Beyond Capricornia: Tropical Sea Slugs (Gastropoda, Heterobranchia) Extend Their Distributions into the Tasman Sea
Abstract
1. Introduction
1.1. An Emerging Trend—Poleward Range Extensions in Sea Slug Distributions
1.2. Recent Poleward Range Extensions in Eastern Australia
2. Materials and Methods
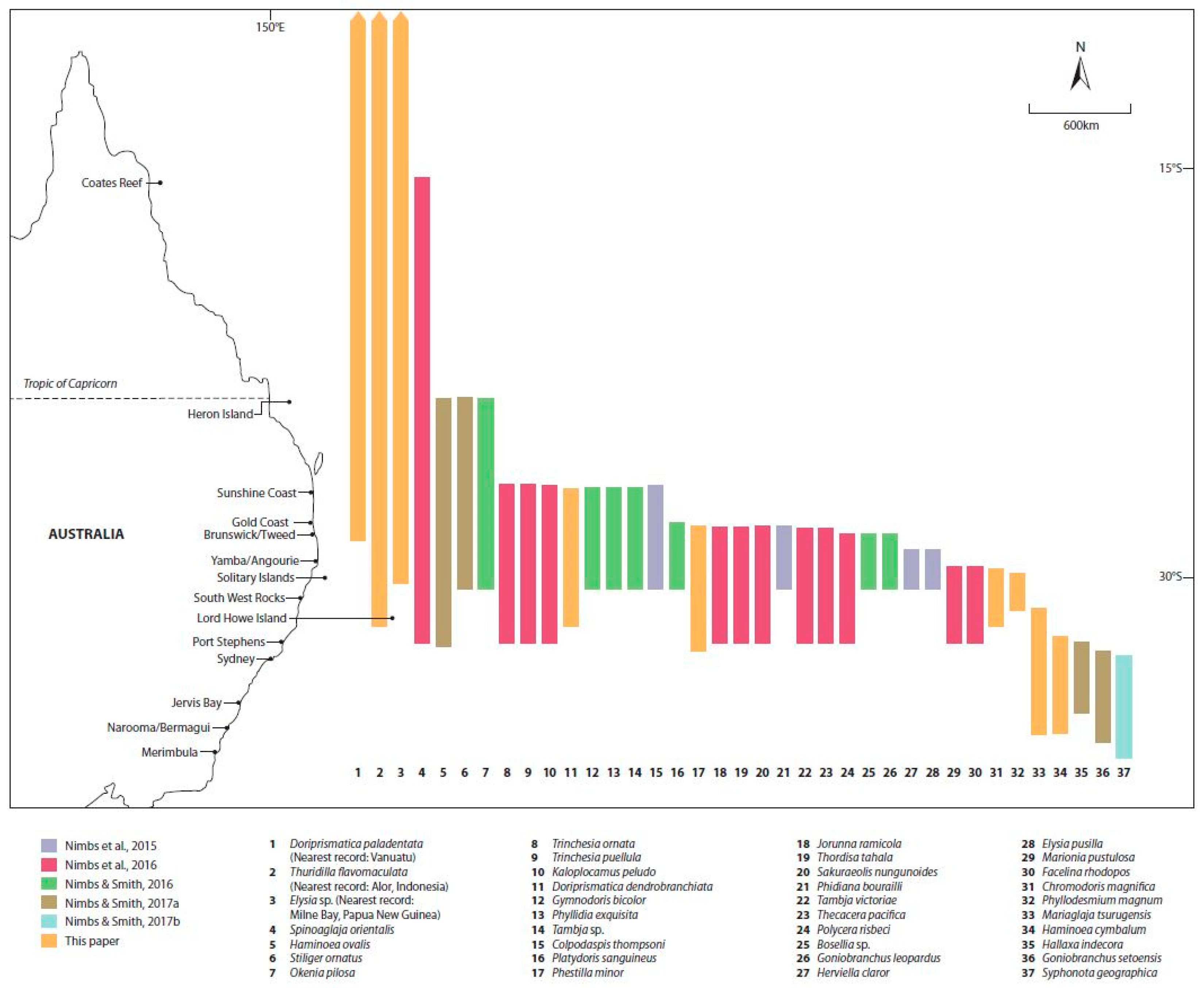
3. Results
Recent Southern Range Shifts in Eastern Australia
- Systematics
- Subclass Heterobranchia
- Order Cephalaspidea Fischer, 1883
- Family Aglajidae Pilsbry, 1895 (1847)
- Genus Mariaglaja Zamora-Silva & Malaquias, 2017
- Mariaglaja tsurugensis (Baba & Abe, 1959) (Figure 2A)
- Synonymy: Chelidonura sandrana Rudman, 1973, Chelidonura babai Gosliner, 1988.
- Family Haminoeidae Pilsbry, 1895
- Genus Haminoea Turton and Kingston [in Carrington], 1830
- Haminoea cymbalum (Quoy and Gaimard, 1832) (Figure 2B)
- Synonymy: Bulla cymbalum Quoy and Gaimard, 1832; Haminea cymbalum (Quoy and Gaimard, 1832); Haminoea simillima Pease, 1868; and, Lamprohaminoea cymbalum (Quoy and Gaimard, 1832) [45].
- Order Sacoglossa
- Family Plakobranchidae Gray, 1840
- Genus Elysia Risso, 1818
- Elysia sp. (Figure 2C)
- Genus Thuridilla Bergh, 1872
- Thuridilla flavomaculata Gosliner, 1995 (Figure 2D)
- Order Nudibranchia
- Family Chromodorididae Bergh, 1891
- Genus Chromodoris Alder & Hancock, 1855
- Chromodoris magnifica (Quoy & Gaimard, 1832) (Figure 2E)
- Synonym: Doris magnifica Quoy & Gaimard, 1832 [45]
- Genus Doriprismatica d’Orbigny, 1839
- Doriprismatica dendrobranchia (Rudman, 1990) (Figure 2F)
- Synonym: Glossodoris dendrobranchia Rudman, 1990 [45]
- Family Facelinidae Bergh, 1889
- Genus Phyllodesmium Ehrenberg, 1831
- Phyllodesmium magnum Rudman, 1991 (Figure 3C)
- Family Trinchesiidae F. Nordsieck, 1972
- Genus Phestilla Bergh, 1874
- Phestilla minor Rudman, 1981 (Figure 3B)
- Synonym: Tenellia minor (Rudman, 1981) [45]
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gosliner, T.M.; Valdés, Á.; Behrens, D.W. Nudibranch and Sea Slug Identification: Indo-Pacific; New World Publications: Jacksonville, FL, USA, 2015. [Google Scholar]
- Kaligis, F.; Eisenbarth, J.H.; Schillo, D.; Dialao, J.; Schäberle, T.F.; Böhringer, N.; Bara, R.; Reumschüssel, S.; König, G.M.; Wägele, H. Second survey of heterobranch sea slugs (Mollusca, Gastropoda, Heterobranchia) from Bunaken National Park, North Sulawesi, Indonesia—How much do we know after 12 years? Mar. Biodivers. Rec. 2018, 11, 2. [Google Scholar] [CrossRef]
- Rudman, W.; Willan, R. Fauna of Australia: Opisthobranchia. In Mollusca: The Southern Synthesis; CSIRO: Melbourne, Australia, 1998. [Google Scholar]
- Burn, R. A checklist and bibliography of the Opisthobranchia (Mollusca: Gastropoda) of Victoria and the Bass Strait area, southeastern Australia. Mus. Vic. Sci. Rep. 2006, 10, 7–13. [Google Scholar] [CrossRef]
- Nimbs, M.J.; Smith, S.D.A. An illustrated inventory of the sea slugs of New South Wales, Australia (Gastropoda: Heterobranchia). Proc. R. Soc. Vic. 2017, 128, 44–113. [Google Scholar] [CrossRef]
- Ridgway, K.R. Long-term trend and decadal variability of the southward penetration of the East Australian Current. Geophys. Res. Lett. 2007, 34, 13. [Google Scholar] [CrossRef]
- Suthers, I.M.; Young, J.W.; Baird, M.E.; Roughan, M.; Everett, J.D.; Brassington, G.B.; Byrne, M.; Condie, S.A.; Hartog, J.R.; Hassler, C.S. The strengthening East Australian Current, its eddies and biological effects—An introduction and overview. Deep Sea Res. Part II Top. Stud. Oceanogr. 2011, 58, 538–546. [Google Scholar] [CrossRef]
- Malcolm, H.A.; Davies, P.L.; Jordan, A.; Smith, S.D.A. Variation in sea temperature and the East Australian Current in the Solitary Islands region between 2001–2008. Deep Sea Res. Part II Top. Stud. Oceanogr. 2011, 58, 616–627. [Google Scholar] [CrossRef]
- Denham, R.N.; Crook, F.G. The Tasman front. N. Z. J. Mar. Freshw. 1976, 10, 15–30. [Google Scholar] [CrossRef]
- Roughan, M.; Middleton, J.H. A comparison of observed upwelling mechanisms off the east coast of Australia. Cont. Shelf Res. 2002, 22, 2551–2572. [Google Scholar] [CrossRef]
- Harriott, V.J.; Smith, S.D.A.; Harrison, P.L. Patterns of coral community structure of subtropical reefs in the Solitary-Islands Marine Reserve, Eastern Australia. Mar. Ecol. Prog. Ser. 1994, 109, 67–76. [Google Scholar] [CrossRef]
- Malcolm, H.A.; Jordan, A.; Smith, S.D.A. Biogeographical and cross-shelf patterns of reef fish assemblages in a transition zone. Mar. Biodivers. 2010, 40, 181–193. [Google Scholar] [CrossRef]
- Smith, S.D.A. Growth and population dynamics of the giant clam Tridacna maxima (Röding) at its southern limit of distribution in coastal, subtropical eastern Australia. Molluscan Res. 2011, 31, 37–41. [Google Scholar]
- Harrison, M.A.; Smith, S.D.A. Cross-shelf variation in the structure of molluscan assemblages on shallow, rocky reefs in subtropical, eastern Australia. Mar. Biodivers. 2012, 42, 203–216. [Google Scholar] [CrossRef]
- Booth, D.J.; Figueira, W.F.; Gregson, M.A.; Brown, L.; Beretta, G. Occurrence of tropical fishes in temperate southeastern Australia: Role of the East Australian Current. Estuar. Coast. Shelf Sci. 2007, 72, 102–114. [Google Scholar] [CrossRef]
- Figueira, W.F.; Booth, D.J. Increasing ocean temperatures allow tropical fishes to survive overwinter in temperate waters. Glob. Chang. Biol. 2010, 16, 506–516. [Google Scholar] [CrossRef]
- Fowler, A.M.; Parkinson, K.; Booth, D.J. New poleward observations of 30 tropical reef fishes in temperate southeastern Australia. Mar. Biodives. 2017, 1–6. [Google Scholar] [CrossRef]
- Padula, V.; Bahia, J.; Vargas, C.; Lindner, A. Mollusca, Nudibranchia: New records and southward range extensions in Santa Catarina, southern Brazil. Check List 2011, 7, 806–808. [Google Scholar] [CrossRef]
- Tibirica, Y.; Pola, M.; Cervera, J.L. Astonishing diversity revealed: An annotated and illustrated inventory of Nudipleura (Gastropoda: Heterobranchia) from Mozambique. Zootaxa 2017, 4359, 1–133. [Google Scholar] [CrossRef] [PubMed]
- Richards, Z.; Kirkendale, L.; Moore, G.; Hosie, A.; Huisman, J.; Bryce, M.; Marsh, L.; Bryce, C.; Hara, A.; Wilson, N. Marine biodiversity in temperate Western Australia: Multi-taxon surveys of Minden and Roe reefs. Diversity 2016, 8, 7. [Google Scholar] [CrossRef]
- Bertsch, H. Biodiversity in La Reserva de la Biósfera Bahía de los Ángeles y Canales de Ballenas y Salsipuedes: naming of a new genus, range extensions and new records, and species list of Heterobranchia (Mollusca: Gastropoda), with comments on biodiversity conservation. Festivus 2014, 46, 158–177. [Google Scholar]
- Schrödl, M.; Hooker, Y. Sea slugs of Peru: Peruvian-Chilean faunal elements. Spixiana 2014, 37, 45–59. [Google Scholar]
- Chichvarkhin, A.Y.; Chichvarkhina, O.V.; Kartavtsev, Y.P. Janolus fuscus O’Donoghue, 1924 (Gastropoda: Proctonotidae), a species of a nudibranch family that is new to the marine fauna of Russia. Russ. J. Mar. Biol. 2016, 42, 20–25. [Google Scholar] [CrossRef]
- Whittaker, D.E. On the occurrence of the nudibranch Geitodoris planata in the North Sea and its addition to the Yorkshire marine fauna, with a note on recent new species. Naturalist 2013, 138, 82. [Google Scholar]
- Uribe, R.A.; Pacheco, A.S. First record of Spurilla neapolitana (Mollusca: Nudibranchia: Aeolidiidae) on the central coast of Peru (Humboldt Current Upwelling Ecosystem). Mar. Biodivers. Rec. 2012, 5. [Google Scholar] [CrossRef]
- Valdés, Á.; Bertsch, H. Redescription and range extension of Bathydoris aioca Marcus, & Marcus, 1962 (Nudibranchia: Gnathodoridoidea). Veliger-Berkeley 2000, 43, 172–178. [Google Scholar]
- Goddard, J.H.R.; Treneman, N.; Pence, W.E.; Mason, D.E.; Dobry, P.M.; Green, B.; Hoover, C. Nudibranch range shifts associated with the 2014 warm anomaly in the Northeast Pacific. Bull. South. Calif. Acad. Sci. 2016, 115, 15–40. [Google Scholar] [CrossRef]
- Goddard, J.H.R.; Schaefer, M.C.; Hoover, C.; Valdés, Á. Regional extinction of a conspicuous dorid nudibranch (Mollusca: Gastropoda) in California. Mar. Biol. 2013, 160, 1497–1510. [Google Scholar] [CrossRef]
- Pecl, G.T.; Araújo, M.B.; Bell, J.D.; Blanchard, J.; Bonebrake, T.C.; Chen, I.C.; Clark, T.D.; Colwell, R.K.; Danielsen, F.; Evengård, B. Biodiversity redistribution under climate change: Impacts on ecosystems and human well-being. Science 2017, 355, eaai9214. [Google Scholar] [CrossRef] [PubMed]
- Goddard, J.H.R.; Gosliner, T.M.; Pearse, J.S. Impacts associated with the recent range shift of the aeolid nudibranch Phidiana hiltoni (Mollusca, Opisthobranchia) in California. Mar. Biol. 2011, 158, 1095–1109. [Google Scholar] [CrossRef] [PubMed]
- Bridle, T. Spurilla braziliana—A new sea slug in South Australia. South Aust. Nat. 2017, 91, 29–33. [Google Scholar]
- Por, F.D. Lessepsian Migration: The Influx of Red Sea Biota into the Mediterranean by Way of the Suez Canal; Springer Science & Business Media: Berlin, Germany, 2012; Volume 23. [Google Scholar]
- Çevik, C.E.M.; Özturk, B. A new Lessepsian mollusc Hypselodoris infucata (Ruppell and Leuckart, 1828) (Gastropoda: Nudibranchia) for the coasts of Turkey. Turk. J. Zool. 2000, 25, 27–30. [Google Scholar]
- Zenetos, A.; Çinar, M.E.; Pancucci-Papadopoulou, M.A.; Harmelin, J.G.; Furnari, G.; Andaloro, F.; Bellou, N.; Streftaris, N.; Zibrowius, H. Annotated list of marine alien species in the Mediterranean with records of the worst invasive species. Mediterr. Mar. Sci. 2005, 6, 63–118. [Google Scholar] [CrossRef]
- Moles, J.; Mas, G.; Figueroa, I.; Fernández-Vilert, R.; Salvador, X.; Giménez, J. As fast as a hare: Colonization of the heterobranch Aplysia dactylomela (Mollusca: Gastropoda: Anaspidea) into the western Mediterranean Sea. Cah. Biol. Mar. 2017, 2017 58, 341–345. [Google Scholar]
- Gavagnin, M.; Carbone, M.; Nappo, M.; Mollo, E.; Roussis, V.; Cimino, G. First chemical study of anaspidean Syphonota geographica: Structure of degraded sterols aplykurodinone-1 and -2. Tetrahedron 2005, 61, 617–621. [Google Scholar] [CrossRef]
- Harasti, D. Range extension and first occurrence of the thorny seahorse Hippocampus histrix in New South Wales, Australia. Mar. Biodivers. Rec. 2015, 8. [Google Scholar] [CrossRef]
- Scott, A.; Harasti, D.; Davis, T.; Smith, S.D.A. Southernmost records of the host sea anemone, Stichodactyla haddoni, and associated commensal shrimps in a climate change hotspot. Mar. Biodivers. 2015, 45. [Google Scholar] [CrossRef]
- Davis, T.R. First records of three fishes, and southern records of a further four fishes, from New South Wales, Australia. Check List 2016, 12, 2008. [Google Scholar] [CrossRef]
- Nimbs, M.J.; Smith, S.D.A. Revision of the southern distribution limit for the tropical marine herbivore Syphonota geographica (A. Adams & Reeve, 1850) (Heterobranchia: Aplysiidae) in a global climate change hot-spot. Aust. Zool. 2017, 38. [Google Scholar] [CrossRef]
- Nimbs, M.J.; Willan, R.C.; Smith, S.D.A. Range extensions for heterobranch sea slugs (formerly opisthobranch) belonging to the families Diaphanidae, Plakobranchidae and Facelinidae on the Eastern Coast of Australia. Mar. Biodivers. Rec. 2015, 8, e76. [Google Scholar] [CrossRef]
- Nimbs, M.J.; Smith, S.D.A. Welcome strangers: Southern range extensions for seven heterobranch sea slugs (Mollusca: Gastropoda) on the subtropical east Australian coast, a climate change hot spot. Reg. Stud. Mar. Sci. 2016, 8, 27–32. [Google Scholar] [CrossRef]
- Nimbs, M.J.; Willan, R.C.; Larkin, M.; Davis, T.R.; Smith, S.D.A. Southern range extensions for twelve heterobranch sea slugs (Gastropoda: Heterobranchia) on the eastern coast of Australia. Mar. Biodivers. Rec. 2016, 9. [Google Scholar] [CrossRef]
- Layton, K.K.; Gosliner, T.M.; Wilson, N.G. Flexible colour patterns obscure identification and mimicry in Indo-Pacific Chromodoris nudibranchs (Gastropoda: Chromodorididae). Mol. Phylogenet. Evol. 2018, 124, 27–36. [Google Scholar] [CrossRef] [PubMed]
- WoRMS Editorial Board. World Register of Marine Species. 2018. Available online: http://www.marinespecies.org at VLIZ (accessed on 4 August 2018).
- Coleman, N. Nudibranchs Encyclopedia; Neville Coleman’s Underwater Geographic Pty, National Library of Australia: Springwood, Australia, 2008; ISBN 9780947325411. [Google Scholar]
- Coleman, N. Nudibranchs Encyclopedia: Catalogue of Asia/Indo Pacific Sea Slugs, 2nd ed.; Neville Coleman’s Underwater Geographic: Springwood, Australia, 2008. [Google Scholar]
- Cobb, G.; Willan, R.C. Undersea Jewels: A Colour Guide to Nudibranchs; ABRS: Canberra, Australia, 2006. [Google Scholar]
- Debelius, H.; Kuiter, R.H. Nudibranchs of the World; Ikan-Unterwasserarchiv: Frankfurt, Germany, 2007; ISBN 3939767069. [Google Scholar]
- Buchanan, C. Species List and Semi-Quantitative Abundance from Buchanan (1989) A Reference List of Opisthobranch Molluscs from the Solitary Islands and Adjacent Coast; Solitary Islands Underwater Research Group: Coffs Harbour Jetty, Australia, 1989. [Google Scholar]
- Pixel, N. Available online: http://www.nudipixel.net (accessed on 3 October 2017).
- Atlas of Living Australia. 2015. Available online: http://www.ala.org.au (accessed on 31 March 2017).
- Rudman, W.B. Sea Slug Forum. Available online: http://www.seaslugforum.net/find/chelfulv (accessed on 6 October 2017).
- Ocean Biogeographic Information System (OBIS). Intergovernmental Oceanographic Commission of UNESCO. Available online: http://www.iobis.org (accessed on 3 October 2017).
- Cobb, G.; Mullins, D. Nudibranchs: Sunshine Coast QLD & Tasmania Australia—Thordisa tahala. Available online: http://www.nudibranch.com.au/pages/CapThorClown-044w.htm (accessed on 7 October 2017).
- Köhler, E. “Doriprismatica” paladentata (Rudman, 1986). Available online: https://www.philippine-sea-slugs.com/Nudibranchia/Euctenidiacea/Doriprismatica_paladentata_02.htm (accessed on 6 October 2017).
- Clarke, L.; Buckland, P. Julian Rocks.net. Available online: http://www.julianrocks.net/index.php (accessed on 19 October 2017).
- Riek, D. Sea Slugs and other Marine Invertebrates of the Tweed-Byron Coast, Australia. Available online: http://www.roboastra.com (accessed on 17 October 2017).
- GBIF.org. GBIF Home Page. Available online: http://www.gbif.org (accessed on 20 April 2017).
- Rudman, W.B. Chelidonura sandrana. Available online: http://www.seaslugforum.net/find/chelsand (accessed on 20 April 2017).
- Rudman, W.B. Re: Chelidonura sandrana—Feeding. Available online: http://www.seaslugforum.net/find/8940 (accessed on 20 April 2017).
- Malaquias, M.A.E.; Cervera, J.L. The genus Haminoea (Gastropoda: Cephalaspidea) in Portugal, with a review of the European species. J. Molluscan Stud. 2006, 72, 89–103. [Google Scholar] [CrossRef]
- Rudman, W.B. Haminoea cymbalum in Southern Queensland. Available online: http://www.seaslugforum.net (accessed on 20 April 2017).
- Carmona, L.; Malaquias, M.A.E.; Gosliner, T.M.; Pola, M.; Cervera, J.L. Amphi-Atlantic distributions and cryptic species in sacoglossan sea slugs. J. Molluscan Stud. 2011, 77, 401–412. [Google Scholar] [CrossRef]
- Gosliner, T.M. The genus Thuridilla (Opisthobranchia: Elysiidae) from the tropical Indo-Pacific, with a revision of the phylogeny and systematics of the Elysiidae. Proc. Calif. Acad. Sci. 1995, 49, 1–54. [Google Scholar]
- Urasawa, T. Thuridilla flavomaculata from the Mariana Islands. Sea Slug Forum. Available online: http://www.seaslugforum.net/showall/thurflav (accessed on 3 October 2017).
- Bala, K. Thuridilla flavomaculata from Bunaken, Indonesia. Available online: http://www.seaslugforum.net/find/15555 (accessed on 3 October 2017).
- Warren, L. Thuridilla flavomaculata from Sulawesi. Available online: http://www.seaslugforum.net/find/2804 (accessed on 3 October 2017).
- Rudman, W.B. Chromodoris magnifica (Quoy & Gaimard, 1832). Available online: http://www.seaslugforum.net/showall/chrmag (accessed on 3 October 2017).
- Bryce, C. Chromodoris magnifica from W. Australia. Available online: http://www.seaslugforum.net/find/805 (accessed on 3 October 2017).
- Reef Life Survey. Available online: http://reeflifesurvey.com (accessed on 23 August 2018).
- Marshall, J.G.; Willan, R.C. Nudibranchs of Heron Island, Great Barrier Reef: A Survey of the Opisthobranchia (Sea Slugs) of Heron and Wistari Reefs; Backhuys: Leiden, The Netherlands, 1999; ISBN 9057820331. [Google Scholar]
- Atlas of Living Australia. Human observation of Chromodoris magnifica Recorded on 2013-06-16. Available online: https://biocache.ala.org.au/occurrences/defbe9d3-3638-49b8-a699-b4f8ea5abb11 (accessed on 3 October 2017).
- Rudman, W.B. Glossodoris dendrobranchia Rudman. 1990. Available online: http://www.seaslugforum.net/factsheet/glosdend (accessed on 5 October 2017).
- Rudman, W.B. The Chromodorididae (Opisthobranchia: Mollusca) of the Indo-West Pacific: further species of Glossodoris, Thorunna and the Chromodoris aureomarginata colour group. Zool. J. Linn. Soc. 1990, 100, 263–326. [Google Scholar] [CrossRef]
- Cobb, G. Glossodoris dendrobranchia in sthn Queensland. Available online: http://www.seaslugforum.net/find/12235 (accessed on 5 October 2017).
- Rudman, W.B. Glossodoris paladentatus Rudman. 1986. Available online: http://www.seaslugforum.net/showall/glospala (accessed on 6 October 2017).
- Rudman, W.B. Glossodoris atromarginata. Available online: http://www.seaslugforum.net/find/glosatro (accessed on 6 October 2017).
- Rudman, W.B. The Chromodorididae (Opisthobranchia: Mollusca) of the Indo-West Pacific: The genus Glossodoris Ehrenbergh (=Casella, H. & A. Adams). Zool. J. Linn. Soc. 1986, 86, 101–184. [Google Scholar]
- Rudman, W.B. Phyllodesmium magnum Rudman. 1991. Available online: http://www.seaslugforum.net/showall/phylmagn (accessed on 7 October 2017).
- Rudman, W.B. The anatomy and biology of alcyonarian-feeding aeolid opisthobranch molluscs and their development of symbiosis with zooxanthellae. Zool. J. Linn. Soc. 1981, 72, 219–262. [Google Scholar] [CrossRef]
- Rudman, W.B. Further studies on the taxonomy and biology of the octocoral-feeding genus Phyllodesmium Ehrenberg, 1831 (Nudibranchia: Aeolidoidea). J. Molluscan Stud. 1991, 57, 167–203. [Google Scholar] [CrossRef]
- Brook, F.J.; Marshall, B.A. Checklist of benthic coastal marine Mollusca from the northern Kermadec Islands. Appendix in: Brook, F.J., The coastal molluscan fauna of northern Kermadec Islands, Southwest Pacific Ocean. J. R. Soc. New Zeal. 1998, 28, 210–233. [Google Scholar] [CrossRef]
- Smith, S.D.A.; Rule, M.J.; Harrison, M.; Dalton, S.J. Monitoring the sea change: Preliminary assessment of the conservation value of nearshore reefs, and existing impacts, in a high-growth, coastal region of subtropical eastern Australia. Mar. Poll. Bull. 2008, 56, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Rudman, W.B. Further studies on the anatomy and ecology of opisthobranch molluscs feeding on the scleractinian coral Porites. Zool. J. Linn. Soc. 1981, 71, 373–412. [Google Scholar] [CrossRef]
- Rudman, W.B. Sea Slug Forum: Phestilla minor. Available online: http://www.seaslugforum.net/find/phesmino (accessed on 7 October 2017).
- Tarrant, D. Cowries of New South Wales; Regal Printing: Hong Kong, China, 2016. [Google Scholar]
- Champion, C.; Hobday, A.J.; Tracey, S.R.; Pecl, G.T. Rapid shifts in distribution and high-latitude persistence of oceanographic habitat revealed using citizen science data from a climate change hotspot. Glob. Chang. Biol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Nimbs, M.J. NudiKey: An illustrated, interactive identification key to the families of Australian heterobranch sea-slugs (Mollusca: Gastropoda). Aust. Zool. 2017, 38, 537–546. [Google Scholar] [CrossRef]
- Giacobbe, S.; De Matteo, S. The potentially invasive opisthobranch Polycera hedgpethi Er. Marcus, 1964 (Gastropoda Nudibranchia), introduced in a Mediterranean coastal lagoon. Biodivers. J. 2013, 4, 359–364. [Google Scholar]
- Wilson, N.G. New record of the nudibranch Polycera hedgpethi Er. Marcus, 1964, in South Australia, with a discussion on its occurrence in Australia. Rec. West. Aust. Mus. 2006, 140, 137–140. [Google Scholar] [CrossRef]
- Przeslawski, R.; Ahyong, S.; Byrne, M.; Woerheide, G.; Hutchings, P.A.T. Beyond corals and fish: The effects of climate change on noncoral benthic invertebrates of tropical reefs. Glob. Chang. Biol. 2008, 14, 2773–2795. [Google Scholar] [CrossRef]
- Balmford, A.; Green, M.J.B.; Murray, M.G. Using higher-taxon richness as a surrogate for species richness: I. Regional tests. Proc. R. Soc. Lond. B 1996, 263, 1267–1274. [Google Scholar] [CrossRef]
- Caro, T.M.; O’Doherty, G. On the use of surrogate species in conservation biology. Conserv. Biol. 1999, 13, 805–814. [Google Scholar] [CrossRef]
- Smith, S.D.A. Rapid assessment of invertebrate biodiversity on rocky shores: Where there’s a whelk there’s a way. Biodivers. Conserv. 2005, 14, 3565–3576. [Google Scholar] [CrossRef]
- Hughes, T.P.; Kerry, J.T.; Álvarez-Noriega, M.; Álvarez-Romero, J.G.; Anderson, K.D.; Baird, A.H.; Babcock, R.C.; Beger, M.; Bellwood, D.R.; Berkelmans, R. Global warming and recurrent mass bleaching of corals. Nature 2017, 543, 373. [Google Scholar] [CrossRef] [PubMed]
- Beger, M.; Sommer, B.; Harrison, P.L.; Smith, S.D.A.; Pandolfi, J.M. Conserving potential coral reef refuges at high latitudes. Divers. Distrib. 2014, 20, 245–257. [Google Scholar] [CrossRef]
- Last, P.R.; White, W.T.; Gledhill, D.C.; Hobday, A.J.; Brown, R.; Edgar, G.J.; Pecl, G. Long-term shifts in abundance and distribution of a temperate fish fauna: a response to climate change and fishing practices. Glob. Ecol. Biogeogr. 2011, 20, 58–72. [Google Scholar] [CrossRef]
- Malcolm, H.A.; Smith, S.D.A. Objective selection of surrogate families to describe reef fish assemblages in a subtropical marine park. Biodivers. Conserv. 2010, 19, 3611–3618. [Google Scholar] [CrossRef]
- Veron, J.E.N. Corals in Space and Time: The Biogeography and Evolution of the Scleractinia; Cornell University Press: New York, NY, USA, 1995; ISBN 0801482631. [Google Scholar]
- Smith, S.D.A.; Nimbs, M.J. Quantifying temporal variation in heterobranch (Mollusca: Gastropoda) sea slug assemblages: Tests of alternate models. Molluscan Res. 2017, 37. [Google Scholar] [CrossRef]
- Larkin, M.; Smith, S.D.A.; Willan, R.C.; Davis, T.R. Diel and seasonal variation in heterobranch sea slug assemblages within an embayment in temperate eastern Australia. Mar. Biodivers. 2017. [Google Scholar] [CrossRef]


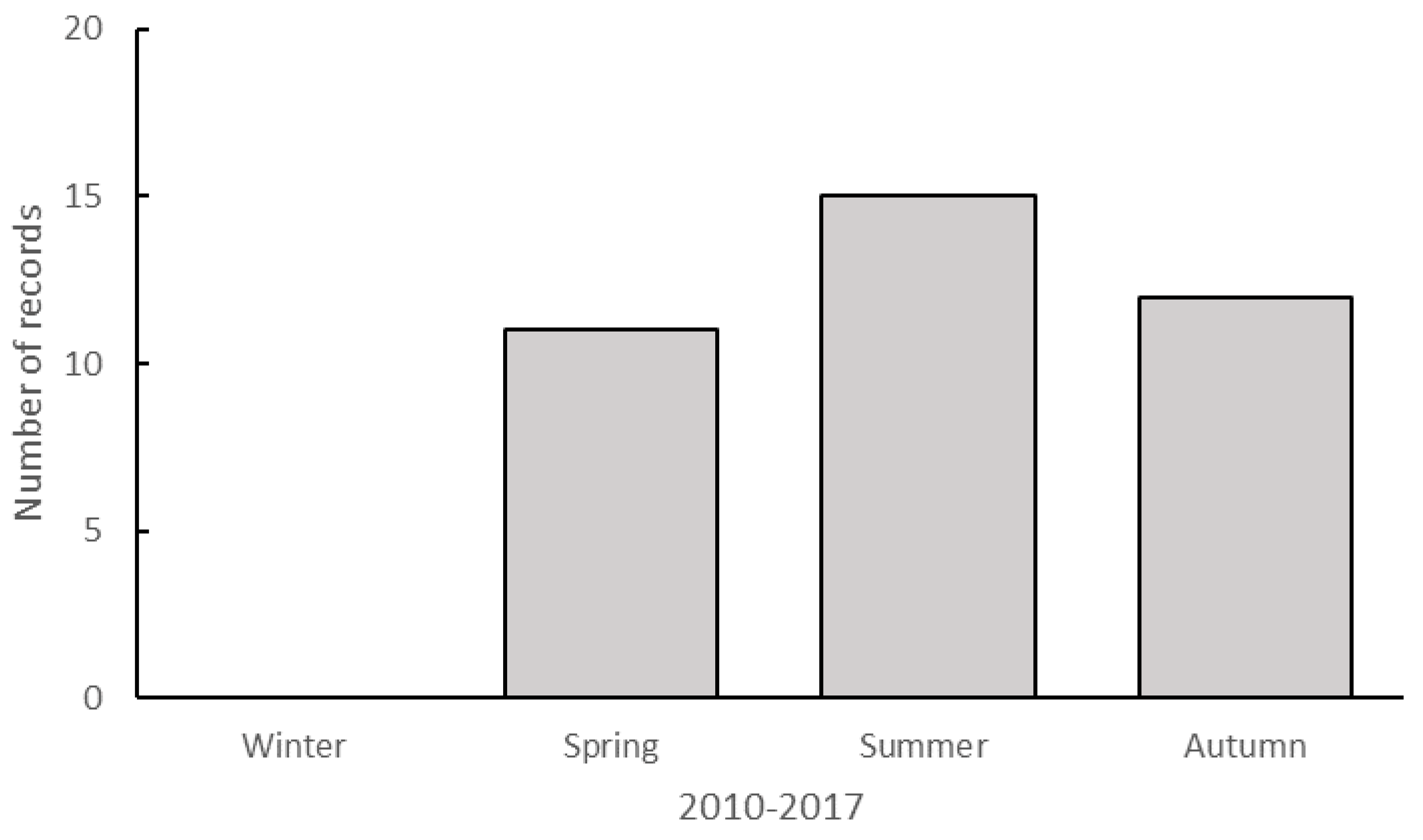
| Source Type | Reference |
|---|---|
| Published literature | Coleman [46,47] |
| Nimbs & Smith [5] | |
| Gosliner et al. [1] | |
| Burn [4] | |
| Cobb & Willan [48] | |
| Debelius & Kuiter [49] | |
| Buchanan [50] | |
| Electronic sources | NudiPixel [51] |
| Atlas of Living Australia [52] | |
| Sea Slug Forum [53] | |
| OBIS [54] | |
| Cobb & Mullins [55] | |
| Köhler [56] | |
| Clarke & Buckland [57] | |
| Riek [58] | |
| GBIF [59] |
| Location | Date | Latitude | Longitude | Reference |
|---|---|---|---|---|
| Lizard Island, QLD | 1979 | 14°40′24″ S | 145°27′05″ E | [61] |
| Angourie, NSW | 2017 | 29°28′53″ S | 153°21′54″ E | This paper |
| Solitary Islands, NSW | 1988 | 30°12′13″ S | 153°16′52″ E | [50] |
| Lord Howe Island, NSW | 1979 | 31°33′20″ S | 159°04′56″ E | [46] |
| Bermagui, NSW | 2017 | 36°25′22″ S | 150°05′13″ E | This paper |
| Location | Date | Latitude | Longitude | Reference |
|---|---|---|---|---|
| Great Detached Reef | 1981 | 11°44′23″ S | 144°03′46″ E | [52] |
| Lizard Island, QLD | 2004 | 14°57′23″ S | 145°44′05″ E | [52] |
| Marble Island, QLD | 2002 | 21°59′03″ S | 150°09′21″ E | [52] |
| Saumarez Reef, QLD | 1983 | 21°50′00″ S | 153°39′59″ E | [52] |
| Heron Island, QLD | 1981 | 23°26′35″ S | 151°54′35″ E | [52] |
| Gold Coast, QLD | 2007 | 28°03′12″ S | 153°26′27″ E | [47] |
| Solitary Islands, NSW | 2015 | 30°12′13″ S | 153°16′52″ E | [5] |
| South West Rocks, NSW | 2014 | 30°55′49″ S | 153°05′33″ E | [52] |
| Port Stephens, NSW | 1987 | 32°43′54″ S | 152°32′00″ E | [52] |
| Bermagui, NSW | 2017 | 36°25′22″ S | 150°05′13″ E | This paper |
| Location | Date | Latitude | Longitude | Reference |
|---|---|---|---|---|
| Milne Bay, Papua New Guinea | 2007 | 10°24′21″ S | 150°36′36″ E | [46,47] |
| North Solitary Island, NSW | 2017 | 29°55′30″ S | 153°23′20″ E | This paper |
| Location | Date | Latitude | Longitude | Reference |
|---|---|---|---|---|
| Saipan, N. Mariana Islands | 2006 | 15°09′15″ N | 145°45′13″ E | [66] |
| Guam | 2010 | 13°26′32″ N | 144°51′35″ E | [65] |
| Marshall Islands | 1995 | 07°07′53″ N | 171°11′04″ E | [65] |
| Bunaken, Indonesia | 2006 | 01°37′51″ N | 124°45′42″ E | [67] |
| Lembeh, Indonesia | 2009 | 01°26′43″ N | 125°12′56″ E | [51] |
| Tukang Besi, Indonesia | 2000 | 05°27′17″ S | 123°39′30″ E | [68] |
| Alor, Indonesia | 2010 | 08°15′01″ S | 124°29′37″ E | [51] |
| Malindi, Kenya | 2010 | 03°13′34″ S | 040°08′38″ E | [51] |
| Lord Howe Island, NSW | 2017 | 31°33′45″ S | 159°05′36″ E | This paper |
| Location | Date | Latitude | Longitude | Reference |
|---|---|---|---|---|
| Cartier & Ashmore Reefs, WA | 1999 | 12°15′26″ S | 123°00′01″ E | [70] |
| Fitzroy Island, QLD | 2015 | 16°55′18″ S | 145°59′00″ E | [71] |
| Heron Island, QLD | 1999 | 23°26′35″ S | 151°54′38″ E | [72] |
| Gneering Shoals, QLD | 2012 | 26°39′00″ S | 153°11′00″ E | [48] |
| Cook Island, NSW | 2013 | 28°11′46″ S | 153°34′35″ E | [73] |
| North Solitary Island, NSW | 2017 | 29°55′50″ S | 153°23′29″ E | This paper |
| Lord Howe Island, NSW | 2009 | 31°33′45″ S | 159°05′36″ E | [51] |
| Location | Date | Latitude | Longitude | Reference |
|---|---|---|---|---|
| Swain Reefs, QLD | 1990 | 21°45′18″ S | 152°22′20″ E | [74] |
| Noosa, QLD | 2004, 2018 | 26°21′33″ S | 153°06′46″ E | [76] |
| Lord Howe Island, NSW | 2017 | 31°33′45″ S | 159°05′36″ E | This paper |
| Location | Date | Latitude | Longitude | Reference |
|---|---|---|---|---|
| Anilao, Philippines | 2010 | 13°42′45″ N | 120°51′10″ E | [56] |
| Vietnam | 2005 | 11°12′06″ N | 109°08′41″ E | [51] |
| Banda Islands, Indonesia | 2010 | 04°43′09″ S | 129°59′35″ E | [51] |
| Milne Bay, PNG | 1982 | 10°02′45″ S | 150°05′02″ E | [79] |
| Espiritu Santo Is., Vanuatu | 1999 | 15°15′59″ S | 166°40′44″ E | [53] |
| Cook Island, NSW | 2017 | 28°11′46″ S | 153°34′35″ E | This paper |
| Location | Date | Latitude | Longitude | Reference |
|---|---|---|---|---|
| Keppel Island, QLD | 1969 | 23°10′30″ S | 150°55′38″ E | [46,47] |
| Heron Island, QLD | 1999 | 23°26′35″ S | 151°54′38″ E | [72] |
| Gneering Shoals, QLD | 2011 | 26°39′00″ S | 153°11′00″ E | [48] |
| Cook Island, NSW | 1977 | 28°11′46″ S | 153°34′35″ E | [46] |
| Julian Rocks, NSW | n.d. | 28°36′42″ S | 153°37′44″ E | [57] |
| Solitary Islands, NSW | 1988 | 30°04′03″ S | 153°20′15″ E | [50] |
| South West Rocks, NSW | 2016 | 30°55′49″ S | 153°05′33″ E | This paper, [5] |
| Location | Date | Latitude | Longitude | Reference |
|---|---|---|---|---|
| Lizard Island, QLD | 1979 | 14°40′24″ S | 145°27′05″ E | [86] |
| Mooloolaba, QLD | 2004 | 26°40′32″ S | 153°08′12″ E | [55] |
| Hastings Point, NSW | 2006 | 28°21′40″ S | 153°34′50″ E | [58] |
| Port Stephens, NSW | 2017 | 32°43′54″ S | 152°32′00″ E | This paper |
| Year | |||||||
| 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 |
| 1 | - | - | 3 | 5 | 13 | 7 | 8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nimbs, M.J.; Smith, S.D.A. Beyond Capricornia: Tropical Sea Slugs (Gastropoda, Heterobranchia) Extend Their Distributions into the Tasman Sea. Diversity 2018, 10, 99. https://doi.org/10.3390/d10030099
Nimbs MJ, Smith SDA. Beyond Capricornia: Tropical Sea Slugs (Gastropoda, Heterobranchia) Extend Their Distributions into the Tasman Sea. Diversity. 2018; 10(3):99. https://doi.org/10.3390/d10030099
Chicago/Turabian StyleNimbs, Matt J., and Stephen D. A. Smith. 2018. "Beyond Capricornia: Tropical Sea Slugs (Gastropoda, Heterobranchia) Extend Their Distributions into the Tasman Sea" Diversity 10, no. 3: 99. https://doi.org/10.3390/d10030099
APA StyleNimbs, M. J., & Smith, S. D. A. (2018). Beyond Capricornia: Tropical Sea Slugs (Gastropoda, Heterobranchia) Extend Their Distributions into the Tasman Sea. Diversity, 10(3), 99. https://doi.org/10.3390/d10030099






