Distribution of Plant Species and Dispersal Traits along Environmental Gradients in Central Mediterranean Summits
Abstract
1. Introduction
2. Materials and Methods
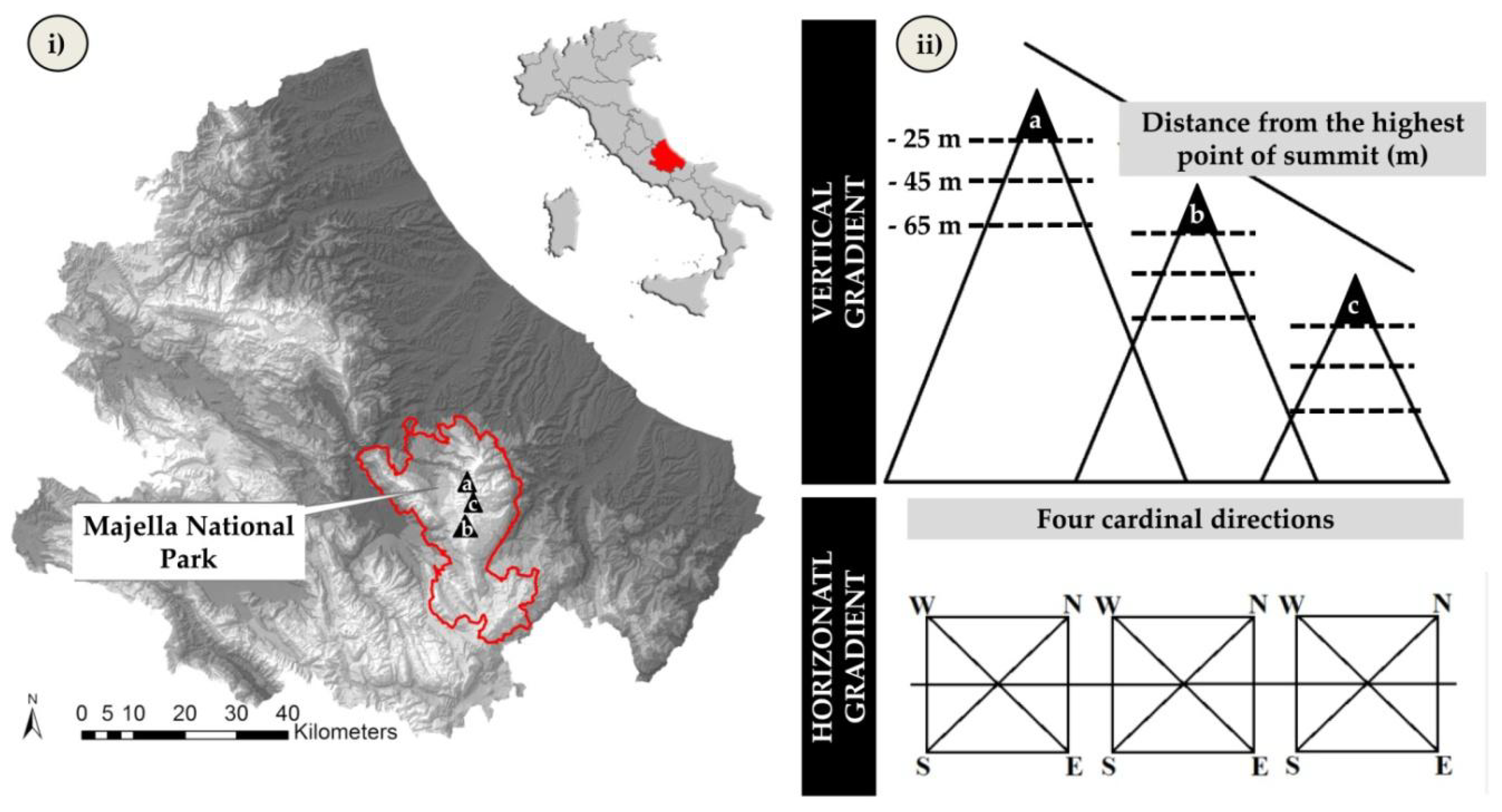
2.1. Study Area
2.2. Data Collection
2.3. Plant Traits
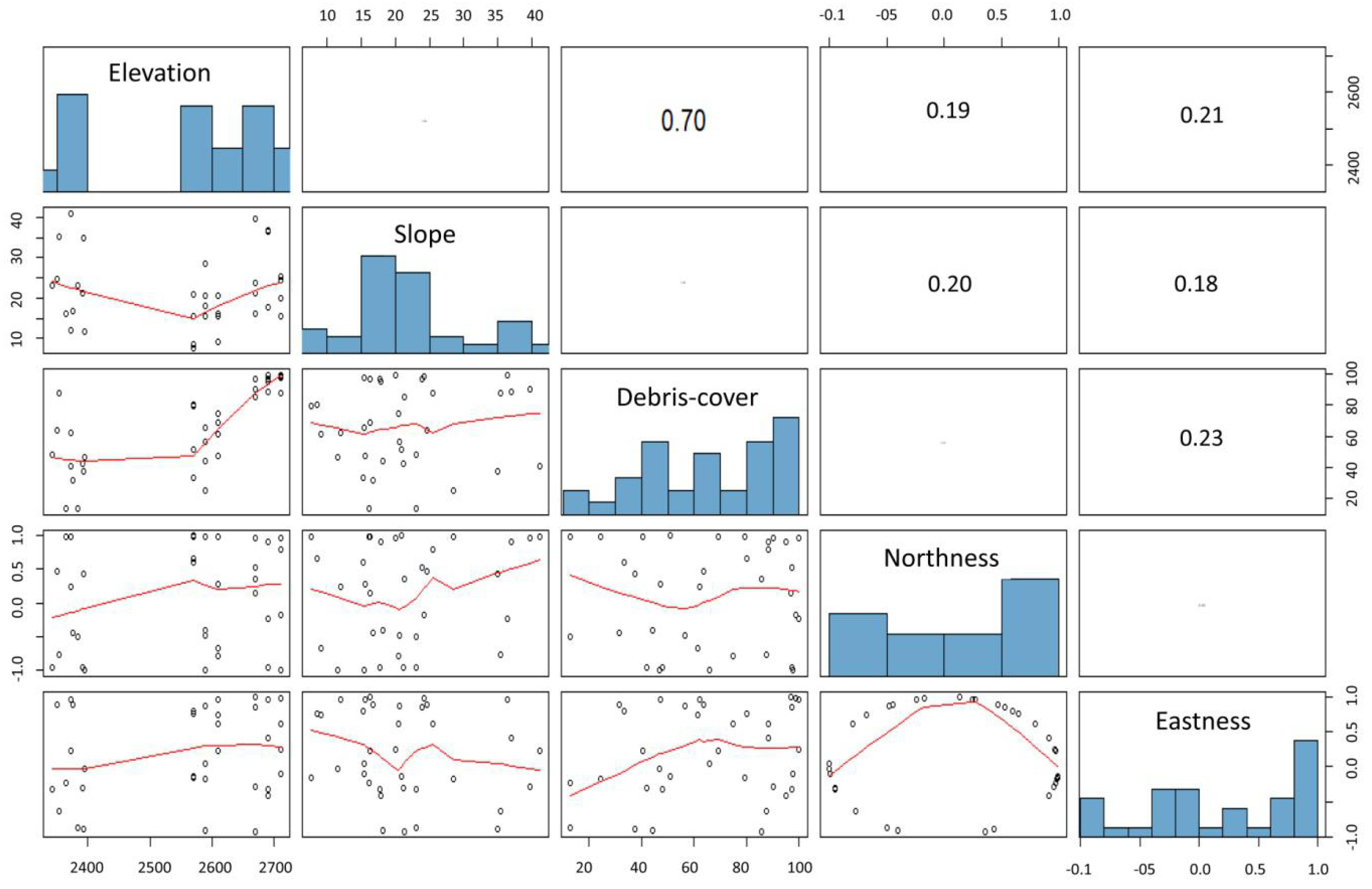
2.4. Data Analysis
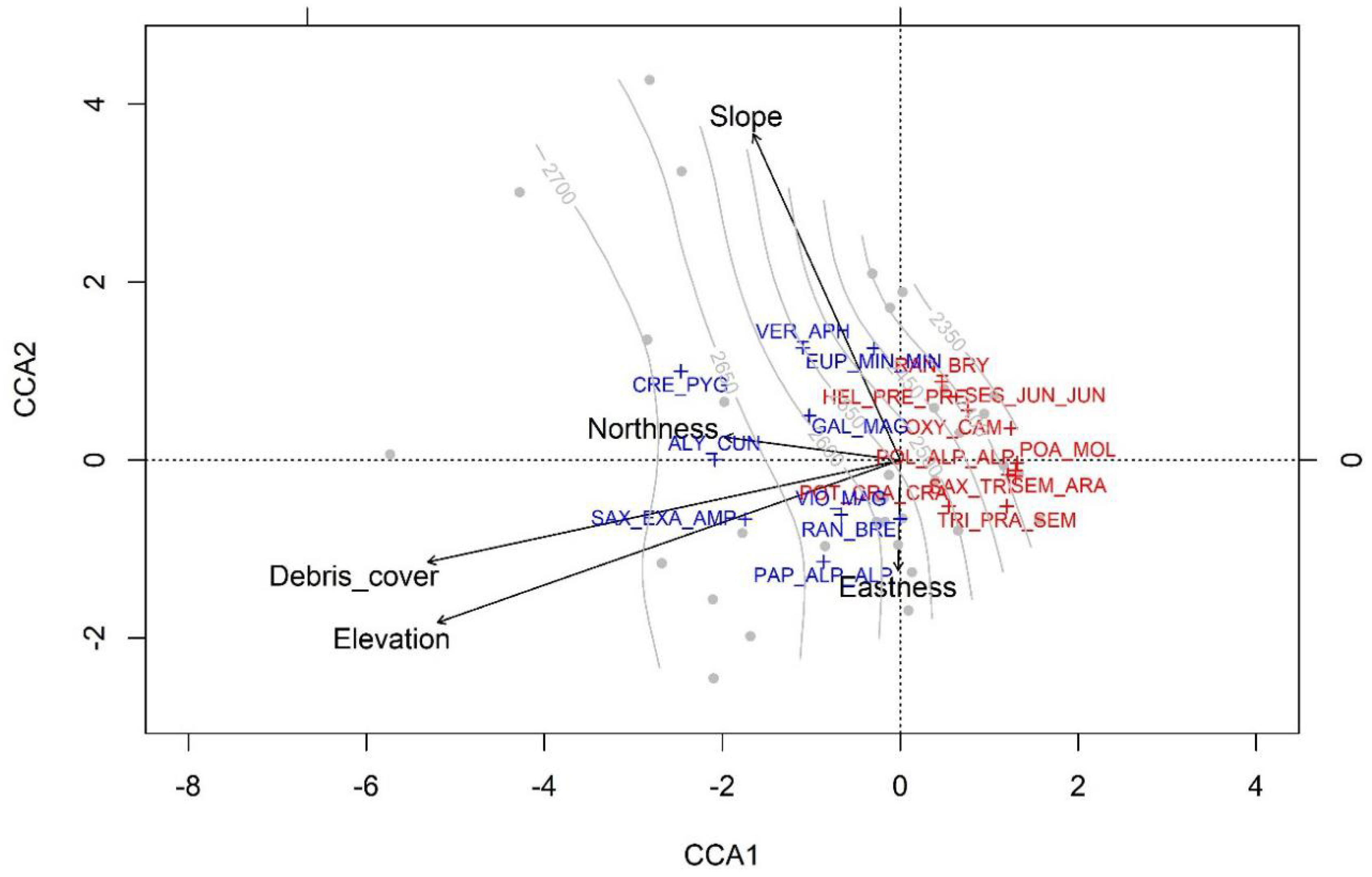
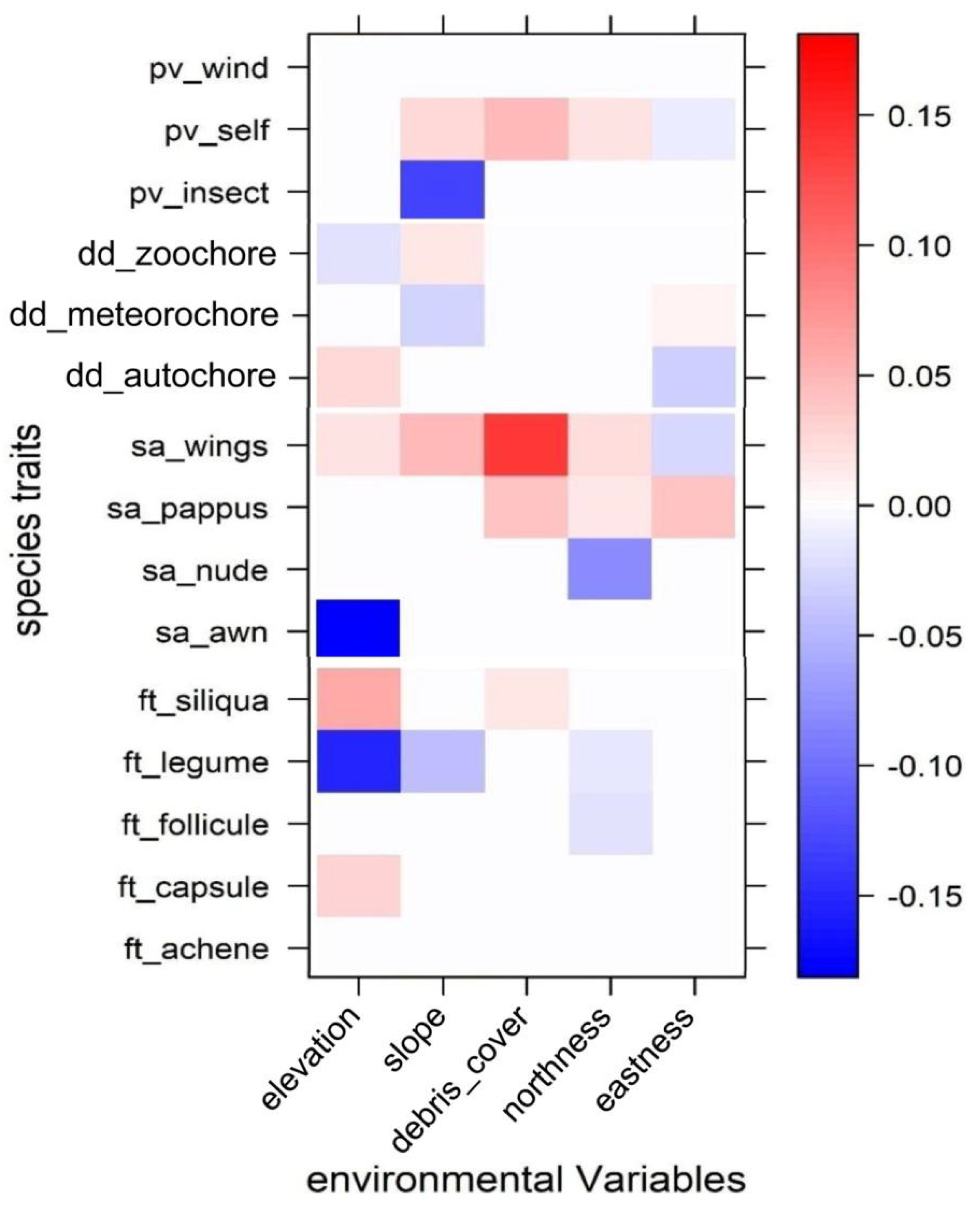
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Species Name | Taxonomic Family | pv | dd | ft | sa |
|---|---|---|---|---|---|
| Achillea barrelieri subsp. barrelieri | Asteraceae | insect | autochory | achene | wings |
| Adonis distorta | Ranunculaceae | insect | zoochory | achene | awn |
| Alyssum cuneifolium | Brassicaceae | insect | autochory | siliqua | wings |
| Androsace villosa subsp. villosa | Primulaceae | insect | autochory | capsule | nude |
| Anthyllis montana subsp. montana | Fabaceae | insect | meteochory | legume | nude |
| Anthyllis vulneraria subsp. pulchella | Fabaceae | insect | meteochory | legume | nude |
| Arabis alpina subsp. caucasica | Brassicaceae | insect | autochory | siliqua | nude |
| Arenaria grandiflora subsp. grandiflora | Caryophyllaceae | insect | autochory | capsule | nude |
| Armeria gracilis | Plumbaginaceae | insect | meteochory | achene | wings |
| Aster alpinus subsp. alpinus | Asteraceae | insect | meteochory | achene | wings |
| Bellis perennis | Asteraceae | insect | autochory | achene | nude |
| Bistorta vivipara | Polygonaceae | insect | autochory | achene | nude |
| Bunium petraeum | Apiaceae | insect | zoochory | achene | nude |
| Campanula cochleariifolia | Campanulaceae | insect | autochory | capsule | nude |
| Campanula scheuchzeri subsp. scheuchzeri | Campanulaceae | insect | autochory | capsule | nude |
| Carduus chrysacanthus | Asteraceae | insect | meteochory | achene | pappus |
| Carex humilis | Cyperaceae | wind | meteochory | achene | nude |
| Carex kitaibeliana | Cyperaceae | wind | zoochory | achene | nude |
| Carex myosuroides | Juncaceae | wind | meteochory | achene | awn |
| Carum heldreichii | Apiaceae | insect | zoochory | achene | nude |
| Cerastium thomasii | Caryophyllaceae | wind | autochory | capsule | nude |
| Cerastium tomentosum | Caryophyllaceae | self | autochory | capsule | nude |
| Clinopodium alpinum subsp. alpinum | Lamiaceae | insect | zoochory | achene | nude |
| Crepis aurea subsp. glabrescens | Asteraceae | insect | meteochory | achene | pappus |
| Crepis magellensis | Asteraceae | insect | meteochory | achene | pappus |
| Crepis pygmaea | Asteraceae | self | meteochory | achene | pappus |
| Doronicum columnae | Asteraceae | insect | meteochory | achene | pappus |
| Draba aizoides subsp. aizoides | Brassicaceae | insect | autochory | siliqua | nude |
| Edraianthus graminifolius subsp. graminifolius | Campanulaceae | insect | autochory | capsule | nude |
| Erigeron epiroticus | Asteraceae | insect | meteochory | achene | pappus |
| Erysimum majellense | Brassicaceae | insect | autochory | siliqua | nude |
| Euphrasia minima subsp. minima | Scrophulariaceae | self | autochory | capsule | nude |
| Festuca violacea subsp. italica | Poaceae | wind | meteochory | achene | awn |
| Galium magellense | Rubiaceae | wind | zoochory | achene | nude |
| Gentiana nivalis | Gentianaceae | self | autochory | capsule | nude |
| Gentiana orbicularis | Gentianaceae | insect | autochory | capsule | nude |
| Gentiana verna subsp. verna | Gentianaceae | insect | autochory | capsule | nude |
| Helianthemum oelandicum subsp. alpestre | Cistaceae | insect | zoochory | capsule | nude |
| Helictochloa praetutiana subsp. praetutiana | Poaceae | wind | meteochory | achene | awn |
| Iberis saxatilis subsp. saxatilis | Brassicaceae | insect | meteochory | siliqua | nude |
| Leontopodium nivale | Asteraceae | insect | meteochory | achene | pappus |
| Leucanthemum tridactylites | Asteraceae | insect | autochory | achene | nude |
| Linaria alpina | Scrophulariaceae | insect | autochory | capsule | nude |
| Luzula spicata subsp. italica | Juncaceae | wind | autochory | capsule | nude |
| Myosotis graui | Boraginaceae | insect | autochory | achene | nude |
| Omalotheca diminuta | Asteraceae | wind | meteochory | achene | pappus |
| Oreojuncus monanthos | Cyperaceae | wind | zoochory | capsule | nude |
| Oxytropis campestris | Fabaceae | insect | autochory | legume | nude |
| Papaver alpinum subsp. alpinum | Caryophyllaceae | insect | meteochory | capsule | nude |
| Paronychia kapela subsp. kapela | Papaveraceae | insect | autochory | achene | nude |
| Pedicularis elegans | Scrophulariaceae | insect | autochory | capsule | nude |
| Phyteuma orbiculare | Campanulaceae | insect | autochory | capsule | nude |
| Pilosella lactucella | Asteraceae | self | meteochory | achene | pappus |
| Plantago atrata subsp. atrata | Plantaginaceae | wind | zoochory | capsule | nude |
| Poa alpina subsp. alpina | Poaceae | wind | meteochory | achene | awn |
| Poa molinerii | Poaceae | wind | meteochory | achene | awn |
| Polygala alpestris subsp. alpestris | Polygalaceae | insect | meteochory | capsule | awn |
| Potentilla crantzii subsp. crantzii | Rosaceae | insect | autochory | achene | awn |
| Pulsatilla alpina subsp. alpina | Ranunculaceae | insect | meteochory | achene | awn |
| Ranunculus brevifolius | Ranunculaceae | insect | meteochory | achene | awn |
| Ranunculus breyninus | Ranunculaceae | insect | meteochory | achene | awn |
| Sabulina verna subsp. verna | Caryophyllaceae | wind | autochory | capsule | nude |
| Salix retusa | Salicaceae | wind | meteochory | capsule | awn |
| Saxifraga adscendens subsp. adscendens | Saxifragaceae | self | autochory | capsule | nude |
| Saxifraga exarata subsp. ampullacea | Saxifragaceae | self | autochory | capsule | nude |
| Saxifraga oppositifolia subsp. oppositifolia | Saxifragaceae | self | autochory | capsule | nude |
| Saxifraga paniculata | Saxifragaceae | self | autochory | capsule | nude |
| Saxifraga tridactylites | Saxifragaceae | self | autochory | capsule | nude |
| Scorzoneroides montana subsp. montana | Asteraceae | insect | meteochory | achene | pappus |
| Sedum atratum | Crassulaceae | insect | autochory | follicule | nude |
| Sempervivum arachnoideum | Crassulaceae | insect | autochory | follicule | nude |
| Senecio squalidus subsp. squalidus | Asteraceae | insect | meteochory | achene | pappus |
| Sesleria juncifolia subsp. juncifolia | Poaceae | wind | meteochory | achene | awn |
| Silene acaulis subsp. acaulis | Caryophyllaceae | insect | autochory | capsule | nude |
| Taraxacum apenninum | Asteraceae | insect | meteochory | achene | pappus |
| Thymus praecox subsp. polytrichus | Lamiaceae | insect | autochory | legume | nude |
| Trifolium pratense subsp. semipurpureum | Fabaceae | insect | zoochory | legume | nude |
| Trifolium thalii | Fabaceae | insect | meteochory | legume | nude |
| Trinia dalechampii | Apiaceae | insect | autochory | achene | nude |
| Valeriana saliunca | Valerianaceae | insect | meteochory | achene | pappus |
| Veronica aphylla | Scrophulariaceae | insect | autochory | capsule | nude |
| Viola eugeniae subsp. eugeniae | Violaceae | insect | zoochory | capsule | nude |
| Viola magellensis | Violaceae | insect | autochory | capsule | nude |
References
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; Da Fonseca, G.A.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Väre, H.; Lampinen, R.; Humphries, C.; Williams, P. Taxonomic diversity of vascular plants in the European alpine areas. In Alpine Biodiversity in Europe; Springer: Berlin, Heidelberg, Germany, 2003; pp. 133–148. [Google Scholar]
- Pauli, H.; Gottfried, M.; Grabherr, G. Effects of climate change on the alpine and nival vegetation of the Alps. J. Mt. Ecol. 2003, 7, 9–12. [Google Scholar]
- Barthlott, W.; Lauer, W.; Placke, A. Global distribution of species diversity in vascular plants: Towards a world map of phytodiversity (globale verteilung der artenvielfalt höherer pflanzen: Vorarbeiten zu einer weltkarte der phytodiversität). Erdkunde 1996, 50, 317–327. [Google Scholar] [CrossRef]
- Pauli, H.; Gottfried, M.; Reiter, K.; Klettner, C.; Grabherr, G. Signals of range expansions and contractions of vascular plants in the high Alps: Observations (1994–2004) at the Gloria master site schrankogel, Tyrol, Austria. Glob. Chang. Biol. 2007, 13, 147–156. [Google Scholar] [CrossRef]
- Kazakis, G.; Ghosn, D.; Vogiatzakis, I.; Papanastasis, V. Vascular plant diversity and climate change in the alpine zone of the Lefka Ori, Crete. Biodivers. Conserv. 2007, 16, 1603–1615. [Google Scholar] [CrossRef]
- Stanisci, A.; Carranza, M.L.; Pelino, G.; Chiarucci, A. Assessing the diversity pattern of cryophilous plant species in high elevation habitats. Plant Ecol. 2011, 212, 595–600. [Google Scholar] [CrossRef]
- Gutiérrez-Girón, A.; Gavilán, R. Plant functional strategies and environmental constraints in mediterranean high mountain grasslands in central Spain. Plant Ecol. Divers. 2013, 6, 435–446. [Google Scholar] [CrossRef]
- Nogués-Bravo, D.; Araújo, M.; Romdal, T.; Rahbek, C. Scale effects and human impact on the elevational species richness gradients. Nature 2008, 453, 216–219. [Google Scholar] [CrossRef] [PubMed]
- Pauli, H.; Gottfried, M.; Dullinger, S.; Abdaladze, O.; Akhalkatsi, M.; Alonso, J.L.B.; Coldea, G.; Dick, J.; Erschbamer, B.; Calzado, R.F.; et al. Recent plant diversity changes on Europe’s mountain summits. Science 2012, 336, 353–355. [Google Scholar] [CrossRef] [PubMed]
- Dullinger, S.; Gattringer, A.; Thuiller, W.; Moser, D.; Zimmerman, N.; Guisan, A. Climate warming, dispersal limitation and extinction debt of European mountain plants. Nat. Clim. Chang. 2012, 2, 619–622. [Google Scholar] [CrossRef]
- Becker, A.B. Predicting Global Change Impacts on Mountain Hydrology and Ecology: Integrated Catchment Hydrology/Altitudinal Gradient Studies Workshop Report: Documentation Resulting from an International Workshop, Kathmandu, Nepal 30 March–2 April 1996; International Geosphere Biosphere Programme: Stockholm, Swedish, 1997. [Google Scholar]
- Beniston, M. Mountain Environments in Changing Climates; Routledge: London, UK, 2002; ISBN 9780415102247. [Google Scholar]
- Chapin, F., III; Körner, C. Patterns, causes, changes, and consequences of biodiversity in arctic and alpine ecosystems. In Arctic and Alpine Biodiversity: Patterns, Causes and Ecosystem Consequences; Springer: Berlin, Heidelberg, Germany, 2013; Volume 113, pp. 313–320. [Google Scholar]
- Guisan, A.; Rahbek, C. Sesam—A new framework integrating macroecological and species distribution models for predicting spatio-temporal patterns of species assemblages. J. Biogeogr. 2011, 38, 1433–1444. [Google Scholar] [CrossRef]
- Byers, D.L.; Chang, S.M. Studying plant–pollinator interactions facing climate change and changing environments. Appl. Plant Sci. 2017, 5, 1700052. [Google Scholar] [CrossRef] [PubMed]
- Matteodo, M.; Wipf, S.; Rixen, C.; Vittoz, P. Elevation gradient of successful plant traits for colonizing alpine summits under climate change. Environ. Res. Lett. 2013, 8, 024043. [Google Scholar] [CrossRef]
- Pescador, D.S.; de Bello, F.; Valladares, F.; Escudero, A. Plant trait variation along an altitudinal gradient in mediterranean high mountain grasslands: Controlling the species turnover effect. PLoS ONE 2015, 10, e0118876. [Google Scholar] [CrossRef] [PubMed]
- Frate, L.; Carranza, M.L.; Evangelista, A.; Stinca, A.; Schaminée, J.H.; Stanisci, A. Climate and land use change impacts on mediterranean high-mountain vegetation in the Apennines since the 1950s. Plant Ecol. Divers. 2018, 1–13. [Google Scholar] [CrossRef]
- Kugler, H. Die verbreitung anemogamer arten in Europa. Berichte Deutschen Botanischen Gesellschaft 1975, 88, 441–450. [Google Scholar] [CrossRef]
- Vittoz, P.; Dussex, N.; Wassef, J.; Guisan, A. Diaspore traits discriminate good from weak colonisers on high-elevation summits. Basic Appl. Ecol. 2009, 10, 508–515. [Google Scholar] [CrossRef]
- Winkler, M.; Lamprecht, A.; Steinbauer, K.; Hülber, K.; Theurillat, J.P.; Breiner, F.; Choler, P.; Ertl, S.; Gutiérrez Girón, A.; Rossi, G.; et al. The rich sides of mountain summits—A pan-European view on aspect preferences of alpine plants. J. Biogeogr. 2016, 43, 2261–2273. [Google Scholar] [CrossRef]
- Jump, A.S.; Penuelas, J. Running to stand still: Adaptation and the response of plants to rapid climate change. Ecol. Lett. 2005, 8, 1010–1020. [Google Scholar] [CrossRef]
- Gentili, R.; Bacchetta, G.; Fenu, G.; Cogoni, D.; Abeli, T.; Rossi, G.; Salvatore, M.C.; Baroni, C.; Citterio, S. From cold to warm-stage refugia for boreo-alpine plants in southern European and Mediterranean mountains: The last chance to survive or an opportunity for speciation? Biodiversity 2015, 16, 247–261. [Google Scholar] [CrossRef]
- Pellissier, L.; Fournier, B.; Guisan, A.; Vittoz, P. Plant traits co-vary with altitude in grasslands and forests in the European Alps. Plant Ecol. 2010, 211, 351–365. [Google Scholar] [CrossRef]
- Weppler, T.; Stoll, P.; Stöcklin, J. The relative importance of sexual and clonal reproduction for population growth in the long-lived alpine plant Geum reptans. J. Ecol. 2006, 94, 869–879. [Google Scholar] [CrossRef]
- Kühn, I.; Bierman, S.M.; Durka, W.; Klotz, S. Relating geographical variation in pollination types to environmental and spatial factors using novel statistical methods. New Phytol. 2006, 172, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Körner, C. Alpine Plant Life: Functional Plant Ecology of High Mountain Ecosystems; With 47 Tables; Springer: Berlin, Heidelberg, Germany, 2003; ISBN 978-3-642-18970-8. [Google Scholar]
- Matteodo, M.; Ammann, K.; Verrecchia, E.P.; Vittoz, P. Snowbeds are more affected than other subalpine–alpine plant communities by climate change in the Swiss Alps. Ecol. Evolut. 2016, 6, 6969–6982. [Google Scholar] [CrossRef] [PubMed]
- Illa, E.; Carrillo, E.; Ninot, J.M. Patterns of plant traits in Pyrenean alpine vegetation. Flora Morphol. Distrib. Funct. Ecol. Plants 2006, 201, 528–546. [Google Scholar] [CrossRef]
- Ninot, J.M.; Grau, O.; Carrillo, E.; Guàrdia, R.; Lluent, A.; Illa, E. Functional plant traits and species assemblage in Pyrenean snowbeds. Folia Geobot. 2013, 48, 23–38. [Google Scholar] [CrossRef]
- Stanisci, A.; Pelino, G.; Blasi, C. Vascular plant diversity and climate change in the alpine belt of the central Apennines (Italy). Biodivers. Conserv. 2005, 14, 1301–1318. [Google Scholar] [CrossRef]
- Faegri, K.; Van Der Pijl, L. Principles of Pollination Ecology; Pergamon Press: Oxford, UK, 1979; ISBN 9781483293035. [Google Scholar]
- Kinlan, B.P.; Gaines, S.D. Propagule dispersal in marine and terrestrial environments: A community perspective. Ecology 2003, 84, 2007–2020. [Google Scholar] [CrossRef]
- Van Gils, H.; Conti, F.; Ciaschetti, G.; Westinga, E. Fine resolution distribution modelling of endemics in Majella National Park, central Italy. Plant Biosyst. 2012, 146, 276–287. [Google Scholar] [CrossRef]
- Giraudi, C. Middle to late holocene glacial variations, periglacial processes and alluvial sedimentation on the higher Apennine massifs (Italy). Quat. Res. 2005, 64, 176–184. [Google Scholar] [CrossRef]
- Pauli, H.; Gottfried, M.; Lamprecht, A.; Niessner, S.; Rumpf, S.; Winkler, M.; Steinbauer, K.; Grabherr, G. The GLORIA Field Manual–Standard Multi-Summit Approach, Supplementary Methods and Extra Approaches; Global Observation Research Initiative in Alpine Environment; GLORIA-Coordination, Austrian Academy of Sciences & University Of Natural Resources and Life Sciences: Vienna, Austria, 2015; Volume 5, ISBN 978-92-79-45694-7. [Google Scholar]
- Bartolucci, F.; Peruzzi, L.; Galasso, G.; Albano, A.; Alessandrini, A.; Ardenghi, N.; Astuti, G.; Bacchetta, G.; Ballelli, S.; Banfi, E. An updated checklist of the vascular flora native to Italy. Plant Biosyst. 2018, 152, 179–303. [Google Scholar] [CrossRef]
- Kleyer, M.; Bekker, R.; Knevel, I.; Bakker, J.; Thompson, K.; Sonnenschein, M.; Poschlod, P.; Van Groenendael, J.; Klimeš, L.; Klimešová, J. The LEDA traitbase: A database of life-history traits of the northwest European flora. J. Ecol. 2008, 96, 1266–1274. [Google Scholar] [CrossRef]
- Evangelista, A.; Frate, L.; Stinca, A.; Carranza, M.; Stanisci, A. VIOLA-the vegetation database of the central Apennines: Structure, current status and usefulness for monitoring Annex I EU habitats (92/43/EEC). Plant Sociol. 2016, 53, 47–58. [Google Scholar] [CrossRef]
- Pignatti, S. Flora D’Italia; Edagricole: Bologna, Italy, 1982; ISBN 8850624499. [Google Scholar]
- Pignatti, S. Valori di Bioindicazione Delle Piante Vascolari Della Flora D’Italia; Dipartimento di Botanica ed Ecologia dell’Università Camerino: Camerino, Italy, 2005. [Google Scholar]
- Landolt, E.; Bäumler, B.; Erhardt, A.; Hegg, O.; Klötzli, F.; Lämmler, W.; Nobis, M.; Rudmann-Maurer, K.; Schweingruber, F.; Theurillat, J. Flora Indicativa–Ecological Indicator Values and Biological Attributes of the Flora of Switzerland and the Alps. Haupt Verlag: Berne, Switzerlad, 2010; ISBN 978-3-258-07461-0. [Google Scholar]
- Gottfried, M.; Pauli, H.; Futschik, A.; Akhalkatsi, M.; Barančok, P.; Alonso, J.L.B.; Coldea, G.; Dick, J.; Erschbamer, B.; Kazakis, G.; et al. Continent-wide response of mountain vegetation to climate change. Nat. Clim. Chang. 2012, 2, 111–115. [Google Scholar] [CrossRef]
- Aeschimann, D.; Lauber, K.; Moser, D.M.; Theurillat, J.-P. Flora Alpina: Ein Atlas Sämtlicher 4500 Gefässpflanzen Der Alpen; Haupt Verlag: Berne, Switzerland, 2004; ISBN 978-8808071590. [Google Scholar]
- Klotz, S.; Kühn, I.; Durka, W.; Briemle, G. Biolflor: Eine Datenbank mit Biologisch-Ökologischen Merkmalen zur Flora von Deutschland; Bundesamt für naturschutz: Bonn, Germany, 2002; Volume 38, ISBN 978-3784335087. [Google Scholar]
- Oksanen, J.; Kindt, R.; Legendre, P.; O’Hara, B.; Stevens, M.H.H.; Oksanen, M.J.; Suggests, M. The vegan package. Community Ecol. Package 2007, 10, 631–637. [Google Scholar]
- Dray, S.; Choler, P.; Doledec, S.; Peres-Neto, P.R.; Thuiller, W.; Pavoine, S.; ter Braak, C.J.F. Combining the fourth-corner and the RLQ methods for assessing trait responses to environmental variation. Ecology 2014, 95, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.M.; Warton, D.I.; Andrew, N.R.; Binns, M.; Cassis, G.; Gibb, H. The fourth-corner solution–using predictive models to understand how species traits interact with the environment. Methods Ecol. Evolut. 2014, 5, 344–352. [Google Scholar] [CrossRef]
- Dolédec, S.; Chessel, D.; Ter Braak, C.; Champely, S. Matching species traits to environmental variables: A new three-table ordination method. Environ. Ecol. Stat. 1996, 3, 143–166. [Google Scholar] [CrossRef]
- Wang, Y.; Naumann, U.; Wright, S.T.; Warton, D.I. Mvabund—An R package for model-based analysis of multivariate abundance data. Methods Ecol. Evolut. 2012, 3, 471–474. [Google Scholar] [CrossRef]
- Hastie, T.; Tibshirani, R.; Friedman, J. Unsupervised learning. In The Elements of Statistical Learning; Springer: Berlin, Heidelberg, Germany, 2009; pp. 485–585. ISBN 978-0-387-84858-7. [Google Scholar]
- Sarkar, D. Lattice: Multivariate Data Visualization with R; Springer Science & Business Media: New York, NY, USA, 2008; ISBN 978-0-387-75969-2. [Google Scholar]
- Beniston, M. Climatic change in mountain regions: A review of possible impacts. Clim. Chang. 2003, 59, pp. 5–31. [CrossRef]
- Gottfried, M.; Pauli, H.; Reiter, K.; Grabherr, G. Potential effects of climate change on alpine and nival plants in the Alps. In Mountain Biodiversity, a Global Assessment; Parthenon: Boca Raton, FL, USA, 2002; pp. 213–223. ISBN 0-884736-02-5. [Google Scholar]
- Pauli, H.; Gottfried, M.; Reiter, K.; Grabherr, G. High mountain summits as sensitive indicators of climate change effects on vegetation patterns: The “multi summit-approach” of GLORIA (GLobal Observation Research Initiative in Alpine environments). In Global Change and Protected Areas; Advances in Global Change Research; Visconti, G., Beniston, M., Iannorelli, E.D., Barba, D., Eds.; Springer: Dordrecht, 2001; volume 9. [Google Scholar]
- Jiménez-Alfaro, B.; Marcenó, C.; Bueno, Á.; Gavilán, R.; Obeso, J.R. Biogeographic deconstruction of alpine plant communities along altitudinal and topographic gradients. J. Veg. Sci. 2014, 25, 160–171. [Google Scholar] [CrossRef]
- Choler, P.; Michalet, R.; Callaway, R.M. Facilitation and competition on gradients in alpine plant communities. Ecology 2001, 82, 3295–3308. [Google Scholar] [CrossRef]
- Mark, A.F.; Dickinson, K.J.; Allen, J.; Smith, R.; West, C.J. Vegetation patterns, plant distribution and life forms across the alpine zone in southern tierra del fuego, argentina. Austral Ecol. 2001, 26, 423–440. [Google Scholar] [CrossRef]
- Pauli, H.; Gottfried, M.; Grabherr, G. Vascular plant distribution patterns at the low-temperature limits of plant life-the alpine-nival ecotone of mount schrankogel (Tyrol, Austria). Phytocoenologia 1999, 29, 297–325. [Google Scholar] [CrossRef]
- Proctor, M.; Yeo, P.; Lack, A. The Natural History of Pollination; Harper Collins Publishers: London, UK, 1996; ISBN 000219905X. [Google Scholar]
- Arroyo, M.T.K.; Primack, R.; Armesto, J. Community studies in pollination ecology in the high temperate andes of central Chile I. Pollination mechanisms and altitudinal variation. Am. J. Bot. 1982, 69, 82–97. [Google Scholar] [CrossRef]
- Berry, P.E.; Calvo, R.N. Wind pollination, self-incompatibility, and altitudinal shifts in pollination systems in the high Andean genus Espeletia (Asteraceae). Am. J. Bot. 1989, 76, 1602–1614. [Google Scholar] [CrossRef]
- Bingham, R.A.; Orthner, A.R. Efficient pollination of alpine plants. Nature 1998, 391, 238–239. [Google Scholar] [CrossRef]
- Lawton, R.O. Wind stress and elfin stature in a montane rain forest tree: An adaptive explanation. Am. J. Bot. 1982, 69, 1224–1230. [Google Scholar] [CrossRef]
- Ridley, H. The Dispersal of Plants throughout the World; L. Reeve & Co. Ltd.: Ashford, Kent, UK, 1930; ISBN 20057003410. [Google Scholar]
- Collins, S.L.; Uno, G.E. Seed predation, seed dispersal, and disturbance in grasslands: A comment. Am. Nat. 1985, 125, 866–872. [Google Scholar] [CrossRef]
- Tackenberg, O.; Stöcklin, J. Wind dispersal of alpine plant species: A comparison with lowland species. J. Veg. Sci. 2008, 19, 109–118. [Google Scholar] [CrossRef]
- Brooker, R.W.; Callaghan, T.V. The balance between positive and negative plant interactions and its relationship to environmental gradients: A model. Oikos 1998, 81, 196–207. [Google Scholar] [CrossRef]
- Gentili, R.; Armiraglio, S.; Sgorbati, S.; Baroni, C. Geomorphological disturbance affects ecological driving forces and plant turnover along an altitudinal stress gradient on alpine slopes. Plant Ecol. 2013, 214, 571–586. [Google Scholar] [CrossRef]
- Willson, M.F.; Whelan, C.J. Variation in postdispersal survival of vertebrate-dispersed seeds: Effects of density, habitat, location, season, and species. Oikos 1990, 57, 191–198. [Google Scholar] [CrossRef]
- Almeida-Neto, M.; Campassi, F.; Galetti, M.; Jordano, P.; Oliveira-Filho, A. Vertebrate dispersal syndromes along the atlantic forest: Broad-scale patterns and macroecological correlates. Glob. Ecol. Biogeogr. 2008, 17, 503–513. [Google Scholar] [CrossRef]
- Van der Pijl, L. Principles of Dispersal; Springer: Berlin, Germany, 1982; ISBN 978-3-642-87925-8. [Google Scholar]
- Morgan, J.; Venn, S. Alpine plant species have limited capacity for long-distance seed dispersal. Plant Ecol. 2017, 218, 813–819. [Google Scholar] [CrossRef]
- Riibak, K.; Reitalu, T.; Tamme, R.; Helm, A.; Gerhold, P.; Znamenskiy, S.; Bengtsson, K.; Rosén, E.; Prentice, H.C.; Pärtel, M.; et al. Dark diversity in dry calcareous grasslands is determined by dispersal ability and stress-tolerance. Ecography 2015, 38, 713–721. [Google Scholar] [CrossRef]
- Navarro, T.; El Oualidi, J.; Taleb, M.S.; Pascual, V.; Cabezudo, B. Dispersal traits and dispersal patterns in an oro-mediterranean thorn cushion plant formation of the eastern high Atlas, Morocco. Flora Morphol. Distrib. Funct. Ecol. Plants 2009, 204, 658–672. [Google Scholar] [CrossRef]
- Wipf, S.; Stöckli, V.; Herz, K.; Rixen, C. The oldest monitoring site of the Alps revisited: Accelerated increase in plant species richness on Piz Linard summit since 1835. Plant Ecol. Divers. 2013, 6, 447–455. [Google Scholar] [CrossRef]
- Rogora, M.; Frate, L.; Carranza, M.; Freppaz, M.; Stanisci, A.; Bertani, I.; Bottarin, R.; Brambilla, A.; Canullo, R.; Carbognani, M.; et al. Assessment of climate change effects on mountain ecosystems through a cross-site analysis in the Alps and Apennines. Sci. Total Environ. 2018, 624, 1429–1442. [Google Scholar] [CrossRef] [PubMed]
- Rogora, M.; Mosello, R.; Arisci, S.; Brizzio, M.C.; Barbieri, A.; Balestrini, R.; Waldner, P.; Schmitt, M.; Stähli, M.; Thimonier, A.; et al. An overview of atmospheric deposition chemistry over the Alps: Present status and long-term trends. Hydrobiologia 2006, 562, 17–40. [Google Scholar] [CrossRef]
| Group of Traits | Traits—References | Trait States |
|---|---|---|
| Fruit type | Simplified from Landolt et al. [43] | Follicule Capsule Achene Legume Siliqua |
| Pollen vector | Simplified from Klotz et al. [46]; Aeschimann et al. [45] | Insect only Selfing Wind |
| Dispersal of diaspores | Simplified from Landolt et al. [43] | Zoochore Autochore Meteochore |
| Diaspore appendages | Klotz et al. [46]; Pignatti [41] | Awn (awn, hooks, calyx) Nude (no appendage) Pappus Wings (wings or fringes) |
| Species | Acronym | CCA1 | CCA2 | pv | dd | ft | sa |
|---|---|---|---|---|---|---|---|
| Crepis pygmaea | CRE_PYG | −2.47 | 0.97 | self | meteochore | achene | pappus |
| Alyssum cuneifolium | ALY_CUN | −2.08 | 0 | insect | autochore | siliqua | wings |
| Saxifraga exarata subsp. ampullacea | SAX_EXA_AMP | −1.77 | −0.65 | self | autochore | capsule | nude |
| Veronica aphylla | VER_APH | −1.1 | 1.26 | insect | meteochore | achene | pappus |
| Galium magellensis | GAL_MAG | −1 | 0.5 | wind | zoochore | achene | nude |
| Papaver alpinum subsp. alpinum | PAP_ALP_ALP | −0.84 | −1.13 | insect | meteochore | capsule | nude |
| Viola magellensis | VIO_MAG | −0.64 | −0.62 | insect | autochore | capsule | nude |
| Euphrasia minima subsp. minima | EUP_MIN_MIN | −0.31 | 1.26 | self | autochore | capsule | nude |
| Ranunculus brevifolius | RAN_BRE | 0 | −0.65 | insect | meteochore | achene | awn |
| Ranunculusbreyninus | RAN_BRY | 0.48 | 0.89 | insect | meteochore | achene | awn |
| Potentilla crantzii subsp. crantzii | POT_CRA_CRA | 0.55 | −0.51 | insect | autochore | achene | awn |
| Sesleria juncifolia subsp. juncifolia | SES_JUN_JUN | 0.63 | 0.72 | wind | meteochore | achene | awn |
| Helictochloa praetutiana subsp. praetutiana | HEL_PRE_PRE | 0.76 | 0.58 | wind | meteochore | achene | awn |
| Trifolium pratensis subsp. semipurpureum | TRI_PRA_SEM | 1.17 | −0.49 | insect | zoochore | legume | nude |
| Sempervivum arachnoideum | SEM_ARA | 1.19 | −0.15 | insect | autochore | follicule | nude |
| Oxytropis campestris | OXY_CAM | 1.23 | 0.36 | insect | autochore | legume | nude |
| Polygala alpestris subsp. alpestris | POL_ALP_ALP | 1.28 | −0.1 | insect | meteochore | capsule | awn |
| Saxifraga tridactylites | SAX_TRI | 1.28 | −0.17 | self | autochore | capsule | nude |
| Poa molinerii | POA_MOL | 1.3 | −0.02 | wind | meteochore | achene | awn |
| Group of Traits | Traits | N | N% | Plots | Plot% |
|---|---|---|---|---|---|
| Pollen vector | insect | 39 | 67.24 | 359 | 62.76 |
| self | 7 | 12.07 | 50 | 8.74 | |
| wind | 12 | 20.69 | 163 | 28.50 | |
| Dispersal of diaspores | meteochore | 24 | 41.38 | 199 | 34.79 |
| autochore | 28 | 48.28 | 309 | 54.02 | |
| zoochore | 6 | 10.34 | 64 | 11.19 | |
| Seed appendages | awn | 12 | 20.69 | 105 | 18.36 |
| nude | 35 | 60.34 | 384 | 67.13 | |
| pappus | 7 | 12.07 | 45 | 7.87 | |
| wings | 4 | 6.90 | 38 | 6.64 | |
| Fruit type | achene | 27 | 46.55 | 250 | 43.71 |
| capsule | 21 | 36.21 | 229 | 40.03 | |
| follicule | 2 | 3.45 | 15 | 2.62 | |
| legume | 5 | 8.62 | 42 | 7.34 | |
| siliqua | 3 | 5.17 | 36 | 6.29 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Musciano, M.; Carranza, M.L.; Frate, L.; Di Cecco, V.; Di Martino, L.; Frattaroli, A.R.; Stanisci, A. Distribution of Plant Species and Dispersal Traits along Environmental Gradients in Central Mediterranean Summits. Diversity 2018, 10, 58. https://doi.org/10.3390/d10030058
Di Musciano M, Carranza ML, Frate L, Di Cecco V, Di Martino L, Frattaroli AR, Stanisci A. Distribution of Plant Species and Dispersal Traits along Environmental Gradients in Central Mediterranean Summits. Diversity. 2018; 10(3):58. https://doi.org/10.3390/d10030058
Chicago/Turabian StyleDi Musciano, Michele, Maria Laura Carranza, Ludovico Frate, Valter Di Cecco, Luciano Di Martino, Anna Rita Frattaroli, and Angela Stanisci. 2018. "Distribution of Plant Species and Dispersal Traits along Environmental Gradients in Central Mediterranean Summits" Diversity 10, no. 3: 58. https://doi.org/10.3390/d10030058
APA StyleDi Musciano, M., Carranza, M. L., Frate, L., Di Cecco, V., Di Martino, L., Frattaroli, A. R., & Stanisci, A. (2018). Distribution of Plant Species and Dispersal Traits along Environmental Gradients in Central Mediterranean Summits. Diversity, 10(3), 58. https://doi.org/10.3390/d10030058







