Cumulative Human Impacts on Coral Reefs: Assessing Risk and Management Implications for Brazilian Coral Reefs
Abstract
1. Introduction
2. Materials and Methods
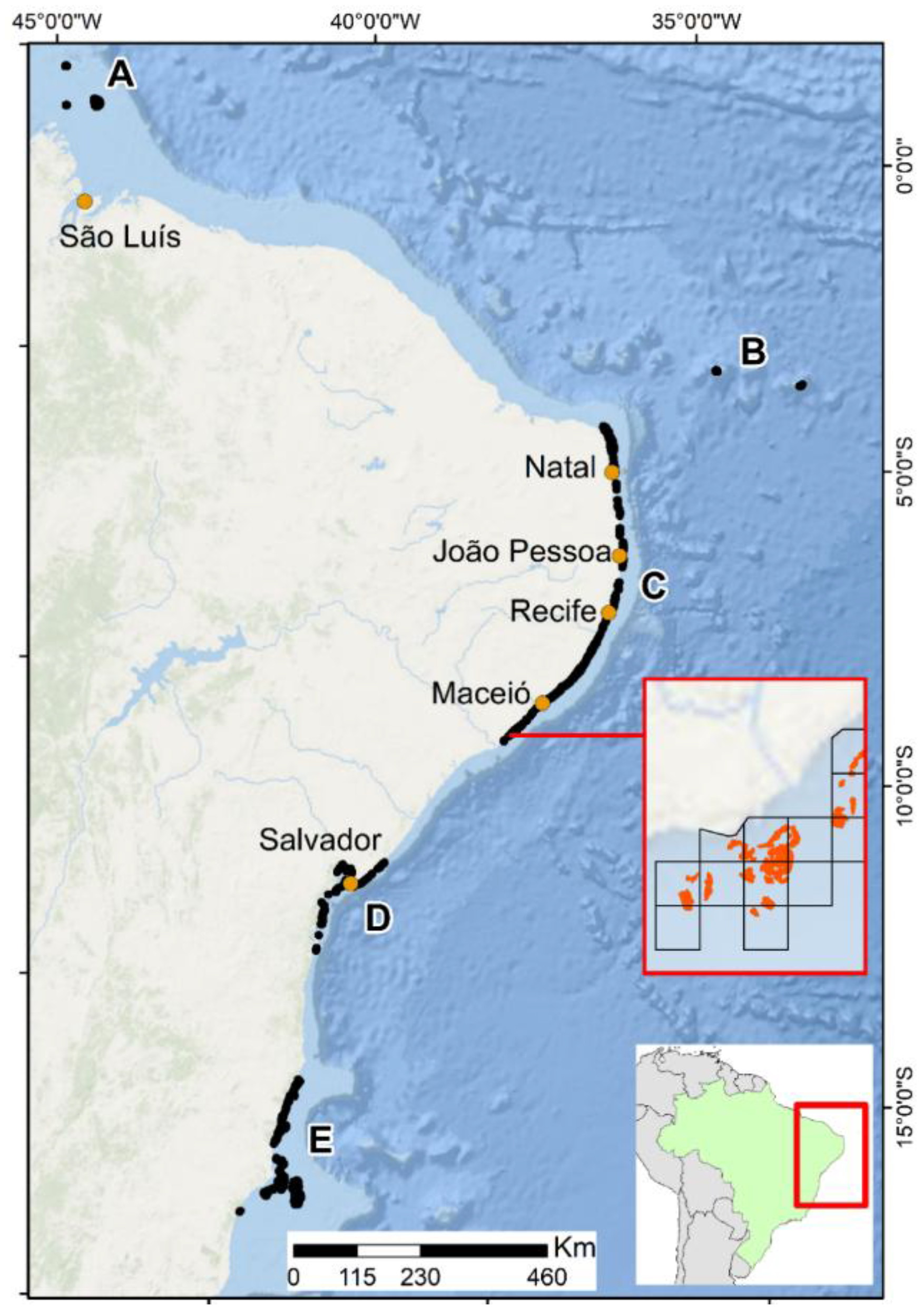
2.1. Mapping the Exposure to Multiple Stressors
2.2. Composite Index of the Cumulative Impact on Coral Reefs
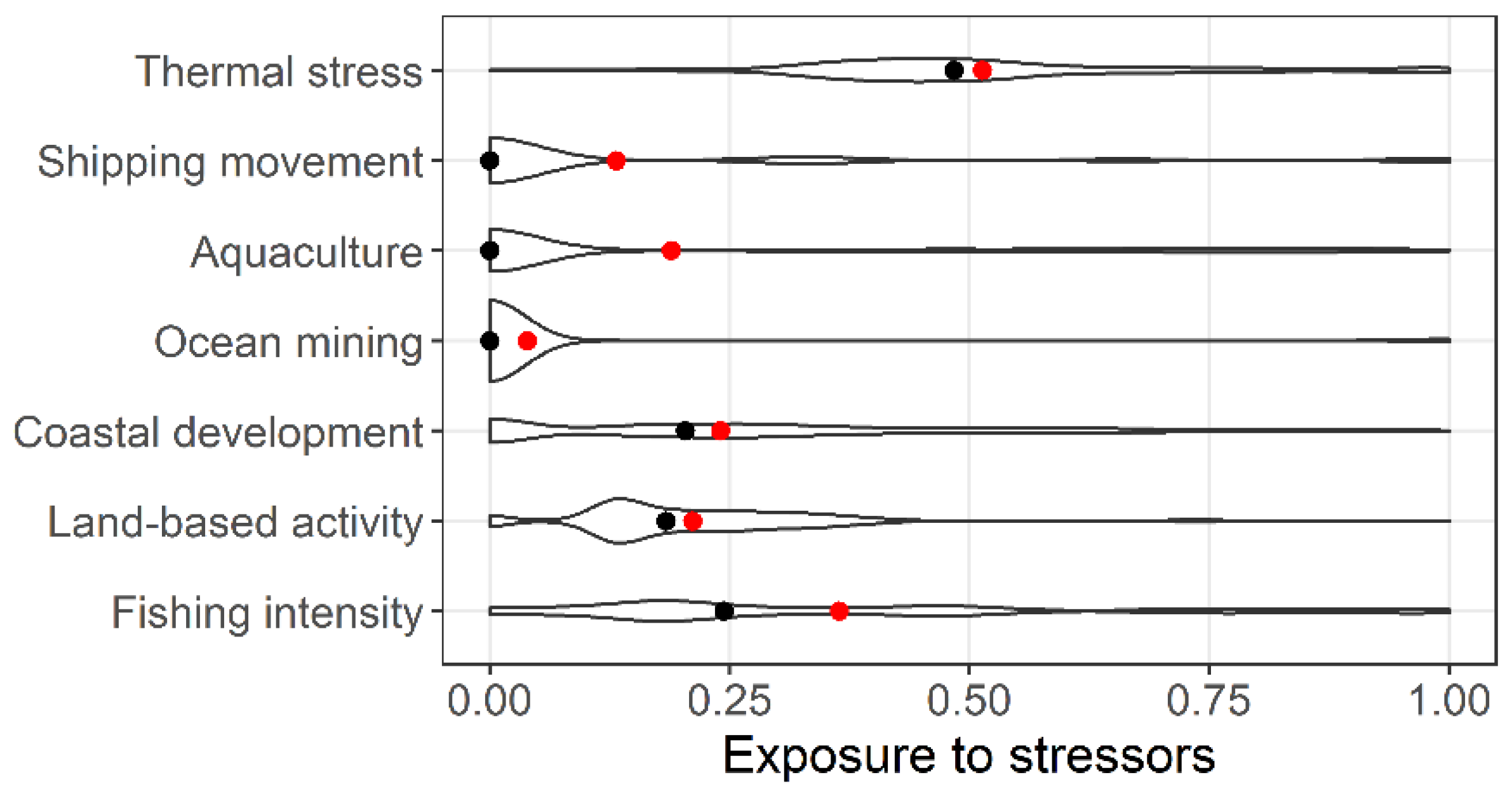
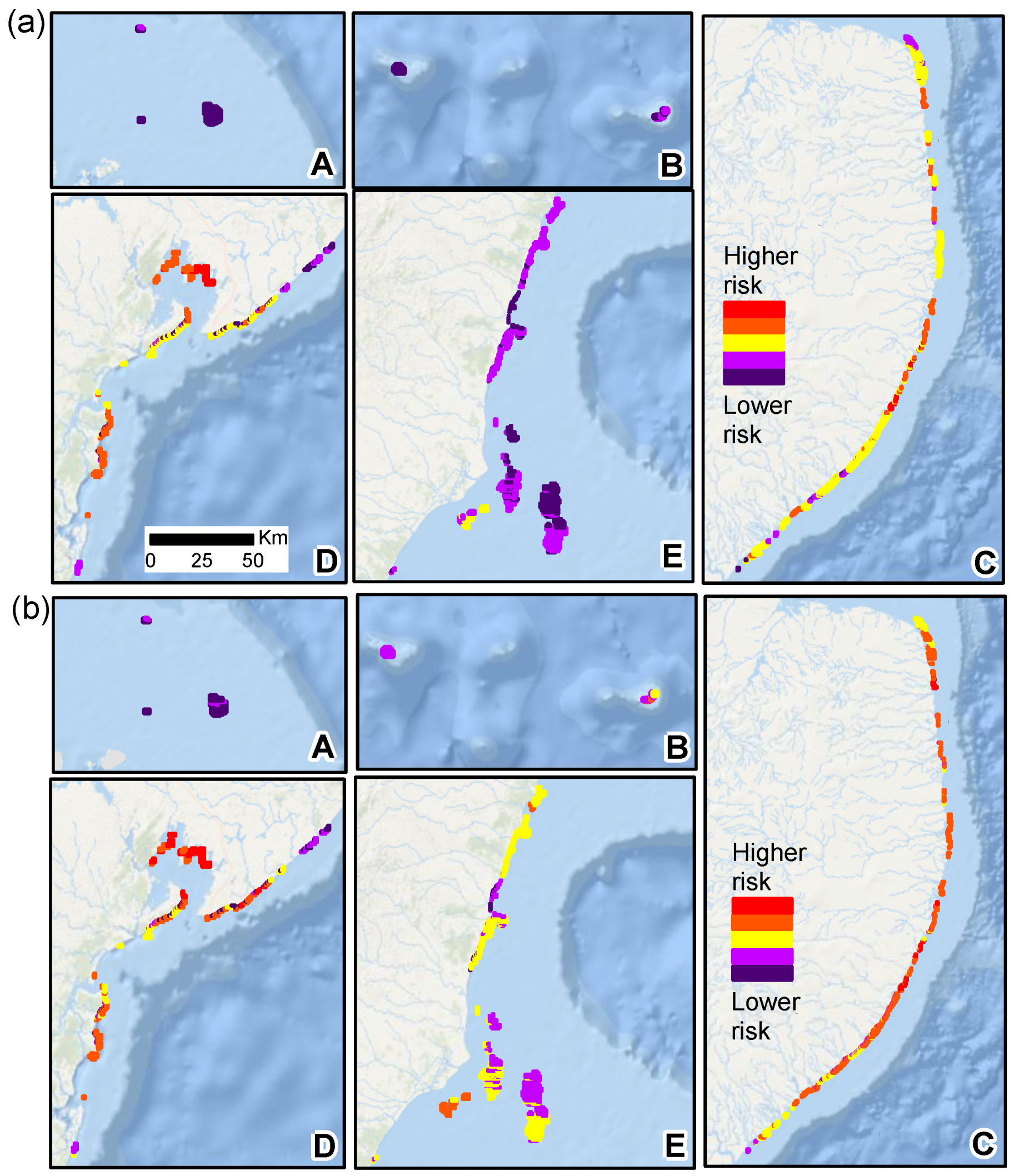
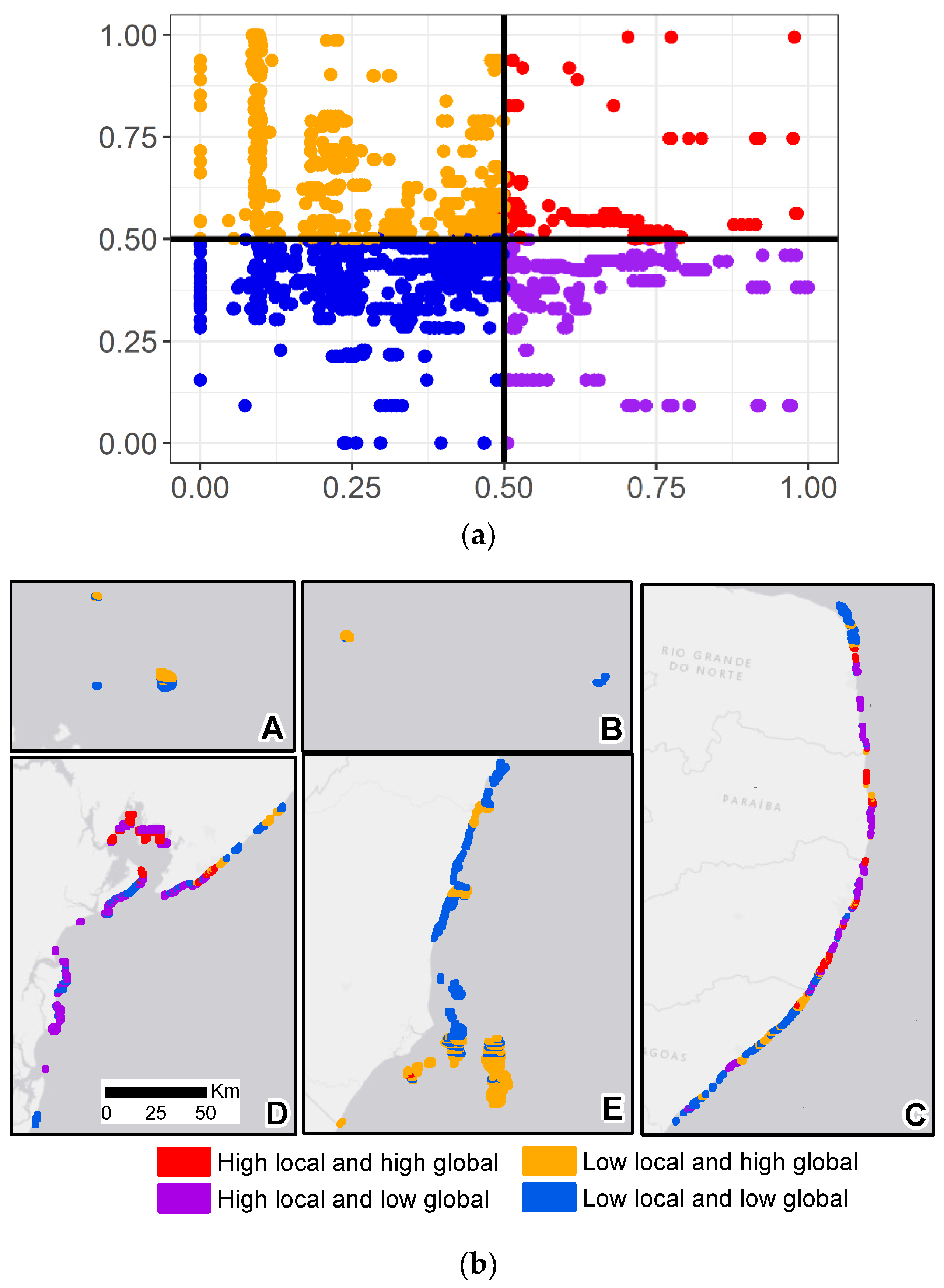
3. Results
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bellwood, D.; Hughes, T.; Folke, C.; Nyström, M. Confronting the coral reef crisis. Nature 2004, 429, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Hoegh-Guldberg, O.; Mumby, P.J.; Hooten, A.J.; Steneck, R.S.; Greenfield, P.; Gomez, E.; Harvell, C.D.; Sale, P.F.; Edwards, A.J.; Caldeira, K.; et al. Coral reefs under rapid climate change and ocean acidification. Science 2007, 318, 1737–1742. [Google Scholar] [CrossRef] [PubMed]
- Hughes, T.P.; Barnes, M.L.; Bellwood, D.R.; Cinner, J.E.; Cumming, G.S.; Jackson, J.B.C.; Kleypas, J.; van de Leemput, I.A.; Lough, J.M.; Morrison, T.H.; et al. Coral reefs in the Anthropocene. Nature 2017, 546, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Mora, C.; Andréfouët, S.; Costello, M.J.; Kranenburg, C.; Rollo, A.; Veron, J.; Gaston, K.J.; Myers, R.A. Coral reefs and the global network of marine protected areas. Science 2006, 312, 1750–1751. [Google Scholar] [CrossRef] [PubMed]
- Cinner, J.E.; Huchery, C.; MacNeil, M.A.; Graham, N.A.J.; McClanahan, T.R.; Maina, J.; Maire, E.; Kittinger, J.N.; Hicks, C.C.; Mora, C.; et al. Bright spots among the world’s coral reefs. Nature 2016, 535, 416–419. [Google Scholar] [CrossRef] [PubMed]
- Van Hooidonk, R.; Maynard, J.; Tamelander, J.; Gove, J.; Ahmadia, G.; Raymundo, L.; Williams, G.; Heron, S.F.; Planes, S. Local-scale projections of coral reef futures and implications of the Paris Agreement. Sci. Rep. 2016, 6, 39666. [Google Scholar] [CrossRef] [PubMed]
- Hughes, T.P.; Baird, A.H.; Bellwood, D.R.; Card, M.; Connolly, S.R.; Folke, C.; Grosberg, R.; Hoegh-Guldberg, O.; Jackson, J.; Kleypas, J. Climate change, human impacts, and the resilience of coral reefs. Science 2003, 301, 929–933. [Google Scholar] [CrossRef] [PubMed]
- Donner, S.D.; Potere, D. The inequity of the global threat to coral reefs. Bioscience 2007, 57, 214–215. [Google Scholar] [CrossRef]
- Pratchett, M.S.; Munday, P.L.; Wilson, S.K.; Graham, N.A.J.; Cinner, J.E.; Bellwood, D.R.; Jones, G.P.; Polunin, N.V.C.; McClanahan, T.R. Effects of climate-induced coral bleaching on coral-reef fishes—Ecological and economic consequences. Oceanogr. Mar. Biol. Annu. Rev. 2008, 46, 251–296. [Google Scholar]
- McClanahan, T.R.; Donner, S.D.; Maynard, J.A.; MacNeil, M.A.; Graham, N.A.; Maina, J.; Baker, A.C.; Beger, M.; Campbell, S.J.; Darling, E.S. Prioritizing key resilience indicators to support coral reef management in a changing climate. PLoS ONE 2012, 7, e42884. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.C.; Glynn, P.W.; Riegl, B. Climate change and coral reef bleaching: An ecological assessment of long-term impacts, recovery trends and future outlook. Estuar. Coast. Shelf Sci. 2008, 80, 435–471. [Google Scholar] [CrossRef]
- Makino, A.; Yamano, H.; Beger, M.; Klein, C.J.; Yara, Y.; Possingham, H.P. Spatio-temporal marine conservation planning to support high-latitude coral range expansion under climate change. Divers. Distrib. 2014, 20, 1–13. [Google Scholar] [CrossRef]
- Bruno, J.F.; Selig, E.R.; Casey, K.S.; Page, C.A.; Willis, B.L.; Harvell, C.D.; Sweatman, H.; Melendy, A.M. Thermal stress and coral cover as drivers of coral disease outbreaks. PLoS Biol. 2007, 5, e124. [Google Scholar] [CrossRef] [PubMed]
- Munday, P.; Leis, J.; Lough, J.; Paris, C.; Kingsford, M.; Berumen, M.; Lambrechts, J. Climate change and coral reef connectivity. Coral Reefs 2009, 28, 379–395. [Google Scholar] [CrossRef]
- Magris, R.A.; Pressey, R.L.; Weeks, R.; Ban, N.C. Integrating connectivity and climate change into marine conservation planning. Biol. Conserv. 2014, 170, 207–221. [Google Scholar] [CrossRef]
- Graham, N.A.; Chabanet, P.; Evans, R.D.; Jennings, S.; Letourneur, Y.; Aaron MacNeil, M.; McClanahan, T.R.; Öhman, M.C.; Polunin, N.V.; Wilson, S.K. Extinction vulnerability of coral reef fishes. Ecol. Lett. 2011, 14, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Darling, E.S.; McClanahan, T.R.; Côté, I.M. Combined effects of two stressors on kenyan coral reefs are additive or antagonistic, not synergistic. Conserv. Lett. 2010, 3, 122–130. [Google Scholar] [CrossRef]
- Humanes, A.; Ricardo, G.F.; Willis, B.L.; Fabricius, K.E.; Negri, A.P. Cumulative effects of suspended sediments, organic nutrients and temperature stress on early life history stages of the coral Acropora tenuis. Sci. Rep. 2017, 7, 44101. [Google Scholar] [CrossRef] [PubMed]
- Côté, I.M.; Darling, E.S.; Brown, C.J. Interactions among ecosystem stressors and their importance in conservation. Proc. R. Soc. B 2016, 283, 20152592. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.J.; Saunders, M.I.; Possingham, H.P.; Richardson, A.J. Interactions between global and local stressors of ecosystems determine management effectiveness in cumulative impact mapping. Divers. Distrib. 2014, 20, 538–546. [Google Scholar] [CrossRef]
- Brown, C.J.; Saunders, M.I.; Possingham, H.P.; Richardson, A.J. Managing for interactions between local and global stressors of ecosystems. PLoS ONE 2013, 8, e65765. [Google Scholar] [CrossRef] [PubMed]
- Halpern, B.S.; Walbridge, S.; Selkoe, K.A.; Kappel, C.V.; Micheli, F.; D’Agrosa, C.; Bruno, J.F.; Casey, K.S.; Ebert, C.; Fox, H.E.; et al. A global map of human impact on marine ecosystems. Science 2008, 319, 948. [Google Scholar] [CrossRef] [PubMed]
- Halpern, B.S.; Frazier, M.; Potapenko, J.; Casey, K.S.; Koenig, K.; Longo, C.; Lowndes, J.S.; Rockwood, R.C.; Selig, E.R.; Selkoe, K.A.; et al. Spatial and temporal changes in cumulative human impacts on the world/’s ocean. Nat. Commun. 2015, 6, 7615. [Google Scholar] [CrossRef] [PubMed]
- Loiola, M.; Oliveira, M.D.; Kikuchi, R.K. Tolerance of Brazilian brain coral Mussismilia braziliensis to sediment and organic matter inputs. Mar. Pollut. Bull. 2013, 77, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Magris, R.; Heron, S.F.; Pressey, R.L. Conservation planning for coral reefs accounting for climate warming disturbances. PLoS ONE 2015, 10, e0140828. [Google Scholar] [CrossRef] [PubMed]
- Tedesco, E.C.; Segal, B.; Calderon, E.N.; Schiavetti, A. Conservation of Brazilian coral reefs in the Southwest Atlantic Ocean: A change of approach. Lat. Am. J. Aquat. Res. 2017, 45, 228–245. [Google Scholar] [CrossRef]
- Bender, M.; Floeter, S.; Mayer, F.; Vila-Nova, D.; Longo, G.; Hanazaki, N.; Carvalho-Filho, A.; Ferreira, C. Biological attributes and major threats as predictors of the vulnerability of species: A case study with brazilian reef fishes. Oryx 2013, 47, 259–265. [Google Scholar] [CrossRef]
- Leão, Z.M.A.N.; Dominguez, J.M.L. Tropical coast of Brazil. Mar. Pollut. Bull. 2000, 41, 112–122. [Google Scholar] [CrossRef]
- Magris, R.A.; Pressey, R.L.; Mills, M.; Vila-Nova, D.A.; Floeter, S. Integrated conservation planning for coral reefs: Designing conservation zones for multiple conservation objectives in spatial prioritisation. Glob. Ecol. Conserv. 2017, 11, 53–68. [Google Scholar] [CrossRef]
- Heron, S.F.; Maynard, J.A.; Ruben van Hooidonk, C. Warming trends and bleaching stress of the world’s coral reefs 1985–2012. Sci. Rep. 2016, 6, 38402. [Google Scholar] [CrossRef] [PubMed]
- Magris, R.A.; Bond-Buckup, G.; Magalhães, C.; Mantelatto, F.L.; Reid, J.W.; Loureiro, L.M.A.E.-m.; Coelho, P.A.; Santana, W.R.A.; Buckup, L.; Rocha, S.S.d. Quantification of extinction risk for crustacean species: An overview of the national red listing process in Brazil. Nauplius 2010, 129–135. [Google Scholar]
- Bender, M.; Floeter, S.; Hanazaki, N. Do traditional fishers recognise reef fish species declines? Shifting environmental baselines in eastern Brazil. Fish. Manag. Ecol. 2013, 20, 58–67. [Google Scholar] [CrossRef]
- Giglio, V.J.; Luiz, O.J.; Schiavetti, A. Recreational diver behavior and contacts with benthic organisms in the Abrolhos national marine park, Brazil. Environ. Manag. 2016, 57, 637–648. [Google Scholar] [CrossRef] [PubMed]
- Mouillot, D.; Graham, N.A.; Villéger, S.; Mason, N.W.; Bellwood, D.R. A functional approach reveals community responses to disturbances. Trends Ecol. Evol. 2013, 28, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Spalding, M.D.; Brown, B.E. Warm-water coral reefs and climate change. Science 2015, 350, 769. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.; Pressey, R.L.; Newton, A.; Burgman, M.; Possingham, H.; Weston, C. Measuring and incorporating vulnerability into conservation planning. Environ. Manag. 2005, 35, 527–543. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Almany, G.; Russ, G.; Sale, P.; Steneck, R.; Van Oppen, M.; Willis, B. Larval retention and connectivity among populations of corals and reef fishes: History, advances and challenges. Coral Reefs 2009, 28, 307–325. [Google Scholar] [CrossRef]
- Magris, R.A.; Treml, E.A.; Pressey, R.L.; Weeks, R. Integrating multiple species connectivity and habitat quality into conservation planning for coral reefs. Ecography 2016, 39, 649–664. [Google Scholar] [CrossRef]
- Erftemeijer, P.L.; Riegl, B.; Hoeksema, B.W.; Todd, P.A. Environmental impacts of dredging and other sediment disturbances on corals: A review. Mar. Pollut. Bull. 2012, 64, 1737–1765. [Google Scholar] [CrossRef] [PubMed]
- Venter, O.; Sanderson, E.W.; Magrach, A.; Allan, J.R.; Beher, J.; Jones, K.R.; Possingham, H.P.; Laurance, W.F.; Wood, P.; Fekete, B.M. Sixteen years of change in the global terrestrial human footprint and implications for biodiversity conservation. Nat. Commun. 2016, 7, 12558. [Google Scholar] [CrossRef] [PubMed]
- Venter, O.; Sanderson, E.W.; Magrach, A.; Allan, J.R.; Beher, J.; Jones, K.R.; Possingham, H.P.; Laurance, W.F.; Wood, P.; Fekete, B.M. Global terrestrial human footprint maps for 1993 and 2009. Sci. Data 2016, 3, 160067. [Google Scholar] [CrossRef] [PubMed]
- Munday, P.L. Habitat loss, resource specialization, and extinction on coral reefs. Glob. Chang. Biol. 2004, 10, 1642–1647. [Google Scholar] [CrossRef]
- Fabricius, K.E. Effects of terrestrial runoff on the ecology of corals and coral reefs: Review and synthesis. Mar. Pollut. Bull. 2005, 50, 125–146. [Google Scholar] [CrossRef] [PubMed]
- Aubrecht, C.; Elvidge, C.; Longcore, T.; Rich, C.; Safran, J.; Strong, A.; Eakin, C.; Baugh, K.; Tuttle, B.; Howard, A. A global inventory of coral reef stressors based on satellite observed nighttime lights. Geocarto Int. 2008, 23, 467–479. [Google Scholar] [CrossRef]
- Pulfrich, A.; Branch, G.M. Effects of sediment discharge from namibian diamond mines on intertidal and subtidal rocky-reef communities and the rock lobster Jasus lalandii. Estuar. Coast. Shelf Sci. 2014, 150, 179–191. [Google Scholar] [CrossRef]
- Hatje, V.; de Souza, M.M.; Ribeiro, L.F.; Eca, G.F.; Barros, F. Detection of environmental impacts of shrimp farming through multiple lines of evidence. Environ. Pollut. 2016, 219, 672–684. [Google Scholar] [CrossRef] [PubMed]
- Magris, R.A.; Barreto, R. Mapping and assessment of protection of mangrove habitats in Brazil. Pan-Am. J. Aquat. Sci. 2010, 5, 546–556. [Google Scholar]
- Grech, A.; Bos, M.; Brodie, J.; Coles, R.; Dale, A.; Gilbert, R.; Hamann, M.; Marsh, H.; Neil, K.; Pressey, R. Guiding principles for the improved governance of port and shipping impacts in the Great Barrier Reef. Mar. Pollut. Bull. 2013, 75, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Rowlands, G.; Purkis, S.; Riegl, B.; Metsamaa, L.; Bruckner, A.; Renaud, P. Satellite imaging coral reef resilience at regional scale. A case-study from Saudi Arabia. Mar. Pollut. Bull. 2012, 64, 1222–1237. [Google Scholar] [CrossRef] [PubMed]
- Ban, N.; Alder, J. How wild is the ocean? Assessing the intensity of anthropogenic marine activities in British Columbia, Canada. Aquat. Conserv. 2008, 18, 55–85. [Google Scholar] [CrossRef]
- Halpern, B.S.; Kappel, C.V.; Selkoe, K.A.; Micheli, F.; Ebert, C.M.; Kontgis, C.; Crain, C.M.; Martone, R.G.; Shearer, C.; Teck, S.J. Mapping cumulative human impacts to California current marine ecosystems. Conserv. Lett. 2009, 2, 138–148. [Google Scholar] [CrossRef]
- Crain, C.M.; Kroeker, K.; Halpern, B.S. Interactive and cumulative effects of multiple human stressors in marine systems. Ecol. Lett. 2008, 11, 1304–1315. [Google Scholar] [CrossRef] [PubMed]
- Halpern, B.S.; Selkoe, K.A.; Micheli, F.; Kappel, C.V. Evaluating and ranking the vulnerability of global marine ecosystems to anthropogenic threats. Conserv. Biol. 2007, 21, 1301–1315. [Google Scholar] [CrossRef] [PubMed]
- Tulloch, V.J.; Tulloch, A.I.; Visconti, P.; Halpern, B.S.; Watson, J.E.; Evans, M.C.; Auerbach, N.A.; Barnes, M.; Beger, M.; Chadès, I. Why do we map threats? Linking threat mapping with actions to make better conservation decisions. Front. Ecol. Environ. 2015, 13, 91–99. [Google Scholar] [CrossRef]
- Leão, Z.; Kikuchi, R.; Oliveira, M.D.M.; Vasconcellos, V. Status of eastern Brazilian coral reefs in time of climate changes. Pan-Am. J. Aquat. Sci. 2010, 5, 224–235. [Google Scholar]
- Francini-Filho, R.B.; Moura, R.L.; Thompson, F.L.; Reis, R.M.; Kaufman, L.; Kikuchi, R.K.; Leão, Z.M. Diseases leading to accelerated decline of reef corals in the largest south atlantic reef complex (Abrolhos bank, eastern Brazil). Mar. Pollut. Bull. 2008, 56, 1008–1014. [Google Scholar] [CrossRef] [PubMed]
- Francini-Filho, R.; Moura, R.L. Evidence for spillover of reef fishes from a no-take marine reserve: An evaluation using the before-after control-impact (BACI) approach. Fish. Res. 2008, 93, 346–356. [Google Scholar] [CrossRef]
- Barreto, R.; Bornatowski, H.; Motta, F.; Santander-Neto, J.; Vianna, G.; Lessa, R. Rethinking use and trade of pelagic sharks from Brazil. Mar. Policy 2017, 85, 114–122. [Google Scholar] [CrossRef]
- Giglio, V.; Luiz, O.; Gerhardinger, L. Depletion of marine megafauna and shifting baselines among artisanal fishers in eastern Brazil. Anim. Conserv. 2015, 18, 348–358. [Google Scholar] [CrossRef]
- Selkoe, K.; Halpern, B.; Ebert, C.; Franklin, E.; Selig, E.; Casey, K.; Bruno, J.; Toonen, R. A map of human impacts to a “pristine” coral reef ecosystem, the Papahānaumokuākea marine national monument. Coral Reefs 2009, 28, 635–650. [Google Scholar] [CrossRef]
- Ban, N.C.; Alidina, H.M.; Ardron, J.A. Cumulative impact mapping: Advances, relevance and limitations to marine management and conservation, using Canada’s Pacific waters as a case study. Mar. Policy 2010, 34, 876–886. [Google Scholar] [CrossRef]
- Stelzenmüller, V.; Lee, J.; Garnacho, E.; Rogers, S. Assessment of a bayesian belief network–gis framework as a practical tool to support marine planning. Mar. Pollut. Bull. 2010, 60, 1743–1754. [Google Scholar] [CrossRef] [PubMed]
- Stelzenmüller, V.; Lee, J.; South, A.; Rogers, S.I. Quantifying cumulative impacts of human pressures on the marine environment: A geospatial modelling framework. Mar. Ecol. Prog. Ser. 2010, 398, 19–32. [Google Scholar] [CrossRef]
- Grech, A.; Coles, R.; Marsh, H. A broad-scale assessment of the risk to coastal seagrasses from cumulative threats. Mar. Policy 2011, 35, 560–567. [Google Scholar] [CrossRef]
- Halpern, B.S.; Fujita, R. Assumptions, challenges, and future directions in cumulative impact analysis. Ecosphere 2013, 4, 1–11. [Google Scholar] [CrossRef]
- Ban, S.S.; Pressey, R.L.; Graham, N.A. Assessing interactions of multiple stressors when data are limited: A bayesian belief network applied to coral reefs. Glob. Environ. Chang. 2014, 27, 64–72. [Google Scholar] [CrossRef]
- Teck, S.J.; Halpern, B.S.; Kappel, C.V.; Micheli, F.; Selkoe, K.A.; Crain, C.M.; Martone, R.; Shearer, C.; Arvai, J.; Fischhoff, B. Using expert judgment to estimate marine ecosystem vulnerability in the California current. Ecol. Appl. 2010, 20, 1402–1416. [Google Scholar] [CrossRef] [PubMed]
- Martin, T.G.; Burgman, M.A.; Fidler, F.; Kuhnert, P.M.; Low-Choy, S.; McBride, M.; Mengersen, K. Eliciting expert knowledge in conservation science. Conserv. Biol. 2012, 26, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Drescher, M.; Perera, A.; Johnson, C.; Buse, L.; Drew, C.; Burgman, M. Toward rigorous use of expert knowledge in ecological research. Ecosphere 2013, 4, 1–26. [Google Scholar] [CrossRef]
- Ban, S.S.; Graham, N.A.; Connolly, S.R. Evidence for multiple stressor interactions and effects on coral reefs. Glob. Chang. Biol. 2014, 20, 681–697. [Google Scholar] [CrossRef] [PubMed]
- Pressey, R.L.; Visconti, P.; Ferraro, P.J. Making parks make a difference: Poor alignment of policy, planning and management with protected-area impact, and ways forward. Phil. Trans. R. Soc. B 2015, 370, 20140280. [Google Scholar] [CrossRef] [PubMed]
- Magris, R.A.; Andrello, M.; Pressey, R.L.; Mouillot, D.; Dalongeville, A.; Jacobi, M.N.; Manel, S. Biologically representative and well-connected marine reserves enhance biodiversity persistence in conservation planning. Conserv. Lett. 2018, 11, e12439. [Google Scholar] [CrossRef]

| Stressor | Justification for Stressor | Source of Dataset | Scaling of Data | Data Model and Attributes |
|---|---|---|---|---|
| Fishing FII | The wholesale removal of reef fish species causes severe depletion of populations and impairs coral reef resilience [37] | Google Earth Pro and government reports described in [38] | Continuous | Linear decay model based on mapping the spatial location of 3610 traditional and 65 industrial active vessels |
| Land-based activities LAI | These activities are proven to increase erosion and deteriorate coastal water quality, changes that cause detrimental impacts on coral reef species [39] | Land-use data, infrastructure, and human access to natural areas in the terrestrial environment described in [40,41] and river plume data (Appendix A) | Continuous | Raster dataset containing the human footprint score derived from several variables (e.g., crop land, extent of built environments, and roads) measuring the direct and indirect human pressures on coastal catchments; Euclidean distance from each reef cell centroid to mouth of the nearest main river |
| Coastal develop-ment CDI | Coastal development results in sewage discharge, urbanisation, presence of engineering structures, and other general uses of the shore that can disrupt coral reef ecosystems [42,43] | DMSP/NOAA/NGDC nighttime satellite imagery as described in [38,44] | Continuous | Relative distance from each reef cell (centroid) to a source of night-time light (based on the metric LPI (light proximity index) |
| Ocean mining OMI | Physical structures for mineral extraction promote habitat loss. Indirect impacts include nutrient loading and sedimentation [45] | Government databases publicly available (Appendix A) | Categorical (presence or absence) | Vector data set of spatial locations of areas converted for mining (n = 70) as well as areas beyond their physical presence under indirect exposure |
| Aqua-culture ADI | An important source of organic and inorganic pollution, often associated with eutrophication of coastal waters [46] | Shrimp farm locations mapped by [47] | Continuous | Euclidean-distance-based model from the centroid of each farm pond (n = 115) that would potentially affect a given reef cell |
| Shipping move-ments SMI | Shipping-associated disturbances such as sediment resuspension and noise pollution lead to habitat degradation [48] | Shipping activity including domestic and international fleet and several vessel types made available by Marine Traffic (Appendix A) | Categorical (low, medium, high, and very high) | Density map classifying levels of ship movement based on historical data on ship positions |
| Thermal stress TSI | Sea-surface temperature is associated with coral bleaching and several other ecological shifts in coral reef ecosystems [2] | Sea surface temperature data obtained from NOAA and described by [25] | Continuous | Rate of warming using non-linear mixed effect models based on monthly climatology of sea surface temperature. |
| Stressor | Constants Used in the Formula Above | Weights |
|---|---|---|
| Fishing intensity | a | 2.5 |
| Land-based activity | b | 2.8 |
| Coastal development | c | 2.9 |
| Ocean mining | d | 0.8 |
| Aquaculture | e | 1.8 |
| Shipping movement | f | 1.5 |
| Thermal stress | g | 2.8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magris, R.A.; Grech, A.; Pressey, R.L. Cumulative Human Impacts on Coral Reefs: Assessing Risk and Management Implications for Brazilian Coral Reefs. Diversity 2018, 10, 26. https://doi.org/10.3390/d10020026
Magris RA, Grech A, Pressey RL. Cumulative Human Impacts on Coral Reefs: Assessing Risk and Management Implications for Brazilian Coral Reefs. Diversity. 2018; 10(2):26. https://doi.org/10.3390/d10020026
Chicago/Turabian StyleMagris, Rafael A., Alana Grech, and Robert L. Pressey. 2018. "Cumulative Human Impacts on Coral Reefs: Assessing Risk and Management Implications for Brazilian Coral Reefs" Diversity 10, no. 2: 26. https://doi.org/10.3390/d10020026
APA StyleMagris, R. A., Grech, A., & Pressey, R. L. (2018). Cumulative Human Impacts on Coral Reefs: Assessing Risk and Management Implications for Brazilian Coral Reefs. Diversity, 10(2), 26. https://doi.org/10.3390/d10020026





