5-(9-(p-Tolyl)-2,3,4,4a,9,9a-hexahydro-1H-1,4-methanocarbazol-6-yl)thiophene-2-carbaldehyde
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Prajapat, K.; Dhonde, M.; Sahu, K.; Bhojane, P.; Murty, V.; Shirage, P.M. The Evolution of Organic Materials for Efficient Dye-Sensitized Solar Cells. J. Photochem. Photobiol. C Photochem. Rev. 2023, 55, 100586. [Google Scholar] [CrossRef]
- Riede, M.; Spoltore, D.; Leo, K. Organic Solar Cells—The Path to Commercial Success. Adv. Energy Mater. 2021, 11, 2002653. [Google Scholar] [CrossRef]
- Sen, A.; Putra, M.H.; Biswas, A.K.; Behera, A.K.; Groβ, A. Insight on the Choice of Sensitizers/Dyes for Dye Sensitized Solar Cells: A Review. Dye. Pigment. 2023, 213, 111087. [Google Scholar] [CrossRef]
- Yahya, M.; Bouziani, A.; Ocak, C.; Seferoğlu, Z.; Sillanpää, M. Organic/Metal-Organic Photosensitizers for Dye-Sensitized Solar Cells (DSSC): Recent Developments, New Trends, and Future Perceptions. Dye. Pigment. 2021, 192, 109227. [Google Scholar] [CrossRef]
- Lee, C.-P.; Li, C.-T.; Ho, K.-C. Use of Organic Materials in Dye-Sensitized Solar Cells. Mater. Today 2017, 20, 267–283. [Google Scholar] [CrossRef]
- Knyazeva, E.A.; Rakitin, O.A. Influence of structural factors on the photovoltaic properties of dye-sensitized solar cells. Russ. Chem. Rev. 2016, 85, 1146–1183. [Google Scholar] [CrossRef]
- Duan, T.; Feng, W.; Li, Y.; Li, Z.; Zhang, Z.; Liang, H.; Chen, H.; Zhong, C.; Jeong, S.; Yang, C.; et al. Electronic Configuration Tuning of Centrally Extended Non-Fullerene Acceptors Enabling Organic Solar Cells with Efficiency Approaching 19%. Angew. Chemie Int. Ed. 2023, 62, e202308832. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Bi, X.; Chen, H.; He, T.; Lin, Y.; Zhang, Y.; Ma, K.; Feng, W.; Ma, Z.; Long, G.; et al. A Rare Case of Brominated Small Molecule Acceptors for High-Efficiency Organic Solar Cells. Nat. Commun. 2023, 14, 4707. [Google Scholar] [CrossRef] [PubMed]
- Wan, Z.; Yang, J.; Xia, J.; Shu, H.; Yao, X.; Luo, J.; Jia, C. Simple Molecular Structure but High Efficiency: Achieving over 9% Efficiency in Dye-Sensitized Solar Cells Using Simple Triphenylamine Sensitizer. J. Power Sources 2021, 506, 230214. [Google Scholar] [CrossRef]
- Singh, M.; Nadendla, S.; Kanaparthi, R.K. Unravelling the Effect of Donor-π-Acceptor Architecture in Designing 1,3-Indanedione Based Sensitizers for DSSC Applications. J. Photochem. Photobiol. A Chem. 2023, 435, 114328. [Google Scholar] [CrossRef]
- Sun, K.; Wang, L.; Mao, L.; Zhang, Y.; Liu, F.; Zhang, J. Influence of π Spacer of Donor-Acceptor-π-Acceptor Sensitizers on Photovoltaic Properties in Dye-Sensitized Solar Cells. Org. Electron. 2020, 76, 105429. [Google Scholar] [CrossRef]
- Marsya, M.A.; Hayati, D.; Han, S.; Long, D.X.; Choi, K.; Hong, J. UV-Harvesting Dyes Featuring a Fluorene Donor for Visibly Transparent and Colorless Dye-Sensitized Solar Cells. Dye. Pigment. 2022, 200, 110131. [Google Scholar] [CrossRef]
- Reddy-Marri, A.; Marchini, E.; Cabanes, V.D.; Argazzi, R.; Pastore, M.; Caramori, S.; Gros, P.C. Panchromatic Light Harvesting and Record Power Conversion Efficiency for Carboxylic/Cyanoacrylic Fe(ii) NHC Co-Sensitized FeSSCs. Chem. Sci. 2023, 14, 4288–4301. [Google Scholar] [CrossRef] [PubMed]
- Mikhailov, M.S.; Ustimenko, O.O.; Knyazeva, E.A.; Rakitin, O.A. 4,4-Bis(2-Ethylhexyl)-6-(9-(2-Ethylhexyl)-2,3,4,4a,9,9a-Hexahydro-1H-Carbazol-6-Yl)-4H-Cyclopenta[2,1-b:3,4-B′]Dithiophene-2-Carbaldehyde. Molbank 2022, 2022, M1486. [Google Scholar] [CrossRef]
- Gudim, N.S.; Mikhailov, M.S.; Knyazeva, E.A.; Almenningen, D.M.; Mikhalchenko, L.V.; Economopoulos, S.P.; Rakitin, O.A. Monitoring the Dependence of the Photovoltaic Properties of Dye-Sensitized Solar Cells from the Structure of D–A–π–A-Type Sensitizers with a 9-(p-Tolyl)-2,3,4,4a,9,9a-Hexahydro-1H-1,4-Methanocarbazole Donor Building Block. Mol. Syst. Des. Eng. 2022, 7, 755–766. [Google Scholar] [CrossRef]
- Chmovzh, T.N.; Kudryashev, T.A.; Alekhina, D.A.; Rakitin, O.A. Palladium-Catalyzed Direct (Het)Arylation Reactions of Benzo[1,2-d:4,5-D′]Bis([1,2,3]Thiadiazole and 4,8-Dibromobenzo[1,2-d:4,5-D′]Bis([1,2,3]Thiadiazole). Molecules 2023, 28, 3977. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Chu, Y.; Xu, M.; Zhang, S.; Ye, H.; Hu, Y.; Hua, J. Effect of π-Bridge Groups Based on Indeno[1,2- b ]Thiophene D–A–π–A Sensitizers on the Performance of Dye-Sensitized Solar Cells and Photocatalytic Hydrogen Evolution. J. Mater. Chem. C 2020, 8, 14864–14872. [Google Scholar] [CrossRef]
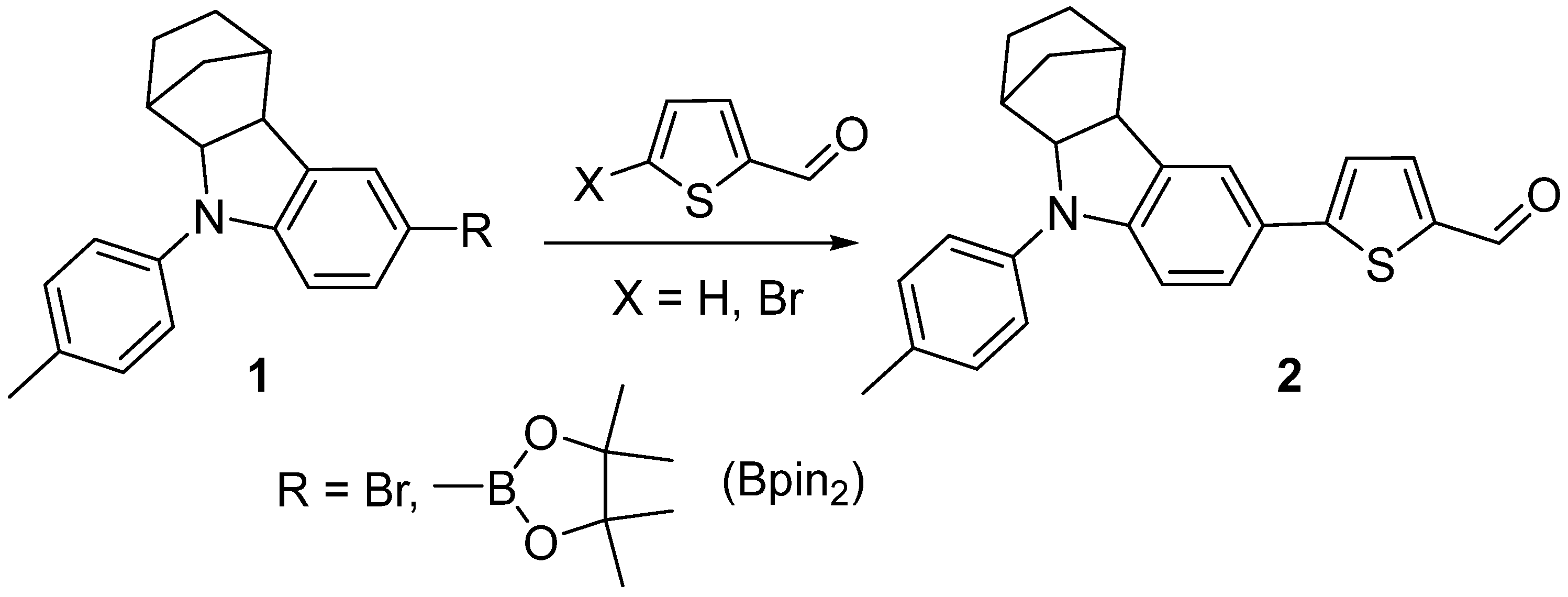
| Entry | R | X | Catalyst | Solvent | Base | Conditions | Yield of 2, % |
|---|---|---|---|---|---|---|---|
| 1 | Br | H | Pd(PPh3)2Cl2 | Toluene | K2CO3/PivOH | 110 °C, 16 h | 0 |
| 2 | Br | H | Pd(OAc)2 | Toluene | K2CO3/PivOH PtBu3·HBF4 | 110 °C, 16 h | 0 |
| 3 | Bpin2 | Br | Pd(PPh3)4 | THF/H2O | K2CO3/PivOH | 66 °C, 8 h | 63 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gudim, N.S.; Knyazeva, E.A.; Rakitin, O.A. 5-(9-(p-Tolyl)-2,3,4,4a,9,9a-hexahydro-1H-1,4-methanocarbazol-6-yl)thiophene-2-carbaldehyde. Molbank 2024, 2024, M1792. https://doi.org/10.3390/M1792
Gudim NS, Knyazeva EA, Rakitin OA. 5-(9-(p-Tolyl)-2,3,4,4a,9,9a-hexahydro-1H-1,4-methanocarbazol-6-yl)thiophene-2-carbaldehyde. Molbank. 2024; 2024(1):M1792. https://doi.org/10.3390/M1792
Chicago/Turabian StyleGudim, Nikita S., Ekaterina A. Knyazeva, and Oleg A. Rakitin. 2024. "5-(9-(p-Tolyl)-2,3,4,4a,9,9a-hexahydro-1H-1,4-methanocarbazol-6-yl)thiophene-2-carbaldehyde" Molbank 2024, no. 1: M1792. https://doi.org/10.3390/M1792
APA StyleGudim, N. S., Knyazeva, E. A., & Rakitin, O. A. (2024). 5-(9-(p-Tolyl)-2,3,4,4a,9,9a-hexahydro-1H-1,4-methanocarbazol-6-yl)thiophene-2-carbaldehyde. Molbank, 2024(1), M1792. https://doi.org/10.3390/M1792







