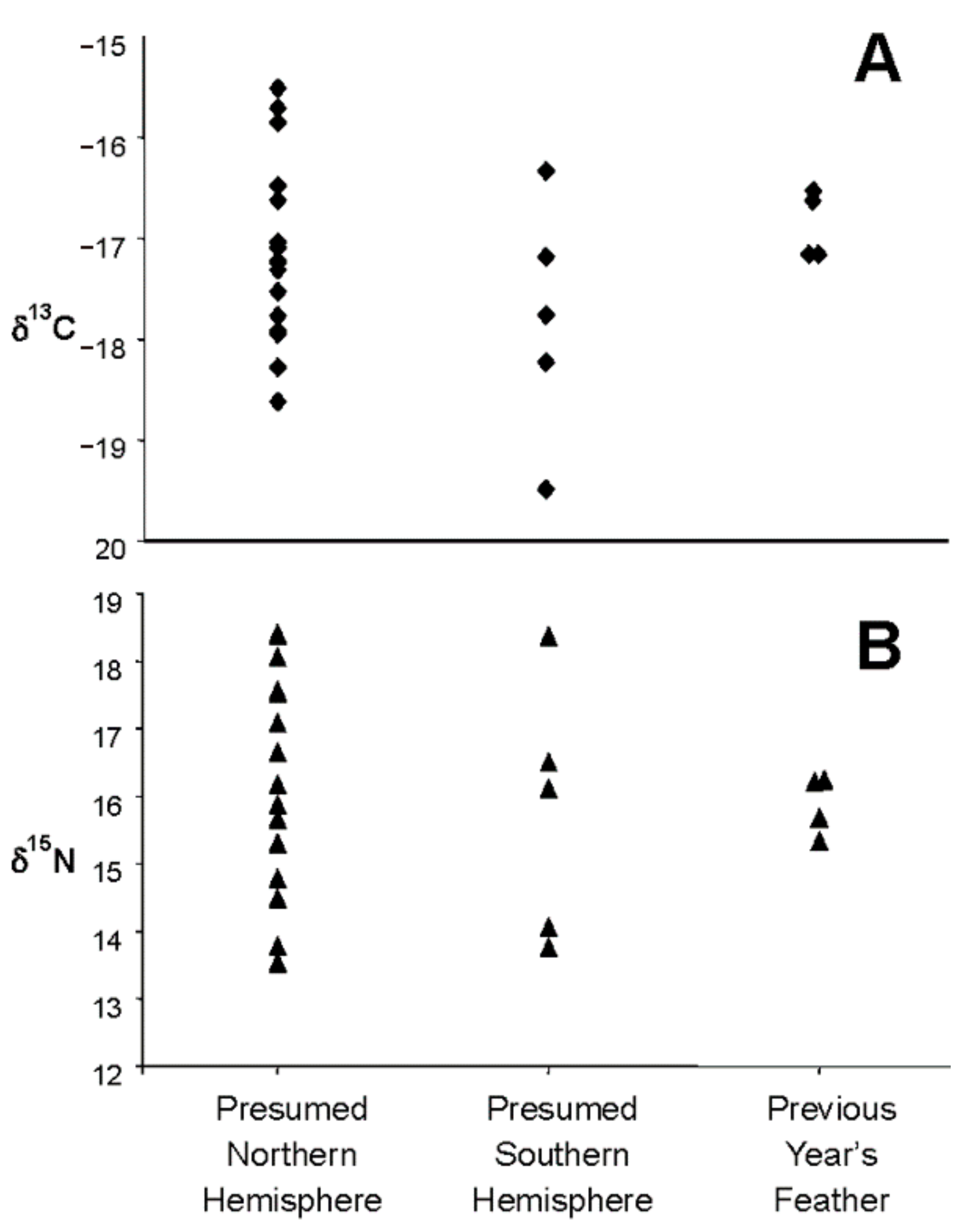
- Great shearwaters that winter in the waters of Massachusetts Bay and Cape Cod may molt the sixth primary (p6) flight feather in either the Southern or Northern Hemisphere.
- The diet of great shearwaters and the portion of the food web they sample varies between years.
- The diet of great shearwaters and the portion of the food web they sample varies seasonally or by location.
- If the molt of the p6 feather is variable and is molted by some birds in the South Atlantic and other birds in the North Atlantic, we should see evidence of a bimodal distribution in the δ13C of the feathers, reflecting the difference in signal between great shearwater feathers grown in northern and Southern Hemispheres. Sixth primary feathers of birds whose molt was completed when captured in July should have molted the p6 feather in the Southern Hemisphere and therefore have relatively enriched δ13C-values, whereas the p6 feathers of birds whose molt was not completed by the July capture should have molted in the Northern Hemisphere and therefore be relatively depleted in 13C [37,47].
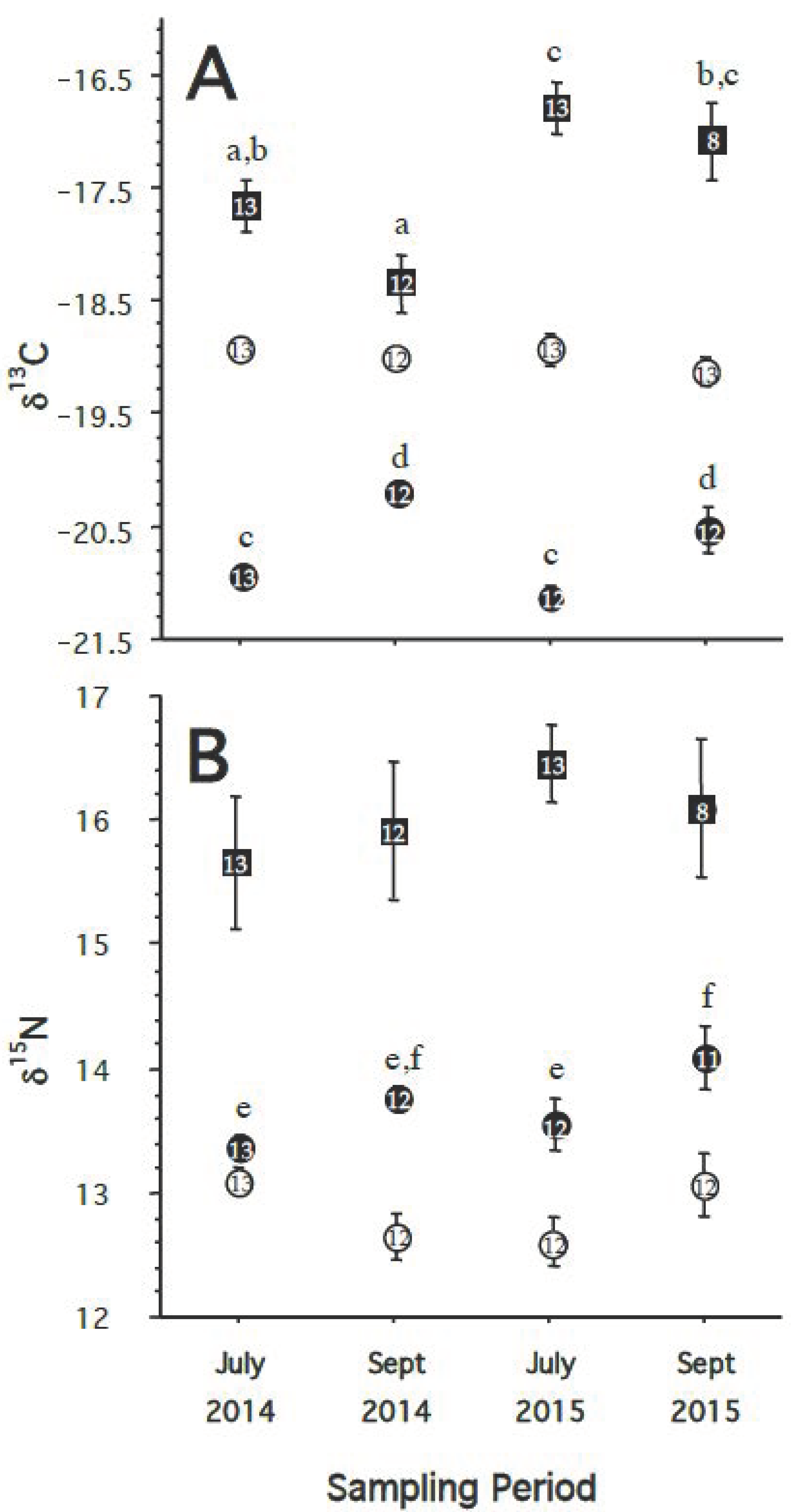
- If the food web and diet of great shearwaters in Massachusetts Bay and the waters off Cape Cod differs from year to year, then we predict there will be a significant difference in both δ13C and δ15N of the RBCs, plasma, and/or feathers of birds caught in different years.
- If there are changes in the diet and food web on a seasonal basis or between individual sampling locations, we would expect to see changes in the δ13C and δ15N of blood plasma (shorter turnover rate) but not in the δ13C and δ15N of RBCs (longer turnover rate). Consequently, when comparing tissues, we predict that the Δ13Cplasma-RBC and Δ15Nplasma-RBC would also differ significantly between capture events. Likewise, we predict that the Δ13Cfeather-RBC and Δ15Nfeather-RBC would differ significantly between capture events.
2. Materials and Methods
2.1. Capture
2.2. Field Data Collection
2.3. Stable Isotope Analysis
2.4. Statistical Analysis
3. Results
3.1. Hypothesis 1: Sixth Primary Feather is Molted in Either the Southern or Northern Hemisphere
3.2. Hypothesis 2: Diet/Food Web Changes between Years
3.3. Hypothesis 3: Diet/Food Web Changes by Season/Location
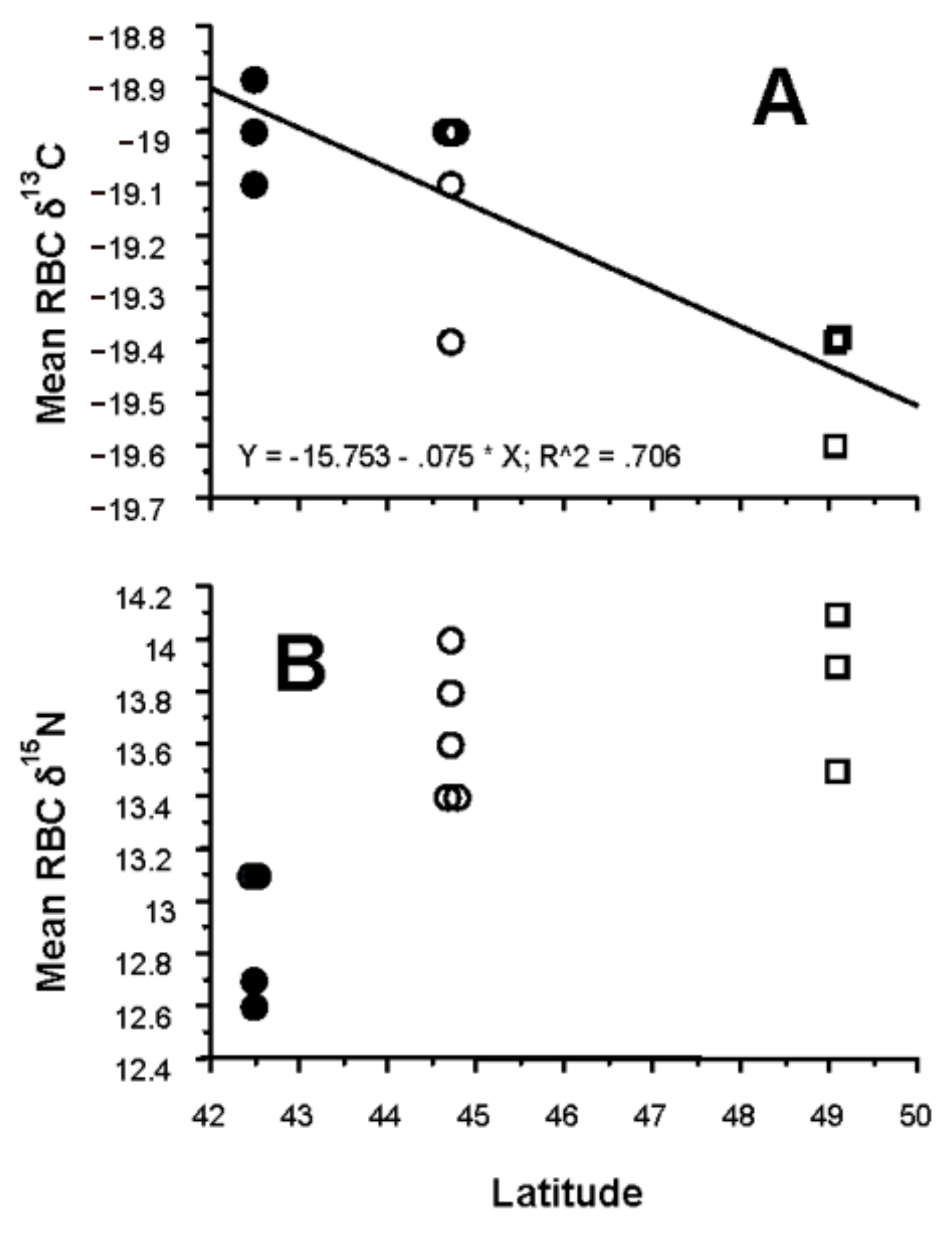
3.4. Latitudinal Gradient
4. Discussion and Conclusions
4.1. Hypothesis 1: Sixth Primary Feather is Molted in Either the Southern or Northern Hemisphere
4.2. Hypothesis 2: Diet/Food Web Changes between Year
4.3. Hypothesis 3: Diet/Food Web Changes by Season/Location
4.4. Latitudinal Gradient
4.5. Food Webs
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thompson, C. State of the Gulf of Maine Report; Gulf of Maine Council on the Marine Environment: Ogunquit, ME, USA, 2010; pp. 1–56. [Google Scholar]
- Boscolo-Galazzo, F.; Crichton, K.A.; Barker, S.; Pearson, P.N. Temperature dependency of metabolic rates in the upper ocean: A positive feedback to global climate change? Glob. Planet. Chang. 2018, 170, 201–212. [Google Scholar] [CrossRef]
- Northeast Multispecies (Large Mesh/Groundfish) Fishery Management Plan; New England Fishery Management Council: Newburyport, MA, USA, 2006; Available online: http://archive.nefmc.org/nemulti/summary/large_mesh_multi.pdf (accessed on 31 August 2018).
- Drinkwater, K.F.; Mueter, F.; Friedland, K.D.; Taylor, M.; Hunt, G.L., Jr.; Hare, J.; Melle, W. Recent climate forcing and physical oceanographic changes in Northern Hemisphere regions: A review and comparison of four marine ecosystems. Prog. Oceanogr. 2009, 81, 10–28. [Google Scholar] [CrossRef]
- Salisbury, J.E.; Jönsson, B.F. Rapid warming and salinity changes in the Gulf of Maine alter surface ocean carbonate parameters and hide ocean acidification. Biogeochemistry 2018, 141, 401–418. [Google Scholar] [CrossRef] [PubMed]
- Polis, G.A.; Strong, D.R. Food web complexity and community dynamics. Am. Nat. 1996, 147, 813–846. [Google Scholar] [CrossRef]
- Ainley, D.G.; Santora, J.A.; Capitolo, P.J.; Field, J.C.; Beck, J.N.; Carle, R.D.; Donnelly-Greenan, E.; McChesney, G.J.; Elliott, M.; Bradley, R.W.; et al. Ecosystem-based management affecting Brandt’s cormorant resources and populations in the Central California current region. Biol. Conserv. 2018, 217, 407–418. [Google Scholar] [CrossRef]
- Perkins, A.; Ratcliffe, N.; Suddaby, D.; Ribbands, B.; Smith, C.; Ellis, P.; Meek, E.; Bolton, M. Combined bottom-up and top-down pressures drive catastrophic population declines of Arctic skuas in Scotland. J. Anim. Ecol. 2018, 87, 1573–1586. [Google Scholar] [CrossRef] [PubMed]
- Donohue, I.; Petchey, O.L.; Kéfi, S.; Génin, A.; Jackson, A.L.; Yang, Q.; O’Connor, N.E. Loss of predator species, not intermediate consumers, triggers rapid and dramatic extinction cascades. Glob. Chang. Biol. 2017, 23, 2962–2972. [Google Scholar] [CrossRef]
- Sanders, D.; Thébault, E.; Kehoe, R.; Frank van Veen, F.J. Trophic redundancy reduces vulnerability to extinction cascades. Proc. Natl. Acad. Sci. USA 2018, 115, 2419–2424. [Google Scholar] [CrossRef]
- Kostrzewa, R. Puffins, climate change and fisheries. Biol. Unserer Zeit 2015, 45, 322–329. [Google Scholar] [CrossRef]
- Berg, S.; Pimenov, A.; Palmer, C.; Emmerson, M.; Jonsson, T. Ecological communities are vulnerable to realistic extinction sequences. Oikos 2015, 124, 486–496. [Google Scholar] [CrossRef]
- Gao, X.; Chen, X.; Li, Y. A review on the methods used in aquatic food web research: Development and applications. J. Fish. Sci. China 2018, 25, 1347–1360. [Google Scholar] [CrossRef]
- Cucherousset, J.; Bouletreau, S.; Martino, A.; Roussel, J.M.; Santoul, F. Using stable isotope analyses to determine the ecological effects of non-native fishes. Fish. Manag. Ecol. 2012, 19, 111–119. [Google Scholar] [CrossRef]
- Franco, M.A.L.; Carvalho, M.C.; de Rezende, C.E. A revision on the use of multiple tools for trophic studies of fish communities. Neotrop. Biol. Conserv. 2014, 9, 172–185. [Google Scholar]
- Layman, C.A.; Araujo, M.S.; Boucek, R.; Hammerschlag-Peyer, C.M.; Harrison, E.; Jud, Z.R.; Matich, P.; Rosenblatt, A.E.; Vaudo, J.J.; Yeager, L.A.; et al. Applying stable isotopes to examine food-web structure: An overview of analytical tools. Biol. Rev. 2012, 87, 545–562. [Google Scholar] [CrossRef]
- Wang, X.; Jiang, H.X.; Zhang, Y.N. Application of stable isotope analyses to avian diets and trophic structure. Shengtai Xuebao Acta Ecol. Sin. 2015, 35, 5556–5569. [Google Scholar]
- Minagawa, M.; Wada, E. Stepwise enrichment of 15N along food chains: Further evidence and the relation between 15N and animal age. Geochim. Cosmochim. Acta 1984, 48, 1135–1140. [Google Scholar] [CrossRef]
- Vanderklift, M.A.; Ponsard, S. Sources of variation in consumer-diet δ15N enrichment: A meta-analysis. Oecologia 2003, 136, 169–182. [Google Scholar] [CrossRef]
- Post, D.M. Using stable isotopes to estimate trophic position: Models, methods, and assumptions. Ecology 2002, 83, 703–718. [Google Scholar] [CrossRef]
- Schoeninger, M.J.; DeNiro, M.J. Nitrogen and carbon isotopic composition of bone collagen from marine and terrestrial animals. Geochim. Cosmochim. Acta 1984, 48, 625–639. [Google Scholar] [CrossRef]
- Hobson, K.A.; Piatt, J.F.; Pitocchelli, J. Using stable isotopes to determine seabird trophic relationships. J. Anim. Ecol. 1994, 63, 786–798. [Google Scholar] [CrossRef]
- Hobson, K.A.; Gilchrist, G.; Falk, K. Isotopic investigations of seabirds of the North Water Polynya: Contrasting trophic relationships between the eastern and western sectors. Condor 2002, 104, 1–11. [Google Scholar] [CrossRef]
- Gagne, T.O.; David Hyrenbach, K.; Hagemann, M.E.; Van Houtan, K.S. Trophic signatures of seabirds suggest shifts in oceanic ecosystems. Sci. Adv. 2018, 4, eaao3946. [Google Scholar] [CrossRef]
- Montevecchi, W.A.; Myers, A. Dietary changes of seabirds indicate shifts in pelagic food webs. Sarsia 1996, 80, 313–322. [Google Scholar] [CrossRef]
- Montevecchi, W.A. Binary dietary responses of northern gannets Sula bassana indicate changing food web and oceanographic conditions. Mar. Ecol. Prog. Ser. 2007, 352, 213–220. [Google Scholar] [CrossRef]
- Furness, R.W. A review of seabird responses to natural or fisheries-induced changes in food supply. Aquat. Predat. Prey 1996, 166–173. [Google Scholar]
- Furness, R.W.; Camphuysen, K. Seabirds as monitors of the marine environment. ICES J. Mar. Sci. 1997, 54, 726–737. [Google Scholar] [CrossRef]
- Lauria, V.; Attrill, M.J.; Brown, A.; Edwards, M.; Votier, S.C. Regional variation in the impact of climate change: Evidence that bottom-up regulation from plankton to seabirds is weak in parts of the Northeast Atlantic. Mar. Ecol. Prog. Ser. 2013, 488, 11–22. [Google Scholar] [CrossRef]
- Lauria, V.; Attrill, M.J.; Pinnegar, J.K.; Brown, A.; Edwards, M.; Votier, S.C. Influence of climate change and trophic coupling across four trophic levels in the Celtic Sea. PLoS ONE 2012, 7, e47408. [Google Scholar] [CrossRef]
- Gagné, T.O.; Hyrenbach, K.D.; Hagemann, M.E.; Bass, O.L.; Pimm, S.L.; MacDonald, M.; Peck, B.; Van Houtan, K.S. Seabird trophic position across three ocean regions tracks ecosystem differences. Front. Mar. Sci. 2018, 5, 317. [Google Scholar] [CrossRef]
- Powers, K.D.; Wiley, D.N.; Allyn, A.J.; Welch, L.J.; Ronconi, R.A. Movements and foraging habitats of great shearwaters Puffinus gravis in the Gulf of Maine. Mar. Ecol. Prog. Ser. 2017, 574, 211–226. [Google Scholar] [CrossRef]
- Cuthbert, R.J. Breeding biology, chick growth and provisioning of great shearwaters (Puffinus gravis) at Gough Island, South Atlantic Ocean. Emu 2005, 105, 305–310. [Google Scholar] [CrossRef]
- Gulka, J.; Carvalho, P.C.; Jenkins, E.; Johnson, K.; Maynard, L.; Davoren, G.K. Dietary niche shifts of multiple marine predators under varying prey availability on the northeast Newfoundland coast. Front. Mar. Sci. 2017, 4, 324. [Google Scholar] [CrossRef]
- Ronconi, R.A.; Koopman, H.N.; McKinstry, C.A.E.; Wong, S.N.P.; Westgate, A.J. Figure. Ecol. Prog. Ser. 2010, 419, 267–282. [Google Scholar] [CrossRef]
- Brown, R.G.B.; Barker, S.P.; Gaskin, D.E.; Sandeman, M.R. The foods of great and sooty shearwaters Puffinus gravis and P. Griseus in Eastern Canadian Waters. Ibis 1981, 123, 19–30. [Google Scholar] [CrossRef]
- Haman, K.H.; Norton, T.M.; Ronconi, R.A.; Nemeth, N.M.; Thomas, A.C.; Courchesne, S.J.; Al, S.; Kevin Keel, M. Great shearwater (Puffinus gravis) mortality events along the eastern coast of the United States. J. Wildl. Dis. 2013, 49, 235–245. [Google Scholar] [CrossRef]
- Lee, D.S. Mass die-offs of greater shearwaters in the western North Atlantic: Effects of weather patterns on mortality of a trans-equatorial migrant. Chat 2009, 73, 37–47. [Google Scholar]
- Ronconi, R.A.; Ryan, P.G.; Ropert-Coudert, Y. Diving of great shearwaters (Puffinus gravis) in cold and warm water regions of the South Atlantic Ocean. PLoS ONE 2010, 5, e15508. [Google Scholar] [CrossRef]
- Woods, R.W. Great shearwater Puffinus gravis breeding in the Falkland Islands. Ibis 1970, 112, 259–260. [Google Scholar] [CrossRef]
- Warham, J. The Petrels, Their Ecology and Breeding Systems; Academic Press: London, UK, 1990. [Google Scholar]
- Voous, K.H.; Wattel, J. Distribution and migration of greater shearwaters. Ardea 1963, 51, 143–157. [Google Scholar]
- Huettmann, F.; Diamond, A.W. Seabird migration in the Canadian Northwest Atlantic Ocean: Moulting locations and movement patterns of immature birds. Can. J. Zool. 2000, 78, 624–647. [Google Scholar] [CrossRef]
- Brown, R.G.B. The wing-moult of fulmars and shearwaters (Procellariidae) in Canadian Atlantic waters. Can. Field-Nat. 1988, 102, 203–208. [Google Scholar]
- Harrison, P. Seabirds: An Identification Guide, 2nd ed.; Houghton Mifflin Company: Boston, MA, USA, 1985. [Google Scholar]
- Bugoni, L.; Naves, L.C.; Furness, R.W. Moult of three Tristan da Cunha seabird species sampled at sea. Antarct. Sci. 2014, 27, 240–251. [Google Scholar] [CrossRef]
- Quillfeldt, P.; Bugoni, L.; McGill, R.A.R.; Masello, J.F.; Furness, R.W. Differences in stable isotopes in blood and feathers of seabirds are consistent across species, age and latitude: Implications for food web studies. Mar. Biol. 2008, 155, 593–598. [Google Scholar] [CrossRef]
- Navarro, J.; González-Solís, J.; Viscor, G. Nutritional and feeding ecology in Cory’s shearwater Calonectris diomedea during breeding. Mar. Ecol. Prog. Ser. 2007, 351, 261–271. [Google Scholar] [CrossRef]
- Paiva, V.H.; Xavier, J.; Geraldes, P.; Ramirez, I.; Garthe, S.; Ramos, J.A. Foraging ecology of Cory’s shearwaters in different oceanic environments of the North Atlantic. Mar. Ecol. Prog. Ser. 2010, 410, 257–268. [Google Scholar] [CrossRef]
- Kramer, M.H.; Harris, D.J. Avian blood collection. J. Exot. Pet. Med. 2010, 19, 82–86. [Google Scholar] [CrossRef]
- Ginn, H.B.; Melville, D.S. Moult in birds. BTO Guide 1983, 19, 1–112. [Google Scholar]
- Bearhop, S.; Waldron, S.; Votier, S.C.; Furness, R.W. Factors that influence assimilation rates and fractionation of nitrogen and carbon stable isotopes in avian blood and feathers. Physiol. Biochem. Zool. 2002, 75, 451–458. [Google Scholar] [CrossRef]
- Bond, A.L.; Jones, I.L. A practical introduction to stable-isotope analysis for seabird biologists: Approaches, cautions and caveats. Mar. Ornithol. 2009, 37, 183–188. [Google Scholar]
- Pyle, P. Identification Guide to North American Birds. Part II: Anatidae to Alcidae; Slate Creek Press: Point Reyes Station, CA, USA, 2008. [Google Scholar]
- Klaassen, M.; Piersma, T.; Korthals, H.; Dekinga, A.; Dietz, M.W. Single-point isotope measurements in blood cells and plasma to estimate the time since diet switches. Funct. Ecol. 2010, 24, 796–804. [Google Scholar] [CrossRef]
- Phillips, D.L.; Eldridge, P.M. Estimating the timing of diet shifts using stable isotopes. Oecologia 2006, 147, 195–203. [Google Scholar] [CrossRef]
- Ainley, D.G.; Lewis, T.J.; Morrell, S. Molt in Leach’s and ashy storm-petrels. Wilson Bull. 1976, 88, 76–95. [Google Scholar]
- Rohwer, S.; Ricklefs, R.E.; Rohwer, V.G.; Copple, M.M. Allometry of the duration of flight feather molt in birds. PLoS Biol. 2009, 7, e1000132. [Google Scholar] [CrossRef]
- Evans Ogden, L.J.; Hobson, K.A.; Lank, D.B. Blood isotopic (δ13c and δ15n) turnover and diet-tissue fractionation factors in captive dunlin (Calidris alpina pacifica). Auk 2004, 121, 170–177. [Google Scholar] [CrossRef]
- Bennettand, P.M.; Harvey, P.H. Active and resting metabolism in birds: Allometry, phylogeny and ecology. J. Zool. 1987, 213, 327–344. [Google Scholar] [CrossRef]
- Nagy, K.A. Field metabolic rate and food requirement scaling in mammals and birds. Ecol. Monogr. 1987, 57, 111–128. [Google Scholar] [CrossRef]
- Carleton, S.A.; del Rio, C.M. The effect of cold-induced increased metabolic rate on the rate of 13C and 15N incorporation in house sparrows (Passer domesticus). Oecologia 2005, 144, 226–232. [Google Scholar] [CrossRef]
- Guglielmo, C.G.; Gerson, A.R.; Price, E.R.; Hays, Q.R. The effects of dietary macronutrients on flight ability, energetics, and fuel metabolism of yellow-rumped warblers Setophaga coronata. J. Avian Biol. 2017, 48, 133–148. [Google Scholar] [CrossRef]
- Nagy, K.A. Field metabolic rate and body size. J. Exp. Biol. 2005, 208, 1621–1625. [Google Scholar] [CrossRef]
- Podlesak, D.W.; McWilliams, S.R.; Hatch, K.A. Stable isotopes in breath, blood, feces and feathers can indicate intra-individual changes in the diet of migratory songbirds. Oecologia 2005, 142, 501–510. [Google Scholar] [CrossRef]
- Hobson, K.A.; Clark, R.G. Turnover of 13C in cellular and plasma fractions of blood: Implications for nondestructive sampling in avian dietary studies. Auk 1993, 110, 638–641. [Google Scholar] [CrossRef]
- Hahn, S.; Hoye, B.J.; Korthals, H.; Klaassen, M. From food to offspring down: Tissue-specific discrimination and turn-over of stable isotopes in herbivorous waterbirds and other avian foraging guilds. PLoS ONE 2012, 7, e30242. [Google Scholar] [CrossRef]
- Lourenço, P.M.; Granadeiro, J.P.; Guilherme, J.L.; Catry, T. Turnover rates of stable isotopes in avian blood and toenails: Implications for dietary and migration studies. J. Exp. Mar. Biol. Ecol. 2015, 472, 89–96. [Google Scholar] [CrossRef]
- Hobson, K.A. Trophic relationships among high arctic seabirds: Insights from tissue-dependent stable-isotope models. Mar. Ecol. Prog. Ser. 1993, 95, 7–18. [Google Scholar] [CrossRef]
- Moody, A.T.; Hobson, K.A.; Gaston, A.J. High-arctic seabird trophic variation revealed through long-term isotopic monitoring. J. Ornithol. 2012, 153, 1067–1078. [Google Scholar] [CrossRef]
- DeNiro, M.J.; Epstein, S. Mechanism of carbon isotope fractionation associated with lipid synthesis. Science 1977, 197, 261–263. [Google Scholar] [CrossRef]
- Speake, B.K.; Murray, A.M.B.; Noble, R.C. Transport and transformations of yolk lipids during development of the avian embryo. Prog. Lipid Res. 1998, 37, 1–32. [Google Scholar] [CrossRef]
- Rowan, M.K. The greater shearwater puffinus gravis at’ its breeding grounds. Ibis 1952, 94, 97–121. [Google Scholar] [CrossRef]
- Danielsen, N.S.T.; Hedeholm, R.B.; Grønkjær, P. Seasonal changes in diet and lipid content of northern sand lance Ammodytes dubius on Fyllas Bank, West Greenland. Mar. Ecol. Prog. Ser. 2016, 558, 97–113. [Google Scholar] [CrossRef]
- Henderson, R.J. Seasonal changes in the lipid composition of herring (Clupea harengus) in relation to gonad maturation. J. Mar. Biol. Assoc. UK 1989, 69, 323–334. [Google Scholar] [CrossRef]
- Hislop, J.R.G.; Harris, M.P.; Smith, J.G.M. Variation in the calorific value and total energy content of the lesser sandeel (Ammodytes marinus) and other fish preyed on by seabirds. J. Zool. 1991, 224, 501–517. [Google Scholar] [CrossRef]
- Lane, H.A.; Westgate, A.J.; Koopman, H.N. Ontogenetic and temporal variability in the fat content and fatty acid composition of atlantic herring (clupea harengus) from the Bay of Fundy, Canada. Fish. Bull. 2011, 109, 113–122. [Google Scholar]
- Bond, A.L.; Jardine, T.D.; Hobson, K.A. Multi-tissue stable-isotope analyses can identify dietary specialization. Methods Ecol. Evol. 2016, 7, 1428–1437. [Google Scholar] [CrossRef]
- Heady, W.N.; Moore, J.W. Tissue turnover and stable isotope clocks to quantify resource shifts in anadromous rainbow trout. Oecologia 2013, 172, 21–34. [Google Scholar] [CrossRef]
- Germain, L.R.; McCarthy, M.D.; Koch, P.L.; Harvey, J.T. Stable carbon and nitrogen isotopes in multiple tissues of wild and captive harbor seals (Phoca vitulina) off the California coast. Mar. Mammal Sci. 2012, 28, 542–560. [Google Scholar] [CrossRef]
- Dalerum, F.; Angerbjorn, A. Resolving temporal variation in vertebrate diets using naturally occurring stable isotopes. Oecologia 2005, 144, 647–658. [Google Scholar] [CrossRef]
- Knoff, A.; Hohn, A.; Macko, S. Ontogenetic diet changes in bottlenose dolphins (Tursiops truncatus) reflected through stable isotopes. Mar. Mammal Sci. 2008, 24, 128–137. [Google Scholar] [CrossRef]
- Tieszen, L.L.; Boutton, T.W.; Tesdahl, K.G.; Slade, N.A. Fractionation and turnover of stable carbon isotopes in animal tissues: Implications for δ13C analysis of diet. Oecologia 1983, 57, 32–37. [Google Scholar] [CrossRef]
- Bearhop, S.; Teece, M.A.; Waldron, S.; Furness, R.W. Influence of lipid and uric acid on δ13C and δ15N values of avian blood: Implications for trophic studies. Auk 2000, 117, 504–507. [Google Scholar]
- Cherel, Y.; Hobson, K.A.; Hassani, S. Isotopic discrimination between food and blood and feathers of captive penguins: Implications for dietary studies in the wild. Physiol. Biochem. Zool. 2005, 78, 106–115. [Google Scholar] [CrossRef]
- Cherel, Y.; Hobson, K.A.; Bailleul, F.; Groscolas, R. Nutrition, physiology, and stable isotopes: New information from fasting and molting penguins. Ecology 2005, 86, 2881–2888. [Google Scholar] [CrossRef]
- Hatch, K.A.; Sacksteder, K.A.; Treichel, I.W.; Cook, M.E.; Porter, W.P. Early detection of catabolic state via change in 13C/12C ratios of blood proteins. Biochem. Biophys. Res. Commun. 1995, 212, 719–726. [Google Scholar] [CrossRef]
- Denton, M.J.; Demopoulos, A.W.J.; Baldwin, J.D.; Smith, B.J.; Hart, K.M. Stable isotope analysis enhances our understanding of diamondback terrapin (Malaclemys terrapin) foraging ecology. Estuaries Coasts 2019, 42, 596–611. [Google Scholar] [CrossRef]
- Horswill, C.; Matthiopoulos, J.; Ratcliffe, N.; Green, J.A.; Trathan, P.N.; McGill, R.A.R.; Phillips, R.A.; O’Connell, T.C. Drivers of intrapopulation variation in resource use in a generalist predator, the macaroni penguin. Mar. Ecol. Prog. Ser. 2016, 548, 233–247. [Google Scholar] [CrossRef]
- MacNeil, M.A.; Skomal, G.B.; Fisk, A.T. Stable isotopes from multiple tissues reveal diet switching in sharks. Mar. Ecol. Prog. Ser. 2005, 302, 199–206. [Google Scholar] [CrossRef]
- Villegas, M.; Newsome, S.D.; Blake, J.G. Seasonal patterns in δ2H values of multiple tissues from Andean birds provide insights into elevational migration. Ecol. Appl. 2016, 26, 2381–2387. [Google Scholar] [CrossRef]
- Bauchinger, U.; McWilliams, S. Carbon turnover in tissues of a passerine bird: Allometry, isotopic clocks, and phenotypic flexibility in organ size. Physiol. Biochem. Zool. 2009, 82, 787–797. [Google Scholar] [CrossRef]
- Hatch, K.A. The use and application of stable isotope analysis to the study of starvation, fasting, and nutritional stress in animals. In The Comparative Physiology of Fasting and Starvation; McCue, M.D., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 337–364. [Google Scholar]
- Endo, Y.; Wiebe, P.H. Euphausiid distribution, abundance and succession in North Atlantic warm-core ring 82b. J. Plankton Res. 2005, 27, 175–188. [Google Scholar] [CrossRef][Green Version]
- Myers, R.A.; Drinkwater, K. The influence of Gulf Stream warm core rings on recruitment of fish in the Northwest Atlantic. J. Mar. Res. 1989, 47, 635–656. [Google Scholar] [CrossRef]
| RBC δ13C | RBC δ15N | |||||
|---|---|---|---|---|---|---|
| Study | Location | n | Mean | SE | Mean | SE |
| This study | Massachusetts Bay, Cape Cod (July 2014) | 13 | −18.9 | 0.06 | 13.1 | 0.1 |
| This study | Massachusetts Bay, Cape Cod (September 2014) | 12 | −19 | 0.07 | 12.7 | 0.19 |
| This study | Massachusetts Bay, Cape Cod (July 2015) | 12 | −18.9 | 0.15 | 12.6 | 0.19 |
| This study | Massachusetts Bay, Cape Cod (September 2015) | 12 | −19.1 | 0.13 | 13.1 | 0.25 |
| Ronconi et al. 2010 | Bay of Fundy (2005) * | 34 | −19.1 | 0.05 | 13.4 | 0.13 |
| Ronconi et al. 2010 | Bay of Fundy (2006) * | 71 | −19 | 0.04 | 13.4 | 0.07 |
| Ronconi et al. 2010 | Bay of Fundy (2007) * | 52 | −19 | 0.04 | 13.6 | 0.07 |
| Ronconi et al. 2010 | Bay of Fundy (2008) * | 53 | −19.4 | 0.06 | 14 | 0.06 |
| Ronconi et al. 2010 | Bay of Fundy (2009) * | 21 | −19 | 0.08 | 13.8 | 0.07 |
| Gulka et al. 2017 | Newfoundland (early summer, 2016) † | 28 | −19.6 | 0.05 | 13.5 | 0.13 |
| Gulka et al. 2017 | Newfoundland midsummer, 2016) † | 14 | −19.4 | 0.08 | 13.9 | 0.14 |
| Gulka et al. 2017 | Newfoundland (late summer, 2016) † | 6 | −19.4 | 0.06 | 14.1 | 0.22 |
| Feather δ13C | Feather δ15N | |||||
| Mean | SD | Mean | SD | |||
| This study | Massachusetts Bay, Cape Cod (July 2014) | 13 | −17.7 | 0.8 | 15.6 | 1.9 |
| This study | Massachusetts Bay, Cape Cod (September 2014) | 12 | −18.4 | 0.9 | 15.9 | 1.9 |
| This study | Massachusetts Bay, Cape Cod (July 2015) | 13 | −16.8 | 0.8 | 16.5 | 1.1 |
| This study | Massachusetts Bay, Cape Cod (September 2015) | 8 | −17.1 | 1 | 16.1 | 1.6 |
| Haman et al. 2013 | Bay of Fundy (2006) | 11 | −16.8 | 1 | 16.1 | 1.7 |
| Haman et al. 2013 | Bay of Fundy (2007) | 20 | −16.6 | 0.7 | 16.3 | 1.7 |
| Haman et al. 2013 | Florida stranded (2008) ‡ | 9 | −18.1 | 1.1 | 16.3 | 1.1 |
| Haman et al. 2013 | Bay of Fundy (2008) | 12 | −17.2 | 0.7 | 15.6 | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, P.; Wiley, D.N.; Powers, K.D.; Michener, R.H.; Kaufman, L.; Hatch, K.A. Stable Isotope Analyses of Multiple Tissues of Great Shearwaters (Ardenna Gravis) Reveals Long-Term Dietary Stability, Short-Term Changes in Diet, and Can be Used as a Tool to Monitor Food Webs. Diversity 2019, 11, 163. https://doi.org/10.3390/d11090163
Hong P, Wiley DN, Powers KD, Michener RH, Kaufman L, Hatch KA. Stable Isotope Analyses of Multiple Tissues of Great Shearwaters (Ardenna Gravis) Reveals Long-Term Dietary Stability, Short-Term Changes in Diet, and Can be Used as a Tool to Monitor Food Webs. Diversity. 2019; 11(9):163. https://doi.org/10.3390/d11090163
Chicago/Turabian StyleHong, Peter, David N. Wiley, Kevin D. Powers, Robert H. Michener, Les Kaufman, and Kent A. Hatch. 2019. "Stable Isotope Analyses of Multiple Tissues of Great Shearwaters (Ardenna Gravis) Reveals Long-Term Dietary Stability, Short-Term Changes in Diet, and Can be Used as a Tool to Monitor Food Webs" Diversity 11, no. 9: 163. https://doi.org/10.3390/d11090163
APA StyleHong, P., Wiley, D. N., Powers, K. D., Michener, R. H., Kaufman, L., & Hatch, K. A. (2019). Stable Isotope Analyses of Multiple Tissues of Great Shearwaters (Ardenna Gravis) Reveals Long-Term Dietary Stability, Short-Term Changes in Diet, and Can be Used as a Tool to Monitor Food Webs. Diversity, 11(9), 163. https://doi.org/10.3390/d11090163





