Graminoid Invasion in an Insular Endemism Hotspot and Its Protected Areas
Abstract
1. Introduction
2. Materials and Methods
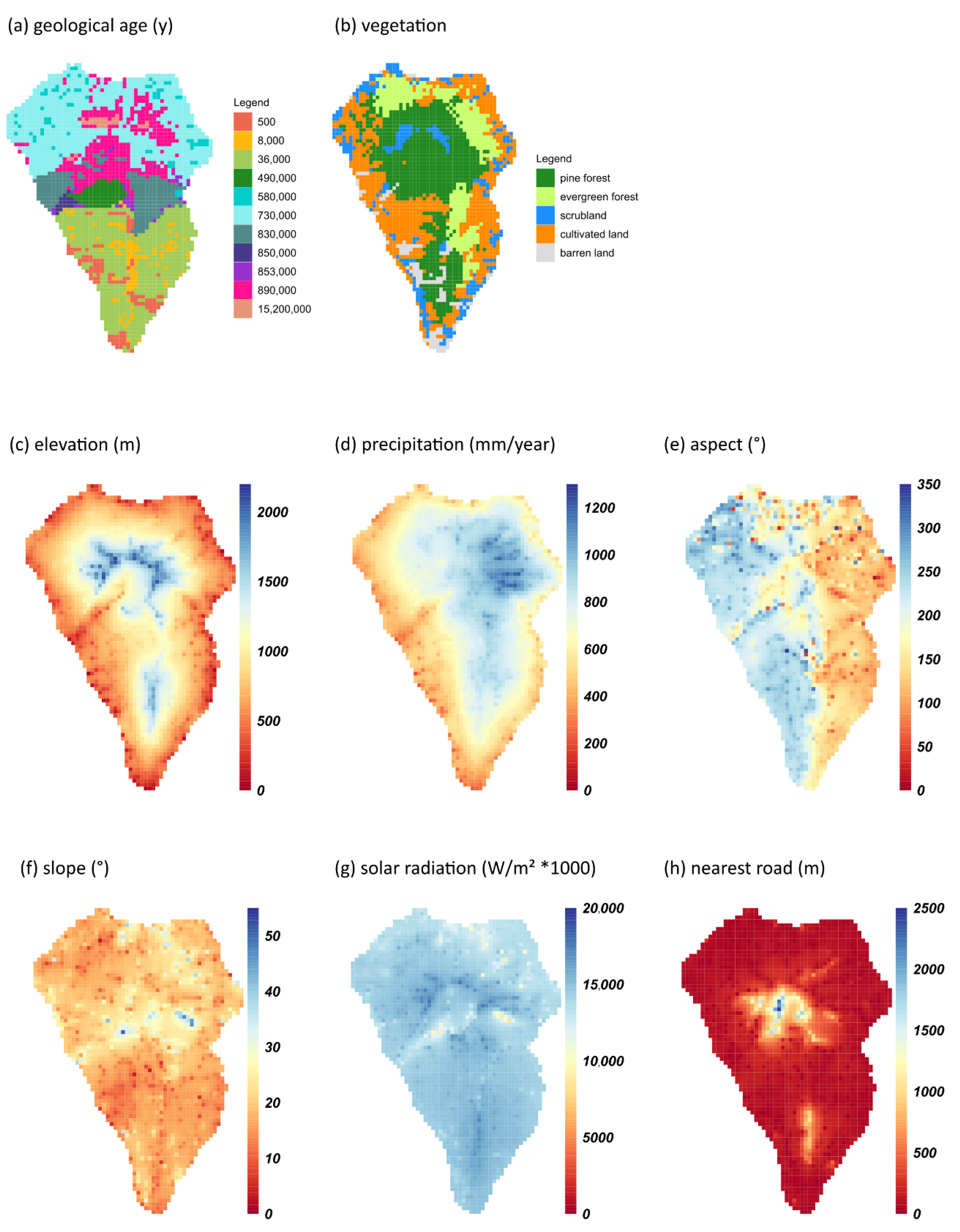
2.1. Compilation of Data Sets
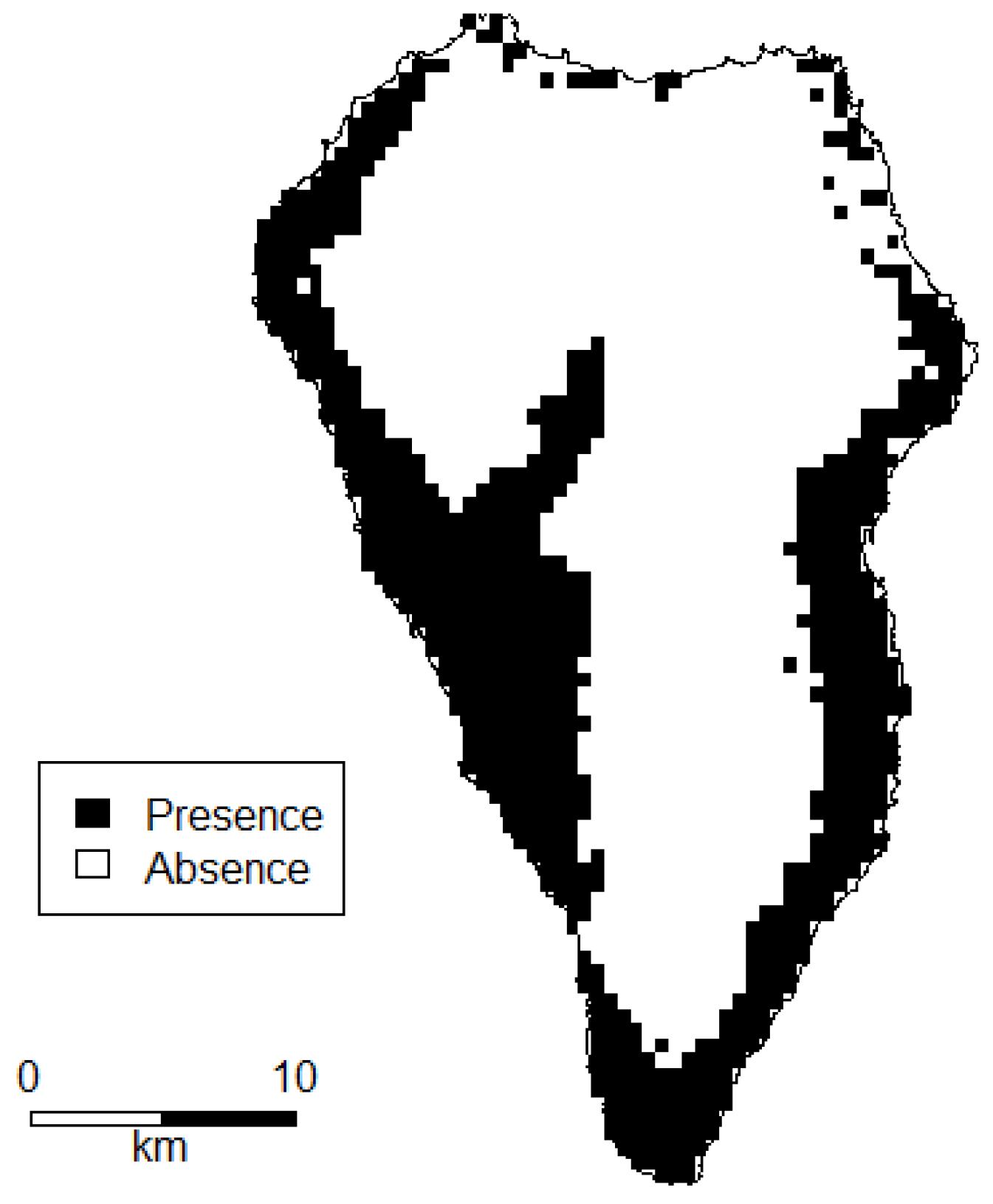
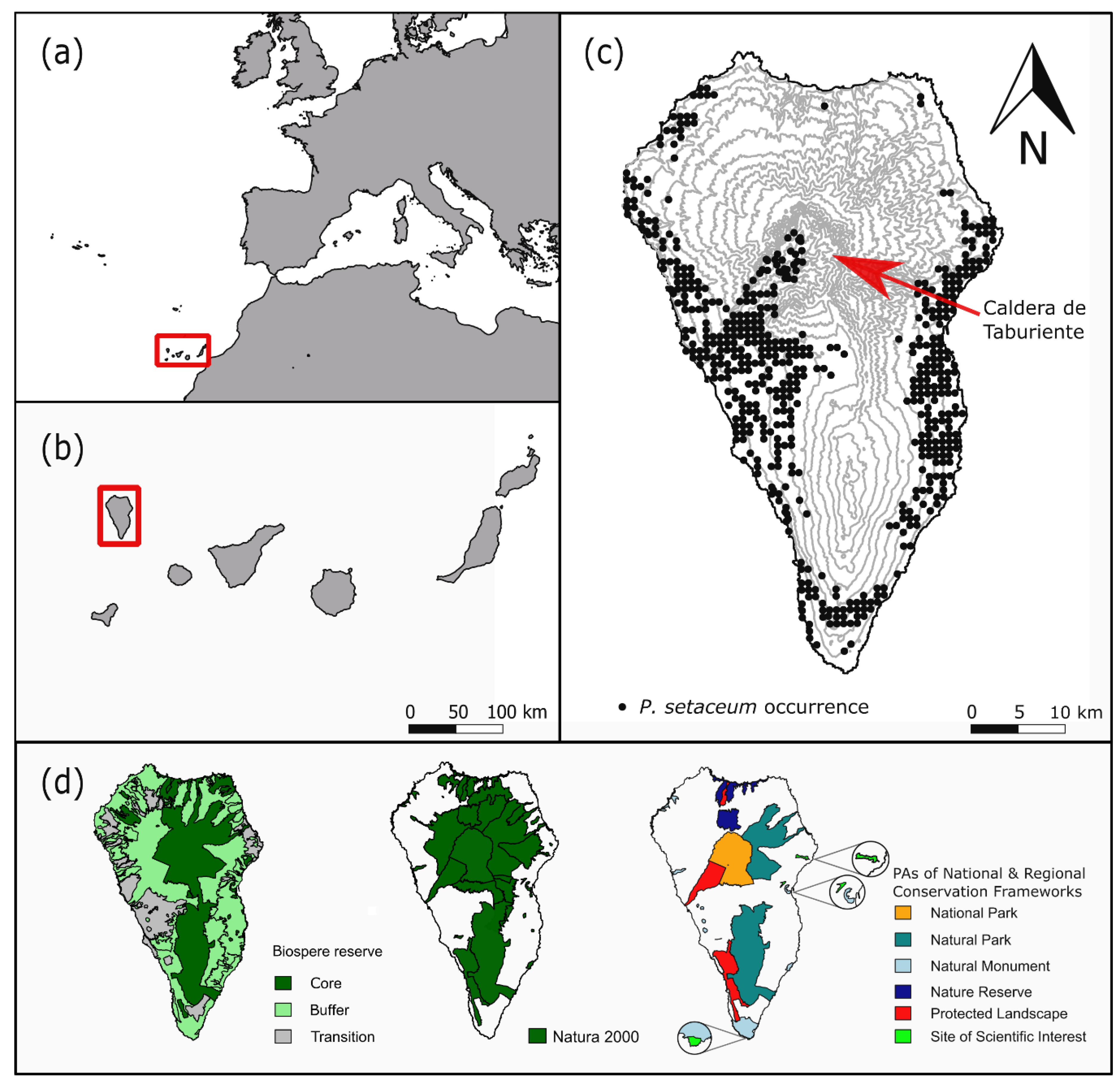
- Occurrence data from the National Park Directorate of La Palma for P. setaceum within the National Park “La Caldera de Taburiente” from 2016, n = 55.
- Fieldwork based occurrence data collected on a yearly basis from 2010 to 2018, n = 19.
- Entries in the GBIF (Global Biodiversity Information Facility) database [50] for P. setaceum (only geo-referenced data; basis of record: observation, human observation) from 2012 till 2017, n = 21.
- Occurrence data from the Atlantis database [51]. From this raster data, we created centroids within each pixel of P. setaceum occurrence with the highest precision level and a resolution of 500 × 500 m to extract occurrence points, n = 691.
2.2. Ensemble Modeling
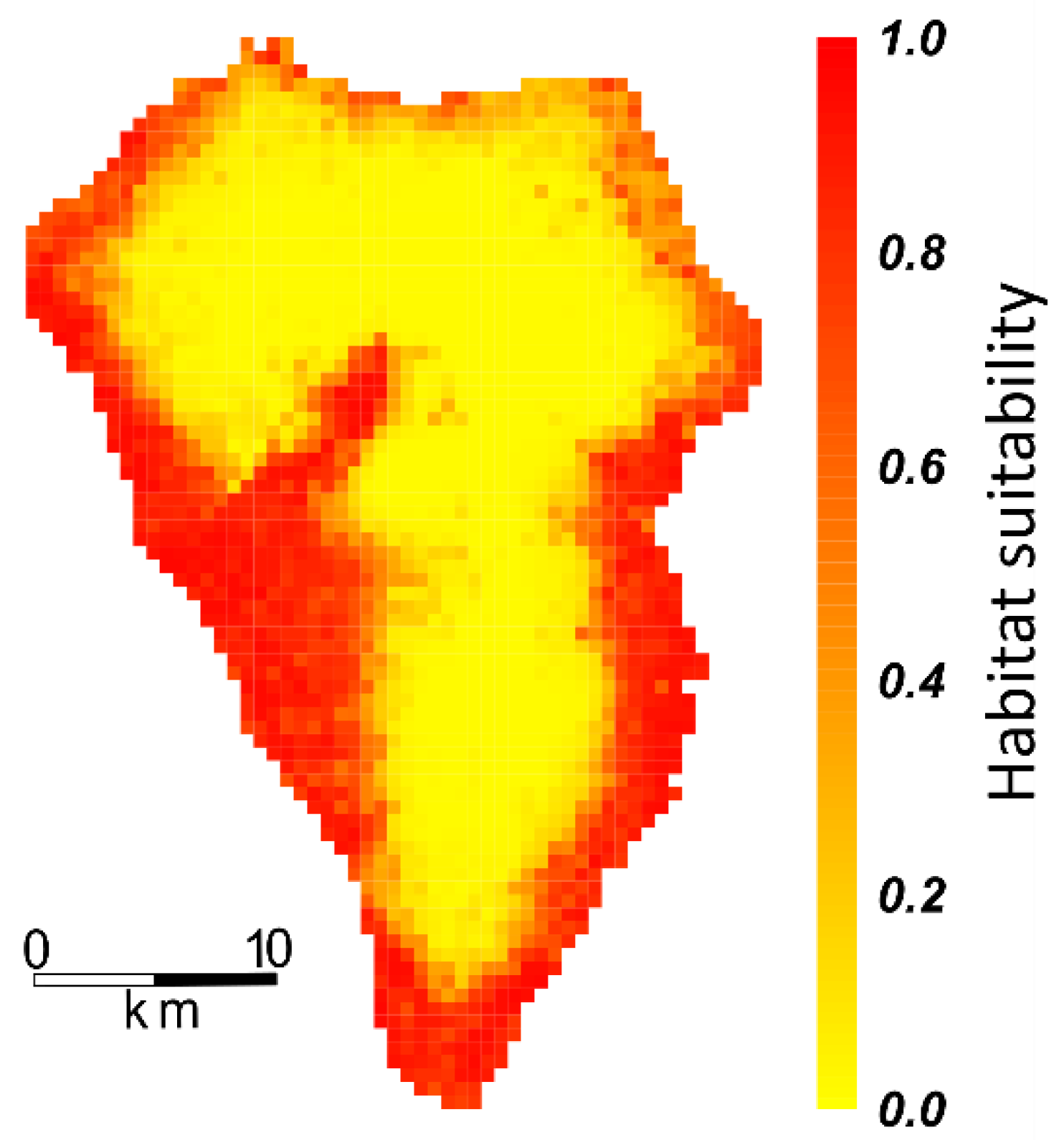
2.3. Projecting Habitat Suitability onto Ecosystems, PAs, and Endemic Plant Species Richness
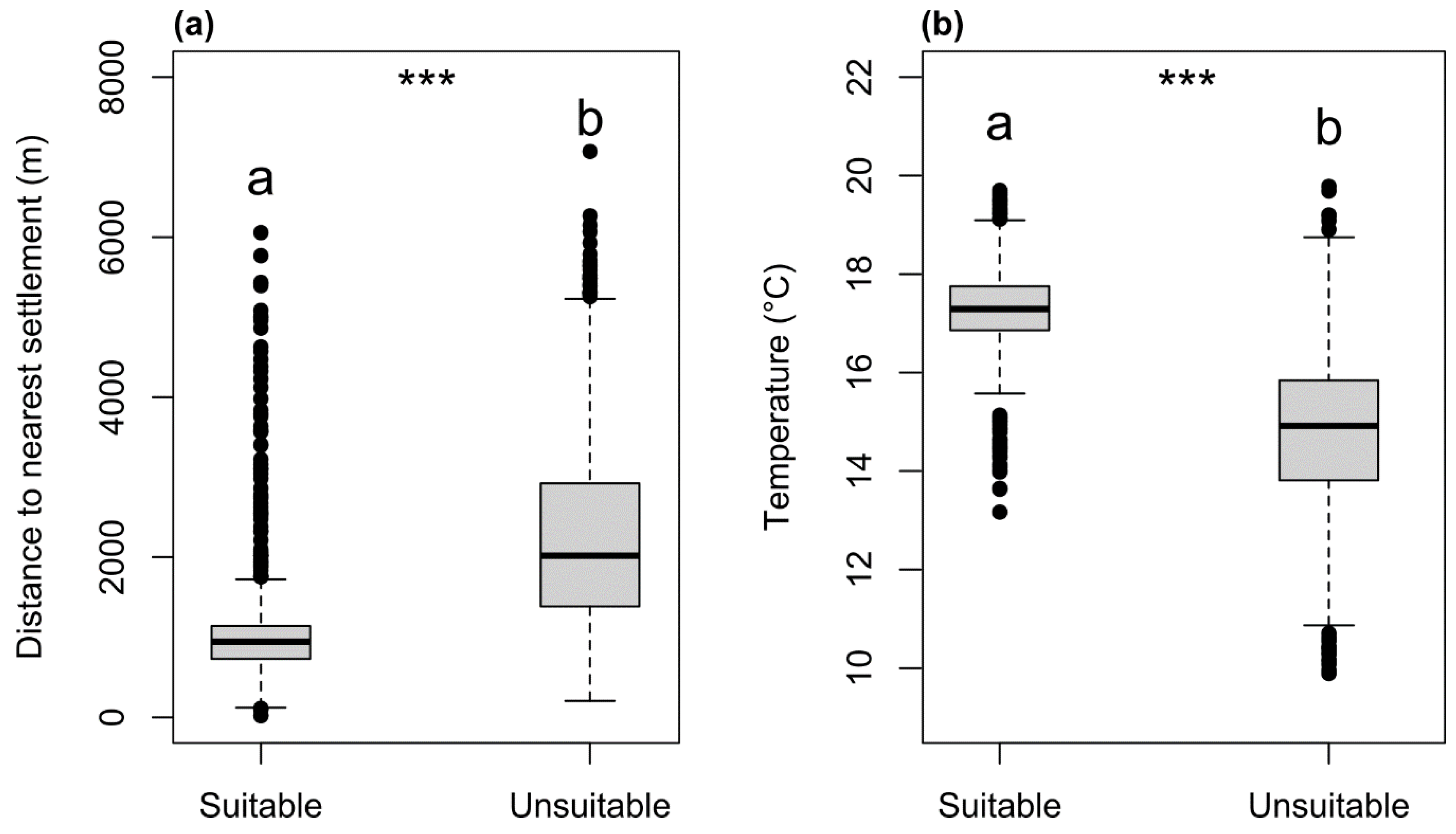
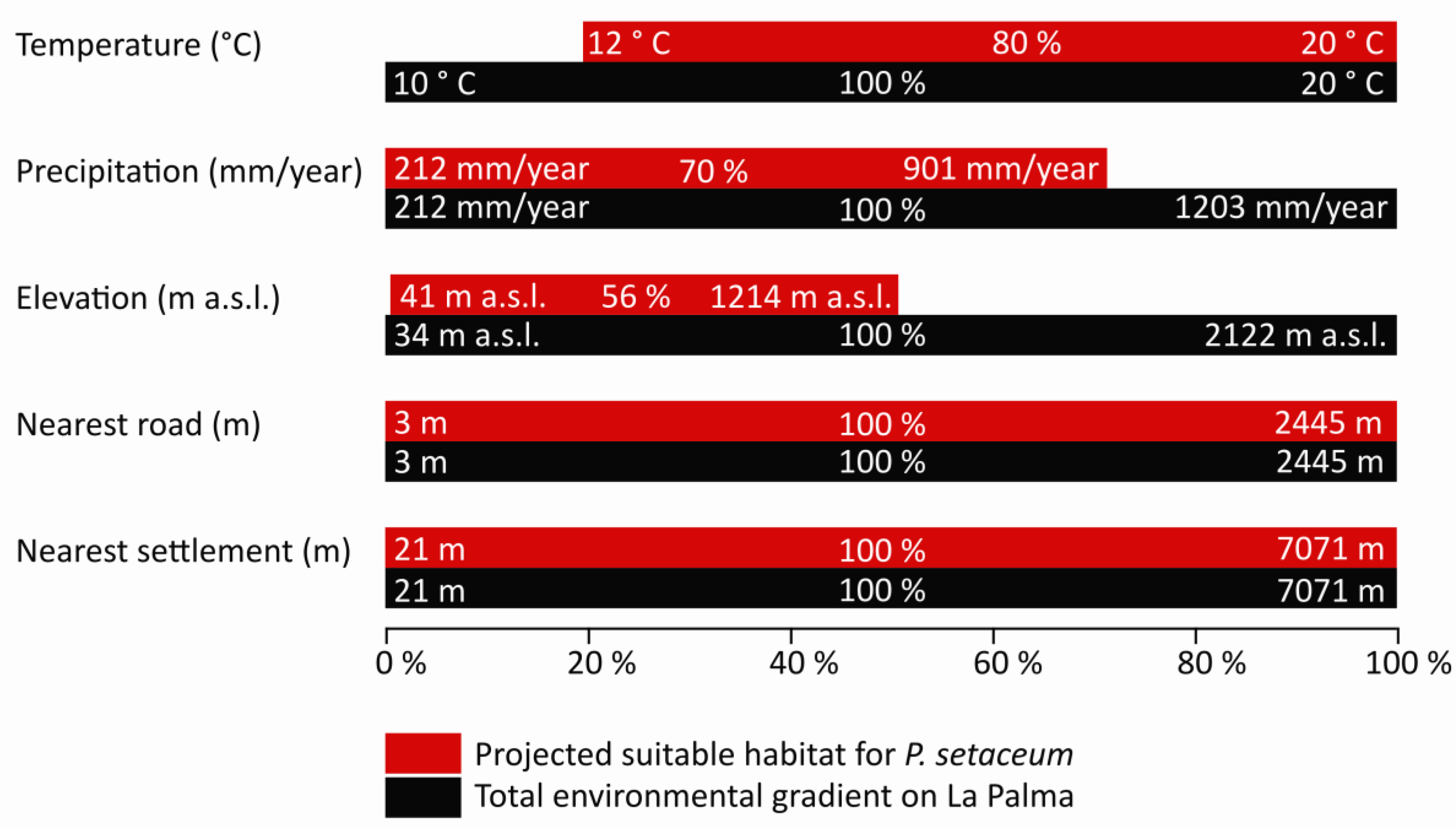
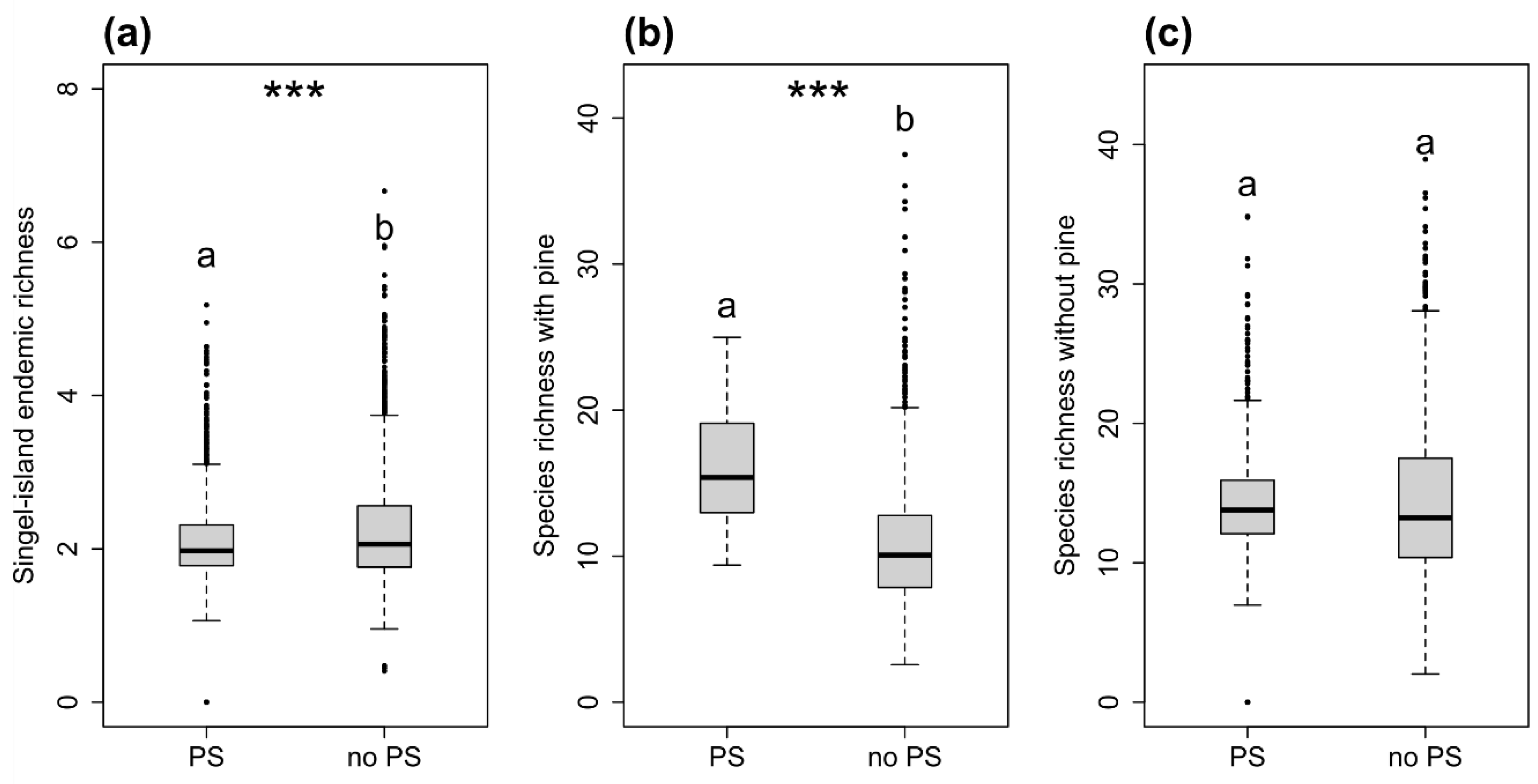
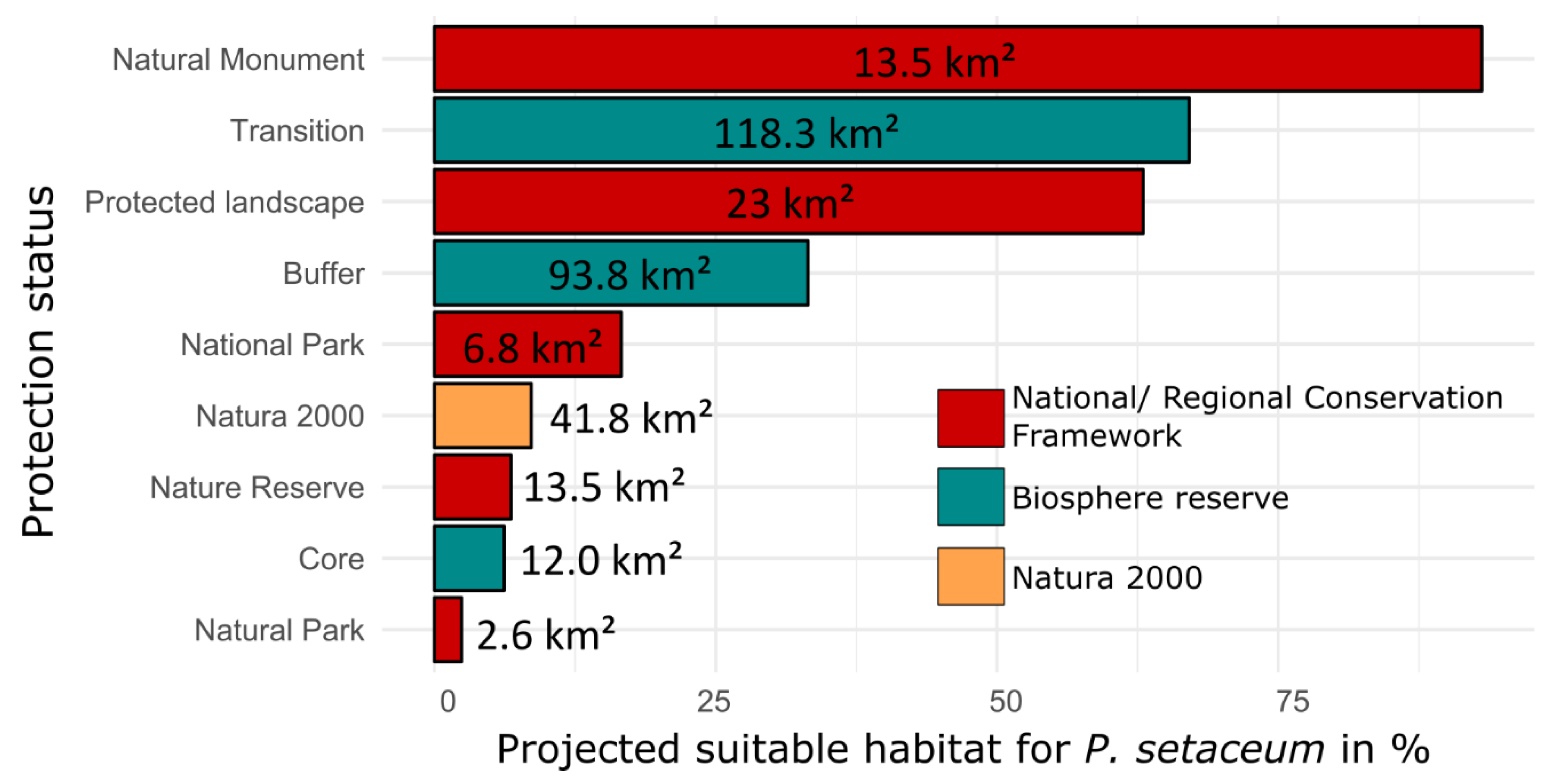
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Whittaker, R.J.; Fernández-Palacios, J.M. Island Biogeography: Ecology, Evolution and Conservation; Oxford University Press: Oxford, UK, 2007; pp. 295–300. [Google Scholar]
- Dawson, W.; Moser, D.; van Kleunen, M.; Kreft, H.; Pergl, J.; Pyšek, P.; Weigelt, P.; Winter, M.; Lenzner, P.; Blackburn, T.M.; et al. Global hotspots and correlates of alien species richness across taxonomic groups. Nat. Ecol. Evol. 2017, 1, 0186. [Google Scholar] [CrossRef]
- Kueffer, C.; Daehler, C.C.; Torres-Santana, C.W.; Lavergne, C.; Meyer, J.Y.; Otto, R.; Silva, L. A global comparison of invasive plant species on oceanic islands. Perspect. Plant Ecol. Syst. 2010, 12, 141–165. [Google Scholar]
- Moser, D.; Lenzner, B.; Weigelt, P.; Dawson, W.; Kreft, H.; Pergl, J.; Pyšek, P.; van Kleunen, M.; Winter, M.; Capinha, C.; et al. Remoteness promotes biological invasions on islands worldwide. PNAS 2018, 115, 9270–9275. [Google Scholar] [CrossRef]
- Whittaker, R.J.; Triantis, K.A.; Ladle, R.J. A general dynamic theory of oceanic island biogeography. J. Biogeogr. 2008, 35, 977–994. [Google Scholar] [CrossRef]
- Kier, G.; Kreft, H.; Lee, T.M.; Jetz, W.; Ibisch, P.L.; Nowicki, C.; Mutke, J.; Barthlott, W. A global assessment of endemism and species richness across island and mainland regions. PNAS 2009, 106, 9322–9327. [Google Scholar] [CrossRef]
- Convention on Biological Diversity (CBD). Global Biodiversity Outlook 3; Secretariat of the Convention on Biological Diversity: Montréal, QC, Canada, 2010. [Google Scholar]
- Tershy, B.R.; Shen, K.W.; Newton, K.M.; Holmes, N.D.; Croll, D.A. The importance of islands for the protection of biological and linguistic diversity. BioScience 2015, 65, 592–597. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; Da Fonseca, G.A.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef]
- Gray, A. The ecology of plant extinction: Rates, traits and island comparisons. Oryx 2018, 53, 424–428. [Google Scholar] [CrossRef]
- Whittaker, R.J.; Fernández-Palacios, J.M.; Matthews, T.J.; Borregaard, M.K.; Triantis, K.A. Island biogeography: Taking the long view of nature’s laboratories. Science 2017, 357, eaam8326. [Google Scholar] [CrossRef]
- Harter, D.E.V.; Irl, S.D.H.; Seo, B.; Steinbauer, M.J.; Gillespie, R.; Triantis, K.A.; Fernández-Palacios, J.M.; Beierkuhnlein, C. Impacts of global climate change on the floras of oceanic islands—Projections, implications and current knowledge. Perspect. Plant Ecol. Syst. 2015, 17, 160–183. [Google Scholar] [CrossRef]
- Millennium Ecosystem Assessment (Program): Ecosystems and Human Well-Being; Island Press: Washington, DC, USA, 2005.
- Caujapé-Castells, J.; Tye, A.; Crawford, D.; Kueffer, C. Conservation of oceanic island floras: Present and future global challenges. Perspect. Plant Ecol. Syst. 2010, 12, 107–129. [Google Scholar] [CrossRef]
- Irl, S.D.H.; Steinbauer, M.J.; Epperlein, L.; Harter, D.E.V.; Jentsch, A.; Pätz, S.; Wohlfart, C.; Beierkuhnlein, C. The Hitchhiker’s guide to island endemism: Biodiversity and endemic perennial plant species in roadside and surrounding vegetation. Biodivers. Conserv. 2014, 23, 2273–2287. [Google Scholar] [CrossRef]
- Irl, S.D.H.; Steinbauer, M.J.; Messinger, J.; Blume-Werry, G.; Palomares-Martínez, Á.; Beierkuhnlein, C.; Jentsch, A. Burned and devoured-introduced herbivores, fire, and the endemic flora of the high-elevation ecosystem on La Palma, Canary Islands. Arct. Antarct. Alp. Res. 2014, 46, 859–869. [Google Scholar] [CrossRef]
- Cubas, J.; Martín-Esquivel, J.L.; Nogales, M.; Irl, S.D.H.; Hernández-Hernández, R.; López-Darias, M.; Marrero-Gómez, M.; del Arco, M.J.; González-Mancebo, J.M. Contrasting effects of invasive rabbits on endemic plants driving vegetation change in a subtropical alpine insular environment. Biol. Invasions 2018, 20, 793–807. [Google Scholar] [CrossRef]
- Cubas, J.; Irl, S.D.H.; Villafuerte, R.; Bello-Rodríguez, V.; Rodríguez-Luengo, J.L.; del Arco, M.; Martín-Esquivel, J.L.; González-Mancebo, J.M. Endemic plant species are more palatable to introduced herbivores than non-endemics. Proc. R. Soc. B. 2019, 286, 20190136. [Google Scholar] [CrossRef]
- Guillera-Arroita, G.; Hauser, C.E.; McCarthy, M.A. Optimal surveillance strategy for invasive species management when surveys stop after detection. Ecol. Evol. 2014, 4, 1751–1760. [Google Scholar] [CrossRef]
- Araújo, M.B.; Guisan, A. Five (or so) challenges for species distribution modelling. J. Biogeogr. 2006, 33, 1677–1688. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J.R.; Emery, W.P.H.; Brown, W.V. Species distribution models: Ecological explanation and prediction across space and time. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 677–697. [Google Scholar] [CrossRef]
- Jiménez-Valverde, A.; Peterson, A.T.; Soberón, J.; Overton, L.M.; Aragón, P.; Lobo, J.M. Use of niche models in invasive species risk assessments. Biol. Invasions 2011, 13, 2785–2797. [Google Scholar] [CrossRef]
- Bosso, L.; De Conno, C.; Russo, D. Modelling the risk posed by the zebra mussel Dreissena polymorpha: Italy as a case study. Environ. Manag. 2017, 60, 304–313. [Google Scholar] [CrossRef]
- Tingley, R.; García-Díaz, P.; Rocha Arantes, C.R.; Cassey, P. Integrating transport pressure data and species distribution models to estimate invasion risk for alien stowaways. Ecography 2017, 41, 635–646. [Google Scholar] [CrossRef]
- Early, R.; Sax, D.F. Climatic niche shifts between species’ native and naturalized ranges raise concern for ecological forecasts during invasions and climate change. Glob. Ecol. Biogeogr. 2014, 23, 1356–1365. [Google Scholar] [CrossRef]
- Atwater, D.Z.; Ervine, C.; Barney, J.N. Climatic niche shifts are common in introduced plants. Nat. Ecol. Evol. 2017, 2, 34–43. [Google Scholar] [CrossRef]
- Briscoe Runquist, R.D.; Lake, T.; Tiffin, P.; Moeller, D.A. Species distribution models throughout the invasion history of palmer amaranth predict regions at risk of future invasion and reveal challenges with modeling rapidly shifting geographic ranges. Sci. Rep. 2019, 9, 2426. [Google Scholar] [CrossRef]
- West, A.M.; Kumar, S.; Brown, C.S.; Stohlgren, T.J.; Bromberg, J. Field validation of an invasive species MaxEnt model. Ecol. Inform. 2016, 36, 126–134. [Google Scholar] [CrossRef]
- Combat Invasive Alien Species—Target 5. Available online: http://ec.europa.eu/environment/nature/biodiversity/strategy/target5/index_en.htm (accessed on 18 October 2017).
- Datasheet Pennisetum setaceum (Fountain Grass). Available online: https://www.cabi.org/isc/datasheet/116202 (accessed on 2 October 2017).
- García Gallo, A.; Wildpret de la Torre, W.; CarquéÁlamo, E.; Jiménez Felipe, M.T. Ornamental flora introduced and naturalized in Tenerife. In Proceedings of the 36th IAVS Symposium, Santa Cruz de Tenerife, Canary Islands, Spain, 12 April 1993; 1997; p. 75. [Google Scholar]
- Fundación Canaria Reserva Mundial de la Biosfera La Palma (FRMB La Palma): Plan de Manejo de Pennisetum setaceum en la isla de La Palma. Fundación Canaria Reserva de Mundial de la Biosfera La Palma y Cabildo Insular de La Palma: Canary Islands, Spain, 2016; 57, unpublished report.
- Irl, S.D.H.; Harter, D.E.V.; Steinbauer, M.J.; Puyol, D.G.; Fernández-Palacios, J.M.; Jentsch, A.; Beierkuhnlein, C. Climate vs. topography—Spatial patterns of plant species diversity and endemism on a high-elevation island. J. Ecol. 2015, 103, 1621–1633. [Google Scholar] [CrossRef]
- Garzón-Machado, V.; Otto, R.; del Arco Aguilar, M.J. Bioclimatic and vegetation mapping of a topographically complex oceanic island applying different interpolation techniques. Int. J. Biometeorol. 2014, 58, 887–899. [Google Scholar] [CrossRef]
- del Arco Aguilar, M.J.; González-González, R.; Garzón-Machado, V.; Pizarro-Hernández, B. Actual and potential natural vegetation on the Canary Islands and its conservation status. Biodivers. Conserv. 2010, 11, 3089–3140. [Google Scholar] [CrossRef]
- Muer, R.; Sauerbier, H.; Cabrera Calixto, F. Die Farn-und Blütenpflanzen der Kanarischen Inseln; Margraf Publishers: Weikersheim, Germany, 2016; p. 20. [Google Scholar]
- Fernández-Palacios, J.M.; Arévalo, J.R.; Balguerías, E.; Barone, R.; de Nascimento, L.; Delgado, J.D.; Bento Elias, R.; Fernández-Lugo, S.; Méndez, J.; Menezes de Sequeira, M.; et al. La Laurisilva. Canarias, Madeira y Azores; Macaronesia Editorial: Santa Cruz de Tenerife, Spain, 2017. [Google Scholar]
- Parson, J. Human influences on the pine and laurel forests of the Canary Islands. Geogr. Rev. 1981, 71, 253–271. [Google Scholar] [CrossRef]
- Santos, A. Flora vascular nativa. In Naturaleza de las Islas Canarias. Ecología y Conservación; Fernández-Palacios, J.M., Martín Esquivel, J.L., Eds.; Publicaciones Turquesa SL: Santa Cruz de Tenerife, Spain, 2001; pp. 185–192. [Google Scholar]
- Evolución del número de turistas que visitaron la isla de La Palma (Canarias) de 2013 a 2018. Available online: https://es.statista.com/estadisticas/524031/numero-de-turistas-que-visitaron-la-isla-de-la-palma-canarias/ (accessed on 8 September 2019).
- Andersen, K.M.; Naylor, B.J.; Endress, B.A.; Parks, C.G. Contrasting distribution patterns of invasive and naturalized non-native species along environmental gradients in a semi-arid montane ecosystem. Appl. Veg. Sci. 2015, 18, 683–693. [Google Scholar] [CrossRef]
- Corona, P.; Badalamenti, E.; Pasta, S.; La Mantia, T. Carbon storage of Mediterranean grasslands. Anales del Jardín Botánico de Madrid 2016, 73, e029. [Google Scholar] [CrossRef]
- Santos, A. Vegetación y Flora de La Palma; Editorial Interinsular Canaria S. A.: Canary Islands, Spain, 1983; p. 348. [Google Scholar]
- Pérez de Paz, P.L.; García Gallo, A.; Heene, A. Control y Erradicación del “Rabo-gato” [Pennisetum setaceum] en la isla de La Palma; Cabildo Insular de La Palma: Canary Islands, Spain, 1999; p. 124. [Google Scholar]
- Emery, W.P.H.; Brown, W.V. Apomixis in the Gramineae: Tribe Andropogoneae: Heteropogon contortus. Madroño 1958, 14, 238–246. [Google Scholar]
- Simpson, C.E.; Bashaw, E.C. Cytology and reproductive characteristics in Pennisetum setaceum. Am. J. Bot. 1969, 56, 31–36. [Google Scholar] [CrossRef]
- Adkins, E.; Cordell, S.; Drake, D.R. Role of fire in the germination ecology of fountain grass (Pennisetum setaceum), an invasive African bunchgrass in Hawai’i. Pac. Sci. 2011, 65, 17–25. [Google Scholar] [CrossRef]
- Poulin, J.; Weller, S.G.; Sakai, A.K. Genetic diversity does not affect the invasiveness of fountain grass (Pennisetum setaceum) in Arizona, California and Hawaii. Divers. Distrib. 2005, 11, 241–247. [Google Scholar] [CrossRef]
- Fountain Grass Control in Hawaii Volcanoes National Park: Management Considerations and Strategies. Available online: https://pdfs.semanticscholar.org/3a9a/2367472594af878c5447e442040a7c998b5c.pdf (accessed on 3 October 2017).
- GBIF Secretariat. Pennisetum Setaceum (Forssk.) Chiov. Available online: https://www.gbif.org/species/2706134 (accessed on 26 September 2017).
- Atlantis 3.3.0. Banco de Datos de Biodiversidad de Canarias. Available online: http://www.biodiversidadcanarias.es/atlantis/ (accessed on 22 October 2018).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: URL https://www.R-project.org/ (accessed on 13 September 2019).
- Aiello-Lammens, M.E.; Boria, R.A.; Radosavljevic, A.; Vilela, B.; Anderson, R.P. spThin: Functions for Spatial Thinning of Species Occurrence Records for Use in Ecological Models. R packageversion 0.1.0. 2014. Available online: https://CRAN.R-project.org/package=spThin (accessed on 13 September 2019).
- Dormann, C.F.; Elith, J.; Bacher, S.; Buchmann, C.; Carl, G.; Carré, G.; García Marquéz, J.R.; Gruber, B.; Lafourcade, B.; Leitão, P.J.; et al. Collinearity: A review of methods to deal with it and a simulation study evaluating their performance. Ecography 2013, 36, 27–46. [Google Scholar] [CrossRef]
- Thuiller, W.; Georges, D.; Engler, R.; Breiner, F. Biomod2: Ensemble Platform for Species Distribution Modeling. R Packageversion 3.3-7. 2016. Available online: https://CRAN.R-project.org/package=biomod2 (accessed on 13 September 2019).
- Seni, G.; Elder, J.F. Ensemble Methods in Data Mining: Improving Accuracy Through Combining Predictions; Morgan & Claypool Publishers: San Rafael, CA, USA, 2010; pp. 2–3. [Google Scholar]
- Araújo, M.B.; Pearson, R.G.; Thuiller, W.; Erhard, M. Validation of species-climate impact models under climate change. Glob. Chang. Biol. 2005, 11, 1504–1513. [Google Scholar] [CrossRef]
- Liu, C.; Berry, P.M.; Dawson, T.P.; Pearson, G. Selecting thresholds of occurrence in the prediction of species distributions. Ecography 2005, 28, 385–393. [Google Scholar] [CrossRef]
- Irl, S.D.H.; Schweiger, A.H.; Medina, F.M.; Fernández-Palacios, J.M.; Harter, D.E.V.; Jentsch, A.; Provenzale, A.; Steinbauer, M.J.; Beierkuhnlein, C. An island view of endemic rarity—Environmental drivers and consequences for nature conservation. Divers. Distrib. 2017, 23, 1132–1142. [Google Scholar] [CrossRef]
- Vera-Galván, M.A.; Samarín-Bello, C.; Delgado-Castro, G.; Viera-Ruíz, G. Natura 2000 en Macaronesia: Azores, Madeira; Gobierno de Canarias: Canary Islands, Spain, 2010; p. 576. [Google Scholar]
- Martín-Esquivel, J.L.; García-Court, H.; Redondo-Rojas, C.E.; García-Fernández, I.; Carralero-Jaime, I. La Red Canaria de Espacios Naturales Protegidos; Gobierno de Canarias: Canary Islands, Spain, 1995; p. 452. [Google Scholar]
- Rahlao, S.J.; Milton, S.J.; Esler, K.L.; Barnard, P. The distribution of invasive Pennisetum setaceum along roadsides in western South Africa: The role of corridor interchanges. Restor. Ecol. 2010, 20, 475–482. [Google Scholar] [CrossRef]
- Rahlao, S.J.; Milton, S.J.; Esler, K.L.; Barnard, P. Performance of invasive alien fountain grass (Pennisetum setaceum) along a climatic gradient through three South African biomes. S. Afr. J. Bot. 2014, 91, 43–48. [Google Scholar] [CrossRef]
- Milton, S.J.; Hoffmann, J.H.; Bowie, R.C.K.; D’Amico, J.; Griffiths, M.; Joubert, D.; Loewenthal, D.; Moinde, N.N.; Seymour, C.; Toral-Granda, M.V.; et al. Invasive fountain grass on the Cape Peninsula. S. Afr. J. Sci. 1998, 94, 57–58. [Google Scholar]
- García-Gallo, A.; Wildpret de la Torre, W.; Rodríguez Delgado, O.; Pérez de Paz, P.L.; León Arencibia, M.C.; Suárez Rodriguez, C.; Reyes-Betancort, J.A. El xenófito Pennisetum setaceum en las islas Canarias (Magnoliophyta, Poaceae). Vieraea 1999, 27, 133–158. [Google Scholar]
- Tackenberg, O.; Poschlod, P.; Bonn, S. Assessment of wind dispersal potential in plant species. Ecol. Monogr. 2003, 73, 191–205. [Google Scholar] [CrossRef]
- Barros, A.; Pickering, C.M. Non-native plant invasion in relation to tourism use of Aconcagua Park, Argentina, the highest protected area in the Southern Hemisphere. Mt. Res. Dev. 2014, 34, 13–27. [Google Scholar] [CrossRef]
- Simberloff, D. Invasive Species. In Conservation Biology for All; Sodhi, N.S., Ehrlich, P.R., Eds.; Oxford University Press: Oxford, UK, 2010; pp. 131–152. [Google Scholar]
- Steinbauer, M.J.; Irl, S.D.H.; González-Mancebo, J.M.; Breiner, F.T.; Hernández-Hernández, R.; Hopfenmüller, S.; Kidane, Y.; Jentsch, A.; Beierkuhnlein, C. Plant invasion and speciation along elevational gradients on the oceanic island La Palma, Canary Islands. Ecol. Evol. 2017, 7, 771–779. [Google Scholar] [CrossRef] [PubMed]
- González-Rodríguez, A.M.; Baruch, Z.; Palomo, D.; Cruz-Trujillo, G.; Jiménez, M.S.; Morales, D. Ecophysiology of the invader Pennisetum setaceum and three native grasses in the Canary Islands. Acta Oecol. 2010, 36, 248–254. [Google Scholar] [CrossRef]
- Hoffmann, S.; Schmitt, T.; Chiarucci, A.; Irl, S.; Rocchini, D.; Vetaas, O.R.; Tanase, M.A.; Mermoz, S.; Bouvet, A.; Beierkuhnlein, C. Remote sensing of beta diversity: Evidence from plant communities in a semi-natural system. Appl. Veg. Sci. 2019, 22, 13–26. [Google Scholar] [CrossRef]
- Garzón-Machado, V.; González-Mancebo, J.M.; Palomares-Martínez, A.; Acevedo-Rodríguez, A.; Fernández-Palacios, J.M.; del-Arco-Aguilar, M.; Pérez-de-Paz, P.L. Strong negative effect of alien herbivores on endemic legumes of the Canary pine forest. Biol. Conserv. 2010, 143, 2685–2694. [Google Scholar] [CrossRef]
- Molina-Terrén, D.M.; Fry, D.L.; Grillo, F.F.; Cardil, A.; Stephens, S.L. Fire history and management of Pinus canariensis forests on the western Canary Islands Archipelago, Spain. For. Ecol. Manag. 2016, 382, 184–192. [Google Scholar] [CrossRef]
- Hartley, S.E.; Fitt, R.N.; McLarnon, E.L.; Wade, R.N. Defending the leaf surface: Intra-and inter-specific differences in silicon deposition in grasses in response to damage and silicon supply. Front. Plant Sci. 2015, 6, 35. [Google Scholar] [CrossRef] [PubMed]
- UNESCO Biosphere Reserves—Learning Sites for Sustainable Development. Available online: http://www.unesco.org/new/en/natural-sciences/environment/ecological-sciences/biosphere-reserves/ (accessed on 14 January 2018).
- Sutherland, W.J.; Pullin, A.S.; Dolman, P.M.; Knight, T.M. The need for evidence-based conservation. Trends Ecol. Evol. 2004, 19, 305–308. [Google Scholar] [CrossRef] [PubMed]

| Category | Variable | Calculation of Variables | Sources |
|---|---|---|---|
| Topography | Elevation | Rasterized at a resolution of 0.5 × 0.5 km deriving from a digital elevation model (DEM) of 2 × 2 m. | Original Data: Cabildo Insular de La Palma, [33] |
| Slope | Calculations were based on a DEM of 2 × 2 m. Resolution was rescaled to 0.5 × 0.5 km. | Original Data: Cabildo Insular de La Palma, Calculations [33] | |
| Aspect (northness, eastness) | Calculations were based on a DEM of 2 × 2 m. Resolution was rescaled to 0.5 × 0.5 km. Northness was calculated as cos(aspect) and eastness as sin(aspect). | Original Data: Cabildo Insular de La Palma, Calculations [33] | |
| Parent material | Geological age | Resolution was rescaled to 0.5 × 0.5 km. | Cabildo Insular de La Palma, [33] |
| Climate data | Mean annual precipitation | Interpolation (Linear regression kriging), using data collected from meteorological stations for the Canary Islands (n = 214). Time span from 1969 to 1998. Resolution was rescaled to 0.5 × 0.5 km. | Original Data: Cabildo Insular de La Palma, Calculations [33] |
| Solar radiation | Calculation from DEM 100 × 100m using a standard diffuse atmosphere and based on latitude, elevation, slope, and aspect. Resolution was rescaled to 0.5 × 0.5 km. | Original Data: Cabildo Insular de La Palma, Calculations [33] | |
| Infrastructure | Nearest road | Resolution was rescaled to 0.5 × 0.5 km. | [33] |
| Land cover | Vegetation types (coniferous forest, broadleaved evergreen forest, scrubland, cultivated land, barren land) | Rasterized at a resolution of 0.5 × 0.5 km | Original Data: Cabildo Insular de La Palma [33] |
| Variable | GLM | MaxEnt | RF | GBM |
|---|---|---|---|---|
| 1. Elevation | 0.963 | 0.573 | 0.480 | 0.676 |
| 2. Precipitation | 0.043 | 0.245 | 0.155 | 0.162 |
| 3. Nearest road | 0.180 | 0.225 | 0.073 | 0.071 |
| 4. Solar radiation | 0.153 | 0.104 | 0.080 | 0.061 |
| 5. Slope | 0.012 | 0.137 | 0.034 | 0.017 |
| 6. Vegetation | 0.022 | 0.051 | 0.027 | 0.006 |
| 7. Northness | 0.000 | 0.070 | 0.021 | 0.008 |
| 8. Eastness | 0.000 | 0.090 | 0.013 | 0.002 |
| 9. Geological age | 0.000 | 0.063 | 0.023 | 0.007 |
| Ecosystem | Total Cover in km² | Suitable Habitat for P. setaceum |
|---|---|---|
| Coniferous forest | 224 km² | 20 km² (9%) |
| Broadleaved evergreen forest | 99 km² | 1 km² (1%) |
| Scrubland | 78 km² | 51 km² (65%) |
| Cultivated land | 214 km² | 130 km² (61%) |
| Bare soil | 34 km² | 22 km² (65%) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Walentowitz, A.J.; Irl, S.D.H.; Acevedo Rodríguez, A.J.; Palomares-Martínez, Á.; Vetter, V.; Zennaro, B.; Medina, F.M.; Beierkuhnlein, C. Graminoid Invasion in an Insular Endemism Hotspot and Its Protected Areas. Diversity 2019, 11, 192. https://doi.org/10.3390/d11100192
Walentowitz AJ, Irl SDH, Acevedo Rodríguez AJ, Palomares-Martínez Á, Vetter V, Zennaro B, Medina FM, Beierkuhnlein C. Graminoid Invasion in an Insular Endemism Hotspot and Its Protected Areas. Diversity. 2019; 11(10):192. https://doi.org/10.3390/d11100192
Chicago/Turabian StyleWalentowitz, Anna J., Severin D. H. Irl, Aurelio Jesús Acevedo Rodríguez, Ángel Palomares-Martínez, Vanessa Vetter, Barbara Zennaro, Félix M. Medina, and Carl Beierkuhnlein. 2019. "Graminoid Invasion in an Insular Endemism Hotspot and Its Protected Areas" Diversity 11, no. 10: 192. https://doi.org/10.3390/d11100192
APA StyleWalentowitz, A. J., Irl, S. D. H., Acevedo Rodríguez, A. J., Palomares-Martínez, Á., Vetter, V., Zennaro, B., Medina, F. M., & Beierkuhnlein, C. (2019). Graminoid Invasion in an Insular Endemism Hotspot and Its Protected Areas. Diversity, 11(10), 192. https://doi.org/10.3390/d11100192






