2-((3-(4-Methoxyphenyl)-4,5-dihydroisoxazol-5-yl)methyl)benzo[d]isothiazol-3(2H)-one1,1-dioxide
Abstract
1. Introduction
2. Results
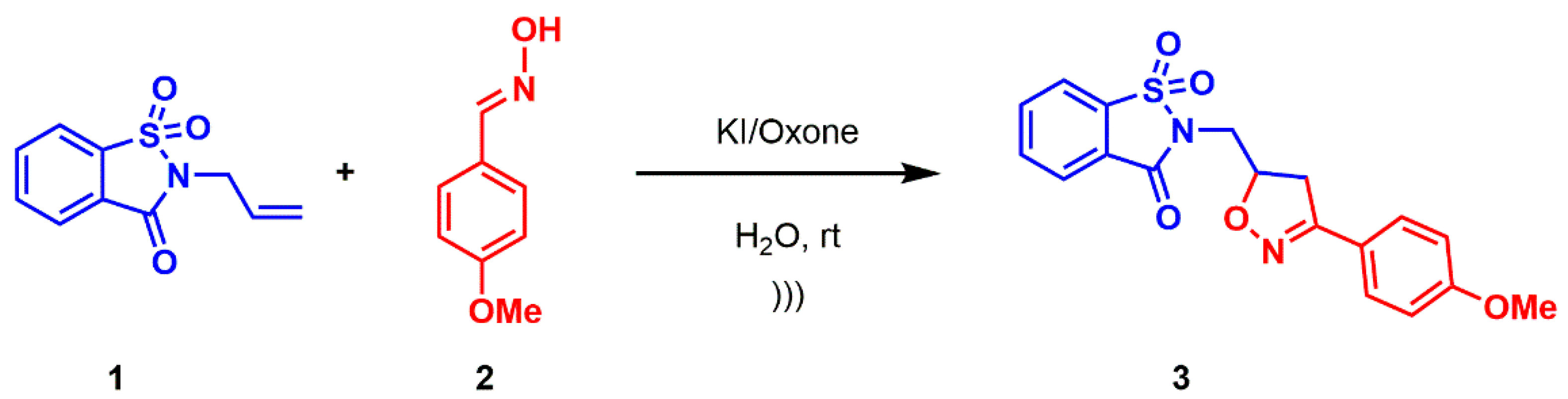
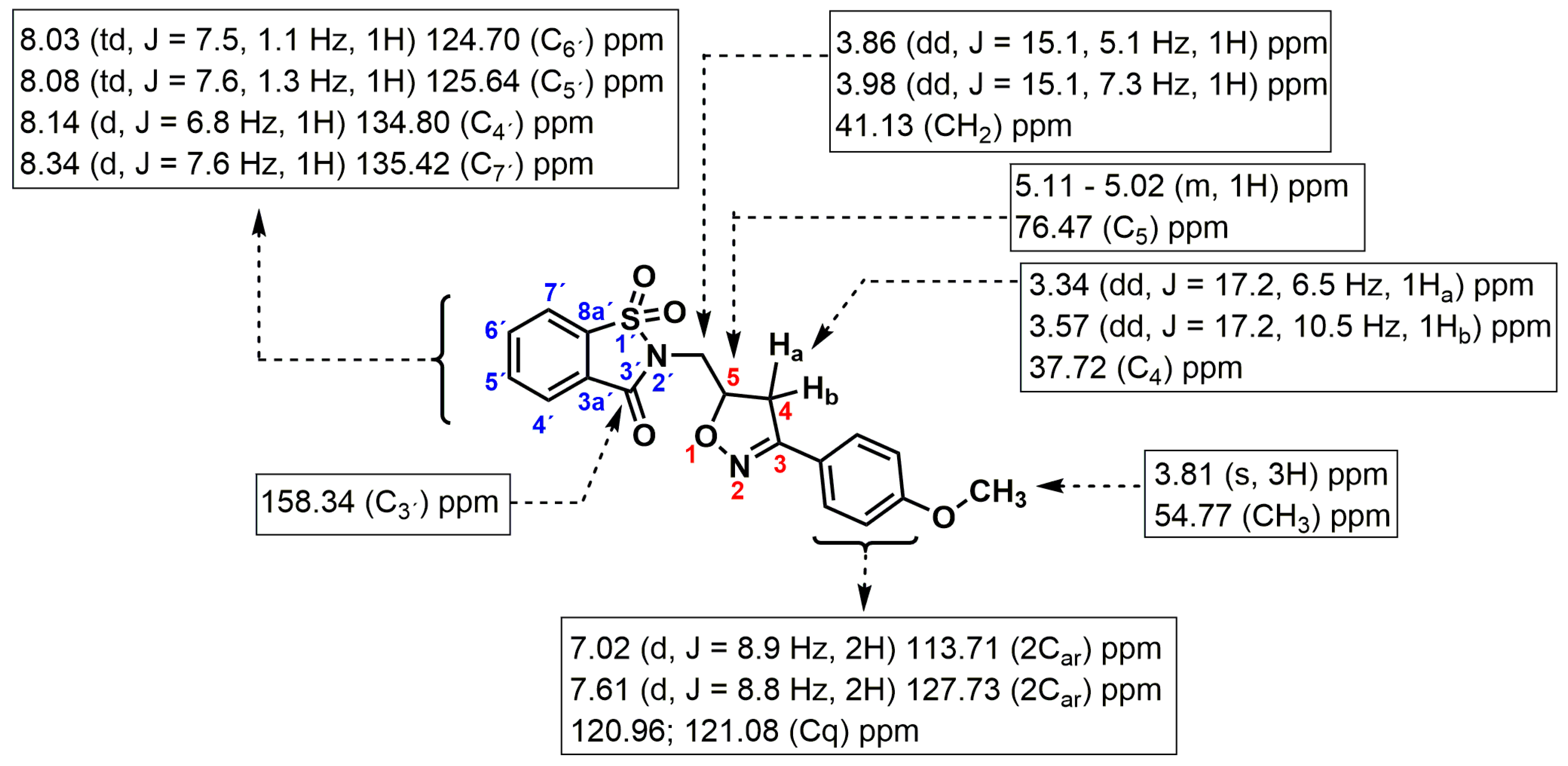
2.1. Synthesis
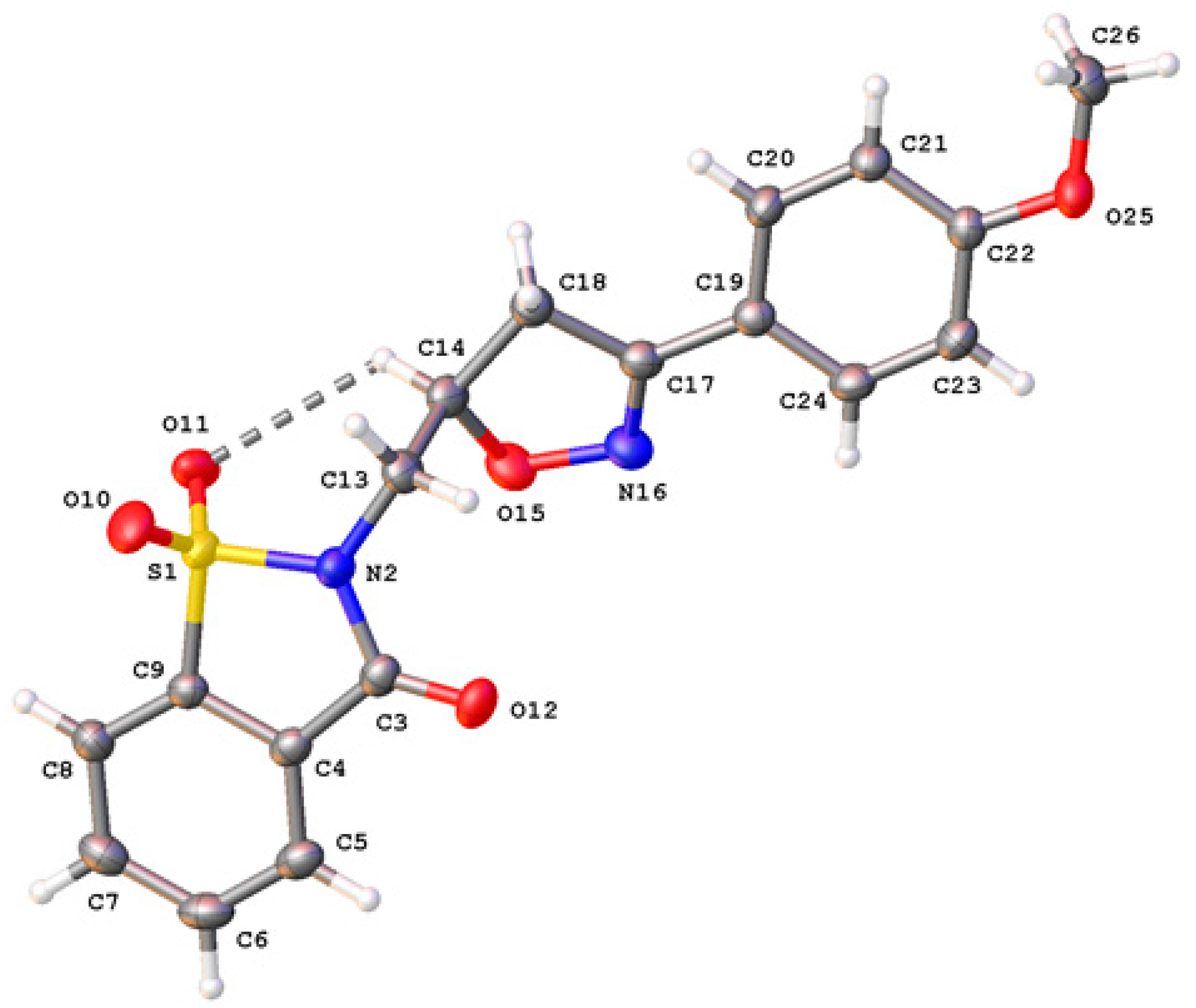
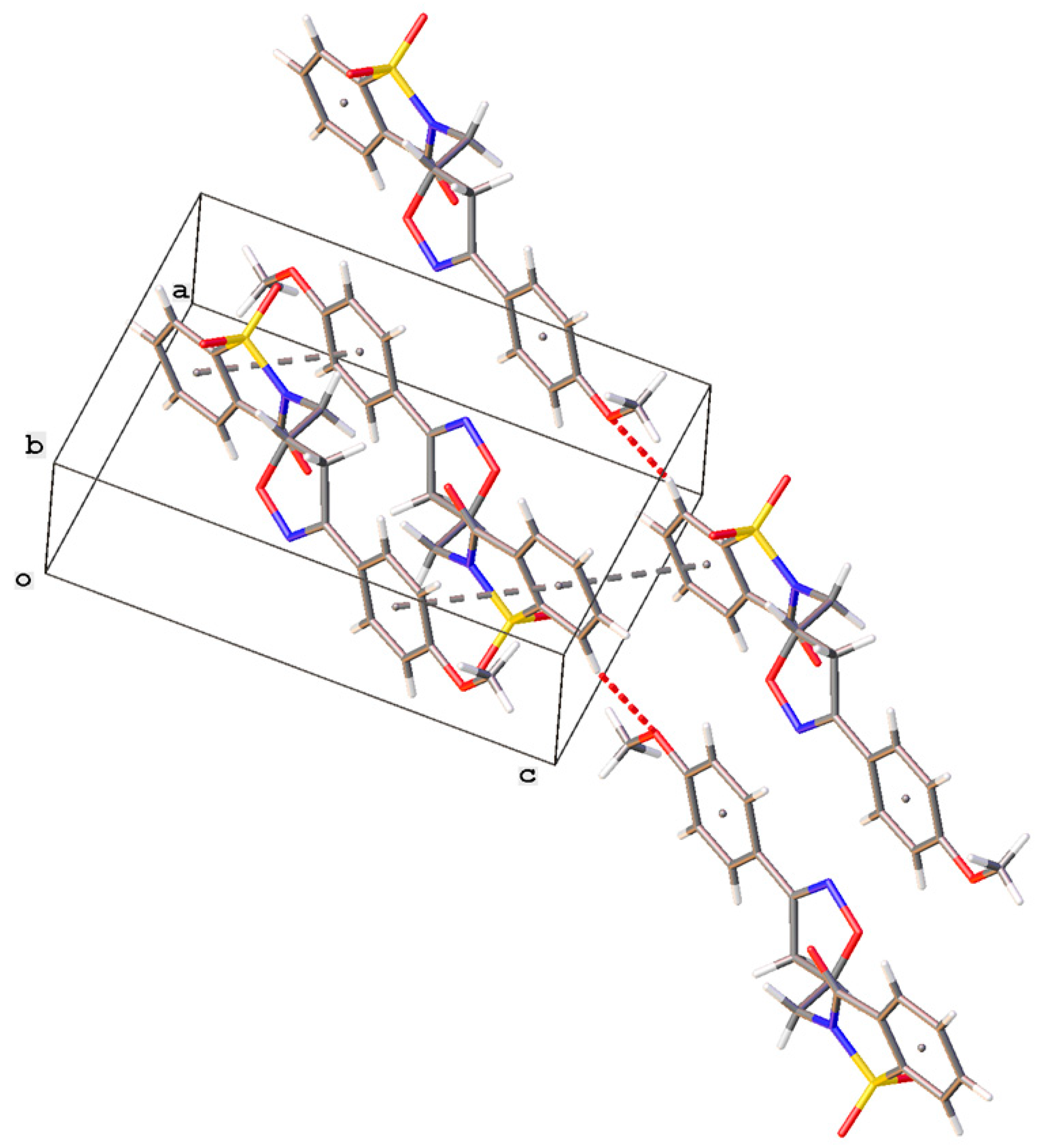
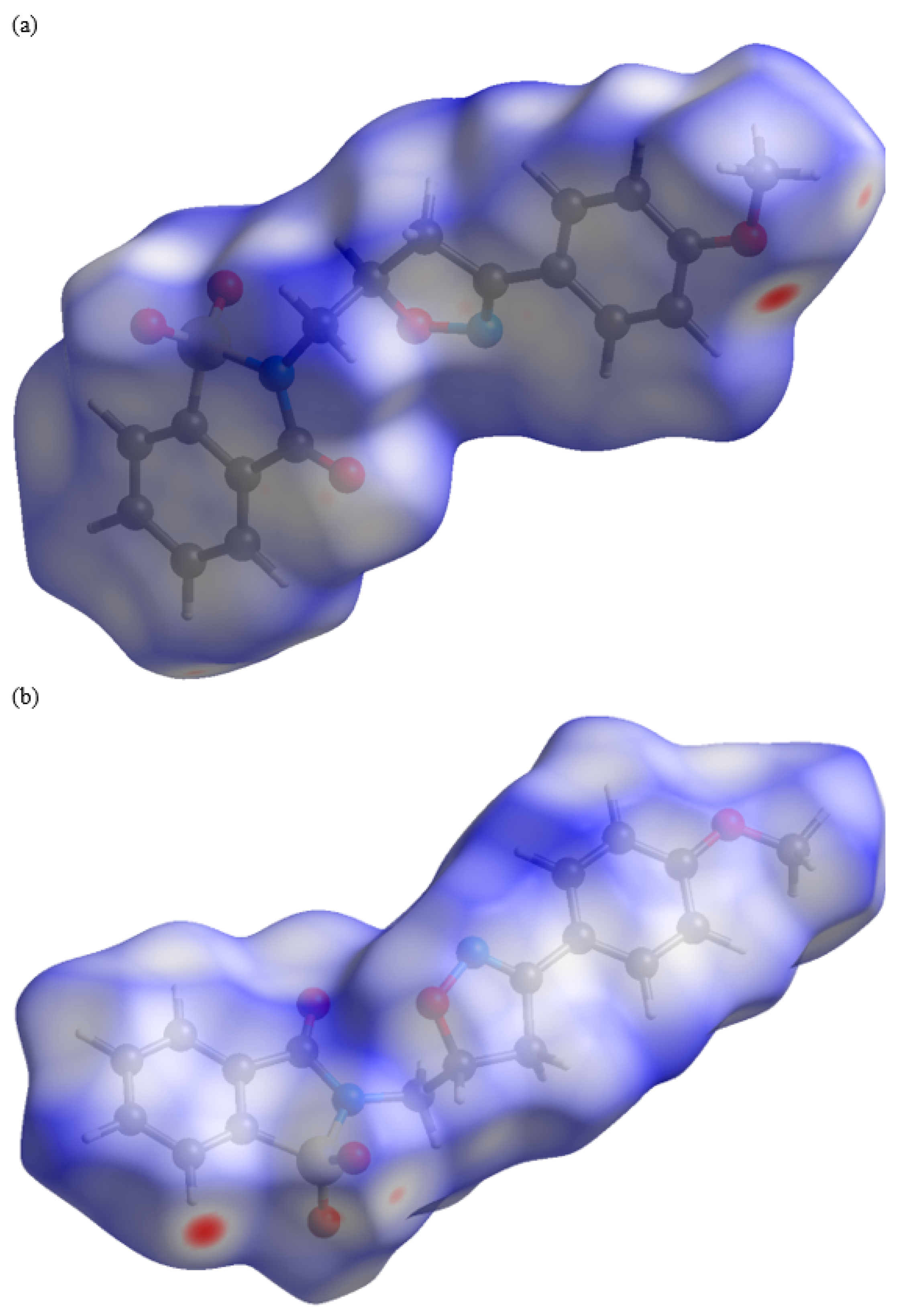
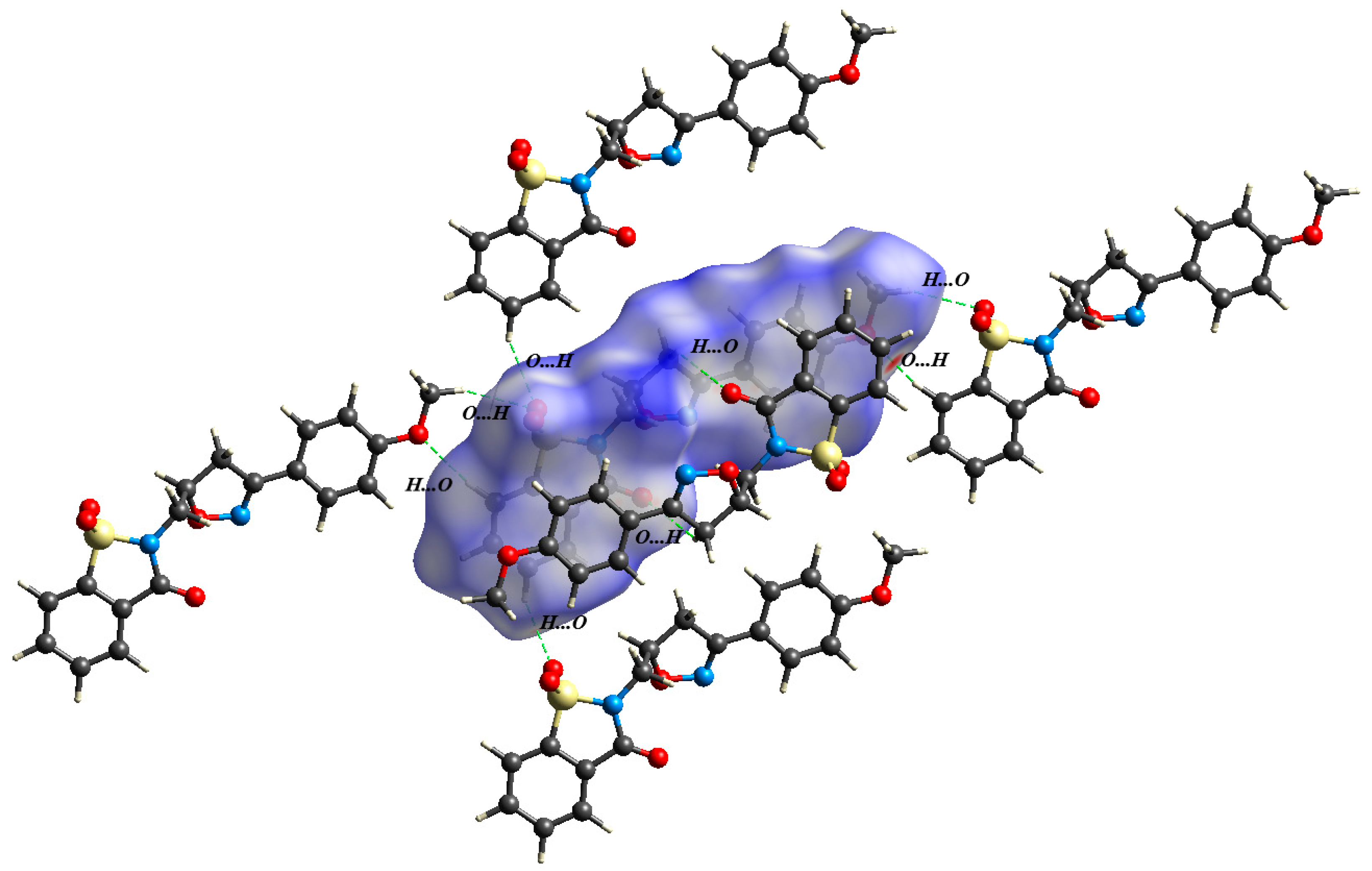
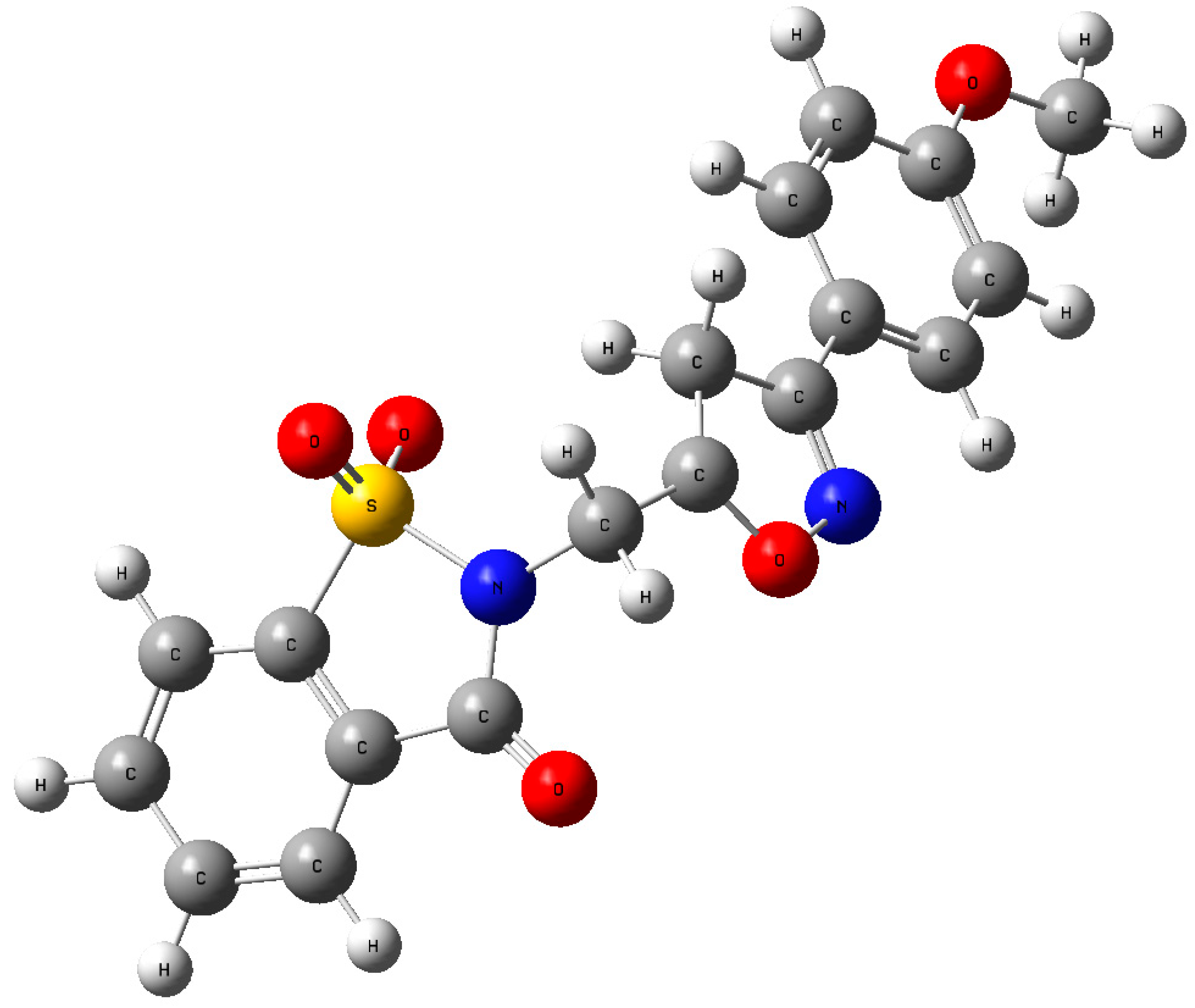
2.2. Crystal Structure Determination
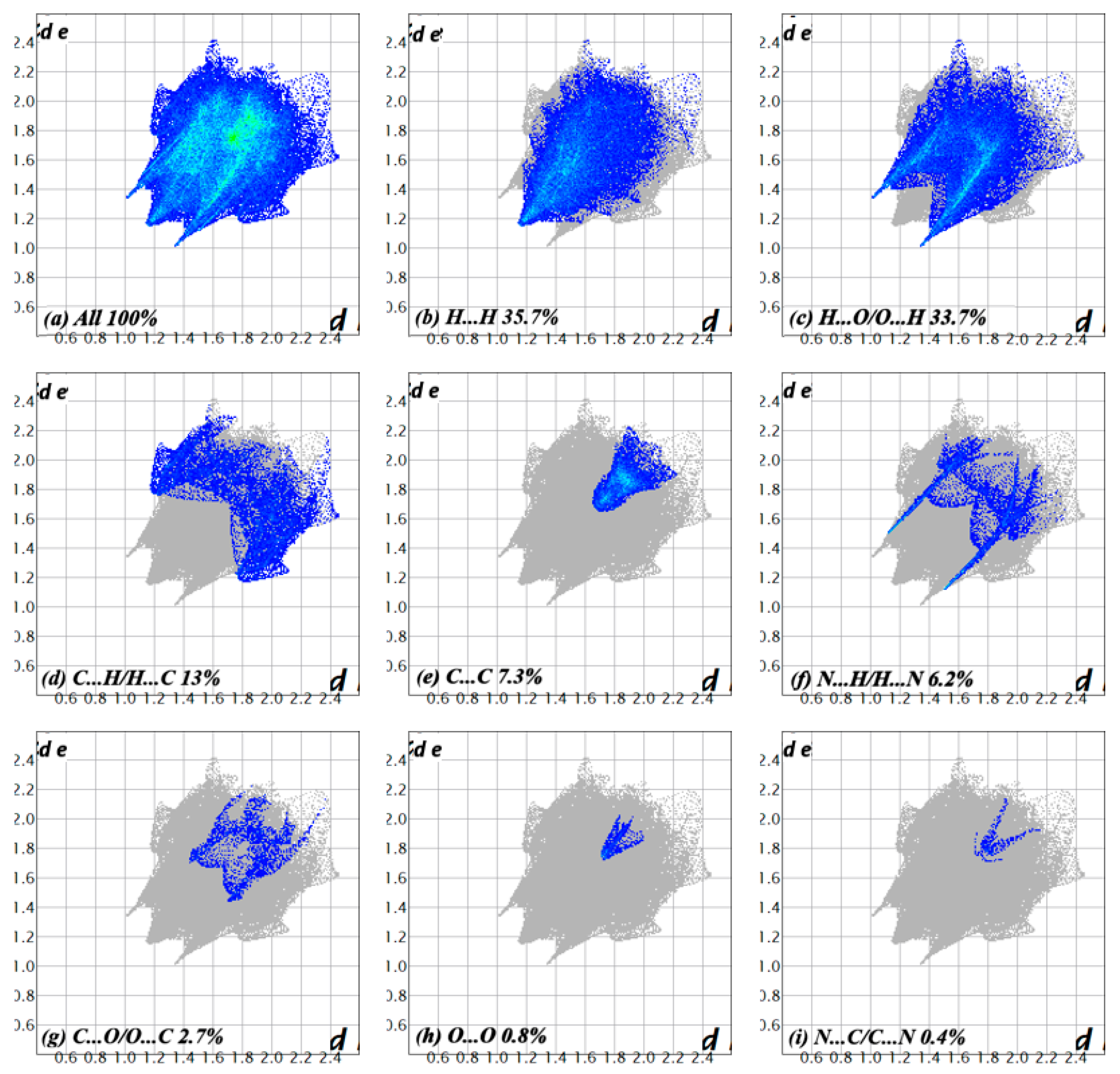
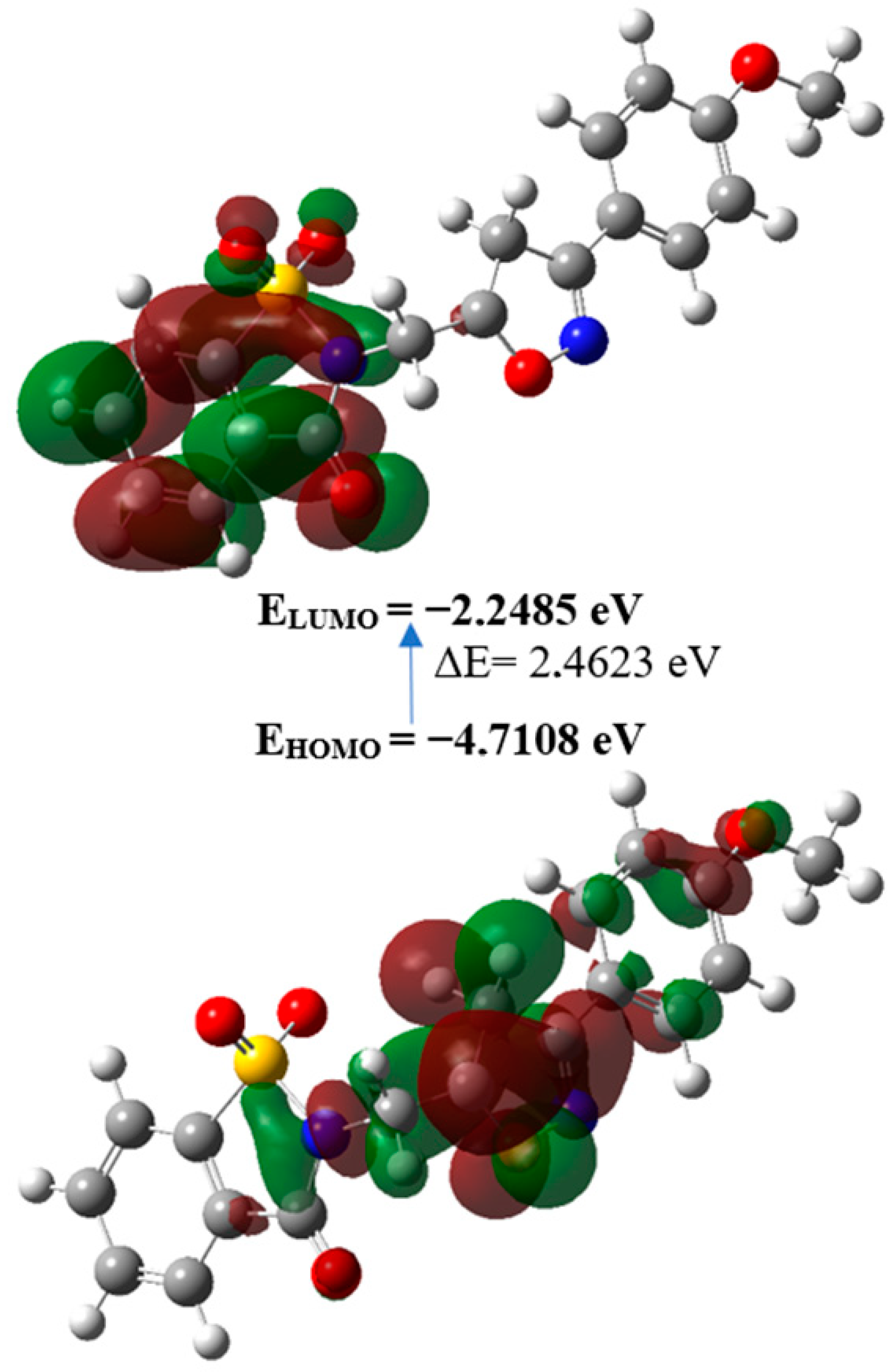
2.3. Density Functional Theory Calculations
3. Experimental Section
3.1. Materials and Methods
3.2. Synthesis of Compound 3
3.3. X-ray Data of Crystal Structure
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barmade, M.A.; Murumkar, P.R.; Kumar Sharma, M.; Ram Yadav, M. Medicinal chemistry perspective of fused isoxazole derivatives. Curr. Top. Med. Chem. 2016, 16, 2863–2883. [Google Scholar] [CrossRef]
- Kumar, G.; Shankar, R. 2-Isoxazolines: A Synthetic and Medicinal Overview. ChemMedChem 2021, 16, 430–447. [Google Scholar] [CrossRef]
- Liao, J.; Ouyang, L.; Jin, Q.; Zhang, J.; Luo, R. Recent advances in the oxime-participating synthesis of isoxazolines. Org. Biomol. Chem. 2020, 18, 4709–4716. [Google Scholar] [CrossRef] [PubMed]
- Ushakov, P.; Ioffe, S.; Sukhorukov, A. Renewed «Isoxazoline Route» for the Synthesis of Densely Functionalized Ketones. Org. Biomol. Chem. 2022. [Google Scholar] [CrossRef]
- Liu, X.; Hong, D.; Sapir, N.G.; Yang, W.; Hersh, W.H.; Leung, P.H.; Dingqiao, Y.; Chen, Y. Iron-catalyzed transfer hydrogenation in aged N-methyl-2-pyrrolidone: Reductive ring-opening of 3, 5-disubstituted isoxazoles and isoxazolines. J. Org. Chem. 2019, 84, 16204–16213. [Google Scholar] [CrossRef]
- Chandrasekhar, B.; Ahn, S.; Ryu, J.S. Stereoselective Synthesis of a Ceramide Transporter Protein (CERT)-Dependent Ceramide-Trafficking Inhibitor,(1R, 3S)-HPA-12, via Gold (I)-Catalyzed Cyclization of a Propargylic N-Hydroxylamine. Synthesis 2017, 49, 1569–1574. [Google Scholar]
- Berthet, M.; Cheviet, T.; Dujardin, G.; Parrot, I.; Martinez, J. Isoxazolidine: A privileged scaffold for organic and medicinal chemistry. Chem. Rev. 2016, 116, 15235–15283. [Google Scholar] [CrossRef]
- Nikam, M.D.; Mahajan, P.S.; Damale, M.G.; Sangshetti, J.N.; Dabhade, S.K.; Shinde, D.W.; Gill, C.H. Synthesis, molecular docking and biological evaluation of some novel tetrazolo [1, 5-a] quinoline incorporated pyrazoline and isoxazoline derivatives. Med. Chem. Res. 2015, 24, 3372–3386. [Google Scholar] [CrossRef]
- Prajapti, S.K.; Shrivastava, S.; Bihade, U.; Gupta, A.K.; Naidu, V.G.M.; Banerjee, U.C.; Babu, B.N. Synthesis and biological evaluation of novel Δ 2-isoxazoline fused cyclopentane derivatives as potential antimicrobial and anticancer agents. MedChemComm 2015, 6, 839–845. [Google Scholar] [CrossRef]
- Goyard, D.; Kónya, B.; Chajistamatiou, A.S.; Chrysina, E.D.; Leroy, J.; Balzarin, S.; Tournier, M.; Tousch, D.; Petit, P.; Duret, C.; et al. Glucose-derived spiro-isoxazolines are anti-hyperglycemic agents against type 2 diabetes through glycogen phosphorylase inhibition. Eur. J. Med. Chem. 2016, 108, 444–454. [Google Scholar] [CrossRef] [PubMed]
- Lopes, E.F.; Penteado, F.; Thurow, S.; Pinz, M.; Reis, A.S.; Wilhelm, E.A.; Luchese, C.; Barcellos, T.; Dalberto, B.; Alves, D.; et al. Synthesis of isoxazolines by the electrophilic chalcogenation of β, γ-unsaturated oximes: Fishing novel anti-inflammatory agents. J. Org. Chem. 2019, 84, 12452–12462. [Google Scholar] [CrossRef] [PubMed]
- Ghidini, E.; Capelli, A.M.; Carnini, C.; Cenacchi, V.; Marchini, G.; Virdis, A.; Italia, A.; Facchinetti, F. Discovery of a novel isoxazoline derivative of prednisolone endowed with a robust anti-inflammatory profile and suitable for topical pulmonary administration. Steroids 2015, 95, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Mota, F.V.B.; de Araújo Neta, M.S.; de Souza Franco, E.; Bastos, I.V.G.A.; da Araújo, L.C.C.; da Silva, S.C.; de Oliveira, T.B.; Souza, E.K.; de Almeida, V.M.; Ximenes, R.M.; et al. Evaluation of anti-inflammatory activity and molecular docking study of new aza-bicyclic isoxazoline acylhydrazone derivatives. Medchemcomm 2019, 10, 1916–1925. [Google Scholar] [CrossRef] [PubMed]
- Maurya, R.; Ahmad, A.; Gupta, P.; Chand, K.; Kumar, M.; Rawat, P.; Rasheed, N.; Palit, G. Synthesis of novel isoxazolines via 1, 3-dipolar cycloaddition and evaluation of anti-stress activity. Med. Chem. Res. 2011, 20, 139–145. [Google Scholar] [CrossRef]
- Aarjane, M.; Slassi, S.; Ghaleb, A.; Tazi, B.; Amine, A. Synthesis, biological evaluation, molecular docking and in silico ADMET screening studies of novel isoxazoline derivatives from acridone. Arab. J. Chem. 2021, 14, 103057. [Google Scholar] [CrossRef]
- Kalaria, P.N.; Satasia, S.P.; Raval, D.K. Synthesis, identification and in vitro biological evaluation of some novel 5-imidazopyrazole incorporated pyrazoline and isoxazoline derivatives. New J. Chem. 2014, 38, 2902–2910. [Google Scholar] [CrossRef]
- Huang, M.; Suk, D.H.; Cho, N.C.; Bhattarai, D.; Kang, S.B.; Kim, Y.; Nim Pae, A.; Rhim, H.; Keum, G. Synthesis and biological evaluation of isoxazoline derivatives as potent M1 muscarinic acetylcholine receptor agonists. Bioorg. Med. Chem. Lett. 2015, 25, 1546–1551. [Google Scholar] [CrossRef] [PubMed]
- Sreenivasa, S.; Shankar, B.J.; Mohan, N.R. Synthesis and Evaluation of Anti-inflammatory and Analgesic Activity of Isoxazoline Bearing Tris (heterocycles). Indo Am. J. Pharma. Res. 2014, 4, 2485–2490. [Google Scholar]
- Liu, Z.; Han, M.; Yan, X.; Cheng, W.; Tang, Z.; Cui, L.; Yang, R.; Guo, Y. Design, Synthesis, and Biological Evaluation of Novel Osthole-Based Isoxazoline Derivatives as Insecticide Candidates. J. Agric. Food Chem. 2022, 70, 7921–7928. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, I.L.; das Neves, G.M.; Kagami, L.P.; Eifler-Lima, V.L.; Merlo, A.A. Discovery, development, chemical diversity and design of isoxazoline-based insecticides. Bioorg. Med. Chem. 2021, 30, 115934. [Google Scholar] [CrossRef]
- Yang, R.; Zhang, Y.; Xu, H. Synthesis of novel isoxazoline-containing podophyllotoxin/2′(2′, 6′)-(di) halogenopodophyllotoxin derivatives and their insecticidal/acaricidal activities. Bioorg. Med. Chem. Lett. 2018, 28, 1410–1416. [Google Scholar] [CrossRef]
- Luo, S.X.L.; Liu, R.Y.; Lee, S.; Swager, T.M. Electrocatalytic Isoxazoline–Nanocarbon Metal Complexes. J. Am. Chem. Soc. 2021, 143, 10441–10453. [Google Scholar] [CrossRef]
- Wu, X.B.; Gao, Q.; Fan, J.J.; Zhao, Z.Y.; Tu, X.Q.; Cao, H.Q.; Yu, J. Anionic Chiral Co (III) Complexes Mediated Asymmetric Halocyclization─ Synthesis of 5-Halomethyl Pyrazolines and Isoxazolines. Org. Lett. 2021, 23, 9134–9139. [Google Scholar] [CrossRef]
- Yu, W.D.; Nie, Y.M.; Yuan, H.; Yan, J.; Yi, H.B. Synthesis and characterization of a highly stable zinc phenylporphyrin Isoxazoline-[60] fullerene dyad: Impact of coordination on the redox and fluorescence properties. Inorg. Chem. Commun. 2017, 84, 134–137. [Google Scholar] [CrossRef]
- Saber, A.; Driowya, M.; Alaoui, S.; Marzag, H.; Demange, L.; Álvarez, E.; Benhida, R.; Bougrin, K. Solvent-Free Regioselective Synthesis of Novel Isoxazoline and Pyrazoline N-Substituted Saccharin Derivatives Under Microwave Irradiation. Chem. Heterocycl. Compd. 2016, 52, 31–40. [Google Scholar] [CrossRef]
- Talha, A.; Favreau, C.; Bourgoin, M.; Robert, G.; Auberger, P.; Ammari, L.E.; Saadi, M.; Benhida, R.; Martin, A.R.; Bougrin, K. Ultrasound-assisted one-pot three-component synthesis of new isoxazolines bearing sulfonamides and their evaluation against hematological malignancies. Ultrason. Sonochem. 2021, 78, 105748. [Google Scholar] [CrossRef]
- Thari, F.Z.; Tachallait, H.; El Alaoui, N.E.; Talha, A.; Arshad, S.; Álvarez, E.; Karrouchi, K.; Bougrin, K. Ultrasound-assisted one-pot green synthesis of new N-substituted-5-arylidene-thiazolidine-2,4-dione-isoxazoline derivatives using NaCl/Oxone/Na3PO4 in aqueous media. Ultrason. Sonochem. 2020, 68, 105222. [Google Scholar] [CrossRef]
- Tachallait, H.; Driowya, M.; Álvarez, E.; Benhida, R.; Bougrin, K. Water Promoted One-pot Three-Step Synthesis of Novel N-Saccharin Isoxazolines/Isoxazoles Using KI/Oxone Under Ultrasonic Activation. Curr. Org. Chem. 2019, 23, 1270–1281. [Google Scholar] [CrossRef]
- Fettach, S.; Thari, F.Z.; Hafidi, Z.; Karrouchi, K.; Bouathmany, K.; Cherrah, Y.; EL Achouri, A.; Benbacer, L.; EL Mzibri, M.; Sefrioui, H.; et al. Biological, toxicological and molecular docking evaluations of isoxazoline-thiazolidine-2, 4-dione analogues as new class of anti-hyperglycemic agents. J. Biomol. Struct. Dyn. 2021, 16, 68. [Google Scholar] [CrossRef]
- Idrissi, A.; Chkirate, K.; Abad, N.; Djerrari, B.; Achour, R.; Essassi, E.M.; Van Meervelt, L. Crystal structure, Hirshfeld surface analysis and density functional theory study of 6-methyl-2-[(5-methylisoxazol-3-yl)methyl]-1H-benzimidazole. Acta Cryst. E 2021, 77, 396–401. [Google Scholar] [CrossRef]
- Abad, N.; Chkirate, K.; Al-Ostoot, F.H.; Van Meervelt, L.; Lahmidi, S.; Ferfra, S.; Ramli, Y.; Essassi, E.M. Crystal structure, Hirshfeld surface analysis and density functional theory study of 1-nonyl-3-phenylquinoxalin-2-one. Acta Cryst. E 2021, 77, 1037–1042. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Cryst. A 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. C 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer17; The University of Western Australia: Crawley, Australia, 2017. [Google Scholar]
- McKinnon, J.J.; Jayatilaka, D.; Spackman, M.A. Towards quantitative analysis of intermolecular interactions with Hirshfeld surfaces. Chem. Commun. 2007, 37, 3814–3816. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. GAUSSIAN09. Revision A.02; Gaussian Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Chkirate, K.; Azgaou, K.; Elmsellem, H.; El Ibrahimi, B.; Sebbar, N.K.; Anouar, E.H.; Benmessaoud, M.; El Hajjaji, S.; Essassi, E.M. Corrosion Inhibition Potential of 2-[(5-methylpyrazol-3-yl)methyl]benzimidazole against carbon steel corrosion in 1M HCl solution: Combining Experimental and Theoretical Studies. J. Mol. Liq. 2021, 321, 114750. [Google Scholar] [CrossRef]
- Bouzian, Y.; Sert, Y.; Karrouchi, K.; Van Meervelt, L.; Chkirate, K.; Mahi, L.; Hamou Ahabchane, N.; Talbaoui, A.; Essassi, E.M. Synthesis, spectroscopic characterization, DFT, molecular docking and in vitro antibacterial potential of novel quinoline derivatives. J. Mol. Struct. 2021, 1246, 131217. [Google Scholar] [CrossRef]
- Chkirate, K.; Akachar, J.; Hni, B.; Hökelek, T.; Anouar, E.H.; Talbaoui, A.; Mague, J.T.; Sebbar, N.K.; Ibrahimi, A.; Essassi, E.M. Synthesis, spectroscopic characterization, crystal structure, DFT, ESI-MS studies, molecular docking and in vitro antibacterial activity of 1,5-benzodiazepin-2-one derivatives. J. Mol. Struct. 2022, 1247, 131188. [Google Scholar] [CrossRef]
| X-ray | B3LYP/6–311G (d,p) | |
|---|---|---|
| S1-N2 | 1.6641 (16) | 1.7163 |
| S1-C9 | 1.7466 (19) | 1.7875 |
| S1-O10 | 1.4213 (17) | 1.458 |
| O15-N16 | 1.419 (2) | 1.4091 |
| S1-O11 | 1.4297 (16) | 1.4589 |
| N16-C17 | 1.284 (2) | 1.2895 |
| N2-C3 | 1.392 (2) | 1.3936 |
| N2-C13 | 1.463 (2) | 1.4941 |
| C3-O12 | 1.206 (2) | 1.2083 |
| C22-O25 | 1.365 (2) | 1.3591 |
| O25-C26 | 1.423 (3) | 1.4225 |
| N2-S1-C9 | 93.00 (8) | 91.7503 |
| O10-S1-N2 | 110.43 (10) | 109.2283 |
| O10-S1-C9 | 112.43 (10) | 112.4224 |
| O10-S1-O11 | 116.94 (10) | 118.6032 |
| O11-S1-C9 | 111.96 (9) | 111.2453 |
| C3-N2-S1 | 115.20 (13) | 114.946 |
| C3-N2-C13 | 123.60 (16) | 122.8963 |
| C13-N2-S1 | 120.90 (13) | 121.519 |
| N2-C3-C4 | 108.62 (16) | 109.0882 |
| O12-C3-N2 | 123.96 (19) | 124.7446 |
| O12-C3-C4 | 127.42 (18) | 126.1588 |
| C4-C9-S1 | 109.89 (14) | 110.0374 |
| C8-C9-S1 | 127.24 (15) | 127.1339 |
| N16-O15-C14 | 108.53 (14) | 109.5655 |
| C17-N16-O15 | 109.10 (17) | 108.6161 |
| N16-C17-C18 | 114.05 (18) | 113.1977 |
| N16-C17-C19 | 121.97 (18) | 121.5156 |
| O25-C22-C21 | 124.07 (18) | 124.6663 |
| O25-C22-C23 | 116.09 (17) | 115.8642 |
| C22-O25-C26 | 117.92 (16) | 118.7364 |
| Molecular Energy | Compound |
|---|---|
| Total Energy TE (eV) | −42,964.3286 |
| EHOMO (eV) | −4.7108 |
| ELUMO (eV) | −2.2485 |
| Gap, ΔE (eV) | 2.4623 |
| Dipole moment, µ (Debye) | 3.5690 |
| Ionization potential, I (eV) | 4.7108 |
| Electron affinity, A | 2.2485 |
| Electronegativity, χ | 3.4797 |
| Hardness, η | 1.2312 |
| Electrophilicity, index ω | 4.9173 |
| Softness, σ | 0.8122 |
| Fraction of electron transferred, ΔN | 1.4296 |
| Crystal Data | |
|---|---|
| Empirical formula | C18H16N2O5S |
| Formula weight | 372.39 |
| Temperature/K | 293(2) |
| Crystal system, Space group | Triclinic, P-1 |
| a, b, c (Å) | 7.3921(3), 8.5605(3), 13.6058(4) |
| α, β, γ (°) | 76.768(3), 86.131(3), 84.776(3) |
| Volume (Å3) | 833.67(5) |
| Z | 2 |
| ρcalc (g/cm3) | 1.483 |
| μ/mm−1 | 0.228 |
| F(000) | 388.0 |
| Crystal size (mm3) | 0.3 × 0.3 × 0.25 |
| Radiation | Mo Kα (λ = 0.71073 Å) |
| 2Θ range for data collection/° | 4.904 to 52.734 |
| Index ranges | −9 ≤ h ≤ 9, −10 ≤ k ≤ 10, −16 ≤ l ≤ 16 |
| Reflections collected | 11964 |
| Independent reflections | 3407 (Rint = 0.0202, Rsigma = 0.0226) |
| Data/restraints/parameters | 3407/0/236 |
| Goodness-of-fit on F2 | 1.028 |
| Final R indexes (I ≥ 2σ (I)) | R1 = 0.0404, wR2 = 0.0987 |
| Final R indexes (all data) | R1 = 0.0519, wR2 = 0.1061 |
| Largest diff. peak/hole/e Å−3 | 0.28/−0.33 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El Mahmoudi, A.; Chkirate, K.; Tachallait, H.; Van Meervelt, L.; Bougrin, K. 2-((3-(4-Methoxyphenyl)-4,5-dihydroisoxazol-5-yl)methyl)benzo[d]isothiazol-3(2H)-one1,1-dioxide. Molbank 2022, 2022, M1488. https://doi.org/10.3390/M1488
El Mahmoudi A, Chkirate K, Tachallait H, Van Meervelt L, Bougrin K. 2-((3-(4-Methoxyphenyl)-4,5-dihydroisoxazol-5-yl)methyl)benzo[d]isothiazol-3(2H)-one1,1-dioxide. Molbank. 2022; 2022(4):M1488. https://doi.org/10.3390/M1488
Chicago/Turabian StyleEl Mahmoudi, Ayoub, Karim Chkirate, Hamza Tachallait, Luc Van Meervelt, and Khalid Bougrin. 2022. "2-((3-(4-Methoxyphenyl)-4,5-dihydroisoxazol-5-yl)methyl)benzo[d]isothiazol-3(2H)-one1,1-dioxide" Molbank 2022, no. 4: M1488. https://doi.org/10.3390/M1488
APA StyleEl Mahmoudi, A., Chkirate, K., Tachallait, H., Van Meervelt, L., & Bougrin, K. (2022). 2-((3-(4-Methoxyphenyl)-4,5-dihydroisoxazol-5-yl)methyl)benzo[d]isothiazol-3(2H)-one1,1-dioxide. Molbank, 2022(4), M1488. https://doi.org/10.3390/M1488






