Synthesis of 3-Bromo-4-phenylisothiazole-5-carboxylic Acid and 3-Bromoisothiazole-5-carboxylic Acid
Abstract
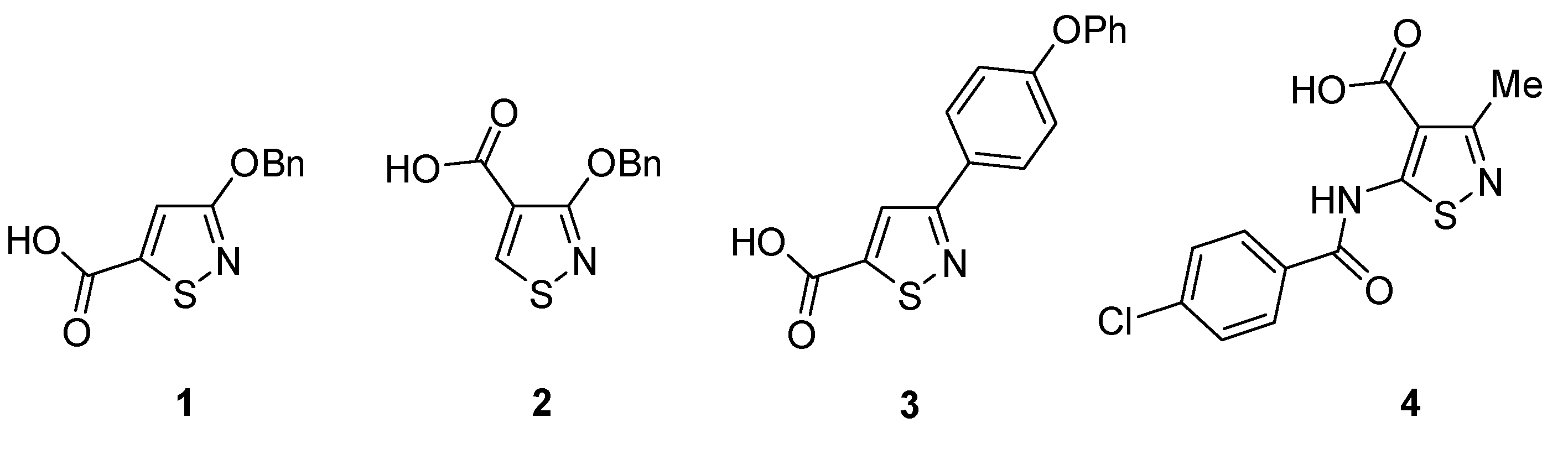
1. Introduction
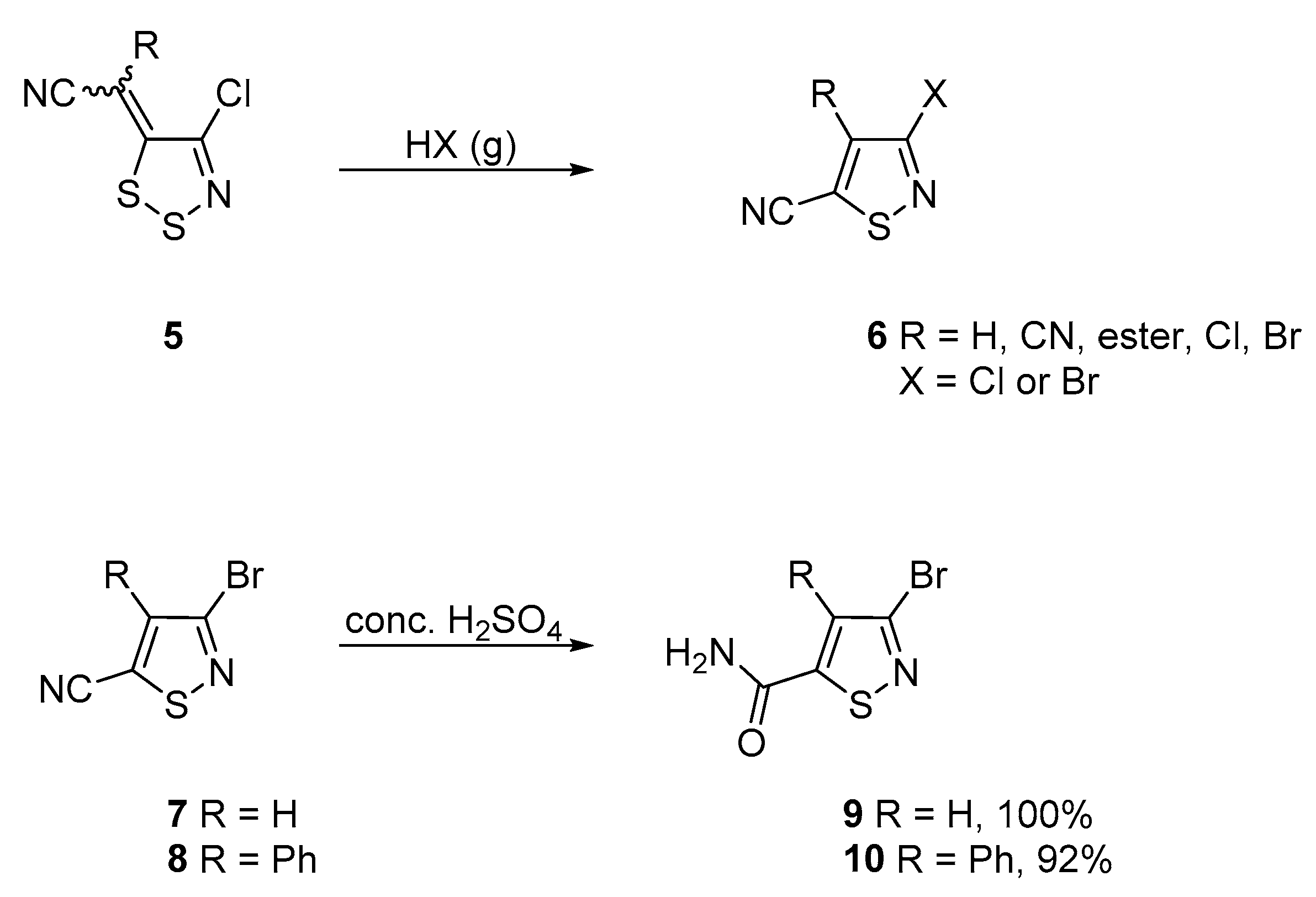
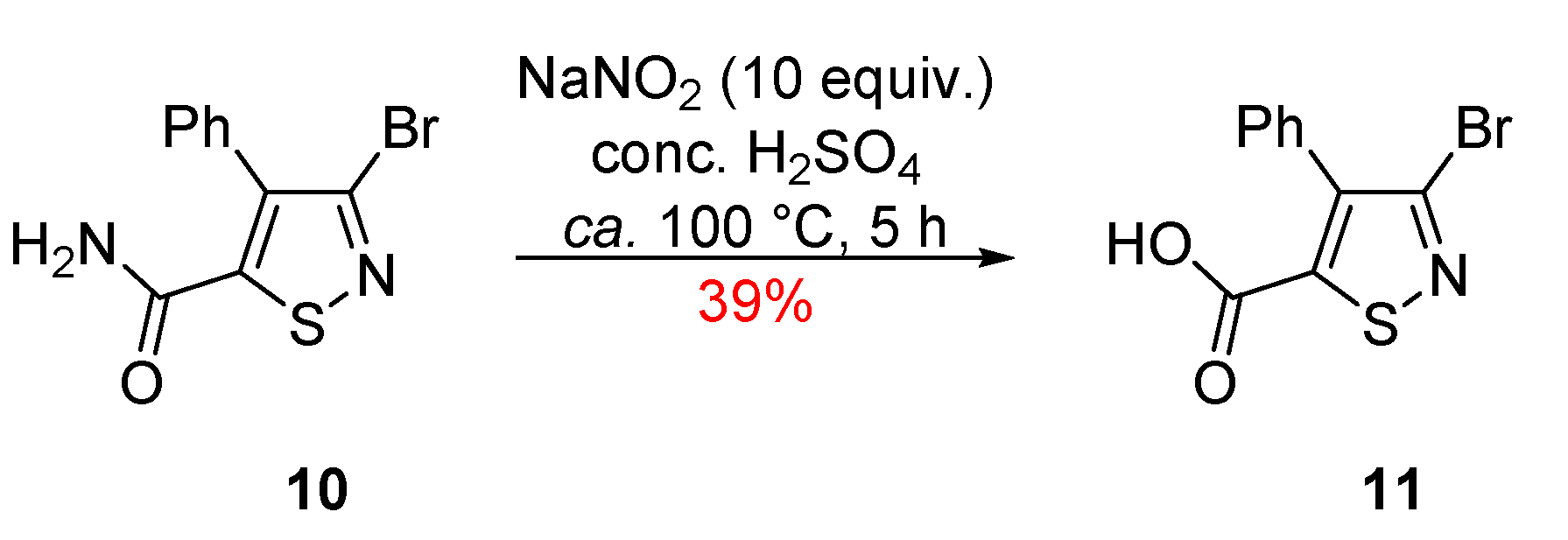
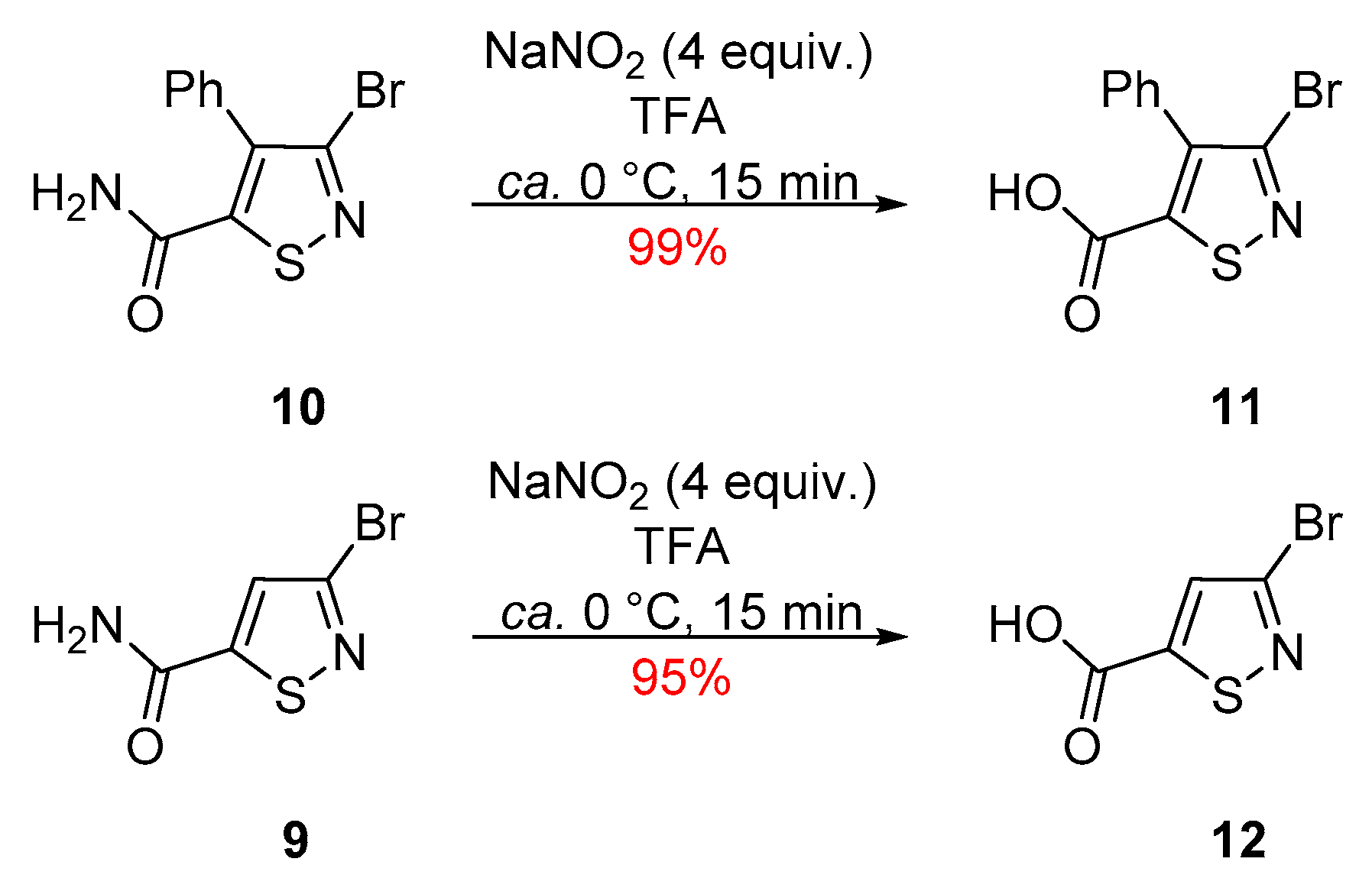
2. Results and Discussion
3. Materials and Methods
3.1. 3-Bromo-4-phenylisothiazole-5-carboxylic Acid (11)
3.2. 3-Bromoisothiazole-5-carboxylic Acid (12)
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Clerici, F.; Gelmi, M.L.; Pellegrino, S.; Joule, J. Comprehensive Heterocyclic Chemistry III; Joule, J., Katritzky, A.R., Ramsden, C.A., Scriven, E.F.V., Taylor, R.J.K., Eds.; Elsevier: Oxford, UK, 2008; Chapter 4.05; Volume 4, pp. 545–633. [Google Scholar]
- Potkin, V.I.; Kletskov, A.V.; Zubkov, F.I. Comprehensive Heterocyclic Chemistry IV; Aitken, R.A., Black, D.S., Cossy, J., Stevens, C.V., Eds.; Elsevier: Oxford, UK, 2022; Chapter 4.05; Volume 4, pp. 482–529. [Google Scholar]
- Larson, E.R.; Noe, M.C.; Gant, T.G. Isothiazole Derivatives Useful as Anticancer Agents. U.S. Patent 6,235,764, 22 May 2001. [Google Scholar]
- Hitoshi, S.; Yanase, Y.; Sekino, T.; Ishikawa, K.; Kuwatsuka, T.; Tanikawa, H.; Kawashima, H.; Tomura, N.; Kanemoto, Y.U.S. Isothiazolecarboxylic Acid Derivatives, Rice Blast Control Agents Containing the Same as Active Ingredients, and Rice Blast Control Method Applying the Control. Agents. Patent 5,240,951, 31 July 1993. [Google Scholar]
- Plant, A.; Boehmer, J.E.; Black, J.; Sparks, T.D. Isoxazolines Derivatives and Its Weedicide Application. CN Patent 101,052,628, 10 October 2007. [Google Scholar]
- Zeng, L.-F.; Zhang, H.-S.; Wang, Y.-H.; Sanchez, T.; Zheng, Y.-T.; Neamati, N.; Long, Y.-Q. Efficient synthesis and utilization of phenyl-substituted heteroaromatic carboxylic acids as aryl diketo acid isosteres in the design of novel HIV-1 integrase inhibitors. Bioorg. Med. Chem. Lett. 2008, 18, 4521–4524. [Google Scholar] [CrossRef] [PubMed]
- Tomisawa, K.; Kameo, K.; Matsunaga, T.; Saito, S.; Hosoda, K.; Asami, Y.; Sota, K. Studies on Hypolipidemic Agents. III: ω-(4-Phenoxybenzyl)-alkanoic Acid Derivatives. Chem. Pharm. Bull. 1986, 34, 701–712. [Google Scholar] [CrossRef] [PubMed]
- Regiec, A.; Machon, Z.; Miedzybrodzki, R.; Szymaniec, S. New Isothiazole Derivatives: Synthesis, Reactivity, Physicochemical Properties and Pharmacological Activity. Arch. Pharm. 2006, 339, 401–413. [Google Scholar] [CrossRef] [PubMed]
- Kalogirou, A.S.; Christoforou, I.C.; Ioannidou, H.A.; Manos, M.; Koutentis, P.A. Ring transformation of (4-chloro-5H-1,2,3-dithiazol-5-ylidene)acetonitriles to 3-haloisothiazole-5-carbonitriles. RSC Adv. 2013, 4, 7735–7748. [Google Scholar] [CrossRef]
- Ioannidou, H.A.; Koutentis, P.A. Silver-mediated palladium-catalyzed direct C-H arylation of 3-bromoisothiazole-4-carbonitrile. Org. Lett. 2011, 13, 1510–1513. [Google Scholar] [CrossRef] [PubMed]
- Kalogirou, A.S.; Koutentis, P.A. Silver mediated direct CH arylation of 3-bromoisothiazole-5-carbonitrile. Tetrahedron 2014, 70, 6796–6802. [Google Scholar] [CrossRef]
- Christoforou, I.C.; Koutentis, P.A. 3,4,5-Triarylisothiazoles via C–C coupling chemistry. Org. Biomol. Chem. 2007, 5, 1381–1390. [Google Scholar] [CrossRef] [PubMed]
- Britcher, S.F.; Lumma, J.; William, C. Diamino Isothiazole-1-oxides and 1,1-dioxides as Gastric Secretion Inhibitors. U.S. Patent 5,171,860, 15 December 1992. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalogirou, A.S.; Koutentis, P.A. Synthesis of 3-Bromo-4-phenylisothiazole-5-carboxylic Acid and 3-Bromoisothiazole-5-carboxylic Acid. Molbank 2023, 2023, M1557. https://doi.org/10.3390/M1557
Kalogirou AS, Koutentis PA. Synthesis of 3-Bromo-4-phenylisothiazole-5-carboxylic Acid and 3-Bromoisothiazole-5-carboxylic Acid. Molbank. 2023; 2023(1):M1557. https://doi.org/10.3390/M1557
Chicago/Turabian StyleKalogirou, Andreas S., and Panayiotis A. Koutentis. 2023. "Synthesis of 3-Bromo-4-phenylisothiazole-5-carboxylic Acid and 3-Bromoisothiazole-5-carboxylic Acid" Molbank 2023, no. 1: M1557. https://doi.org/10.3390/M1557
APA StyleKalogirou, A. S., & Koutentis, P. A. (2023). Synthesis of 3-Bromo-4-phenylisothiazole-5-carboxylic Acid and 3-Bromoisothiazole-5-carboxylic Acid. Molbank, 2023(1), M1557. https://doi.org/10.3390/M1557







