(1RS,3SR)-1-(4-Methylbenzyl)-7-phenyl-5-oxa-6-azaspiro[2.4]hept-6-en-4-one
Abstract
1. Introduction
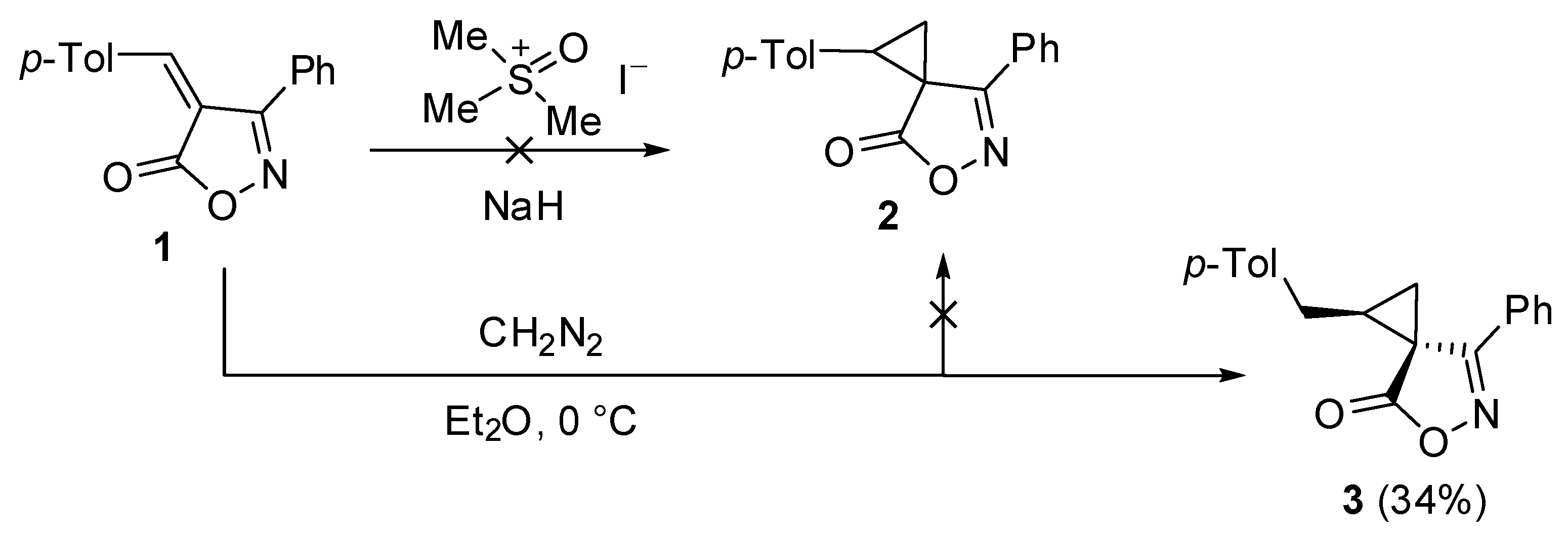
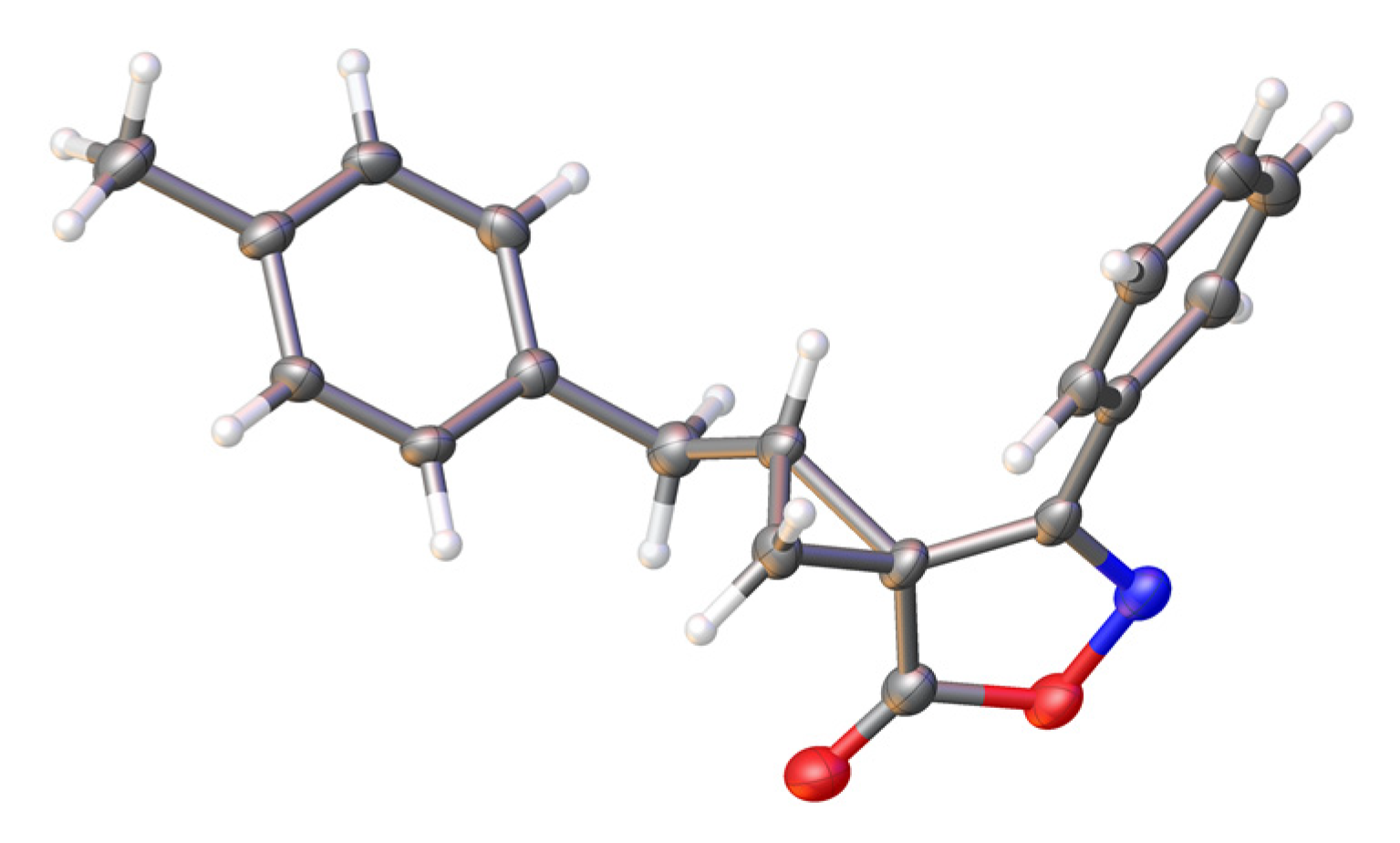
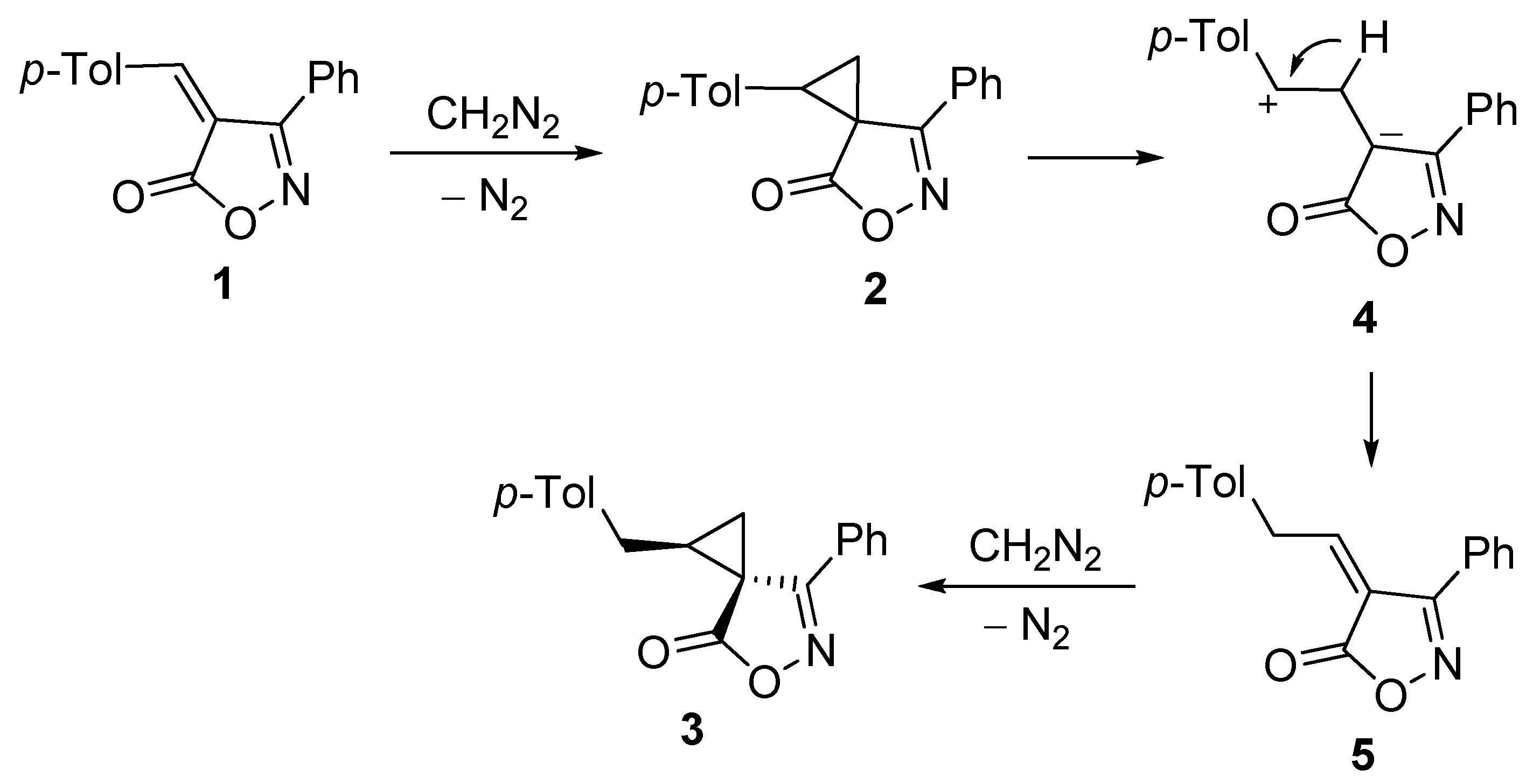
2. Results and Discussion
3. Materials and Methods
3.1. General Instrumentation
3.2. (1RS,3SR)-1-(4-Methylbenzyl)-7-phenyl-5-oxa-6-azaspiro[2.4]hept-6-en-4-one (3)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Grover, H.K.; Emmett, M.R.; Kerr, M.A. Carbocycles from donor-acceptor cyclopropanes. Org. Biomol. Chem. 2015, 13, 655–671. [Google Scholar] [CrossRef]
- Ivanova, O.A.; Trushkov, I.V. Donor–acceptor cyclopropanes in the synthesis of carbocycles. Chem. Rec. 2019, 19, 2189–2208. [Google Scholar] [CrossRef]
- Singh, P.; Varshnaya, R.K.; Dey, R.; Banerjee, P. Donor-Acceptor Cyclopropanes as an Expedient Building Block Towards the Construction of Nitrogen-Containing Molecules: An Update. Adv. Synth. Catal. 2020, 362, 1447–1484. [Google Scholar] [CrossRef]
- Schneider, T.F.; Kaschel, J.; Werz, D.B. A New Golden Age for Donor-Acceptor Cyclopropanes. Angew. Chem. Int. Ed. 2014, 53, 5504–5523. [Google Scholar] [CrossRef]
- Gharpure, S.J.; Nanda, L.N. Application of oxygen/nitrogen substituted donor-acceptor cyclopropanes in the total synthesis of natural products. Tetrahedron Lett. 2017, 58, 711–720. [Google Scholar] [CrossRef]
- Tomilov, Y.V.; Menchikov, L.G.; Novikov, R.A.; Ivanova, O.A.; Trushkov, I.V. Methods for the synthesis of donor-acceptor cyclopropanes. Russ. Chem. Rev. 2018, 87, 201–250. [Google Scholar] [CrossRef]
- Zaytsev, S.V.; Ivanov, K.L.; Skvortsov, D.A.; Bezzubov, S.I.; Melnikov, M.Y.; Budynina, E.M. Nucleophilic Ring Opening of Donor-Acceptor Cyclopropanes with the Cyanate Ion: Access to Spiro[pyrrolidone-3,3′-pxindoles]. J. Org. Chem. 2018, 83, 8695–8709. [Google Scholar] [CrossRef]
- Zhang, D.; Cheng, Q.; Chen, L.; Deng, H.; Cai, H.; Zhang, Q. Lewis-acid-catalyzed [3+2] annulation of oxindole based spirocyclic donor-acceptor cyclopropanes with ynamides. Org. Biomol. Chem. 2021, 19, 9645–9648. [Google Scholar] [CrossRef]
- Cao, Z.; Zhou, F.; Yu, Y.; Zhou, J. A Highly Diastereo- and Enantioselective Hg(II)-Catalyzed Cyclopropanation of Diazooxindoles and Alkenes. Org. Lett. 2013, 15, 42–45. [Google Scholar] [CrossRef]
- Mikhaylov, A.A.; Kuleshov, A.V.; Solyev, P.N.; Korlyukov, A.A.; Dorovatovskii, P.V.; Mineev, K.S.; Baranov, M.S. Imidazol-5-one as an Acceptor in Donor–Acceptor Cyclopropanes: Cycloaddition with Aldehydes. Org. Lett. 2020, 22, 2740–2745. [Google Scholar] [CrossRef]
- Arenal, I.; Bernabe, M.; Fernandez-Alvarez, E.; Penades, S. Synthesis of (E)- and (Z)-1-Amino-2-aryl(methyl)-cyclopropanecarboxylic Acid via Spirooxazolones. Synthesis 1985, 8, 773–775. [Google Scholar] [CrossRef]
- Mustafa, A.; Asker, W.; Harhash, A.H.; Fleifel, A.M. Reactivity of unsaturated centres in heterocycles and chalkones toward diazoalkanes. Tetrahedron 1965, 21, 2215–2229. [Google Scholar] [CrossRef]
- Croce, P.D. Heterocycles from ylides. Part V (1). Reactivity of 4-arylmethylene-Δ2-isoxazolin-5-ones with stabilized sulfur ylides. J. Heterocycl. Chem. 1976, 13, 1109–1110. [Google Scholar] [CrossRef]
- Vereshchagin, A.N.; Elinson, M.N.; Korshunov, A.D.; Anisina, Y.E.; Novikov, R.A.; Goloveshkin, A.S.; Bushmarinov, I.S.; Zlotin, S.G.; Egorov, M.P. Stereoselective Michael Halogenation Initiated Ring Closure (MHIRC) Synthesis of Spirocyclopropanes from Benzylidenemalononitriles and 3-Arylisoxazol-5(4H)-ones. Synlett 2016, 27, 2489–2493. [Google Scholar] [CrossRef]
- Kumar, P.; Kaur, N.; Kumar, R.; Banerjee, P. α,β-Unsaturated Carbonyls for One Pot Transition-Metal-Free Access to 3,6-Dihydro-2H-pyrans. J. Org. Chem. 2022, 87, 7167–7178. [Google Scholar] [CrossRef]
- Senatore, R.; Malik, M.; Langer, T.; Holzer, W.; Pace, V. Consecutive and Selective Double Methylene Insertion of Lithium Carbenoids to Isothiocyanates: A Direct Assembly of Four-Membered Sulfur-Containing Cycles. Angew. Chem. Int. Ed. 2021, 60, 24854–24858. [Google Scholar] [CrossRef]
- Butova, E.D.; Barabash, A.V.; Petrova, A.A.; Kleiner, C.M.; Schreiner, P.R.; Fokin, A.A. Stereospecific Consecutive Epoxide Ring Expansion with Dimethylsulfoxonium Methylide. J. Org. Chem. 2010, 75, 6229–6235. [Google Scholar] [CrossRef]
- Mukherjee, P.; Pettersson, M.; Dutra, J.K.; Xie, L.; Ende, C.W. Trifluoromethyl Oxetanes: Synthesis and Evaluation as a tert-Butyl Isostere. ChemMedChem 2017, 12, 1574–1577. [Google Scholar] [CrossRef]
- Vasilchenko, D.S.; Agafonova, A.V.; Simdianov, I.V.; Koronatov, A.N.; Sakharov, P.A.; Romanenko, I.A.; Rostovskii, N.V.; Khlebnikov, A.F.; Novikov, M.S. 2H-1,2,3-triazole-4-carboxylic acids via Ru(II)-catalyzed rearrangement of 4-hydrazonoisoxazol-5-ones. Tetrahedron Lett. 2023, 123, 154580. [Google Scholar] [CrossRef]
- Filippov, I.P.; Novikov, M.S.; Khlebnikov, A.F.; Rostovskii, N.V. One-Pot Synthesis of Multifunctionalized 1-Pyrrolines from 2-Alkyl-2H-azirines and Diazocarbonyl Compounds. J. Org. Chem. 2022, 87, 8835–8840. [Google Scholar] [CrossRef]
- Tiuftiakov, N.Y.; Strelnikova, J.O.; Filippov, I.P.; Khaidarov, A.R.; Khlebnikov, A.F.; Bunev, A.S.; Novikov, M.S.; Rostovskii, N.V. Rhodium-Catalyzed Synthesis of 2-Aroylpyrimidines via Cascade Heteropolyene Rearrangement. Org. Lett. 2021, 23, 6998–7002. [Google Scholar] [CrossRef]
- Titov, G.D.; Antonychev, G.I.; Novikov, M.S.; Khlebnikov, A.F.; Rogacheva, E.V.; Kraeva, L.A.; Rostovskii, N.V. Gold vs Light: Chemodivergent Reactivity of Diazoesters toward 2H-Azirine-2-carboxylic Acids. Org. Lett. 2023, 25, 2707–2712. [Google Scholar] [CrossRef]
- Dias-Jurberg, I.; Gagosz, F.; Zard, S.Z. Unusual Approach to Branched 3-Alkynylamides and to 1,5-Dihydropyrrol-2-ones. Org. Lett. 2010, 12, 416–419. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A Complete Structure Solution, Refinement and Analysis Program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Cryst. 2015, A71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Titov, G.D.; Rostovskii, N.V. (1RS,3SR)-1-(4-Methylbenzyl)-7-phenyl-5-oxa-6-azaspiro[2.4]hept-6-en-4-one. Molbank 2024, 2024, M1799. https://doi.org/10.3390/M1799
Titov GD, Rostovskii NV. (1RS,3SR)-1-(4-Methylbenzyl)-7-phenyl-5-oxa-6-azaspiro[2.4]hept-6-en-4-one. Molbank. 2024; 2024(2):M1799. https://doi.org/10.3390/M1799
Chicago/Turabian StyleTitov, Gleb D., and Nikolai V. Rostovskii. 2024. "(1RS,3SR)-1-(4-Methylbenzyl)-7-phenyl-5-oxa-6-azaspiro[2.4]hept-6-en-4-one" Molbank 2024, no. 2: M1799. https://doi.org/10.3390/M1799
APA StyleTitov, G. D., & Rostovskii, N. V. (2024). (1RS,3SR)-1-(4-Methylbenzyl)-7-phenyl-5-oxa-6-azaspiro[2.4]hept-6-en-4-one. Molbank, 2024(2), M1799. https://doi.org/10.3390/M1799





