Using Growth Forms to Predict Epiphytic Lichen Abundance in a Wide Variety of Forest Types
Abstract
:1. Introduction
2. Material and Methods
2.1. Literature Search and Selection
2.2. Data Analyses
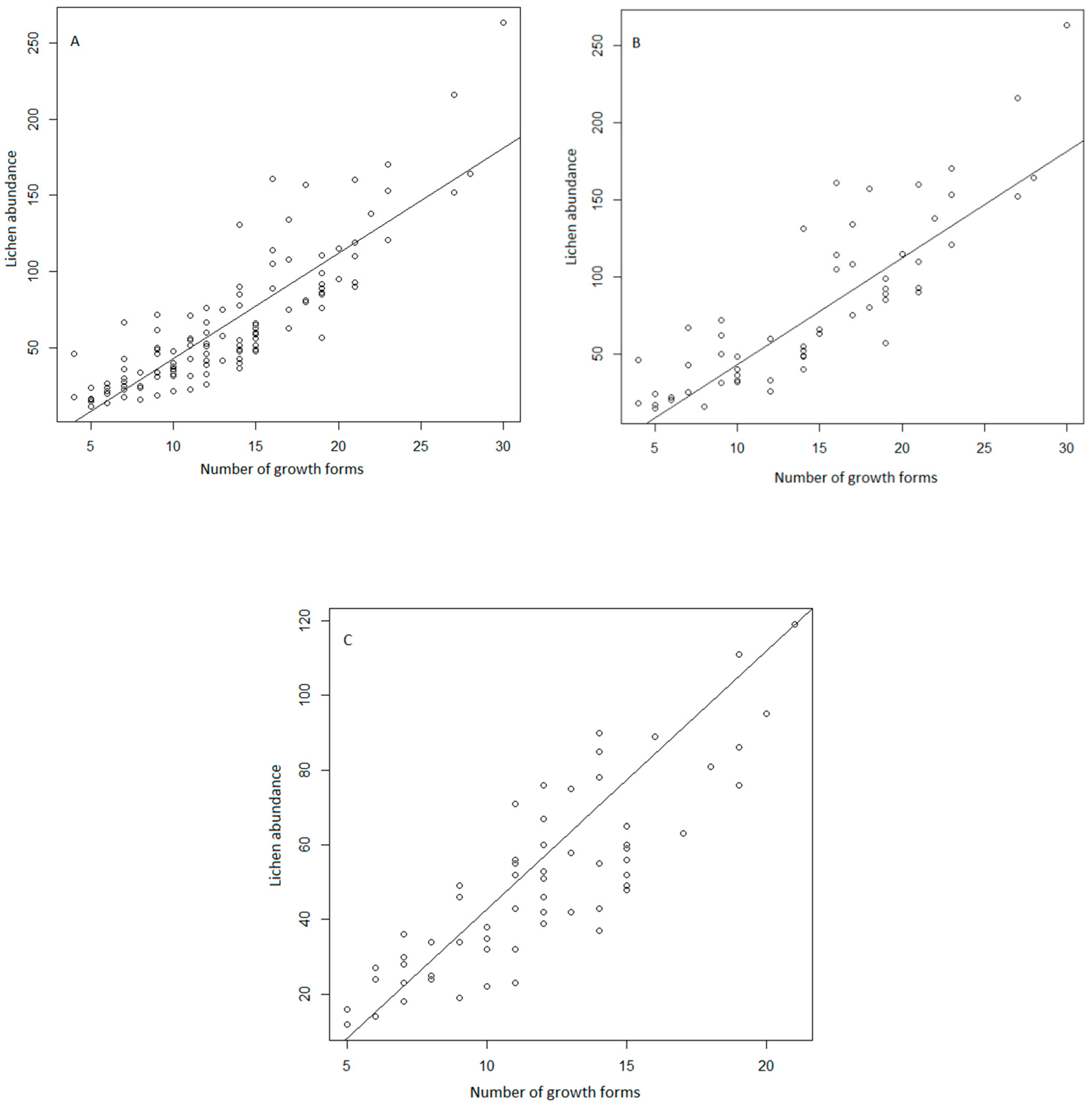
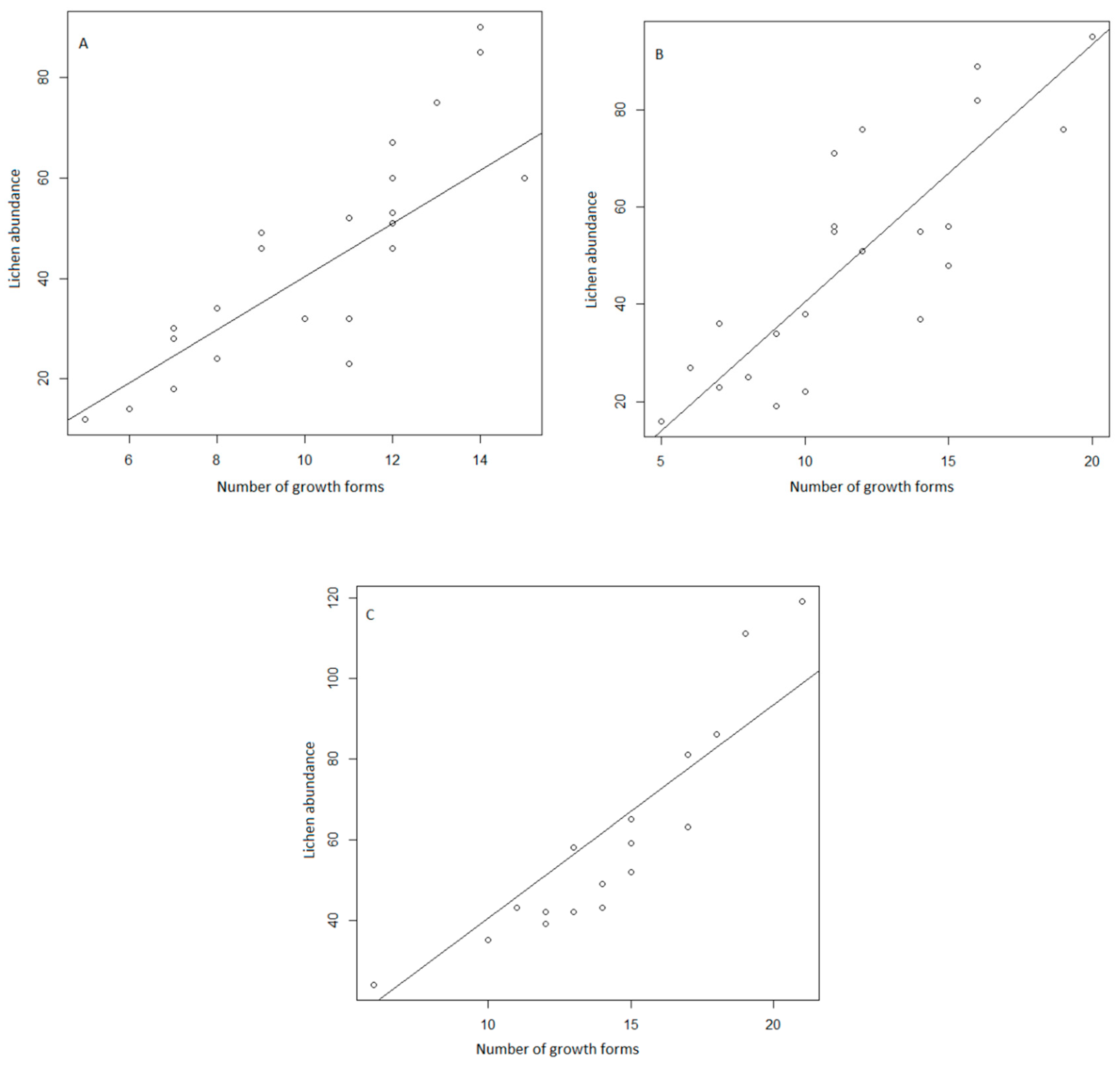
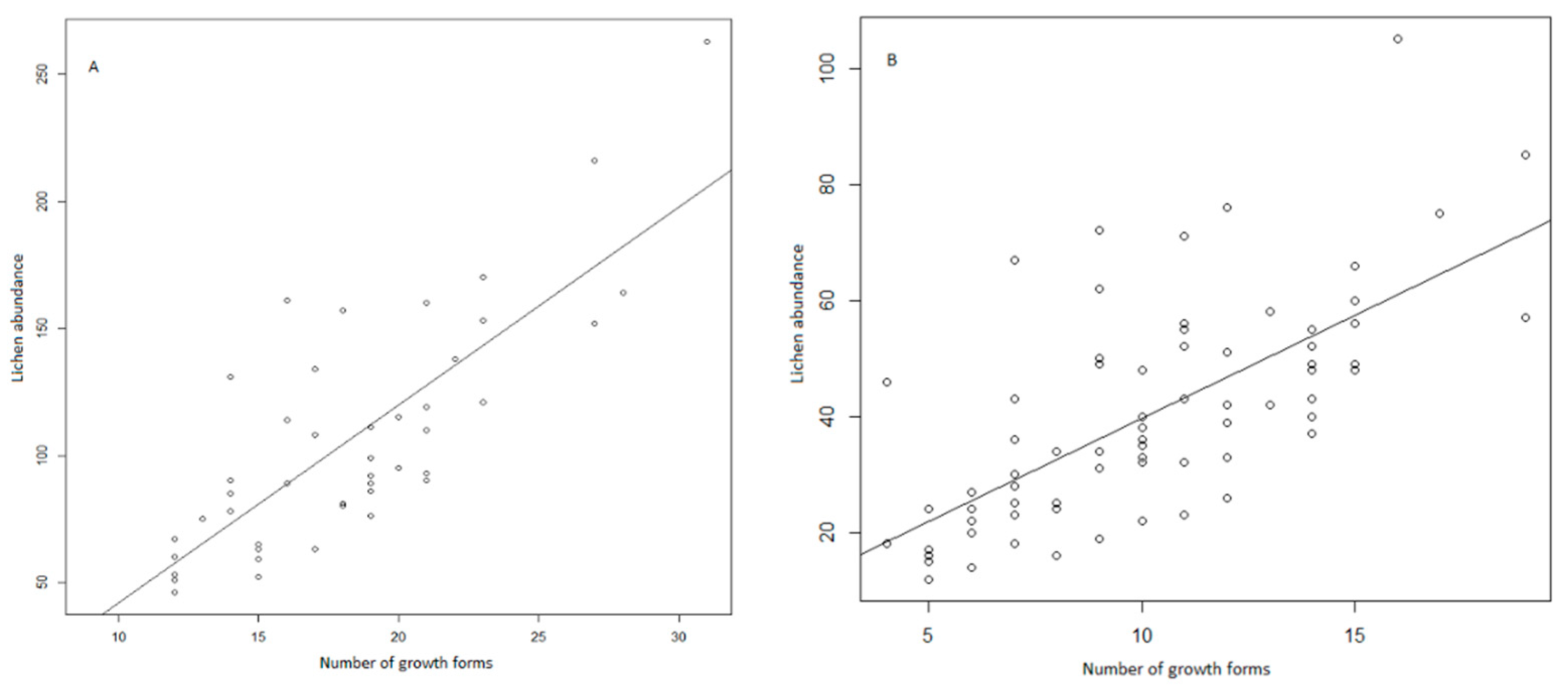
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Aragón, G.; Martínez, I.; Izquierdo, P.; Belinchón, R.; Escudero, A. Effects of forest management on epiphytic lichen diversity in Mediterranean forests. Appl. Veg. Sci. 2010, 13, 183–194. [Google Scholar] [CrossRef]
- Benítez, A.; Aragón, G.; González, Y.; Prieto, M. Functional traits of epiphytic lichens in response to forest disturbance and as predictors of total richness and diversity. Ecol. Ind. 2018, 86, 18–26. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; Da Fonseca, G.A.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Gibson, L.; Lee, T.M.; Koh, L.P.; Brook, B.W.; Gardner, T.A.; Barlow, J.; Peres, C.A.; Bradshaw, C.J.; Laurance, W.F.; Lovejoy, T.E.; et al. Primary forests are irreplaceable for sustaining tropical biodiversity. Nature 2011, 478, 378–381. [Google Scholar] [CrossRef] [PubMed]
- Chazdon, R.L. Beyond deforestation: Restoring forests and ecosystem services on degraded lands. Science 2008, 320, 1458–1460. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, H.K.; Ruesch, A.S.; Achard, F.; Clayton, M.K.; Holmgren, P.; Ramankutty, N.; Foley, J.A. Tropical forests were the primary sources of new agricultural land in the 1980s and 1990s. Proc. Natl. Acad. Sci. USA 2010, 107, 16732–16737. [Google Scholar] [CrossRef] [PubMed]
- Bergamini, A.; Scheidegger, C.; Stofer, S.; Carvalho, P.; Davey, S.; Dietrich, M.; Dubs, F.; Farkas, E.; Groner, U.; Kärkkäinen, K.; et al. Performance of macrolichens and lichen genera as indicators of lichen species richness and composition. Conserv. Biol. 2005, 19, 1051–1062. [Google Scholar] [CrossRef]
- Kelly, L.J.; Hollingsworth, P.M.; Coppins, B.J.; Ellis, C.J.; Harrold, P.; Tosh, J.; Yahr, R. DNA barcoding of lichenized fungi demonstrates high identification success in a floristic context. New Phytol. 2011, 191, 288–300. [Google Scholar] [CrossRef] [PubMed]
- Giordani, P.; Brunialti, G.; Benesperi, R.; Rizzi, G.; Frati, L.; Modenesi, P. Rapid biodiversity assessment in lichen diversity surveys: Implications for quality assurance. J. Environ. Monit. 2009, 11, 730–735. [Google Scholar] [CrossRef] [PubMed]
- Aragón, G.; Belinchón, R.; Martínez, I.; Prieto, M. Estimating epiphytic lichen richness by simple families in Mediterranean forests. For. Ecol. Manag. 2013, 310, 187–193. [Google Scholar] [CrossRef]
- Kranner, I.; Beckett, R.; Hochman, A.; Nash, T., III. Desiccation-Tolerance in Lichens: A Review. Bryologist 2008, 111, 576–593. [Google Scholar] [CrossRef]
- Giordani, P.; Brunialti, G.; Bacaro, G.; Nascimbene, J. Functional traits of epiphytic lichens as potential indicators of environmental conditions in forest ecosystems. Ecol. Ind. 2012, 18, 413–420. [Google Scholar] [CrossRef]
- Pinho, P.; Bergamini, A.; Carvalho, P.; Branquinho, C.; Stofer, S.; Scheidegger, C.; Maguas, C. Lichen functional groups as ecological indicators of the effects of land-use in Mediterranean ecosystems. Ecol. Ind. 2012, 15, 36–42. [Google Scholar] [CrossRef]
- Büdel, B.; Scheidegger, C. Thallus morphology and anatomy. In Lichen Biology, 2nd ed.; Nash, T.H., III, Ed.; Cambridge University Press: Cambridge, UK, 2008; pp. 40–68. [Google Scholar]
- Asplund, J.; Wardle, D.A. How lichens impact on terrestrial community and ecosystem properties. Biol. Rev. 2016, 92, 1720–1738. [Google Scholar] [CrossRef]
- Aragón, G.; Belinchón, R.; Martínez, I.; Prieto, M. A survey method for assessing the richness of epiphytic lichens using growth forms. Ecol. Ind. 2016, 62, 101–105. [Google Scholar] [CrossRef]
- Aragón, G.; López, R.; Martínez, I. Effects of Mediterranean dehesa management on epiphytic lichens. Sci. Total Environ. 2010, 409, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Friedel, A.; Oheimb, G.V.; Dengler, J.; Härdtle, W. Species diversity and species composition of epiphytic bryophytes and lichens—A comparison of managed and unmanaged beech forests in NE Germany. Feddes Repert. 2006, 117, 172–185. [Google Scholar] [CrossRef]
- Nascimbene, J.; Marini, L. Oak forests exploitation and black locust invasion caused severe shifts in epiphytic lichen communities in northern Italy. Sci. Tot. Environ. 2010, 408, 5506–5512. [Google Scholar] [CrossRef]
- Estrabou, C.; Stiefkens, L.; Hadid, M.; Rodríguez, J.; Pérez, A. Estudio comparativo de la comunidad liquénica en cuatro ecosistemas de la provincia de Córdoba. Bol. Soc. Argent. Bot. 2005, 40, 3–12. [Google Scholar]
- Quiroga, G.; Estrabou, C.; Rodríguez, J.M. Lichen community response to different management situations in Cerro Colorado protected forest (Córdoba, Argentina). Lazaroa 2008, 29, 131–138. [Google Scholar]
- García, R.; Rosato, V. Nuevas citas de líquenes para la Reserva Natural de Punta Lara, provincia de Buenos Aires, Argentina. Rev. Mus. Argent. Cienc. Nat. 2013, 15, 169–174. [Google Scholar] [CrossRef]
- Flakus, A. Contribution to the knowledge of the lichen biota of Bolivia. 2. Pol. Bot. J. 2008, 53, 145–153. [Google Scholar]
- Flakus, A.; Oset, M.; Jablonska, A.; Rodríguez, P.; Kurka, M. Contribution to the knowledge of the lichen biota of Bolivia. 3. Pol. Bot. J. 2011, 56, 159–183. [Google Scholar]
- Flakus, A.; Etayo, J.; Schiefelbein, U.; Ahti, T.; Jablonska, A.; Oset, M.; Bach, K.; Rodríguez, P.; Kukwa, M. Contribution to the knowledge of the lichen biota of Bolivia. 4. Pol. Bot. J. 2012, 57, 427–461. [Google Scholar]
- Flakus, A.; Sipman, H.J.M.; Bach, K.; Rodríguez, P.; Knudsen, K.; Ahti, T.; Schiefelbein, U.; Palice, Z.; Jablonska, A.; Oset, M.; et al. Contribution to the knowledge of the lichen biota of Bolivia. 5. Pol. Bot. J. 2013, 58, 697–733. [Google Scholar] [CrossRef]
- Flakus, A.; Sipman, H.J.M.; Rodríguez, P.; Schiefelbein, U.; Jablonska, A.; Oset, M.; Kukwa, M. Contribution to the knowledge of the lichen biota of Bolivia. 6. Pol. Bot. J. 2014, 59, 63–83. [Google Scholar] [CrossRef]
- Flakus, A.; Sipman, H.J.M.; Rodríguez, P.; Jablonska, A.; Oset, M.; Meneses, R.I.; Kukwa, M. Contribution to the knowledge of the lichen biota of Bolivia. 7. Pol. Bot. J. 2015, 60, 81–98. [Google Scholar] [CrossRef]
- Flakus, A.; Oset, M.; Rykaczewski, M.; Schiefelbein, U.; Kukwa, M. Contribution to the knowledge of the lichen biota of Bolivia. 8. Pol. Bot. J. 2016, 61, 107–126. [Google Scholar] [CrossRef]
- Silva-Cáceres, M.E.; Aptroot, A. Lichens from the Brazilian Amazon, with special reference to the genus Astrothelium. Bryologist 2017, 120, 166–182. [Google Scholar] [CrossRef]
- Silva-Cáceres, M.E.; Mota, N.; Alves, L.; de Almeida Pereira, T.; Aptroot, A. New records to Brazil and Southern Hemisphere f corticolous and saxicolous lichens from the semiarid region in Ceará State. Iheringia 2017, 72, 239–245. [Google Scholar] [CrossRef]
- Barrero, A.; Anjos, A.; de Sousa, L.; Aptroot, A.; Lücking, R.; Monique, V.; Silva-Cáceres, M.E. Epiphytic micdrolichens as indicators of phytosociological differentiation between Caatinga and Brejos Altitude. Acta Bot. Braslilica 2015, 29, 457–466. [Google Scholar] [CrossRef]
- Koch, N.M.; de Azevedo, S.M.; Lucheta, F.; Müller, S.C. Functional diversity and traits assembly patterns of lichens as indicators of succesional stages in a tropical rainforest. Ecol. Ind. 2013, 34, 22–30. [Google Scholar] [CrossRef]
- Käffer, M.I.; Dantas, R.V.; de Azevedo Martins, S.M. Characterization of the epiphytic lichen vegetation in a riparian forest in southern Brazil. Plant Ecol. Evol. 2016, 149, 92–100. [Google Scholar] [CrossRef]
- Silva-Cáceres, M.E.; Lücking, R.; Rambold, G. Phorophyte specificity and environmental parameters versus stocasticity as determinants for species composition of corticolous crustose lichen communities in the Atlantic rain forests of northeastern Brazil. Mycol. Prog. 2007, 6, 117–136. [Google Scholar] [CrossRef]
- Silva-Cáceres, M.E. Corticolous Crustose and Microfoliose Lichens of Northeastern Brazil; Libri botanici; IHW-Verlag: Eching, Postfach, 2007; Volume 22, pp. 1–168. ISBN 9783930167685. [Google Scholar]
- Rubio, C.; Saavedra, M.; Cuéllar, M.; Díaz, R.; Quilhot, W. Epiphytic lichens of Conguillío National Park, southern Chile. Gayana Bot. 2013, 70, 66–81. [Google Scholar] [CrossRef]
- Gatica, A.; Pereira, I.; Vallejos, O. Líquenes epífitos: Una herramienta para estudiar la continuidad ecológica en Isla Mocha, Chile. Gayana Bot. 2011, 68, 226–235. [Google Scholar] [CrossRef]
- Pereira, I. Micobiota liquenizada del Parque Katalapi, X Región, Chile. Gayana Bot. 2007, 64, 192–200. [Google Scholar] [CrossRef]
- Quilhot, W.; Cuellar, M.; Díaz, R.; Riquelme, F.; Rubio, C. Estudio preliminar de la flora liquénica de Isla Mocha, sur de Chile. Gayana Bot. 2010, 67, 206–212. [Google Scholar] [CrossRef]
- Pereira, I.; Müller, F.; Moya, M. Influence of Nothofagus bark pH on the lichen and bryophytes richness, Central Chile. Gayana Bot. 2014, 71, 120–130. [Google Scholar] [CrossRef]
- Wolf, J.H.D. Epiphyte communities of tropical montane rain forests in the northern Andes I. Lower montane communities. Phytocoenologia 1993, 22, 1–52. [Google Scholar] [CrossRef]
- Soto, E.; Bolaños, A.C. Diversidad de líquenes corticolas en el bosque subandino de la finca Zíngara (Cali, valle del Cauca). Rev. Cienc. 2010, 14, 35–44. [Google Scholar]
- Ramírez-Morán, N.A.; León-Gómez, M.; Lücking, R. Uso de biotipos de líquenes como bioindicadores de perturbación en fragmentos de bosque altoandino (Reserva Biológica “Encenillo”, Colombia). Caldasia 2016, 38, 31–52. [Google Scholar] [CrossRef]
- Holz, I.; Gradstein, S.R. Cryptogamic epiphytes in primary and recovering upper montane oak forests of Costa Rica—Species richness, community composition and ecology. Plant Ecol. 2005, 178, 89–109. [Google Scholar] [CrossRef]
- Rosabal, D.; Burgaz, A.R.; Reyes, O.J. Diversidad y distribución vertical de líquenes corticícolas en la pluvisilva montana de la Gran Piedra, Cuba. Bot. Compl. 2012, 36, 19–30. [Google Scholar] [CrossRef]
- Rosabal, D.; Aragón, G. Líquenes epífitos en el matorral costero de la Reserva Ecológica Siboney-Juticí (cuba). Bot. Compl. 2010, 34, 21–25. [Google Scholar]
- Benítez, A.; Prieto, M.; Aragon, G. Large trees and dense canopies: Key factors for maintaining high epiphytic diversity on trunk bases (bryophytes and lichens) in tropical montane forests. Forestry 2015, 88, 521–527. [Google Scholar] [CrossRef]
- Benítez, A.R.; Aragón, G.; Prieto, M. Lichen Diversity in Tropical Dry Forests Is Highly Influenced by Host Tree Traits (doc. ined). Chapter 4. Ph.D. Thesis, University Rey Juan Carlos, Móstoles, Spain, 2016. [Google Scholar]
- Nöske, N.M.; Sipman, H.J.M. Cryptogams of the Reserva Biológica San Francisco Province Zamora-Chinchipe, Southern Ecuador. II. Lichens. Cryptogam. Mycol. 2004, 25, 91–100. [Google Scholar]
- Pérez-Pérez, R.E.; Miramontes-Rojas, N.; Aguilar-Rosales, J.; Quiroz-Castelán, H. Macrolíquenes cortícolas de dos especies de coníferas del Parque Nacional lagunas de Zempoala. Acta Univ. 2008, 18, 33–39. [Google Scholar]
- Etayo, J.; Aptroot, A. Líquenes epífitos y hongos liquenícolas de Bahía Honda (Veraguas, Panamá). In Estudios Sobre la Biodiversidad de la Región de Bahía Honda (Veraguas, Panamá); Ibáñez, A., Castroviejo, S., Eds.; CSIC: Madrid, Spain, 2005; pp. 63–94. ISBN 978-84-00-08405-9. [Google Scholar]
- Ramos, D. Lista de especies de líquenes y hongos liquenícolas del Perú. Glalia 2014, 6, 1–49. [Google Scholar]
- Lugo, G. Preliminary inventory of corticolous lichens of the Sierra de San Luis, Venezuela. Rev. Biodivers. Neotrop. 2013, 3, 98–105. [Google Scholar] [CrossRef]
- Marcano, V.; Molares, A.; Sipman, H.; Calderón, L. A first checklist of the lichen-forming fungi of the Venezuelan Andes. Trop. Bryol. 1996, 12, 193–235. [Google Scholar] [CrossRef]
- Aragón, G.; Martínez, I.; Burgaz, A.R.; Belinchón, R.; Prieto, M.; Otálora, M.A.G.; Garcia, A.; Lopez, R.; de Paz, G.A. Inventariación de la diversidad liquénica del Parque Nacional de los Picos de Europa. In Proyectos de Investigación en Parques Nacionales, 2003–2006; Ramírez, L., Asensio, B., Eds.; Organismo Autónomo Parques Nacionales: Madrid, Spain, 2007; pp. 79–106. ISBN 978-84-8014-722-4. [Google Scholar]
- Aragón, G.; Martínez, I.; Belinchón, R. Aportación al conocimiento de los líquenes epífitos de Pinus halepensis, P. nigra y P. sylvestris en la Península Ibérica. Bot. Compl. 2006, 30, 61–70. [Google Scholar]
- Aragón, G.; Rico, V.J.; Belinchón, R. Lichen diversity from Cazorla, Segura and Las Villas Biosphere Reserve (SE Spain). Nova Hedwigia 2006, 82, 31–50. [Google Scholar] [CrossRef]
- Fos, S. Líquenes epífitos de los alcornocales ibéricos. Correlaciones bioclimáticas, anatómicas y densimétricas con el corcho de reproducción. Guineana 1998, 4, 1–507. [Google Scholar]
- Aragón, G.; Martínez, I. Contribución al conocimiento de los líquenes epifíticos de la Sierra de Alcaraz (Albacete, España). Cryptogam. Mycol. 1999, 20, 57–70. [Google Scholar] [CrossRef]
- Aragón, G.; Martínez, I. Contribución al conocimiento de los líquenes epifíticos de los Montes de Toledo (Toledo, España). Cryptogam. Bryol. Lichenol. 1997, 18, 63–75. [Google Scholar]
- Lohmus, P.; Lohmus, A. Snags, and their lichen flora in old Estonian peatland forets. Ann. Bot. Fennici 2001, 38, 265–280. [Google Scholar]
- Marmor, L.; Torra, T.; Saag, T.; Leppik, E.; Randlane, T. Lichens on Picea abies and Pinus sylvestris—From the bottom to the top. Lichenologist 2013, 45, 51–63. [Google Scholar] [CrossRef]
- Kivistö, L.; Kuusinen, M. Edge effects on the epiphytic lichen flora of Picea abies in middle boreal Finland. Lichenologist 2000, 32, 387–398. [Google Scholar] [CrossRef]
- Bricaud, O.; Roux, C. Deux associations lichéniques corticoles nouvelles, mésoméditerranéennes, sciaphiles: Le Ramonio-Striguletum mediterraneae et le Striguletum affinis. Lichenologist 1994, 26, 113–134. [Google Scholar] [CrossRef]
- Muggia, L.; Kati, V.; Rohrer, A.; Hallet, J.; Mayrhofer, H. Species diversity of lichens in the sacred groves of Epirus (Greece). Herzogia 2018, 31, 231–244. [Google Scholar] [CrossRef]
- Christensen, S.N. Lichens associated with Pinus nigra on Mt. Trapezitsa, Epirus, NW Greece. Acta Bot. Fennica 1994, 150, 11–20. [Google Scholar]
- Pirintsos, S.A.; Diamantopoulos, J.; Stamou, G.P. Analysis of the vertical distribution of epiphytic lichens on Pinus nigra (Mount Olympos, Greece) along an altitudinal gradient. Vegetatio 1993, 109, 63–70. [Google Scholar] [CrossRef]
- Király, I.; Nascimbene, J.; Tinya, F.; Ódor, P. Factors influencing epiphytic bryophyte and lichen species at different spatial scales in managed temperate forests. Biodivers. Conserv. 2013, 22, 209–223. [Google Scholar] [CrossRef]
- Loppi, S.; Pirintsos, S.A.; Olivieri, N.; Pacioni, G. Distribution of epiphytic lichens on Quercus pubescens along an altitudinal gradient on the Adriatic side of Central Italy. Stud. Geobot. 1999, 17, 85–90. [Google Scholar]
- Brunialti, G.; Frati, L.; Loppi, S. Fragmentation of mediterranean oak forests affects the diversity of epiphytic lichens. Nova Hedwig. 2012, 96, 265–278. [Google Scholar] [CrossRef]
- Loppi, S.; Pritintsos, S.A.; de Dominicis, V. Analysis of the distribution of epiphytic lichens on Quercus pubescens along an altitudinal gradient in a mediterranean area (Tuscany, ventral Italy). Isr. J. Plant Sci. 1997, 45, 53–58. [Google Scholar] [CrossRef]
- Nascimbene, J.; Nimis, P.L.; Dainese, M. Epiphytic lichen conservation in the Italian Alps: The role of forest type. Fungal Ecol. 2014, 11, 164–172. [Google Scholar] [CrossRef]
- Kubiak, D. Lichens of read oak Quercus rubra in the forest environment in the Olsztyn Lake District (NE Poland). Acta Mycol. 2006, 41, 319–328. [Google Scholar] [CrossRef]
- Fritz, O.; Heilmann-Clausen, J. Rot holes create key microhabitats for epiphytic lichens and bryophytes on beech (Fagus sylvatica). Biol. Conserv. 2010, 143, 1008–1016. [Google Scholar] [CrossRef]
- Bäcklund, A.; Jönsson, M.; Strengbom, J.; Frisch, A.; Thor, G. A pine is a pine and a spruce is a spruce—The effect of tree species and stand age on epihytic lichen communities. PLoS ONE 2016, 11, e0147004. [Google Scholar] [CrossRef]
- Özturk, Ş.; Oran, S.; Güvenç, Ş.; Dalkiran, N. Analysis of the distribution of epiphytic lichens in the oriental beech (Fagus orientalis Lipsky) forests along an altitudinal gradient in Uludag mountain, Bursa—Turkey. Pak. J. Bot. 2010, 42, 2661–2670. [Google Scholar]
- Güvenc, S.; Öztürk, S. Difference in epiphytic lichen communities on Quercus cerris from urban and rural areas in Bursa (Turkey). Pak. J. Bot. 2017, 49, 631–637. [Google Scholar]
- Dymytrova, L.; Nadyeina, O.; Naumovych, A.; Keller, C.; Scheidegger, C. Primeval Beech Forests of Ukrainian Carpathians are Sanctuaries for Rare and Endangered Epiphytic Lichens. Herzogia 2013, 26, 73–89. [Google Scholar] [CrossRef]
- Nimis, P.L.; Martellos, S. ITALIC-The Information System on Italian Lichens. 4.0. University of Trieste, Dept. of Biology, IN4.0/1. 2008. Available online: http://dbiodbs.univ.trieste.it/ (accessed on 26 November 2018).
- Rivas-Plata, E.; Lücking, R.; Sipman, H.J.; Mangold, A.; Kalb, K.; Lumbsch, H.T. A world-wide key to the thelotremoid Graphidaceae, excluding the Ocellularia-Myriotrema-Stegobolus clade. Lichenologist 2010, 42, 139–185. [Google Scholar] [CrossRef]
- Lakatos, M.; Rascher, U.; Büdel, B. Functional characteristics of corticolouslichens in the understory of a tropical lowland rain forest. New Phytol. 2006, 172, 679–695. [Google Scholar] [CrossRef]
- Smith, C.W.; Aptroot, A.; Coppins, B.J.; Fletcher, A.; Gilbert, O.L.; James, P.W.; Wolseley, P.A. The Lichens of Great Britain and Ireland; British Lichen Society: London, UK, 2009. [Google Scholar]
- San Miguel-Ayanz, J.; de Rigo, D.; Caudullo, G.; Houston, T.D.; Mauri, A. European Atlas of Forest Tree Species; European Commission: Luxembourg, 2016. [Google Scholar]
- Nimis, P.L.; Scheidegger, C.; Wolseley, P.A. Monitoring with Lichens: Monitoring Lichens; Kluwer Academic Published in Association with the NATO Scientific Affairs Division; Springer: Dordrecht, The Netherlands; London, UK, 2002. [Google Scholar]
- Svoboda, D.; Peksa, O.; Veselà, J. Epiphytic lichen diversity in central European oak forests: Assessment of the effects of natural environmental factors and human influences. Environ. Pollut. 2010, 158, 812–819. [Google Scholar] [CrossRef]
- Matos, P.; Pinho, P.; Aragón, G.; Martínez, I.; Nunes, A.; Soares, A.M.V.M.; Branquinho, C. Lichen traits responding to aridity. J. Ecol. 2015, 103, 451–458. [Google Scholar] [CrossRef]
- Prieto, M.; Martínez, I.; Aragón, G.; Verdú, M. Phylogenetic and functional structure of lichen communities under contrasting environmental conditions. J. Veg. Sci. 2017, 28, 871–881. [Google Scholar] [CrossRef]
- Ellis, C.J. Lichen epiphyte diversity: A species, community and trait-based review. Perspect. Plant Ecol. Evol. Syst. 2012, 14, 131–152. [Google Scholar] [CrossRef]
- Concostrina-Zubiri, L.; Pescador, D.S.; Martínez, I.; Escudero, A. Climate and small-scale factors determine functional diversity shifts of biological soil crusts in Iberian drylands. Biodiv. Conserv. 2014, 23, 1757–1770. [Google Scholar] [CrossRef]
- Newmaster, S.G.; Bell, F.W. The effects of silvicultural disturbances on cryptogam diversity in the boreal-mixed wood forests. Can. J. For. Res. 2002, 32, 38–51. [Google Scholar] [CrossRef]
- Benítez, A.; Prieto, M.; González, Y.; Aragón, G. Effects of tropical montane forest disturbance on epiphytic macrolichens. Sci. Total Environ. 2012, 441, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Marini, L.; Nascimbene, J.; Nimis, P.L. Large-scale patterns of epiphytic lichen species richness: Photobiont-dependent response to climate and forest structure. Sci. Total Environ. 2011, 409, 4381–4386. [Google Scholar] [CrossRef]
- Li, S.; Liu, W.Y.; Li, D.W. Bole epiphytic lichens as potential indicators of environmental change in subtropical forest ecosystems in Southwest China. Ecol. Indic. 2013, 29, 93–104. [Google Scholar] [CrossRef]
- Nash, T.H. Lichen Biology; Cambridge University Press: New York, NY, USA, 2008. [Google Scholar]
- Sillett, S.C.; Antoine, M.E. Lichens and bryophytes in forest canopies. In Forest Canopies; Lowman, M.D., Inker, H.B., Eds.; Elsevier Academic Press: Oxford, UK, 2004; pp. 151–174. ISBN 9780080491349. [Google Scholar]
- Keddy, P.A. Assembly and response rules: Two goals for predictive community ecology. J. Veg. Sci. 1992, 3, 157–164. [Google Scholar] [CrossRef]
- Weiher, E.; Clarke, G.D.P.; Keddy, P.A. Community assembly rules, morphological dispersion, and the coexistence of plant species. OIKOS 1998, 81, 309–322. [Google Scholar] [CrossRef]
- Grime, J.P. Trait convergence and trait divergence in herbaceous plant communities: Mechanisms and consequences. J. Veg. Sci. 2006, 17, 255–260. [Google Scholar] [CrossRef]
- Petchey, O.L.; Gaston, K.J. Functional diversity (FD), species richness and community composition. Ecol. Lett. 2002, 5, 402–411. [Google Scholar] [CrossRef]
- FAO. El Estado de los Bosques del Mundo. Los Bosques y la Agricultura: Desafíos y Oportunidades; FAO: Rome, Italy, 2016; 36p, ISBN 9789253092086. [Google Scholar]
| Bibliographic Reference | Country | Alt (m) | Forest Type |
|---|---|---|---|
| Central-South America | |||
| [20] | Argentina | 500–1350 | Chaco, semiarid lowland forest |
| [21] | Argentina | 1000 | Belt of mountain woodland and the transition with the Chaco lowland forest |
| [22] | Argentina | 4 | Riverside forest |
| [23,24,25,26,27,28,29] | Bolivia | 155–4500 | Chiquitano-Chaqueno forest, lowland Amazon forest, Tucumano-boliviano montane forest, Yungas montane forest, Podocarpus, Polylepis |
| [30] | Brazil | 80 | Tropical rainforest |
| [31] | Brazil | 130–250 | Caatinga vegetation |
| [32] | Brazil | 400–600 | Brejos and Caatinga |
| [33] | Brazil | 300–800 | Atlantic rainforest |
| [34] | Brazil | 240–872 | Riparian forest |
| [35] | Brazil | 800–900 | Atlantic rainforest |
| [36] | Brazil | 0–800 | Atlantic rainforest, Caatinga |
| [37] | Chile | 1000–1500 | Andean-Patagonian forests and the Andean deciduous forest |
| [38] | Chile | 8–308 | Valdivian temperate rainforest (without Nothofagus spp.) |
| [39] | Chile | 1100–1150 | Montane coniferous forest (Fitzroya cupressoides) |
| [40] | Chile | 0–349 | Valdivian temperate rainforest |
| [41] | Chile | 1100–1150 | Temperate forest (Nothofagus dombeyi, N. nitida, and N. obliqua) |
| *Rodríguez et al. (in prep.) | Chile | 930–1050 | Temperate forest (Nothofagus pumilio) |
| [42] | Colombia | 700–4000 | Lowland forest, lower montane rainforest, tropical montane rainforest, and Polylepis forest |
| [43] | Colombia | 1900–2000 | Premontane forest |
| [44] | Colombia | 2800–3200 | High Andean forest |
| [45] | Costa Rica | 2900 | Oak forest |
| [46] | Cuba | 1000 | Tropical montane rainforest |
| [47] | Cuba | 20 | Coastal sclerophyllous forest |
| [48] | Ecuador | 2196–2848 | Tropical montane rainforest (primary, secondary, and monospecific Alnus acuminata) |
| [49] | Ecuador | 0–300 | Tropical dry forest |
| [50] | Ecuador | 1800–2650 | Tropical montane rainforest |
| *Aragón et al. (in prep.) | Ecuador | 80–300 | Amazonian forest |
| [51] | Mexico | 2714–2775 | Coniferous forest (Pinus ayacahuite, Abies religiosa) |
| [52] | Panama | 0–160 | Lowland and coastal forest |
| [53] | Peru | 300–1500 | Tropical dry forest |
| [54] | Venezuela | 106–1400 | Tropical rainforest |
| [55] | Venezuela | 800–5000 | Montane forest (lower and upper) and Polylepis sericea forest |
| Europe | |||
| *Hurtado et al. (in prep) | Austria | 750–1120 | Temperate forest (Fagus sylvatica) |
| [56] | Spain | 800 | Temperate forest (Fagus sylvatica) |
| [57] | Spain | 870–1100 | Mediterranean pine forest (Pinus pinaster, P. nigra) |
| [58] | Spain | 1280–1560 | Mediterranean pine forest |
| [59] | Spain | 300–700 | Oak forest (Quercus suber) |
| [60] | Spain | 1550 | Mediterranean pine forest (Pinus nigra, Ilex, Acer) |
| [61] | Spain | 1000 | Oak forest (Quercus ilex, Juniperus oxycedrus) |
| *Aragón et al. (in prep.) | Spain | 710 | Oak forest (Quercus faginea) |
| *Hurtado et al. (in prep) | Spain | 856 | Temperate forest (Fagus sylvatica) |
| [62] | Estonia | 30–40 | Coniferous forest (Picea abies, Pinus sylvestris) |
| [63] | Estonia | 170–200 | Coniferous forest (Picea abies, Pinus sylvestris) |
| [64] | Finland | 170 | Coniferous forest (Picea abies) |
| [65] | France | 350–500 | Oak forest (Quercus ilex) |
| *Hurtado et al. (in prep) | France | 1180–1272 | Temperate forest (Fagus sylvatica) |
| [66] | Greece | 570–1210 | Oak forest (Quercus cerris, Q. coccifera, Q. frainetto, Q. petraea, Q. pubescens, Q. trojana, Pinus nigra) |
| [67] | Greece | 800–1500 | Mediterranean pine forest (Pinus nigra) |
| [68] | Greece | 750–1510 | Mediterranean pine forest (Pinus nigra) |
| [69] | Hungary | 250-350 | Temperate forest (Fagus, Quercus, Carpinus, Pinus) |
| [70] | Italy | 0–1000 | Oak forest (Quercus pubescens) |
| [71] | Italy | 50–390 | Oak forest (Quercus ilex, Q. cerris, Q. pubescens) |
| [72] | Italy | 0–900 | Oak forest (Quercus pubescens) |
| [73] | Italy | 400–1900 | Oak forest (Quercus) and coniferous forest (Pinus sylvestris, Abies alba) |
| *Hurtado et al. (in prep) | Italy | 1077–1213 | Temperate forest (Fagus sylvatica) |
| [74] | Poland | 88–150 | Oak forest (Quercus rubra) |
| *Hurtado et al. (in prep) | Slovakia | 1233 | Temperate forest (Fagus sylvatica) |
| [75] | Sweden | 25–170 | Temperate forest (Fagus sylvatica) |
| [76] | Sweden | 260–583 | Coniferous forest (Pinus contorta, P. sylvestris, Picea abies) |
| *Hurtado et al. (in prep) | Sweden | 107–161 | Temperate forest (Fagus sylvatica) |
| [77] | Turkey | 900–1400 | Temperate forest (Fagus orientalis) |
| [78] | Turkey | 21–717 | Oak forest (Quercus cerris) |
| [79] | Ukraine | 400–1350 | Temperate forest (Fagus sylvatica) |
| Growth Forms (Examples) |
|---|
| Leprarioid (Lepraria incana) |
| Crustose |
| Conspicuous (Pertusaria albescens) |
Inconspicuous
|
| Granular crustose (Agonimia octospora) |
| Crustose ecorticate (Herpothallon rubrocinctum) |
| Crustose placodioid (Pyxine berteriana) |
| Squamulose |
| Flat squamulose (Normandina pulchella) |
| Convex squamulose (Phyllopsora furfuracea) |
| Thallus Psoroma-type (Psoroma hypnorum) |
| Squamulose-foliose (Pannaria rubiginosa) |
| Foliose |
| Foliose placodioid (Pectenia plumbea) |
| Foliose umbilicate (Dictyonema glabratum) |
| Foliose narrow-lobed (Physconia venusta) |
| Foliose broad-lobed (Parmelina tiliacea) |
| Large foliose tomentose (Erioderma leylandii) |
| Large foliose glabrae and appressed (Peltigera horizontalis) |
| Large foliose glabrae and ascending (Sticta weigelii) |
| Foliose gelatinous swollen (Collema nigrescens) |
| Foliose gelatinous flat (Leptogium cyanescens) |
| Filamentous (Coenogonium linkii) |
| Fruticose |
| Fruticose cylindrical light-colored (Usnea glabrescens) |
| Fruticose cylindrical dark-colored (Bryoria implexa) |
| Fruticose dorsiventral light-colored (Ramalina fraxinea) |
| Fruticose dorsiventral dark-colored (Pseudevernia furfuracea) |
| Mixed (Cladonia fimbriata) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aragón, G.; Martínez, I.; Hurtado, P.; Benítez, Á.; Rodríguez, C.; Prieto, M. Using Growth Forms to Predict Epiphytic Lichen Abundance in a Wide Variety of Forest Types. Diversity 2019, 11, 51. https://doi.org/10.3390/d11040051
Aragón G, Martínez I, Hurtado P, Benítez Á, Rodríguez C, Prieto M. Using Growth Forms to Predict Epiphytic Lichen Abundance in a Wide Variety of Forest Types. Diversity. 2019; 11(4):51. https://doi.org/10.3390/d11040051
Chicago/Turabian StyleAragón, Gregorio, Isabel Martínez, Pilar Hurtado, Ángel Benítez, Clara Rodríguez, and María Prieto. 2019. "Using Growth Forms to Predict Epiphytic Lichen Abundance in a Wide Variety of Forest Types" Diversity 11, no. 4: 51. https://doi.org/10.3390/d11040051
APA StyleAragón, G., Martínez, I., Hurtado, P., Benítez, Á., Rodríguez, C., & Prieto, M. (2019). Using Growth Forms to Predict Epiphytic Lichen Abundance in a Wide Variety of Forest Types. Diversity, 11(4), 51. https://doi.org/10.3390/d11040051







