Does Thinning Homogenous and Dense Regrowth Benefit Bats? Radio-Tracking, Ultrasonic Detection and Trapping
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Species
2.2. Study Area
2.3. Bat Activity
2.4. Radio-Tracking
2.5. Roost Tree Availability
2.6. Data Analyses
3. Results
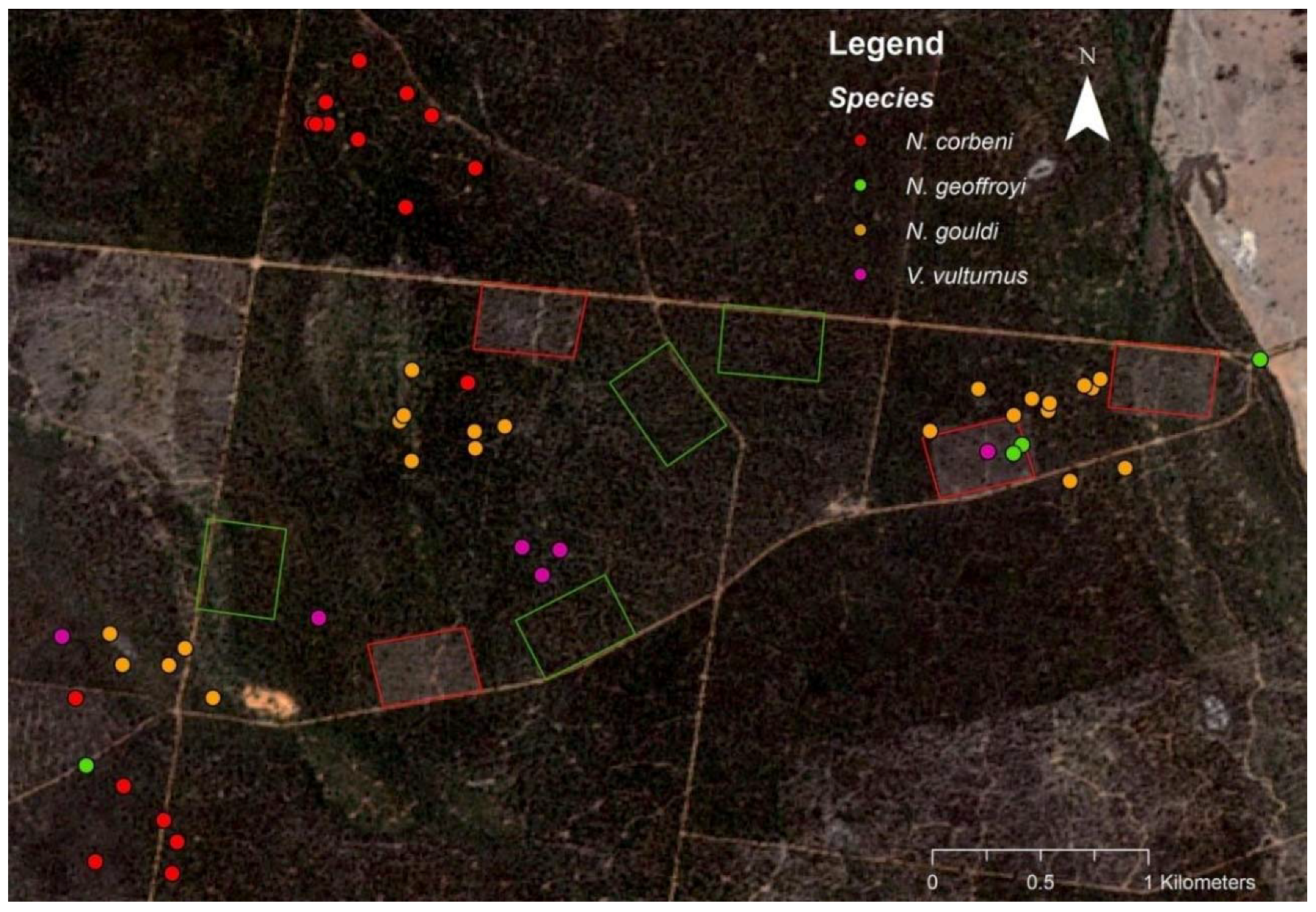
3.1. Roosting
3.2. Stem Density
3.3. Night Foraging
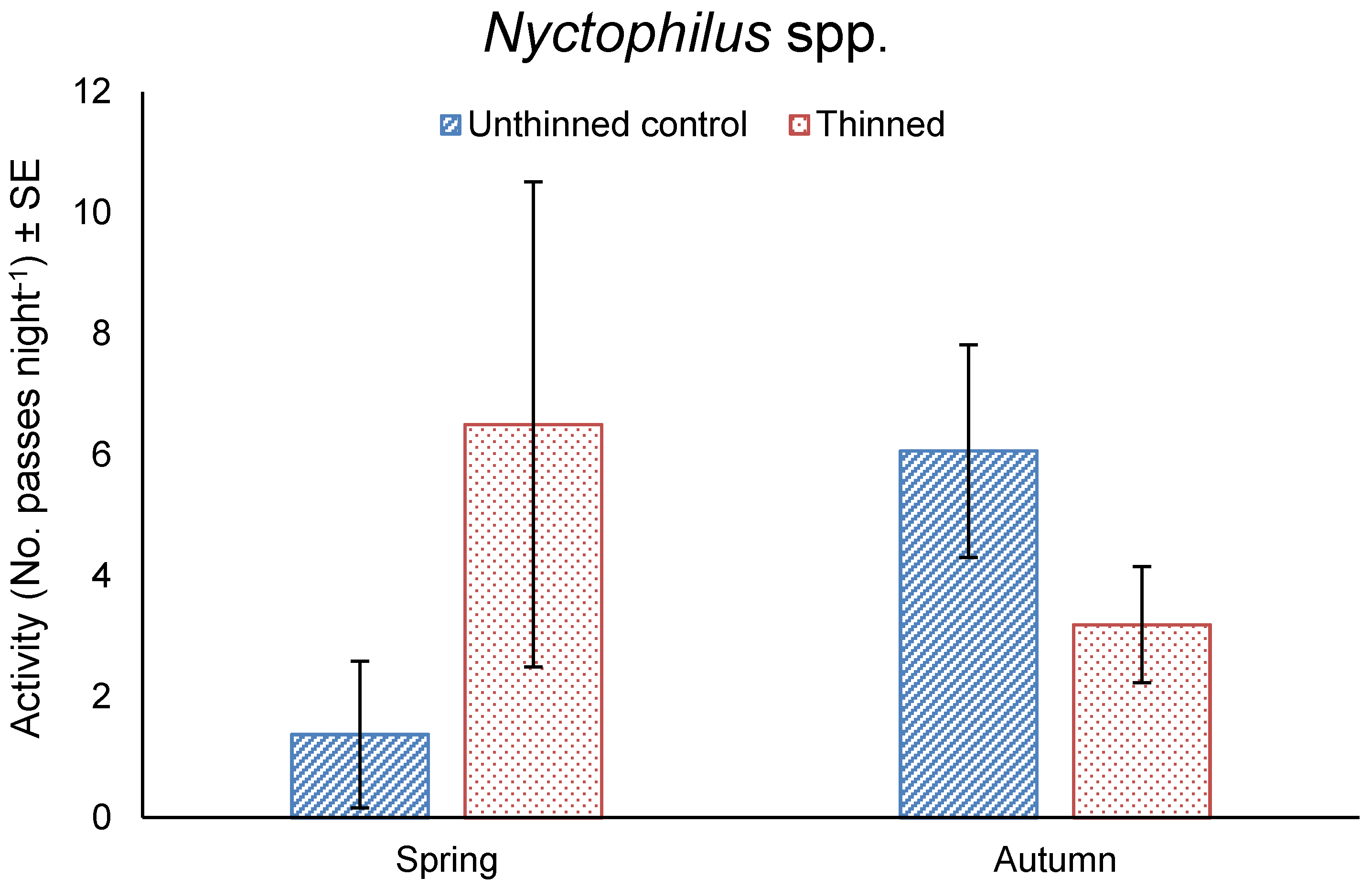
3.4. Bat Activity
3.5. Harp Trapping Captures
4. Discussion
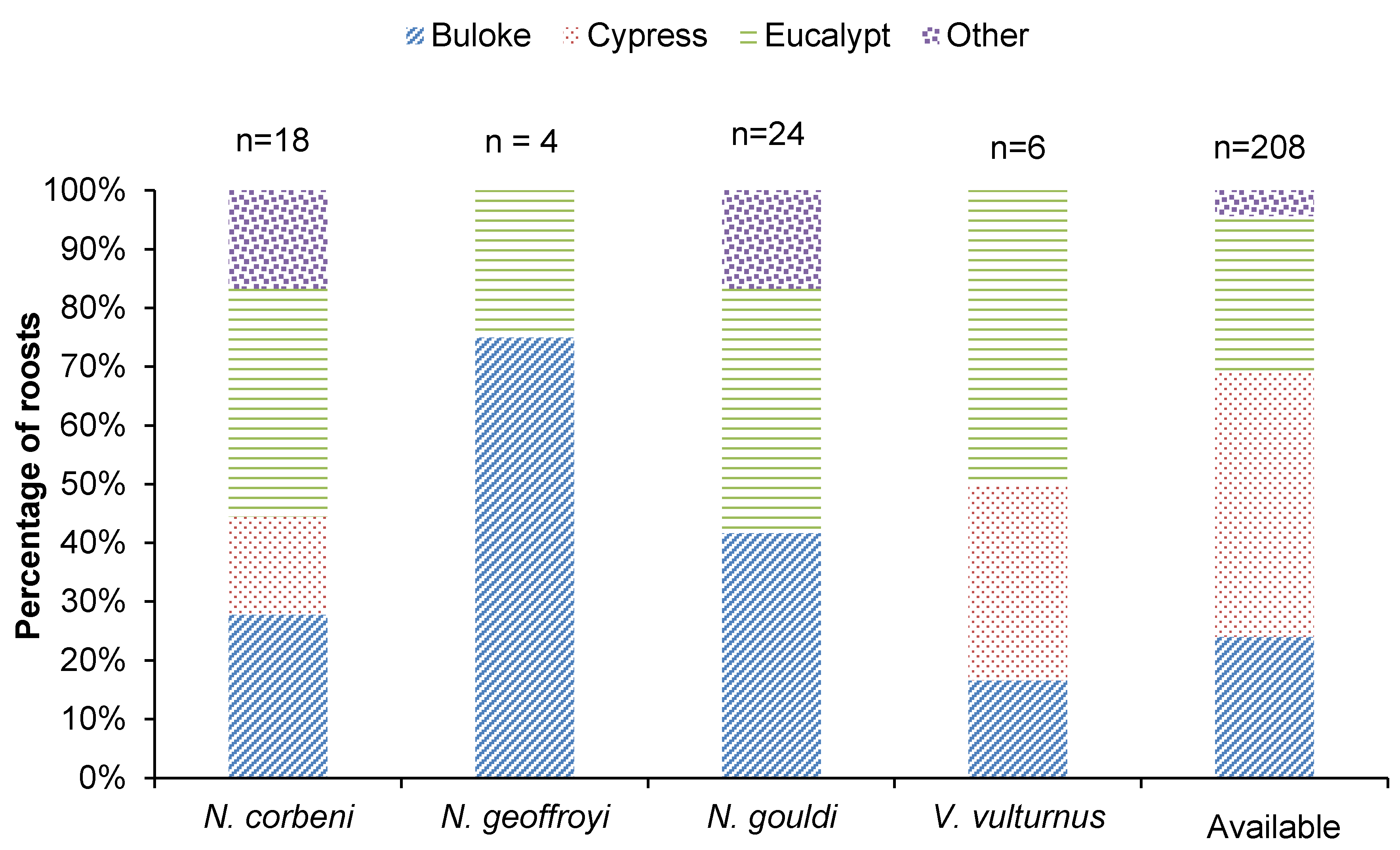
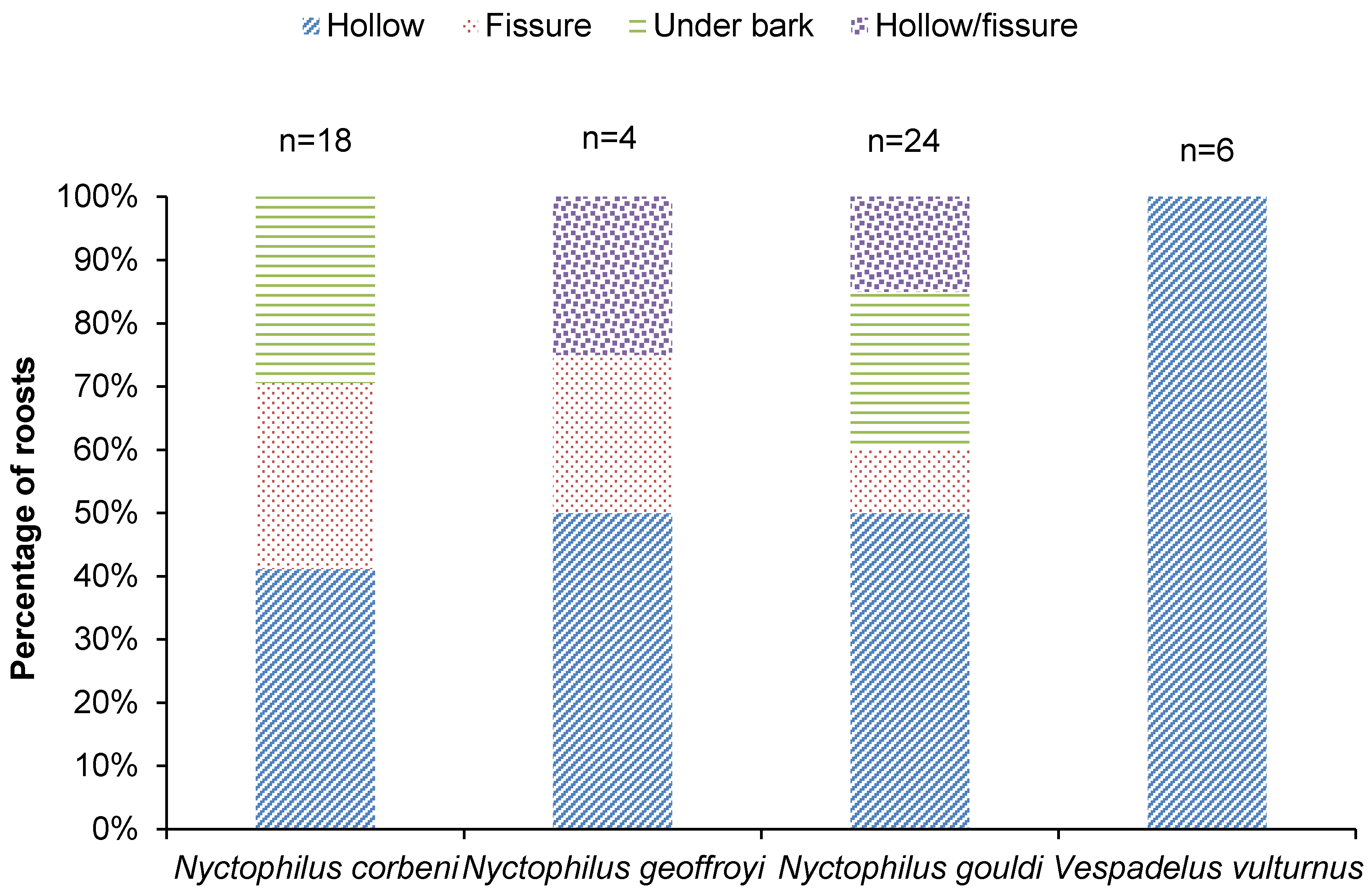
4.1. Roost Selection
4.2. Nocturnal Activity
4.3. Management Implications
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bowman, D.M.; Garnett, S.T.; Barlow, S.; Bekessy, S.A.; Bellairs, S.M.; Bishop, M.J.; Bradstock, R.A.; Jones, D.N.; Maxwell, S.L.; Pittock, J.; et al. Renewal ecology: Conservation for the Anthropocene. Restor. Ecol. 2017, 25, 674–680. [Google Scholar] [CrossRef]
- Montoya, D.; Rogers, L.; Memmott, J. Emerging perspectives in the restoration of biodiversity-based ecosystem services. TREE 2012, 27, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Beaudry, S.; Duchesne, L.C.; Côté, B. Short-term effects of three forestry practices on carabid assemblages in a jack pine forest. Can. J. For. Res. 1997, 27, 2065–2071. [Google Scholar] [CrossRef]
- Gustafsson, L.; Baker, S.C.; Bauhus, J.; Beese, W.J.; Brodie, A.; Kouki, J.; Lindenmayer, D.B.; Lõhmus, A.; Pastur, G.M.; Messier, C.; et al. Retention forestry to maintain multifunctional forests: A world perspective. BioScience 2012, 62, 633–645. [Google Scholar] [CrossRef]
- Law, B.S.; Park, K.J.; Lacki, M.J. Insectivorous bats and silviculture: Balancing timber production and bat conservation. In Bats in the Anthropocene: Conservation of Bats in a Changing World; Voigt, C.C., Kingston, T., Eds.; Springer: Cham, Switzerland, 2016; pp. 105–150. [Google Scholar]
- Bauhus, J.; Puettmann, K.; Messier, C. Silviculture for old-growth attributes. For. Ecol. Manag. 2009, 258, 525–537. [Google Scholar] [CrossRef]
- Verschuyl, J.; Riffell, S.; Miller, D.; Wigley, T.B. Biodiversity response to intensive biomass production from forest thinning in North American forests–a meta-analysis. For. Ecol. Manag. 2011, 261, 221–232. [Google Scholar] [CrossRef]
- Fuller, R.J. Searching for biodiversity gains through woodfuel and forest management. J. Appl. Ecol. 2013, 50, 1295–1300. [Google Scholar] [CrossRef]
- Eyre, T.J.; Ferguson, D.J.; Kennedy, M.; Rowland, J.; Maron, M. Long term thinning and logging in Australian cypress pine forest: Changes in habitat attributes and response of fauna. Biol. Conserv. 2015, 186, 83–96. [Google Scholar] [CrossRef]
- Blakey, R.V.; Law, B.S.; Kingsford, R.T.; Stoklosa, J.; Tap, P.; Williamson, K. Bat communities respond positively to large-scale thinning of forest regrowth. J. Appl. Ecol. 2016, 53, 1694–1703. [Google Scholar] [CrossRef]
- McKenzie, N.L.; Rolfe, J.K. Structure of bat guilds in the Kimberley mangroves, Australia. J. Anim. Ecol. 1986, 55, 401–420. [Google Scholar] [CrossRef]
- Aldridge, H.D.J.N.; Rautenbach, I.L. Morphology, echolocation and resource partitioning in insectivorous bats. J. Anim. Ecol. 1987, 56, 763–778. [Google Scholar] [CrossRef]
- Norberg, U.M.; Rayner, J.M. Ecological morphology and flight in bats (Mammalia; Chiroptera): Wing adaptations, flight performance, foraging strategy and echolocation. Phil. Trans. R. Soc. B 1987, 316, 335–427. [Google Scholar] [CrossRef]
- Crome, F.H.J.; Richards, G.C. Bats and gaps: Microchiropteran community structure in a Queensland rain forest. Ecology 1988, 69, 1960–1969. [Google Scholar] [CrossRef]
- Bullen, R.; McKenzie, N.L. Bat airframe design: Flight performance, stability and control in relation to foraging ecology. Aust. J. Zool. 2001, 49, 235–261. [Google Scholar] [CrossRef]
- Hanspach, J.; Fischer, J.; Ikin, K.; Stott, J.; Law, B.S. Using trait-based filtering as a predictive framework for conservation: A case study of bats on farms in southeastern Australia. J. Appl. Ecol. 2012, 49, 842–850. [Google Scholar] [CrossRef]
- Denzinger, A.; Schnitzler, H.U. Bat guilds, a concept to classify the highly diverse foraging and echolocation behaviors of microchiropteran bats. Front. Physiol. 2013, 4, 164. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.E.; Purvis, A.; Gittleman, J.L. Biological correlates of extinction risk in bats. Am. Nat. 2003, 161, 601–614. [Google Scholar] [CrossRef] [PubMed]
- Safi, K.; Kerth, G. A comparative analysis of specialization and extinction risk in temperate-zone bats. Cons. Biol. 2004, 18, 1293–1303. [Google Scholar] [CrossRef]
- Brigham, R.M.; Francis, R.L.; Hamdorf, S. Microhabitat use by two species of Nyctophilus bats: A test of ecomorphology theory. Aust. J. Zool. 1997, 45, 553–560. [Google Scholar] [CrossRef]
- Pavey, C.R.; Grunwald, J.E.; Neuweiler, G. Foraging habitat and echolocation behaviour of Schneider’s leafnosed bat, Hipposideros speoris, in a vegetation mosaic in Sri Lanka. Behav. Ecol. Sociobiol. 2001, 50, 209–218. [Google Scholar] [CrossRef]
- Humes, M.L.; Hayes, J.P.; Collopy, M.W. Bat activity in thinned, unthinned, and old-growth forests in western Oregon. J. Wildl. Manag. 1999, 63, 553–561. [Google Scholar] [CrossRef]
- Patriquin, K.J.; Barclay, R.M. Foraging by bats in cleared, thinned and unharvested boreal forest. J. Appl. Ecol. 2003, 40, 646–657. [Google Scholar] [CrossRef]
- Müller, J.; Mehr, M.; Bässler, C.; Fenton, M.B.; Hothorn, T.; Pretzsch, H.; Klemmt, H.J.; Brandl, R. Aggregative response in bats: Prey abundance versus habitat. Oecologia 2012, 169, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Montemayor, E.; Goulson, D.; Cavin, L.; Wallace, J.M.; Park, K.J. Fragmented woodlands in agricultural landscapes: The influence of woodland character and landscape context on bats and their insect prey. Agric. Ecosys. Environ. 2013, 172, 6–15. [Google Scholar] [CrossRef]
- Blakey, R.V.; Kingsford, R.T.; Law, B.S.; Stoklosa, J. Floodplain habitat is disproportionately important for bats in a large river basin. Biol. Conserv. 2017, 215, 1–10. [Google Scholar] [CrossRef]
- Kunz, T.H.; Lumsden, L. Ecology of cavity and foliage roosting bats. In Bat Ecology; Kunz, T.H., Fenton, M.B., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 3–89. [Google Scholar]
- Law, B.; Gonsalves, L.; Chidel, M.; Brassil, T. Subtle use of a disturbance mosaic by the south-eastern long-eared bat (Nyctophilus corbeni): An extinction-prone, narrow-space bat. Wildl. Res. 2016, 43, 153–168. [Google Scholar] [CrossRef]
- Perry, R.W.; Thill, R.E.; Carter, S.A. Sex-specific roost selection by adult red bats in a diverse forested landscape. For. Ecol. Manag. 2007, 253, 48–55. [Google Scholar] [CrossRef]
- Lacey, C.J. Factors Influencing Occurrence of Cypress Pine Regeneration in New South Wales; Technical Paper No. 21; Forestry Commission of New South Wales: Sydney, Australia, 1972.
- Lacey, C.J. Silvicultural Characteristics of White Cypress Pine; Research Note No. 26; Forestry Commision of New South Wales: Taree, Australia, 1973.
- Forestry Commission of New South Wales. Managing the State Forests—The Pilliga Management Area; Forestry Commission of NSW: Sydney, Australia, 1988.
- Thompson, W.A.; Eldridge, D.J. White cypress pine (Callitris glaucophylla): A review of its roles in landscape and ecological processes in eastern Australia. Aust. J. Bot. 2005, 53, 555–570. [Google Scholar] [CrossRef]
- Read, J. Recruitment characteristics of the white cypress pine (Callitris glaucophylla) in arid South Australia. Rangel. J. 1995, 17, 228–240. [Google Scholar] [CrossRef]
- Hill, D.A.; Greenaway, F. Effectiveness of an acoustic lure for surveying bats in British woodlands. Mamm. Rev. 2005, 35, 116–122. [Google Scholar] [CrossRef]
- Reinhold, L.; Law, B.; Ford, G.; Pennay, M. Key to the Bat Calls of South-East Queensland and North-East New South Wales; Forest Ecosystem Research and Assessment Technical Paper 2001-07; Department of Natural Resources and Mines: Queensland, Australia, 2001.
- Turbill, C.; Ellis, M. Distribution and abundance of the south-eastern form of the greater long-eared bat Nyctophilus timoriensis. Aust. Mammal. 2006, 28, 1–6. [Google Scholar] [CrossRef]
- Churchill, S. Australian Bats, 2nd ed.; Allen and Unwin: Sydney, Australia, 2008. [Google Scholar]
- Duncan, A.; Baker, G.B.; Montgomery, N. The Action Plan for Australian Bats; Environment Australia: Canberra, Australia, 1999.
- Campbell, S.; Lumsden, L.F.; Kirkwood, R.; Coulson, G. Day roost selection by female little forest bats (Vespadelus vulturnus) within remnant woodland on Phillip Island, Victoria. Wildl. Res. 2005, 32, 183–191. [Google Scholar] [CrossRef]
- Law, B.S.; Chidel, M.; Penman, T. Do young eucalypt plantations benefit bats in an intensive agricultural landscape? Wildl. Res. 2011, 38, 173–187. [Google Scholar] [CrossRef]
- Gonsalves, L.; Law, B.; Webb, C.; Monamy, V. Foraging ranges of insectivorous bats shift relative to changes in mosquito abundance. PLoS ONE 2013, 8, e64081. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, M.G.; Taylor, R.J. Observations on the flight patterns and foraging behavior of Tasmanian bats. Wildl. Res. 1986, 13, 427–432. [Google Scholar] [CrossRef]
- Rhodes, M.P. Assessment of sources of variance and patterns of overlap in microchiropteran wing morphology in southeast Queensland, Australia. Can. J. Zool. 2002, 80, 450–460. [Google Scholar] [CrossRef]
- Rolls, E. A Million Wild Acres: Two Hundred Years of Man and an Australian Forest; Thomas Nelson: Melbourne, Australia, 1981. [Google Scholar]
- van Kempen, E. A History of the Pilliga Cypress Pine Forests; State Forests of New South Wales: Pennant Hills, Australia, 1997.
- Forestry Commission of NSW. Management Plan for the Pilliga Management Area; Forestry Commission of New South Wales: Sydney, Australia, 1986. [Google Scholar]
- Binns, D.; Beckers, D. Floristic patterns in the Pilliga. In Perfumed Pineries: Environmental History of Australia’s Callitris Forests; Dargavel, J., Hart, D., Libbis, B., Eds.; Australian National University: Canberra, Australia, 2001; pp. 104–110. [Google Scholar]
- Whipp, R.K.; Lunt, I.D.; Spooner, P.G.; Bradstock, R.A. Changes in forest structure over 60 years: Tree densities continue to increase in the Pilliga forests, New South Wales, Australia. Aust. J. Bot. 2012, 60, 1–8. [Google Scholar] [CrossRef]
- Adams, M.D.; Law, B.S.; Gibson, M.S. Reliable automation of bat call identification for eastern New South Wales, Australia, using classification trees and AnaScheme software. Acta Chiropterol. 2010, 12, 231–245. [Google Scholar] [CrossRef]
- Tidemann, C.R.; Woodside, D.P. A collapsible bat-trap and a comparison of results obtained with the trap and with mist-nets. Wildl. Res. 1978, 5, 355–362. [Google Scholar] [CrossRef]
- Gibbons, P.; Lindenmayer, D.B.; Barry, S.C.; Tanton, M.T. Hollow formation in eucalypts from temperate forests in southeastern Australia. Pac. Conserv. Biol. 2000, 6, 218–228. [Google Scholar] [CrossRef]
- Pollard, J.H. On distance estimators of density in randomly distributed forests. Biometrics 1971, 27, 991–1002. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. PRIMER v6; PRIMER-E Ltd.: Plymouth, UK, 2001. [Google Scholar]
- Lunney, D.; Barker, J.; Priddel, D.; Oconnell, M. Roost Selection by Goulds Long-Eared Bat, Nyctophilus gouldi Tomes (Chiroptera, Vespertilionidae), in Logged Forest on the South Coast of New South-Wales. Wildl. Res. 1988, 15, 375–384. [Google Scholar] [CrossRef]
- Webala, P.W.; Craig, M.D.; Law, B.S.; Wayne, A.F.; Bradley, J.S. Roost site selection by southern forest bat Vespadelus regulus and Gould’s long-eared bat Nyctophilus gouldi in logged jarrah forests; south-western Australia. For. Ecol. Manag. 2010, 260, 1780–1790. [Google Scholar] [CrossRef]
- Threlfall, C.G.; Law, B.; Banks, P.B. Roost selection in suburban bushland by the urban sensitive bat Nyctophilus gouldi. J. Mamm. 2013, 94, 307–319. [Google Scholar] [CrossRef]
- Burgar, J.M.; Craig, M.D.; Stokes, V.L. The importance of mature forest as bat roosting habitat within a production landscape. For. Ecol. Manag. 2015, 356, 112–123. [Google Scholar] [CrossRef]
- Lumsden, L.F.; Bennett, A.F.; Silins, J.E. Selection of roost sites by the lesser long-eared bat (Nyctophilus geoffroyi) and Gould’s wattled bat (Chalinolobus gouldii) in south-eastern Australia. J. Zool. 2002, 257, 207–218. [Google Scholar] [CrossRef]
- Rueegger, N.; Law, B.; Goldingay, R. Interspecific differences and commonalities in maternity roosting by tree cavity-roosting bats over a maternity season in a timber production landscape. PLoS ONE 2018, 13, e0194429. [Google Scholar] [CrossRef] [PubMed]
- Waters, C.M.; Gonsalves, L.; Law, B.; Melville, G.; Toole, I.; Brassil, T.; Tap, P. The effect of thinning on structural attributes of a low rainfall forest in eastern Australia. For. Ecol. Manag. 2018, 409, 571–583. [Google Scholar] [CrossRef]
- Lacki, M.J.; Baker, M.D.; Johnson, J.S. Temporal dynamics of roost snags of long-legged myotis in the Pacific Northwest, USA. J. Wildl. Manag. 2012, 76, 1310–1316. [Google Scholar] [CrossRef]
- Fuller, N.C. Landscape Genetics and Sociobiology of Gould’s Long-Eared (Nyctophilus gouldi) and the Lesser Long-Eared Bat (N. geoffroyi) in Fragmented Populations of South-Eastern Australia. Ph.D Thesis, University of Adelaide, Adelaide, Australia, 2013. [Google Scholar]
- Lumsden, L.F.; Bennett, A.F.; Silins, J.E. Location of roosts of the lesser long-eared bat Nyctophilus geoffroyi and Gould’s wattled bat Chalinolobus gouldii in a fragmented landscape in south-eastern Australia. Biol. Conserv. 2002, 106, 237–249. [Google Scholar] [CrossRef]
- Lewis, S.E. Roost fidelity of bats: A review. J. Mammal. 1995, 76, 481–496. [Google Scholar] [CrossRef]
- Law, B.; Tap, P.; Chidel, M. Bat activity in ephemeral stream-beds in the Pilliga forests: Clarifying the importance of flyways and buffer widths in open forest and woodland. In The Biology and Conservation of Australasian Bats; Law, B., Eby, P., Lunney, D., Lumsden, L., Eds.; Royal Zoological Society of New South Wales: Sydney, Australia, 2011; pp. 308–321. [Google Scholar]
- Fenton, M.B. The foraging behaviour and ecology of animal-eating bats. Can. J. Zool. 1990, 68, 411–422. [Google Scholar] [CrossRef]
- Law, B.S.; Chidel, M. Tracks and riparian zones facilitate the use of Australian regrowth forest by insectivorous bats. J. Appl. Ecol. 2002, 39, 605–617. [Google Scholar] [CrossRef]
- Arlettaz, R. Habitat selection as a major resource partitioning mechanism between the two sympatric sibling bat species Myotis myotis and Myotis blythii. J. Anim. Ecol. 1999, 68, 460–471. [Google Scholar] [CrossRef]
- McConville, A.; Law, B.; Penman, T.; Mahony, M. Contrasting habitat use of morphologically similar bat species with differing conservation status in south-eastern Australia. Aust. Ecol. 2013, 39, 83–94. [Google Scholar] [CrossRef]
- Gonsalves, L.; Law, B.; Brassil, T.; Waters, C.; Toole, I.; Tap, P. Ecological outcomes for multiple taxa from silvicultural thinning of regrowth forest. For. Ecol. Manag. 2018, 425, 177–188. [Google Scholar] [CrossRef]
- Gonsalves, L.; Law, B.; Blakey, R. Experimental evaluation of the initial effects of large-scale thinning on structure and biodiversity of river red gum (Eucalyptus camaldulensis) forests. Wildl. Res. 2018, in press. [Google Scholar]
- Law, B.S.; Dickman, C.R. The use of habitat mosaics by terrestrial vertebrate fauna: Implications for conservation and management. Biodivers. Conserv. 1998, 7, 323–333. [Google Scholar] [CrossRef]
- Kavanagh, R.; Law, B.; Lemckert, F.; Stanton, M.; Chidel, M.; Brassil, T.; Towerton, A.; Penman, T. Conservation Value of Eucalypt Plantations Established for Wood Production and Multiple Environmental Benefits in Agricultural Landscapes; Final Report for NAP/NHT2 Eucalypt Plantations project. SLA 0013, R3 NAP; Industry & Investment NSW, Forest Science Centre: West Pennant Hills, Australia, 2010.
- Rueegger, N. Bat boxes—A review of their use and application, past, present and future. Acta Chiropterol. 2016, 18, 279–299. [Google Scholar] [CrossRef]



| Trapping/Roost Data | 2016 | 2017 |
|---|---|---|
| No. of bats trapped | 141 | 94 |
| No. of bats tracked | 12 | 9 |
| Species tracked | Nc (3), Nge (2), Ngo (4), Vv (3) | Nc (3), Nge (2), Ngo (2), Vv (2) |
| No. of roost trees | 21 | 31 |
| Roost tree DBHOB (cm) | 27.3 ± 1.9 | 31.6 ± 4.4 |
| Roost tree height (m) | 15.1 ± 1.4 | 11.9 ± 1.2 |
| Dead roost trees (% of all roost trees) | 76.2 | 93.1 |
| Average length of stay at roosts (days) | 1.3 ± 0.2 | 1.3 ± 0.2 |
| Species | Unthinned (n = 35 Trap Nights) | Thinned (n = 65 Trap Nights) |
|---|---|---|
| Chalinolobus gouldii | 7 | 12 |
| Chalinolobus picatus | 0 | 3 |
| Mormopterus petersi | 0 | 1 |
| M. planiceps | 1 | 2 |
| Nyctophilus corbeni | 3 | 8 |
| N. geoffroyi | 7 | 36 |
| N. gouldi | 43 | 23 |
| Saccolaimus flaviventris | 0 | 1 |
| Scotorepens balstoni | 0 | 5 |
| S. greyii | 3 | 27 |
| Scotorepens sp. | 0 | 6 |
| Vespadelus vulturnus | 7 | 42 |
| All bats | 71 | 166 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Law, B.; Gonsalves, L.; Brassil, T.; Hill, D. Does Thinning Homogenous and Dense Regrowth Benefit Bats? Radio-Tracking, Ultrasonic Detection and Trapping. Diversity 2018, 10, 45. https://doi.org/10.3390/d10020045
Law B, Gonsalves L, Brassil T, Hill D. Does Thinning Homogenous and Dense Regrowth Benefit Bats? Radio-Tracking, Ultrasonic Detection and Trapping. Diversity. 2018; 10(2):45. https://doi.org/10.3390/d10020045
Chicago/Turabian StyleLaw, Bradley, Leroy Gonsalves, Traecey Brassil, and David Hill. 2018. "Does Thinning Homogenous and Dense Regrowth Benefit Bats? Radio-Tracking, Ultrasonic Detection and Trapping" Diversity 10, no. 2: 45. https://doi.org/10.3390/d10020045
APA StyleLaw, B., Gonsalves, L., Brassil, T., & Hill, D. (2018). Does Thinning Homogenous and Dense Regrowth Benefit Bats? Radio-Tracking, Ultrasonic Detection and Trapping. Diversity, 10(2), 45. https://doi.org/10.3390/d10020045





