The Western Amazonian Richness Gradient for Squamate Reptiles: Are There Really Fewer Snakes and Lizards in Southwestern Amazonian Lowlands?
Abstract
1. Introduction
2. Materials and Methods
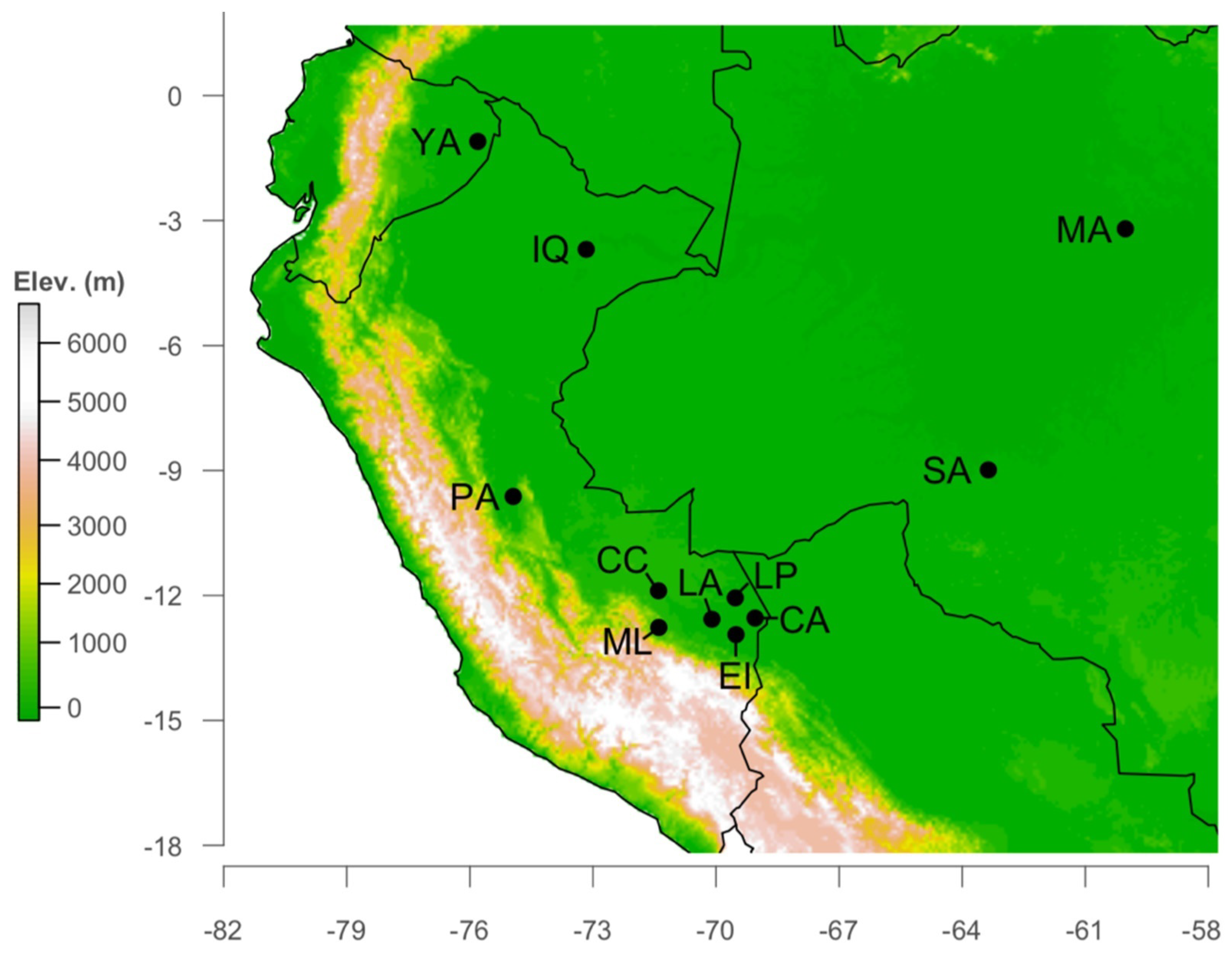
2.1. Study Site
2.2. Data Collection
2.3. Data Tabulation and Analysis
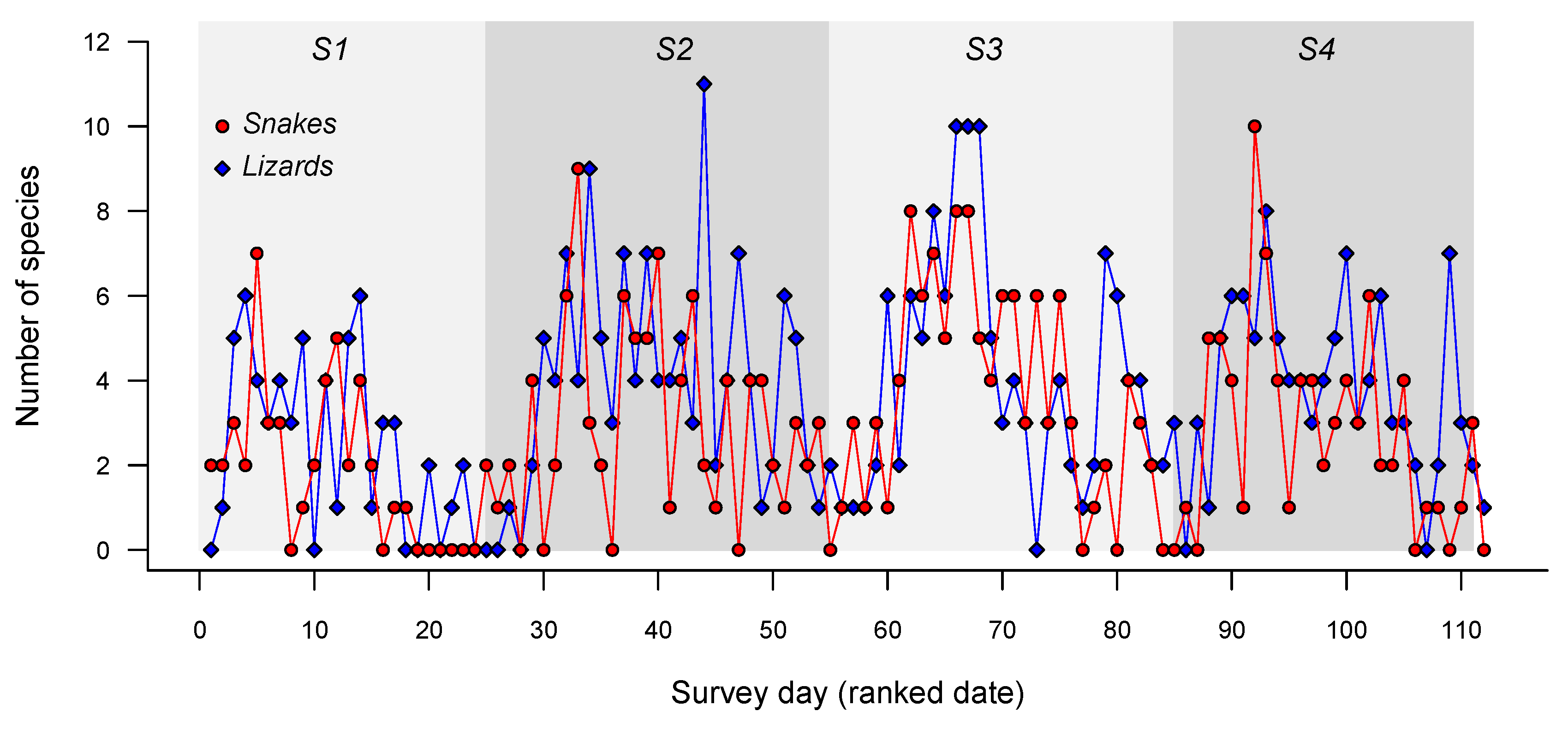
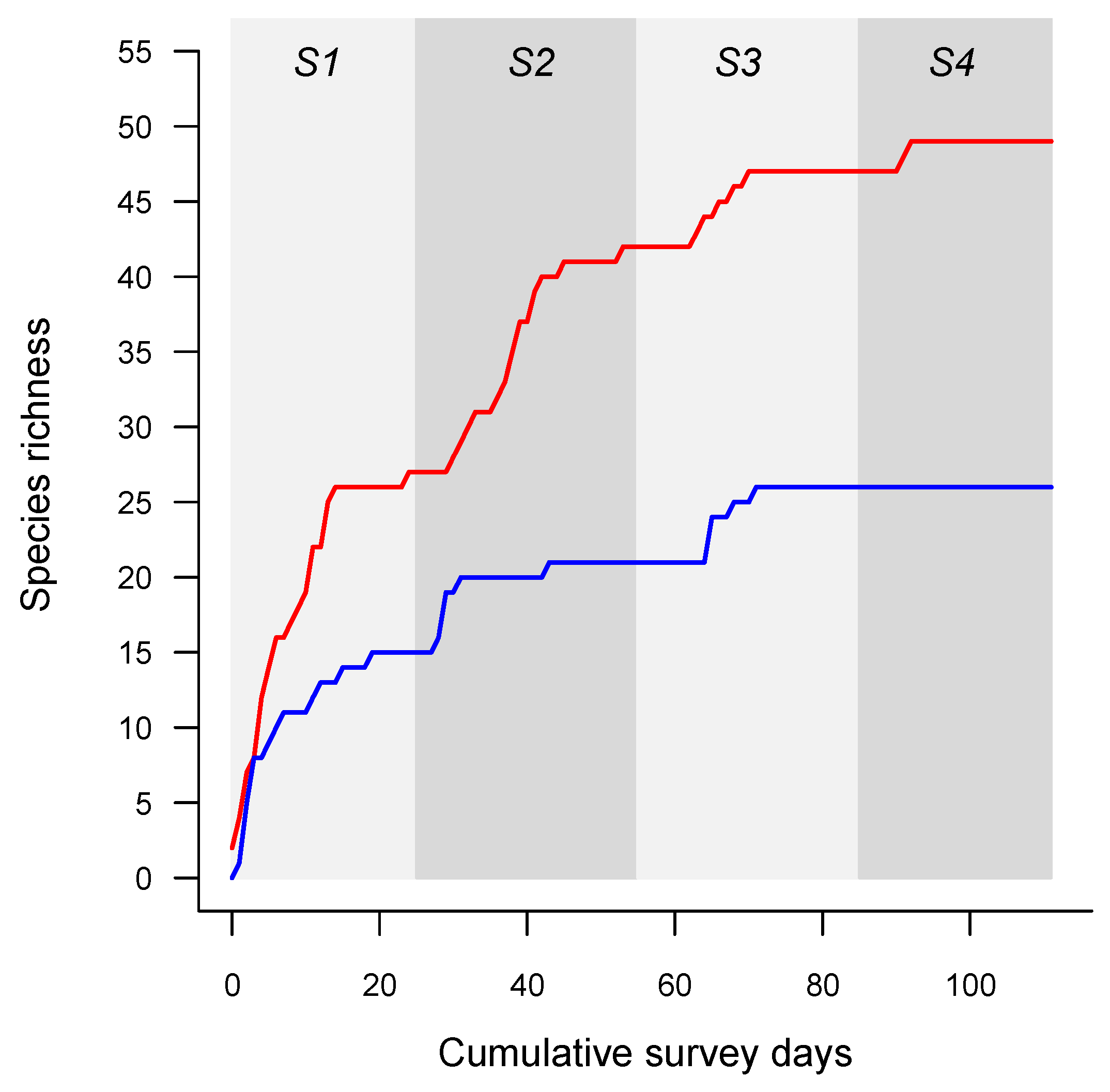
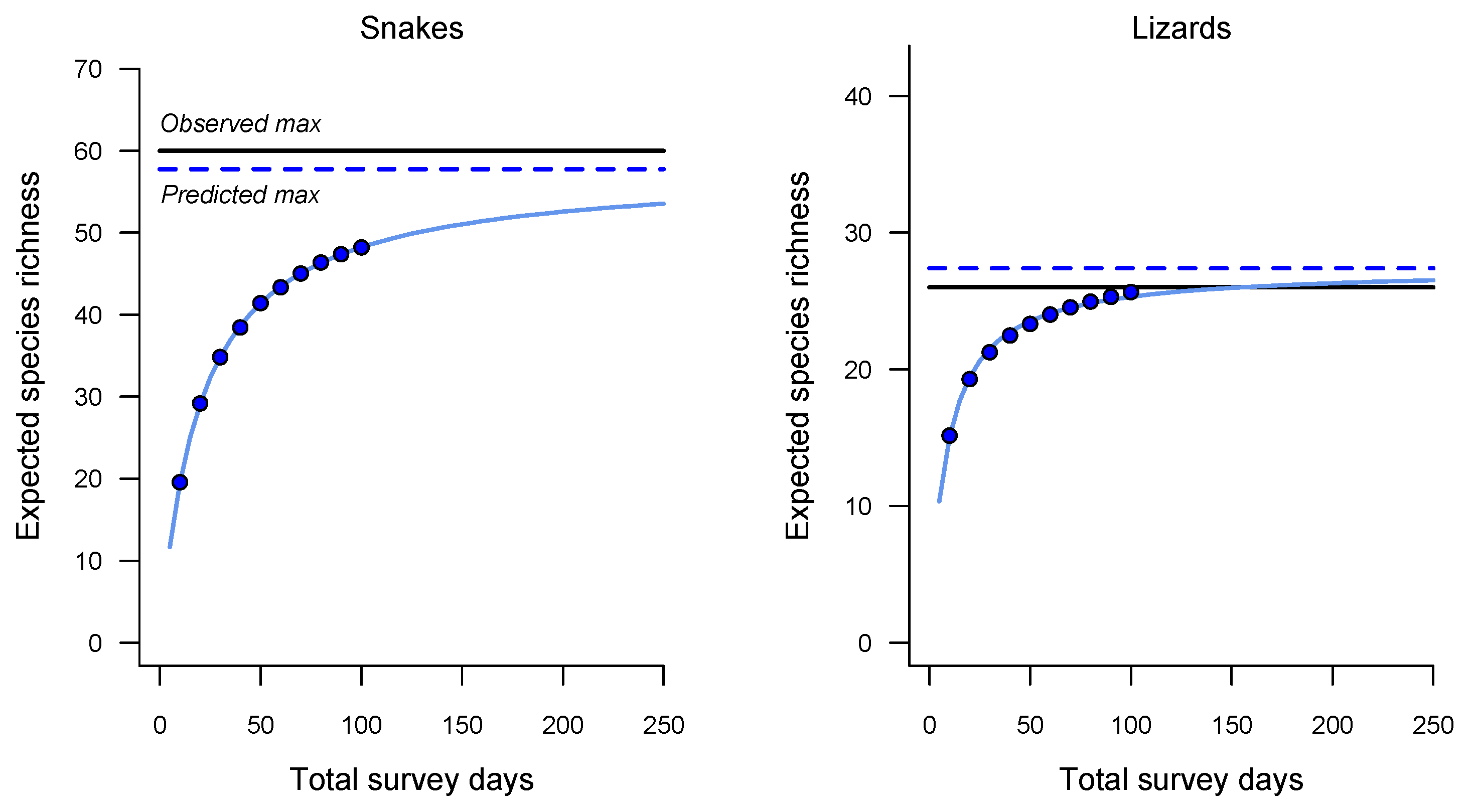
3. Results
4. Discussion
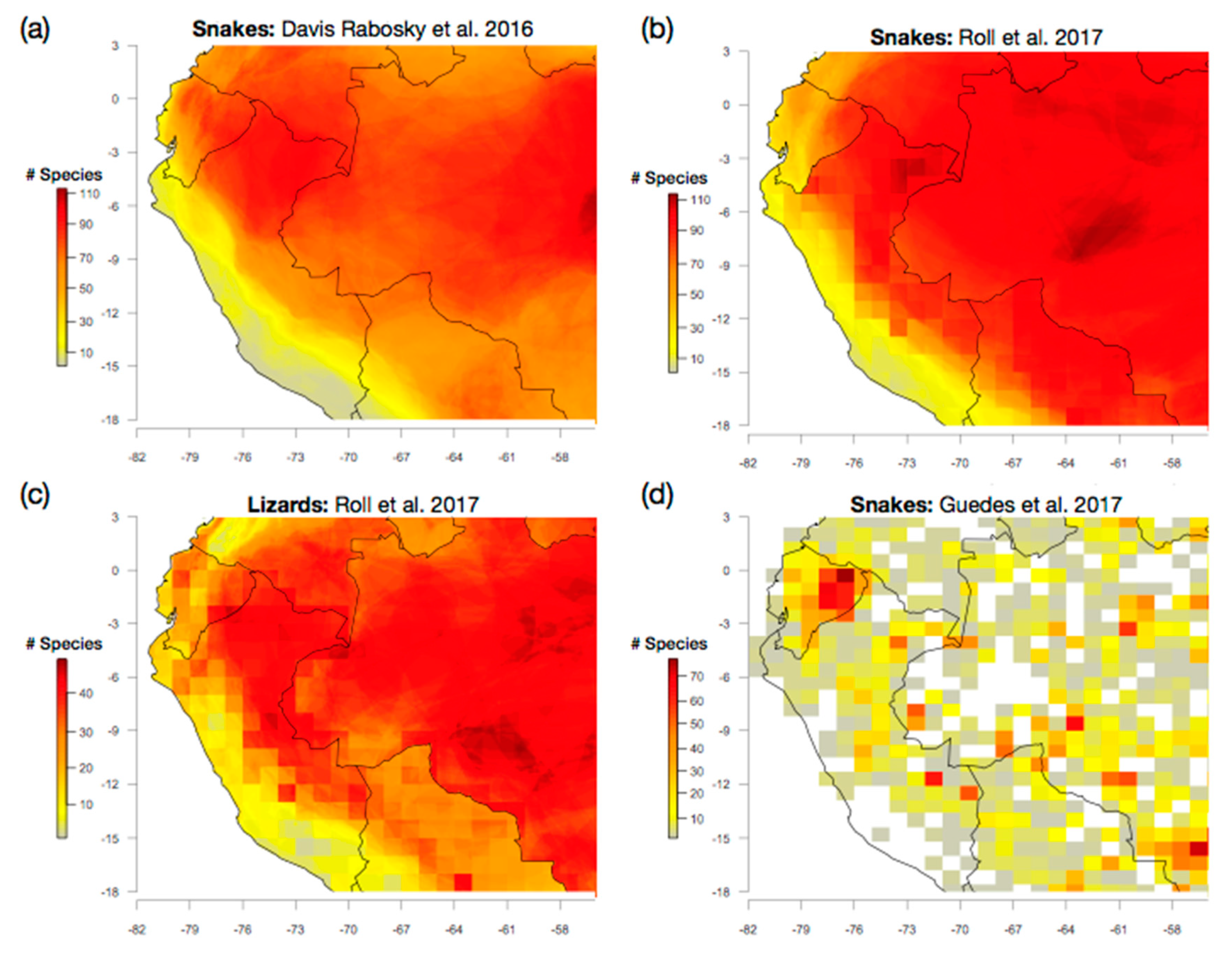
4.1. A Western Amazonian Richness Gradient: Does It Exist?
4.2. Taxonomic Issues
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Clade | Species | LABS | LABS-R | LABS-X | Material Examined |
|---|---|---|---|---|---|
| Amphisbaenidae (lizard) | Amphisbaena alba | X 1 | |||
| Amphisbaena fuliginosa | X | X | X | ||
| Anguidae (lizard) | Diploglossus fasciatus | X 2 | |||
| Gekkota (lizard) | Gonatodes hasemani | X | X | X | RAB 2799, MUSM 38982 |
| Gonatodes humeralis | X | X | X | RAB 3434, MUSM 38984 | |
| Pseudogonatodes guianensis | X | X | X | RAB 2508, MUSM 39869 | |
| Thecadactylus solimoensis | X | X | X | RAB 3277, MUSM 39869 | |
| Scincidae (lizard) | Copeoglossum nigropunctatum | X | X | ||
| Exila nigropalmata | X | X | X 3 | RAB 3514, MUSM 39156 | |
| Varzea altamazonica | X | X | |||
| Teioidea (lizard) | Alopoglossus angulatus | X | X | X | UMMZ 246710, MUSM 36813 |
| Ameiva ameiva | X | X | X | RAB 2924, MUSM 39781 | |
| Bachia dorbignyi | X | X | X | RAB 3497, MUSM 39788 | |
| Bachia trisanale | X | X | X3 | RAB 3469, MUSM 38919 | |
| Cercosaura argulus | X | X | X | RAB 2701, MUSM 38930 | |
| Cercosaura bassleri | X | X | X | UMMZ 245038, MUSM 38937 | |
| Cercosaura eigenmanni | X | X | X | UMMZ 246735, MUSM 36933 | |
| Dracaena guianensis | X 1 | ||||
| Iphisa elegans | X | X | X 3 | RAB 2931, MUSM 39020 | |
| Kentropyx altamazonica | X 1,2 | ||||
| Kentropyx pelviceps | X | X | X | UMMZ 246751, MUSM 37269 | |
| Potamites ecpleopus | X 1,2 | ||||
| Ptychoglossus brevifrontalis | X | X | X | UMMZ 246762, MUSM 39120 | |
| Tupinambis teguixin | X | X | X | RAB 3430 | |
| Iguania (lizard) | Anolis fuscoauratus | X | X | X | RAB 2868, MUSM 36859 |
| Anolis ortonii | X | X | X | UMMZ 248371 | |
| Anolis punctatus | X | X | X | RAB 2537, MUSM 39782 | |
| Anolis tandai | X | X | X | RAB 2660, MUSM 38909 | |
| Enyalioides palpebralis | X | X | X | MUSM 38981 | |
| Plica plica | X | X | X | RAB 2704, MUSM 39860 | |
| Plica umbra | X | X | X | RAB 2682, MUSM 39099 | |
| Polychrus liogaster | X 1 | ||||
| Stenocercus fimbriatus | X 1,2 | ||||
| Stenocercus roseiventris | X 1,2 | ||||
| Uracentron azureum | X | X | X | ||
| Uracentron flaviceps | X | X | X | MUSM 39154 | |
| Typhlopidae (snake) | Amerotyphlops reticulatus | X | X | X | UMMZ 248453, MUSM 36160 |
| Leptotyphlopidae (snake) | Epictia diaplocia | X | X | X | RAB 2932, MUSM 39046 |
| Aniliidae (snake) | Anilius scytale | X | X | X 3 | RAB 2499, MUSM 36848 |
| Boidae (snake) | Boa constrictor | X | X | X | |
| Corallus batesii | X | X | X | ||
| Corallus hortulanus | X | X | X | UMMZ 248448, MUSM 36965 | |
| Epicrates cenchria | X | X | X | RAB 2903, MUSM 37120 | |
| Eunectes murinus | X | X | X | RAB 3417 | |
| Colubrinae (snake) | Chironius carinatus / exoletus | X | X | X 3 | UMMZ 246087, MUSM 38943 |
| Chironius fuscus | X | X | X | UMMZ 245047, MUSM 36957 | |
| Chironius multiventris | X | X | X | UMMZ 245049, MUSM 36962 | |
| Chironius scurrulus | X 1,7 | ||||
| Chironius sp. | X | X | X 3,7 | UMMZ 248360 | |
| Dendrophidion dendrophis | X | X | X | MUSM 36979 | |
| Drymarchon corais | X | X | X 3 | RAB 2739, MUSM 36145 | |
| Drymobius rhombifer | X | X | X 3 | UMMZ 245052, MUSM 39824 | |
| Drymoluber dichrous | X | X | X | UMMZ 246799, MUSM 38977 | |
| Leptophis ahaetulla | X | X | X | UMMZ 246823, MUSM 37345 | |
| Oxybelis aeneus | X 1 | ||||
| Oxybelis fulgidus | X 1 | ||||
| Phrynonax poecilonotus | X | X | X 3 | RAB 3532, MUSM 39115 | |
| Rhinobothryum lentiginosum | X | X | X | ||
| Spilotes pullatus | X | X | X | ||
| Spilotes sulphureus | X 1 | ||||
| Tantilla melanocephala | X | X | X 3 | UMMZ 246845, MUSM 39145 | |
| Dipsadinae (snake) | Apostolepis nigroterminata | X | X | X 3 | RAB 3393, MUSM 39783 |
| Atractus elaps | X | X | X 3 | RAB 3414, MUSM 39784 | |
| Atractus major | X 1 | ||||
| Atractus snethlageae | X | X | X 4 | RAB 2666, MUSM 35703 | |
| Atractus sp. | X | X | X 3 | RAB 2924, MUSM 38913 | |
| Clelia clelia | X | X | X | ||
| Dipsas catesbyi | X | X | X | RAB 2802, MUSM 38970 | |
| Dipsas indica | X | X | X 3 | RAB 3561, MUSM 38973 | |
| Drepanoides anomalus | X | X | X | RAB 3408, MUSM 38974 | |
| Erythrolampus dorsocorallinus6 | X | X | X 3 | MUSM 36049 | |
| Erythrolampus oligolepis6 | X | X | X 3 | RAB 3411, MUSM 39049 | |
| Erythrolampus reginae6 | X | X | X | RAB 3436, MUSM 39837 | |
| Erythrolampus sp. | X | X | X | RAB 2711 | |
| Erythrolampus taeniogaster6 | X | X | X | ||
| Helicops angulatus | X | X | X | UMMZ 245053, MUSM 38989 | |
| Helicops leopardinus | X | X | X | ||
| Helicops polylepis | X | X | |||
| Hydrops triangularis | X | X | X 3 | RAB 3500, MUSM 38992 | |
| Imantodes cenchoa | X | X | X | UMMZ 246814, MUSM 35857 | |
| Imantodes lentiferus | X | X | X | RAB 2900, MUSM 39834 | |
| Leptodeira annulata | X | X | X | UMMZ 246822, MUSM 39043 | |
| Oxyrhopus formosus | X | X | X | UMMZ 248365, MUSM 39077 | |
| Oxyrhopus melanogenys | X | X | X | UMMZ 246832, MUSM 39855 | |
| Oxyrhopus petolarius | X | X | X | UMMZ 245072, MUSM 39081 | |
| Philodryas argentea | X | ||||
| Philodryas viridissima | X | X | |||
| Pseudoboa coronata | X | ||||
| Pseudoeryx plicatilis | X | X | X | MUSM 24359 | |
| Siphlophis cervinus | X | X | X 3 | MUSM 39141 | |
| Siphlophis compressus | X | X | X | MUSM 39142 | |
| Taeniophallus brevirostris | X | X | X 3 | RAB 2462, MUSM 39143 | |
| Taeniophallus occipitalis | X | X | X | RAB 3391, MUSM 37708 | |
| Xenodon rhabdocephalus | X | X | X 3 | UMMZ 246850, MUSM 39158 | |
| Xenodon severus | X | X | X | RAB 2712 | |
| Xenopholis scalaris | X | X | X | UMMZ 245077, MUSM 39889 | |
| Elapidae (snake) | Micrurus annellatus | X | X | X | UMMZ 248451, MUSM 39056 |
| Micrurus hemprichii | X | X | X 3,5 | ||
| Micrurus lemniscatus | X | X | X | UMMZ 248456, MUSM 39057 | |
| Micrurus obscurus | X | X | X | UMMZ 248458, MUSM 39846 | |
| Micrurus surinamensis | X | X | X 3 | UMMZ 246861, MUSM 37353 | |
| Viperidae (snake) | Bothrops atrox | X | X | X | RAB 2814, MUSM 35721 |
| Bothrops bilineatus | X | X | X | UMMZ 246865, MUSM 36919 | |
| Bothrops brazili | X | X | X | MUSM 36922 | |
| Lachesis muta | X | X | X | UMMZ 248454, MUSM 36149 |
References
- Rosenzweig, M.L. Species Diversity in Space and Time; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Couvreur, T.L.P. Odd man out: Why are there fewer plant species in African rain forests? Plant Syst. Evol. 2015, 301, 1299–1313. [Google Scholar] [CrossRef]
- Pianka, E.R. Zoogeography andspeciation of Australian desert lizards. Copeia 1972, 127–145. [Google Scholar] [CrossRef]
- Pianka, E.R. Ecology and Natural History of Desert Lizards; Princeton University Press: Princeton, NJ, USA, 1986. [Google Scholar]
- Morton, S.R.; James, C.D. The diversity and abundance of lizards in arid Australia—A new hypothesis. Am. Nat. 1988, 132, 237–256. [Google Scholar] [CrossRef]
- Rabosky, D.L.; Cowan, M.A.; Talaba, A.L.; Lovette, I.J. Species interactions mediate phylogenetic community structure in a hyperdiverse lizard assemblage from arid Australia. Am. Nat. 2011, 178, 579–595. [Google Scholar] [CrossRef] [PubMed]
- Bass, M.S.; Finer, M.; Jenkins, C.N.; Kreft, H.; Cisneros-Heredia, D.F.; McCracken, S.F.; Pitman, N.C.A.; English, P.H.; Swing, K.; Villa, G.; et al. Global Conservation Significance of Ecuador’s Yasuni National Park. PLoS ONE 2010, 5, e8767. [Google Scholar] [CrossRef]
- Jenkins, C.N.; Pimm, S.L.; Joppa, L.N. Global patterns of terrestrial vertebrate diversity and conservation. Proc. Natl. Acad. Sci. USA 2013, 110, E2602–E2610. [Google Scholar] [CrossRef] [PubMed]
- Pitman, N.C.A.; Terborgh, J.W.; Silman, M.R.; Nunez, P.; Neill, D.A.; Ceron, C.E.; Palacios, W.A.; Aulestia, M. A comparison of tree species diversity in two upper Amazonian forests. Ecology 2002, 83, 3210–3224. [Google Scholar] [CrossRef]
- Magnusson, W.E. Snakes and Other Lizards; Open Science Publishers: New York, NY, USA, 2019. [Google Scholar]
- Da Silva, N.J.; Sites, J.W. Patterns of diversity of neotropical squamate reptile species with emphasis on the brazilian amazon and the conservation potential of indigenous reserves. Conserv. Biol. 1995, 9, 873–901. [Google Scholar] [CrossRef]
- Guedes, T.B.; Sawaya, R.J.; Zizka, A.; Laffan, S.; Faurby, S.; Pyron, R.A.; Bernils, R.S.; Jansen, M.; Passos, P.; Prudente, A.L.C.; et al. Patterns, biases and prospects in the distribution and diversity of Neotropical snakes. Glob. Ecol. Biogeogr. 2018, 27, 14–21. [Google Scholar] [CrossRef]
- Rabosky, A.R.D.; Cox, C.L.; Rabosky, D.L.; Title, P.O.; Holmes, I.A.; Feldman, A.; McGuire, J.A. Coral snakes predict the evolution of mimicry across New World snakes. Nat. Commun. 2016, 7, 11484. [Google Scholar] [CrossRef]
- Roll, U.; Feldman, A.; Novosolov, M.; Allison, A.; Bauer, A.M.; Bernard, R.; Bohm, M.; Castro-Herrera, F.; Chirio, L.; Collen, B.; et al. The global distribution of tetrapods reveals a need for targeted reptile conservation. Nat. Ecol. Evol. 2017, 1, 1677–1682. [Google Scholar] [CrossRef] [PubMed]
- Duellman, W. Cusco Amazonico; Comstock Publishing: Ithaca, NY, USA, 2005. [Google Scholar]
- Whitworth, A.; Downie, R.; von May, R.; Villacampa, J.; MacLeod, R. How much potential biodiversity and conservation value can a regenerating rainforest provide? A ‘best-case’ approach from the Peruvian Amazon. Trop. Conserv. Sci. 2016, 9, 224–245. [Google Scholar] [CrossRef]
- De Fraga, R.; Stow, A.J.; Magnusson, W.E.; Lima, A.P. The Costs of Evaluating Species Densities and Composition of Snakes to Assess Development Impacts in Amazonia. PLoS ONE 2014, 9, e105453. [Google Scholar] [CrossRef] [PubMed]
- Greene, H.W. Snakes: The Evolution of Mystery in Nature; University of California Press: Berkeley, CA, USA, 1997. [Google Scholar]
- Catenazzi, A.; Lehr, E.; von May, R. The amphibians and reptiles of Manu National Park and its buffer zone, Amazon basin and eastern slopes of the Andes, Peru. Biota Neotrop. 2013, 13, 269–283. [Google Scholar] [CrossRef]
- Von May, R.; Donnelly, M.A. Do trails affect relative abundance estimates of rainforest frogs and lizards? Austral Ecol. 2009, 34, 613–620. [Google Scholar] [CrossRef]
- Von May, R.; Jacobs, J.M.; Santa-Cruz, R.; Valdivia, J.; Huaman, J.M.; Donnelly, M.A. Amphibian community structure as a function of forest type in Amazonian Peru. J. Trop. Ecol. 2010, 26, 509–519. [Google Scholar] [CrossRef]
- Pitman, N.C.A. An Overview of the Los Amigos Watershed, Madre de Dios, Southeastern Peru; (Unpublished Report); Amazon Conservation Association: Washington, DC, USA, 2010. [Google Scholar]
- Silman, M.R.; Terborgh, J.W.; Kiltie, R.A. Population regulation of a dominant-rain forest tree by a major seed-predator. Ecology 2003, 84, 431–438. [Google Scholar] [CrossRef]
- Griscom, B.W.; Ashton, P.M.S. A self-perpetuating bamboo disturbance cycle in a neotropical forest. J. Trop. Ecol. 2006, 22, 587–597. [Google Scholar] [CrossRef]
- Olivier, J. Bambusiform grasses (Poaceae) from the Los Amigos River, Madre de Dios, Peru. Rev. Peru. Biol. 2008, 15, 121–126. [Google Scholar]
- Thompson, G.G.; Thompson, S.A. Comparative temperature in funnel and pit traps. Aust. J. Zool. 2009, 57, 311–316. [Google Scholar] [CrossRef]
- Ascenso, A.C.; Costa, J.C.L.; Prudente, A.L.C. Taxonomic revision of the Erythrolamprus reginae species group, with description of a new species from Guiana Shield (Serpentes: Xenodontinae). Zootaxa 2019, 4586, 65–97. [Google Scholar] [CrossRef]
- Colwell, R.K.; Coddington, J.A. Estimating terrestrial biodiversity through extrapolation. Philos. Trans. R. Soc. B 1994, 345, 101–118. [Google Scholar]
- De Oliveira, L.; Araujo, I.; Prudente, A.L.C.; de Fraga, R.; de Almeida, A.; Ascenso, A.C. New distributional records of the toad-headed pitviper Bothrocophias hyoprora (Amaral, 1935) in Brazil. Amphib. Reptile Conserv. 2018, 12, 1–4. [Google Scholar]
- Colwell, R.K.; Winkler, D.W. A null model for null models in biogeography. In Ecological Communities: Conceptual Issues and the Evidence; Strong, D.R., Simberloff, D., Abele, L.G., Thistle, A.B., Eds.; Princeton University Press: Princeton, NJ, USA, 1984; pp. 344–359. [Google Scholar]
- Dixon, J.R.; Soini, P. The Reptiles of the Upper Amazon Basin, Iquitos Region, Peru I–VII; Milwaukee Public Museum: Milwaukee, WI, USA, 1986. [Google Scholar]
- Doan, T.M.; Arriaga, W. Microgeographic variation in species composition of the herpetofaunal communities of Tambopata Region, Peru. Biotropica 2002, 34, 101–117. [Google Scholar] [CrossRef]
- Schluter, A.; Icochea, J.; Perez, J.M. Amphibians and reptiles of the lower Rio Llullapichis, Amazonian Peru: Updated species list with ecological and biogeographic notes. Salamandra 2004, 40, 141–146. [Google Scholar]
- Francisco, B.C.S.; Pinto, R.R.; Fernandes, D.S. Taxonomy of Epictia munoai (Orejas-Miranda, 1961) (Squamata: Serpentes: Leptotyphlopidae). Zootaxa 2012, 3512, 42–52. [Google Scholar] [CrossRef]
- Kreft, H.; Koster, N.; Kuper, W.; Nieder, J.; Barthlott, W. Diversity and biogeography of vascular epiphytes in western Amazonia, Yasuní, Ecuador. J. Biogeogr. 2004, 31, 1463–1476. [Google Scholar] [CrossRef]
- Aceby, A.; Kromer, T. Diversidad y distribución vertical de epífitas en los alrededores del campamento Río Eslabón y de la laguna Chalalán, Parque Nacional Madidi, Dpto. La Paz, Bolivia. Rev. Soc. Boliv. Botánica 2001, 3, 104–123. [Google Scholar]
- Burnham, R.J.; Romero-Saltos, H.G. Diversity and distribution of lianas in Yasuní, Ecuador. In Ecology of Lianas; Schnitzer, S.A., Bongers, F., Burnham, R.J., Putz, F.E., Eds.; Wiley: West Sussex, UK, 2015; pp. 50–64. [Google Scholar]
- Karr, J.R.; Robinson, S.K.; Blake, J.G.; Bierregaard, R.O.J. Birds of four neotropical forests. In Four Neotropical Rainforests; Gentry, A.H., Ed.; Yale University Press: New Haven, CT, USA, 1990. [Google Scholar]
- Sullivan, B.L.; Wood, C.L.; Ilif, M.J.; Bonney, R.E.; Fink, D.; Kelling, S. eBird: A citizen-based bird observation network in the biological sciences. Biol. Conserv. 2009, 142, 2288–2292. [Google Scholar] [CrossRef]
- Janson, C.H.; Emmons, L.H. Ecological structure of the nonflying mammal community at Cocha Cashu Biological Station, Manu National Park, Peru. In Four Neotropical Rainforests; Gentry, A.H., Ed.; Yale University Press: New Haven, CT, USA, 1990; pp. 314–338. [Google Scholar]
- Peres, C.A. Primate community structure at twenty western Amazonian flooded and unflooded forests. J. Trop. Ecol. 1997, 13, 381–405. [Google Scholar] [CrossRef]
- Pitman, R.L. Checklist de los Mamíferos no Voladores de Cocha Cashu y Pakitza. 2008. Available online: https://cochacashu.sandiegozooglobal.org/species-lists/ (accessed on 5 September 2019).
- Hoorn, C.; Wesselingh, F.P.; ter Steege, H.; Bermudez, M.A.; Mora, A.; Sevink, J.; Sanmartin, I.; Sanchez-Meseguer, A.; Anderson, C.L.; Figueiredo, J.P.; et al. Amazonia through time: Andean uplift, climate change, landscape evolution, and biodiversity. Science 2010, 330, 927–931. [Google Scholar] [CrossRef] [PubMed]
- Rull, V. Neotropical biodiversity: Timing and potential drivers. Trends Ecol. Evol. 2011, 26, 508–513. [Google Scholar] [CrossRef] [PubMed]
- De Souza, J.R.D.; Venancio, N.M.; de freitas, M.A.; de Souza, M.B.; Verissimo, D. First record of Bothrops taeniatus Wagler, 1824 (Reptilia, Viperidae) for the state of Acre, Brazil. Check List 2013, 9, 430–431. [Google Scholar] [CrossRef][Green Version]
- Avila-Pires, T.C.S.; Vitt, L.J.; Sartorius, S.S.; Zani, P.A. Squamata (Reptilia) from four sites in southern Amazonia, with a biogeographic analysis of Amazonian lizards. Bol. Mus. Para. Emílio Goeldi. Cienc. Nat. Belém 2009, 4, 99–118. [Google Scholar]
- Da Silva, N. The snakes from Samuel hydroelectric power plant and vicinity, Rondonia, Brazil. Herpetol. Nat. Hist. 1993, 1, 37–86. [Google Scholar]
- Hamdan, B.; Pereira, A.G.; Loss-Oliveira, L.; Rodder, D.; Schrago, C.G. Evolutionary analysis of Chironius snakes unveils cryptic diversity and provides clues to diversification in the Neotropics. Mol. Phylogenet. Evol. 2017, 116, 108–119. [Google Scholar] [CrossRef]
- Nunes, P.M.S.; Fouquet, A.; Curcio, F.F.; Kok, P.J.R.; Rodrigues, M.T. Cryptic species in Iphisa elegans Gray, 1851 (Squamata: Gymnophthalmidae) revealed by hemipenial morphology and molecular data. Zool. J. Linn. Soc. 2012, 166, 361–376. [Google Scholar] [CrossRef]
- Prudente, A.L.C.; Passos, P. New Cryptic Species of Atractus (Serpentes: Dipsadidae) from Brazilian Amazonia. Copeia 2010, 010, 397–404. [Google Scholar] [CrossRef]
- Sturaro, M.J.; Avila-Pires, T.C.S.; Rodrigues, M.T. Molecular phylogenetic diversity in the widespread lizard Cercosaura ocellata (Reptilia: Gymnophthalmidae) in South America. Syst. Biodivers. 2017, 15, 532–540. [Google Scholar] [CrossRef]
- Torres-Carvajal, O.; Echevarria, L.Y.; Lobos, S.E.; Venegas, P.J.; Kok, P.J.R. Phylogeny, diversity and biogeography of Neotropical sipo snakes (Serpentes: Colubrinae: Chironius). Mol. Phylogenet. Evol. 2019, 130, 315–329. [Google Scholar] [CrossRef]
- Meneses-Pelayo, E.; Passos, P. New Polychromatic Species of Atractus (Serpentes: Dipsadidae) from the Eastern Portion of the Colombian Andes. Copeia 2019, 107, 250–261. [Google Scholar] [CrossRef]
- Passos, P.; Scanferla, A.; Melo-Sampaio, P.R.; Brito, J.; Almendariz, A. A giant on the ground: Another large-bodied Atractus (Serpentes: Dipsadinae) from Ecuadorian Andes, with comments on the dietary specializations of the goo-eaters snakes. An. Acad. Bras. Cienc. 2019, 91. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.; Oliveira, M.E. Natural history of snakes in forests of the Manaus region, central Amazonia, Brazil. Herpetol. Nat. Hist. 1999, 6, 78–150. [Google Scholar]
| Locality | Scale | Latitude | Precipitation | Snakes | Lizards | Reference |
|---|---|---|---|---|---|---|
| Manu Learning Center (MLC) 1 | local | −12.8 | 3000 | 40 | 21 | [16] |
| Los Amigos: LABS | local | −12.6 | 2600 | 60 | 27 | This paper |
| Los Amigos: LABS-R | regional | −12.6 | 2600 | 62 | 28 | This paper |
| Los Amigos: LABS-X | regional | −12.6 | 2600 | 68 | 36 | This paper |
| Cusco Amazonico (CA) | local | −12.5 | 2400 | 52 | 28 | [15,32] |
| Tambopata (EI) | local | −12.8 | 2500 | 50 | 26 | [32] |
| Cocha Cashu | local | −11.9 | 2500 | 32 | 25 | [16] |
| Panguana | local | −9.6 | 2500 | 44 | 27 | [33] |
| Yasuni 2 | regional | −0.6 | 3100 | 78 | 33 | [7] |
| Tiputini | local | −0.6 | 3100 | 63 | 31 | [7] |
| Iquitos 3 | regional | −3.7 | 2900 | 89 | 39 | [11] |
| Taxon | Richness (North) | Richness (South) | Diversity Inflation (%) | Scale | Reference |
|---|---|---|---|---|---|
| Trees | 239 | 174 | 40 | local | [9] |
| Trees | 1017 | 693 | 50 | regional 1 | [9] |
| Trees | 1356 | 1004 | 40 | regional 2 | [9] |
| Vascular epiphytes 3 | 313 | 152 | 110 | regional | [35,36] |
| Lianas | 203 | 115 | 80 | local | [37]; Burnham, pers comm |
| Birds | 550–600 | 550–600 | 0 | regional | [7,38,39] |
| Snakes | 90 | 68 | 30 | regional | This study |
| Lizards | 39 | 36 | 10 | regional | This study |
| Frogs | 141 | 100 | 40 | regional | [7,32] |
| Primates | 12–14 | 12–14 | 0 | regional | [7,40,41,42] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rabosky, D.L.; von May, R.; Grundler, M.C.; Davis Rabosky, A.R. The Western Amazonian Richness Gradient for Squamate Reptiles: Are There Really Fewer Snakes and Lizards in Southwestern Amazonian Lowlands? Diversity 2019, 11, 199. https://doi.org/10.3390/d11100199
Rabosky DL, von May R, Grundler MC, Davis Rabosky AR. The Western Amazonian Richness Gradient for Squamate Reptiles: Are There Really Fewer Snakes and Lizards in Southwestern Amazonian Lowlands? Diversity. 2019; 11(10):199. https://doi.org/10.3390/d11100199
Chicago/Turabian StyleRabosky, Daniel L., Rudolf von May, Michael C. Grundler, and Alison R. Davis Rabosky. 2019. "The Western Amazonian Richness Gradient for Squamate Reptiles: Are There Really Fewer Snakes and Lizards in Southwestern Amazonian Lowlands?" Diversity 11, no. 10: 199. https://doi.org/10.3390/d11100199
APA StyleRabosky, D. L., von May, R., Grundler, M. C., & Davis Rabosky, A. R. (2019). The Western Amazonian Richness Gradient for Squamate Reptiles: Are There Really Fewer Snakes and Lizards in Southwestern Amazonian Lowlands? Diversity, 11(10), 199. https://doi.org/10.3390/d11100199






