Lingering Impacts of Hurricane Hugo on Rhizophora mangle (Red Mangrove) Population Genetics on St. John, USVI
Abstract
1. Introduction
2. Materials and Methods
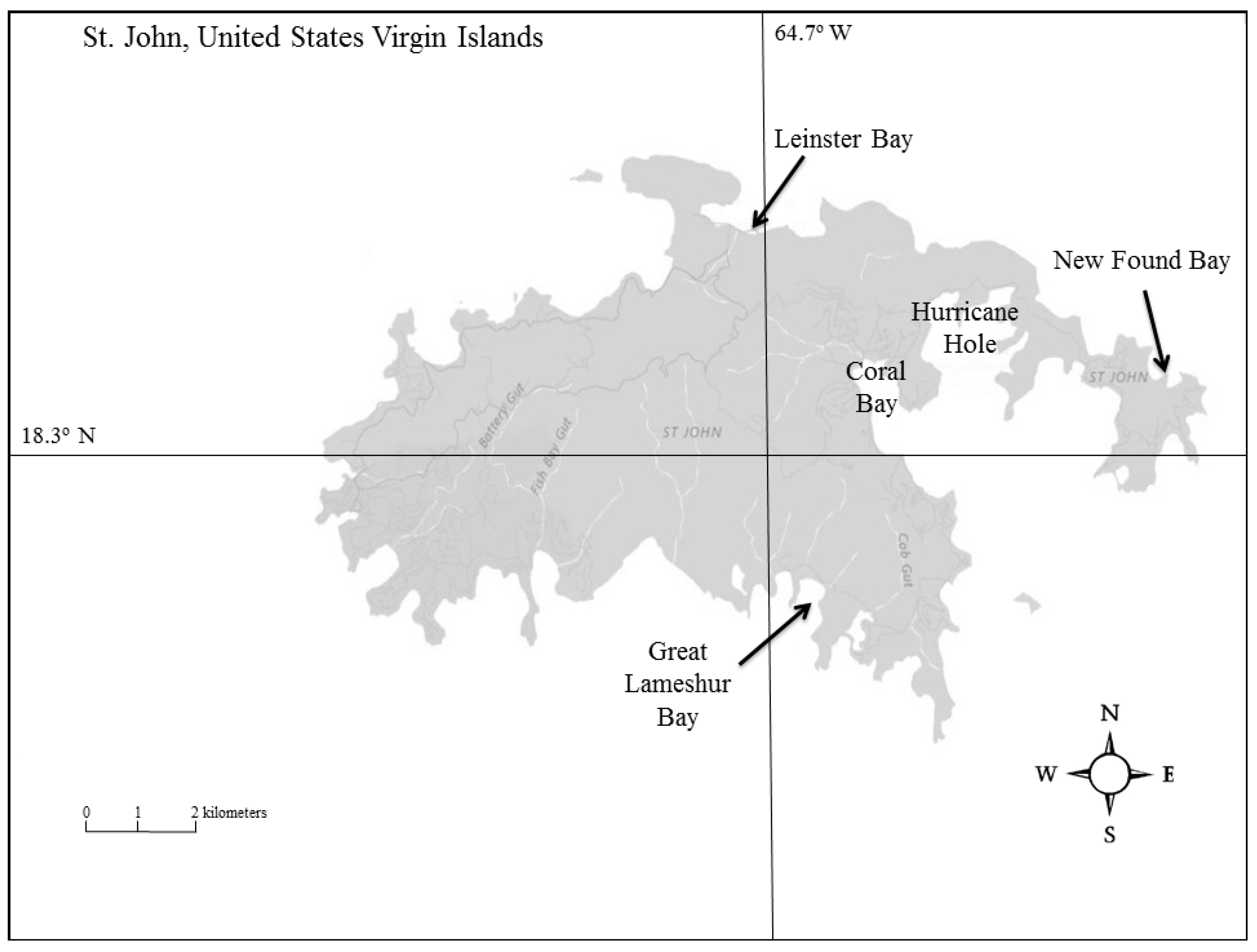
2.1. Study Sites
2.2. Plants Collection
2.3. DNA Extraction and Microsatellite Amplification
2.4. Microsatellite Allele Size Analysis
2.5. Statistical Analysis of Data
3. Results
3.1. Allelic Frequency Data and Diversity
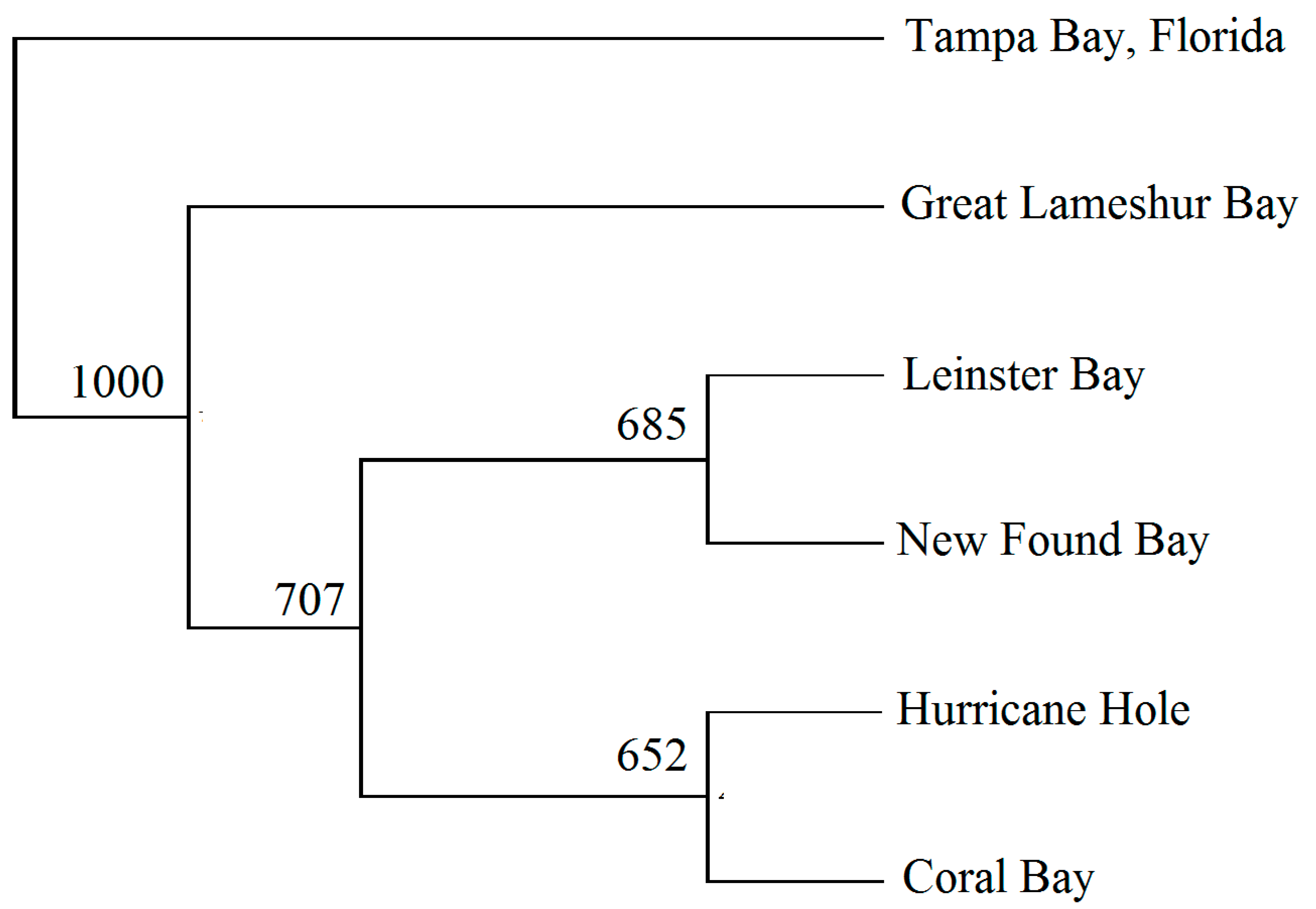
3.2. Genetic Distance and Bottlenecks
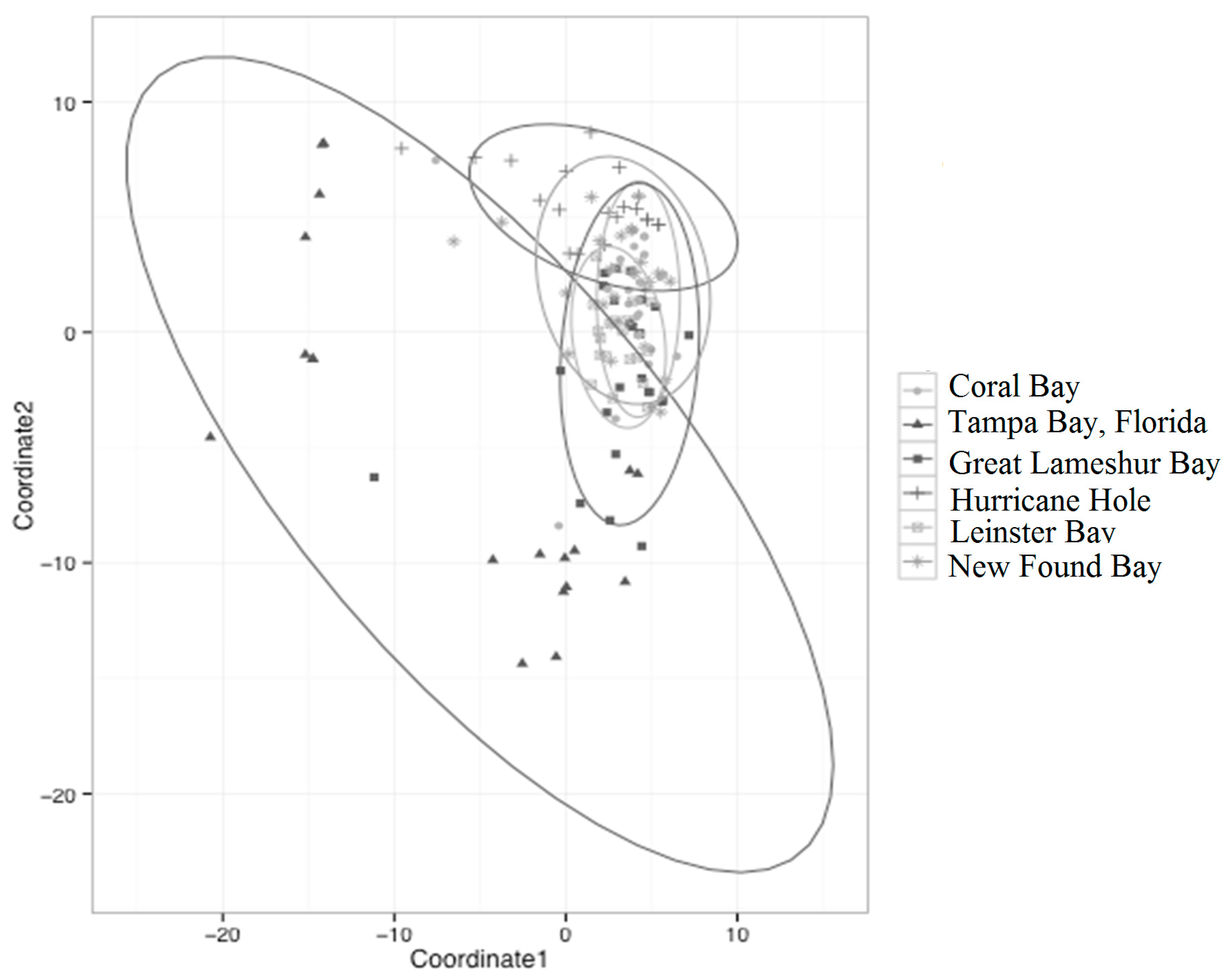
3.3. Fixation and Connectivity
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hogarth, P.J. The Biology of Mangroves and Seagrasses, 3rd ed.; Biology of habitats; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Field, C.D. Impact of Expected Climate Change on Mangroves. Hydrobiologia 1995, 295, 75–81. [Google Scholar] [CrossRef]
- Gilman, E.L.; Ellison, J.; Duke, N.C.; Field, C. Threats to Mangroves from Climate Change and Adaptation Options: A Review. Aquat. Bot. 2008, 89, 237–250. [Google Scholar] [CrossRef]
- Valiela, I.; Bowen, J.L.; York, J.K. Mangrove Forests: One of the World’s Threatened Major Tropical Environments. BioScience 2001, 51, 807. [Google Scholar] [CrossRef]
- Lovelock, C.E.; Cahoon, D.R.; Friess, D.A.; Guntenspergen, G.R.; Krauss, K.W.; Reef, R.; Rogers, K.; Saunders, M.L.; Sidik, F.; Swales, A.; et al. The Vulnerability of Indo-Pacific Mangrove Forests to Sea-Level Rise. Nature 2015, 526, 559–563. [Google Scholar] [CrossRef]
- Hamilton, S.E.; Casey, D. Creation of a High Spatio-Temporal Resolution Global Database of Continuous Mangrove Forest Cover for the 21st Century (CGMFC-21): CGMFC-21. Glob. Ecol. Biogeogr. 2016, 25, 729–738. [Google Scholar] [CrossRef]
- Mumby, P.J.; Edwards, A.J.; Ernesto Arias-González, J.; Lindeman, K.C.; Blackwell, P.G.; Gall, A.; Gorczynska, M.I.; Harborne, A.R.; Pescod, C.L.; Renken, H.; et al. Mangroves Enhance the Biomass of Coral Reef Fish Communities in the Caribbean. Nature 2004, 427, 533–536. [Google Scholar] [CrossRef]
- Alongi, D.M. Present State and Future of the World’s Mangrove Forests. Environ. Conserv. 2002, 29, 331–349. [Google Scholar] [CrossRef]
- Alongi, D.M. Mangrove Forests: Resilience, Protection from Tsunamis, and Responses to Global Climate Change. Est. Coastal Shelf Sci. 2008, 76, 1–13. [Google Scholar] [CrossRef]
- Lewis, R.R. Ecological Engineering for Successful Management and Restoration of Mangrove Forests. Ecol. Eng. 2005, 24, 403–418. [Google Scholar] [CrossRef]
- Ellison, A.M. Mangrove Restoration: Do We Know Enough? Restor. Ecol. 2000, 8, 219–229. [Google Scholar] [CrossRef]
- Bozzano, M.; Jalonen, R.; Thomas, E.; Boshier, D.; Gallo, L.; Cavers, S.; Bordács, S.; Smith, P.; Loo, J. (Eds.) Genetic Considerations in Ecosystem Restoration Using Native Tree Species. State of the World’s Forest Genetic Resources – Thematic Study; FAO and Biodiversity International: Rome, Italy, 2014. [Google Scholar]
- Thomas, E.; Jalonen, R.; Loo, J.; Boshier, D.; Gallo, L.; Cavers, S.; Bordács, S.; Smith, P.; Bozzano, M. Genetic Considerations in Ecosystem Restoration Using Native Tree Species. For. Ecol. Manag. 2014, 333, 66–75. [Google Scholar] [CrossRef]
- Campanella, J.J.; Bologna, P.A.X.; Carvalho, M.; Smalley, J.V.; Elakhrass, M.; Meredith, R.W.; Zaben, N. Clonal Diversity and Connectedness of Turtle Grass (Thalassia testudinum) Populations in a UNESCO Biosphere Reserve. Aquat. Bot. 2015, 123, 76–82. [Google Scholar] [CrossRef]
- Puotinen, M.; Maynard, J.A.; Beeden, R.; Radford, B.; Williams, G.J. A robust Operational Model for Predicting Where Tropical Cyclone Waves Damage Coral Reefs. Sci. Rep. 2016, 6, 26009. [Google Scholar] [CrossRef]
- Williams, S. Disturbance and Recovery of a Deep-Water Caribbean Seagrass Bed. Mar. Ecol. Prog. Ser. 1988, 42, 63–71. [Google Scholar] [CrossRef]
- Lewis, R.R.; Streever, B. Restoration of Mangrove Habitat, WRP Technical Notes Collection (ER TN-WRP-VN-RS-3.2); U.S. Army Engineer Research and Development Center: Vicksburg, MS, USA, 2000.
- Reilly, A.E. The Effects of Hurricane Hugo in Three Tropical Forests in the U.S. Virgin Islands. Biotropica 1991, 23, 414. [Google Scholar] [CrossRef]
- Rogers, C.S.; McLain, L.N.; Tobias, C.R. Effects of Hurricane Hugo (1989) on a coral reef in St. John, USVI. Mar. Ecol. Prog. Ser. 1991, 78, 189–199. [Google Scholar] [CrossRef]
- Cohn, J.P. Paradise in peril. Americas 2003, 55, 6–13. [Google Scholar]
- Devine, B.; Blondeau, J. Restoration of Wetlands at Lameshur Bay. Mapping, Assessment, Planting and Monitoring Report; Center for Marine and Environmental Sciences: St. Thomas, VI, USA, 2004; p. 32. [Google Scholar]
- Bologna, P.A.X. Mangrove Loss Leads to Fish Hyperutilization of Seagrass Beds in a UNESCO Biosphere Reserve. J. Fish Biol. 2014, 84, 1620–1625. [Google Scholar] [CrossRef]
- Campanella, J.J.; Bologna, P.A.; Smalley, J.V.; Rosenzweig, E.B.; Smith, S.M. Population Structure of Zostera marina (Eelgrass) on the Western Atlantic Coast Is Characterized by Poor Connectivity and Inbreeding. J. Hered. 2010, 101, 61–70. [Google Scholar] [CrossRef]
- Ribeiro, D.O.; Vinson, C.C.; Nascimento, D.S.S.; Mehlig, U.; Menezes, M.P.M.; Sampaio, I.; Silva, M.B. Isolation of Microsatellite Markers for the Red Mangrove, Rhizophora mangle (Rhizophoraceae). Appl. Plant Sci. 2013, 1, 1300003. [Google Scholar] [CrossRef]
- Campanella, J.J.; Bologna, P.A.X.; Smalley, J.V.; Avila, D.N.; Lee, K.N.; Areche, E.C.; Slavin, L.J. An Analysis of the Population Genetics of Restored Zostera marina Plantings in Barnegat Bay, New Jersey. Pop. Ecol. 2013, 55, 121–133. [Google Scholar] [CrossRef]
- Koskinen, M.T.; Hirvonen, H.; Landry, P.-A.; Primmer, C.R. The Benefits of Increasing the Number of Microsatellites Utilized in Genetic Population Studies: An Empirical Perspective: Increasing the Number of Microsatellites in Genetic Population Studies. Hereditas 2004, 141, 61–67. [Google Scholar] [CrossRef]
- Kennedy, J.P.; Pil, M.W.; Proffitt, C.E.; Boeger, W.A.; Stanford, A.M.; Devlin, D.J. Postglacial Expansion Pathways of Red Mangrove, Rhizophora Mangle, in the Caribbean Basin and Florida. Am. J. Bot. 2016, 103, 260–276. [Google Scholar] [CrossRef]
- Sandoval-Castro, E.; Dodd, R.S.; Riosmena-Rodríguez, R.; Enríquez-Paredes, L.M.; Tovilla-Hernández, C.; López-Vivas, J.M.; Aguilar-May, B.; Muñiz-Salazar, R. Post-Glacial Expansion and Population Genetic Divergence of Mangrove Species Avicennia germinans (L.) Stearn and Rhizophora mangle L. along the Mexican Coast. PLoS ONE 2014, 9, e93358. [Google Scholar] [CrossRef]
- Peakall, R.; Smouse, P.E. GenAlEx 6.5: Genetic Analysis in Excel. Population Genetic Software for Teaching and Research--an Update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef]
- Minch, E.; Ruiz-Linares, A.; Goldstein, D.; Feldman, M.; Cavalli-Sforza, L.L. MICROSAT: A Computer Program for Calculating Various Statistics on Microsatellite Allele Data, VERSION 1.5d; Stanford University: Palo Alto, CA, USA, 1997. [Google Scholar]
- Baum, B.R. PHYLIP: Phylogeny Inference Package. Version 3.2. Joel Felsenstein. Q. Rev. Biol. 1989, 64, 539–541. [Google Scholar] [CrossRef]
- Piry, S.; Luikart, G.; Cornuet, J.-M. Computer Note. BOTTLENECK: A Computer Program for Detecting Recent Reductions in the Effective Size Using Allele Frequency Data. J. Hered. 1999, 90, 502–503. [Google Scholar] [CrossRef]
- Cornuet, J.M.; Luikart, G. Description and power analysis of two tests for detecting recent population bottlenecks from allele frequency data. Genetics 1996, 144, 2001–2014. [Google Scholar]
- Wang, C.; Schroeder, K.B.; Rosenberg, N.A. A Maximum-Likelihood Method to Correct for Allelic Dropout in Microsatellite Data with No Replicate Genotypes. Genetics 2012, 192, 651–669. [Google Scholar] [CrossRef]
- Nei, M. Genetic Distance between Populations. Am. Nat. 1972, 106, 283–292. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Wilkinson, L. Ggplot2: Elegant Graphics for Data Analysis by WICKHAM, H. Biometrics 2011, 67, 678–679. [Google Scholar] [CrossRef]
- Cavalli-Sforza, L.L.; Edwards, A.W.F. Phylogenetic Analysis: Models and Estimation Procedures. Evolution 1967, 21, 550. [Google Scholar] [CrossRef] [PubMed]
- Luikart, G. Distortion of Allele Frequency Distributions Provides a Test for Recent Population Bottlenecks. J. Hered. 1998, 89, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Di Rienzo, A.; Peterson, A.C.; Garza, J.C.; Valdes, A.M.; Slatkin, M.; Freimer, N.B. Mutational Processes of Simple-Sequence Repeat Loci in Human Populations. Proc. Nat. Acad. Sci. 1994, 91, 3166–3170. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, S.; Borges, A.V.; Castañeda-Moya, E.; Diele, K.; Dittmar, T.; Duke, N.C.; Kristensen, E.; Lee, S.Y.; Marchand, C.; Middelburg, J.J.; et al. Mangrove Production and Carbon Sinks: A Revision of Global Budget Estimates: GLOBAL MANGROVE CARBON BUDGETS. Glob. Biogeochem. Cycles 2008, 22. [Google Scholar] [CrossRef]
- Siikamaki, J.; Sanchirico, J.N.; Jardine, S.L. Global Economic Potential for Reducing Carbon Dioxide Emissions from Mangrove Loss. Proc. Nat. Acad. Sci. 2012, 109, 14369–14374. [Google Scholar] [CrossRef] [PubMed]
- Webster, P.J. Changes in Tropical Cyclone Number, Duration, and Intensity in a Warming Environment. Science 2005, 309, 1844–1846. [Google Scholar] [CrossRef]
- Knutson, T.R.; McBride, J.L.; Chan, J.; Emanuel, K.; Holland, G.; Landsea, C.; Held, I.; Kossin, J.P.; Srivastava, A.K.; Sugi, M. Tropical Cyclones and Climate Change. Nat. Geosci. 2010, 3, 157–163. [Google Scholar] [CrossRef]
- Jennerjahn, T.C.; Gilman, E.; Krauss, K.W.; Lacerda, L.D.; Nordhaus, I.; Wolanski, E. Mangrove Ecosystems under Climate Change. In Mangrove Ecosystems: A Global Biogeographic Perspective: Structure, Function, and Services; Victor, H.R.-M., Lee, S.Y., Erik, K., Robert, R.T.C., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 211–244. [Google Scholar] [CrossRef]
- Takayama, K.; Tamura, M.; Tateishi, Y.; Webb, E.L.; Kajita, T. Strong Genetic Structure over the American Continents and Transoceanic Dispersal in the Mangrove Genus Rhizophora (Rhizophoraceae) Revealed by Broad-Scale Nuclear and Chloroplast DNA Analysis. Am. J. Bot. 2013, 100, 1191–1201. [Google Scholar] [CrossRef]
- Kennedy, J.P.; Garavelli, L.; Truelove, N.K.; Devlin, D.J.; Box, S.J.; Chérubin, L.M.; Feller, I.C. Contrasting Genetic Effects of Red Mangrove (Rhizophora Mangle L.) Range Expansion along West and East Florida. J. Biogeogr. 2017, 44, 335–347. [Google Scholar] [CrossRef]
- Colautti, R.I.; Alexander, J.M.; Dlugosch, K.M.; Keller, S.R.; Sultan, S.E. Invasions and Extinctions through the Looking Glass of Evolutionary Ecology. Philos. Trans. R Soc. Lond. B Biol. Sci. 2017, 372, 20160031. [Google Scholar] [CrossRef]
- Smith, T.J.; Robblee, M.B.; Wanless, H.R.; Doyle, T.W. Mangroves, Hurricanes, and Lightning Strikes. BioScience 1994, 44, 256–262. [Google Scholar] [CrossRef]
- Hebert, P.J. Freezes and hurricanes in Florida. Nat. Weath. Dig. 1993, 18, 1–11. [Google Scholar]
- Lewis, R.R. Time Zero Plus 60 Months Report for the Cross Bayou Mangrove Restoration Site, Pinellas County, Florida; Prepared for the Cross Bayou Project Review Group, Tampa, Florida; Lewis Environmental Services: Salt Springs, Marion County, FL, USA, 2004. [Google Scholar]
- Osland, M.J.; Spivak, A.C.; Nestlerode, J.A.; Lessmann, J.M.; Almario, A.E.; Heitmuller, P.T.; Russell, M.J.; Krauss, K.W.; Alvarez, F.; Dantin, D.D.; et al. Ecosystem Development After Mangrove Wetland Creation: Plant–Soil Change Across a 20-Year Chronosequence. Ecosystems 2012, 15, 848–866. [Google Scholar] [CrossRef]
- Engelhardt, K.A.M.; Lloyd, M.W.; Neel, M.C. Effects of Genetic Diversity on Conservation and Restoration Potential at Individual, Population, and Regional Scales. Biolog. Conserv. 2014, 179, 6–16. [Google Scholar] [CrossRef]
- Kettenring, K.M.; Mercer, K.L.; Reinhardt Adams, C.; Hines, J. EDITOR’S CHOICE: Application of Genetic Diversity-Ecosystem Function Research to Ecological Restoration. J. Appl. Ecol. 2014, 51, 339–348. [Google Scholar] [CrossRef]
- Nadia, T.L.; Machado, I.C. Wind Pollination and Propagule Formation in Rhizophora Mangle L. (Rhizophoraceae): Resource or Pollination Limitation? Anais da Academia Brasileira de Ciências 2014, 86, 229–238. [Google Scholar] [CrossRef]
- Takezaki, N.; Nei, M. Genetic distances and reconstruction of phylogenetic trees from microsatellite DNA. Genetics 1996, 144, 389–399. [Google Scholar]
- Polidoro, B.A.; Carpenter, K.E.; Collins, L.; Duke, N.C.; Ellison, A.M.; Ellison, J.C.; Farnsworth, E.J.; Fernando, E.S.; Kathiresan, K.; Koedam, N.E.; et al. The Loss of Species: Mangrove Extinction Risk and Geographic Areas of Global Concern. PLoS ONE 2010, 5, e10095. [Google Scholar] [CrossRef]
- Krauss, K.W.; Lovelock, C.E.; McKee, K.L.; López-Hoffman, L.; Ewe, S.M.L.; Sousa, W.P. Environmental Drivers in Mangrove Establishment and Early Development: A Review. Aquat. Bot. 2008, 89, 105–127. [Google Scholar] [CrossRef]
- Kontos, C.C.; Bologna, P.A.X. Assessment of fish and decapod distributions between mangrove and seagrass habitats in St. John, USVI. Bull. N. J. Acad. Sci. 2008, 53, 7–11. [Google Scholar]
- McKee, K.L. Growth and Physiological Responses of Neotropical Mangrove Seedlings to Root Zone Hypoxia. Tree Physiol. 1996, 16, 883–889. [Google Scholar] [CrossRef]
- Padilla, C.; Fortes, M.D.; Duarte, C.M.; Terrados, J.; Kamp-Nielsen, L. Recruitment, Mortality and Growth of Mangrove (Rhizophora Sp.) Seedlings in Ulugan Bay, Palawan, Philippines. Trees 2004, 18. [Google Scholar] [CrossRef]
- Imbert, D.; Rousteau, A.; Scherrer, P. Ecology of Mangrove Growth and Recovery in the Lesser Antilles: State of Knowledge and Basis for Restoration Projects. Restor. Ecol. 2000, 8, 230–236. [Google Scholar] [CrossRef]
- Edward Proffitt, C.; Milbrandt, E.C.; Travis, S.E. Red Mangrove (Rhizophora mangle) Reproduction and Seedling Colonization after Hurricane Charley: Comparisons of Charlotte Harbor and Tampa Bay. Est. Coasts 2006, 29, 972–978. [Google Scholar] [CrossRef]
- Harris, R.J.; Milbrandt, E.C.; Everham, E.M.; Bovard, B.D. The Effects of Reduced Tidal Flushing on Mangrove Structure and Function Across a Disturbance Gradient. Est. Coasts 2010, 33, 1176–1185. [Google Scholar] [CrossRef]
- Van Loon, A.F.; Te Brake, B.; Van Huijgevoort, M.H.J.; Dijksma, R. Hydrological Classification, a Practical Tool for Mangrove Restoration. PLoS ONE 2016, 11, e0150302. [Google Scholar] [CrossRef]
- Kodikara, K.A.S.; Mukherjee, N.; Jayatissa, L.P.; Dahdouh-Guebas, F.; Koedam, N. Have Mangrove Restoration Projects Worked? An in-Depth Study in Sri Lanka: Evaluation of Mangrove Restoration in Sri Lanka. Restor. Ecol. 2017, 25, 705–716. [Google Scholar] [CrossRef]
- Balke, T.; Friess, D.A. Geomorphic Knowledge for Mangrove Restoration: A Pan-Tropical Categorization: Geomorphic Knowledge for Mangrove Restoration. Earth Surf. Proc. Landforms 2016, 41, 231–239. [Google Scholar] [CrossRef]
- Daley, B.; Coldren, S. Rapid Health Assessment of Coral Bay Mangrove Forests; Post Irma and Maria Conditions; Geographic Consulting: Christiansted, VI, USA, 2018; p. 14. [Google Scholar]
| Population | RB 18 | RB 19 | RB 20 | RB 25 | RB 50 | RB 59 | RB 64 | Mean | |
|---|---|---|---|---|---|---|---|---|---|
| Great Lameshur Bay | N | 25 | 25 | 25 | 25 | 25 | 25 | 25 | |
| Na | 3 | 5 | 5 | 3 | 2 | 2 | 3 | 3.286 | |
| Ho | 0.080 | 0.040 | 0.040 | 0.280 | 0.200 | 0.280 | 0.000 | 0.129 | |
| He | 0.486 | 0.318 | 0.558 | 0.498 | 0.241 | 0.449 | 0.150 | 0.386 | |
| Fis | 0.835 | 0.874 | 0.928 | 0.438 | 0.169 | 0.376 | 1.000 | 0.660 | |
| Coral Bay | N | 27 | 27 | 27 | 27 | 27 | 27 | 27 | |
| Na | 5 | 2 | 3 | 4 | 2 | 5 | 4 | 3.571 | |
| Ho | 0.519 | 0.037 | 0.593 | 0.444 | 0.000 | 0.222 | 0.074 | 0.270 | |
| He | 0.581 | 0.105 | 0.551 | 0.486 | 0.071 | 0.354 | 0.173 | 0.331 | |
| Fis | 0.107 | 0.647 | −0.076 | 0.085 | 1.000 | 0.372 | 0.571 | 0.387 | |
| Hurricane Hole | N | 22 | 22 | 22 | 22 | 22 | 22 | 22 | |
| Na | 4 | 5 | 3 | 3 | 2 | 2 | 6 | 3.571 | |
| Ho | 0.045 | 0.136 | 0.227 | 0.227 | 0.227 | 0.000 | 0.045 | 0.131 | |
| He | 0.170 | 0.286 | 0.280 | 0.526 | 0.449 | 0.087 | 0.454 | 0.322 | |
| Fis | 0.733 | 0.523 | 0.188 | 0.568 | 0.494 | 1.000 | 0.900 | 0.630 | |
| New Found Bay | N | 25 | 25 | 25 | 25 | 25 | 25 | 25 | |
| Na | 6 | 4 | 5 | 3 | 4 | 4 | 9 | 5.000 | |
| Ho | 0.120 | 0.000 | 0.080 | 0.440 | 0.440 | 0.120 | 0.160 | 0.194 | |
| He | 0.750 | 0.285 | 0.287 | 0.530 | 0.422 | 0.369 | 0.442 | 0.441 | |
| Fis | 0.840 | 1.000 | 0.721 | 0.170 | −0.044 | 0.675 | 0.638 | 0.571 | |
| Leinster Bay | N | 20 | 18 | 20 | 20 | 20 | 20 | 18 | |
| Na | 5 | 4 | 1 | 2 | 5 | 6 | 11 | 4.857 | |
| Ho | 0.850 | 0.222 | 0.000 | 0.550 | 0.900 | 0.700 | 0.556 | 0.540 | |
| He | 0.750 | 0.296 | 0.000 | 0.399 | 0.639 | 0.668 | 0.715 | 0.495 | |
| Fis | −0.133 | 0.250 | N/C | −0.379 | −0.409 | −0.049 | 0.222 | −0.083 | |
| Tampa Bay, FL | N | 20 | 20 | 20 | 20 | 20 | 20 | 20 | |
| Na | 4 | 7 | 7 | 6 | 4 | 3 | 2 | 4.714 | |
| Ho | 0.150 | 0.100 | 0.300 | 0.050 | 0.550 | 0.350 | 0.000 | 0.214 | |
| He | 0.404 | 0.583 | 0.774 | 0.774 | 0.669 | 0.629 | 0.480 | 0.616 | |
| Fis | 0.628 | 0.828 | 0.612 | 0.935 | 0.178 | 0.443 | 1.000 | 0.662 |
| Population | Stepwise Mutation | Infinite Allele | Two Phase Mutation | Mean |
|---|---|---|---|---|
| Great Lameshur | 0.027 | 0.289 | 0.148 | 0.155 |
| Coral Bay | 0.007 | 0.019 | 0.007 | 0.011 |
| Hurricane Hole | 0.011 | 0.027 | 0.019 | 0.019 |
| New Found Bay | 0.007 | 0.019 | 0.019 | 0.015 |
| Leinster Bay | 0.039 | 0.078 | 0.039 | 0.052 |
| Tampa Bay, FL | 0.039 | 0.168 | 0.234 | 0.147 |
| Populations | Great Lameshur | Coral Bay | Hurricane Hole | New Found Bay | Leinster Bay | Tampa Bay |
|---|---|---|---|---|---|---|
| Great Lameshur | 0.000 | |||||
| Coral Bay | 0.033 | 0.000 | ||||
| Hurricane Hole | 0.113 | 0.078 | 0.000 | |||
| New Found Bay | 0.044 | 0.044 | 0.046 | 0.000 | ||
| Leinster Bay | 0.072 | 0.095 | 0.112 | 0.049 | 0.000 | |
| Tampa Bay, FL | 0.104 | 0.155 | 0.194 | 0.129 | 0.119 | 0.000 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bologna, P.A.X.; Campanella, J.J.; Restaino, D.J.; Fetske, Z.A.; Lourenco, M.; Smalley, J.V. Lingering Impacts of Hurricane Hugo on Rhizophora mangle (Red Mangrove) Population Genetics on St. John, USVI. Diversity 2019, 11, 65. https://doi.org/10.3390/d11040065
Bologna PAX, Campanella JJ, Restaino DJ, Fetske ZA, Lourenco M, Smalley JV. Lingering Impacts of Hurricane Hugo on Rhizophora mangle (Red Mangrove) Population Genetics on St. John, USVI. Diversity. 2019; 11(4):65. https://doi.org/10.3390/d11040065
Chicago/Turabian StyleBologna, Paul. A. X., James J. Campanella, Dena J. Restaino, Zachary A. Fetske, Matthew Lourenco, and John V. Smalley. 2019. "Lingering Impacts of Hurricane Hugo on Rhizophora mangle (Red Mangrove) Population Genetics on St. John, USVI" Diversity 11, no. 4: 65. https://doi.org/10.3390/d11040065
APA StyleBologna, P. A. X., Campanella, J. J., Restaino, D. J., Fetske, Z. A., Lourenco, M., & Smalley, J. V. (2019). Lingering Impacts of Hurricane Hugo on Rhizophora mangle (Red Mangrove) Population Genetics on St. John, USVI. Diversity, 11(4), 65. https://doi.org/10.3390/d11040065





