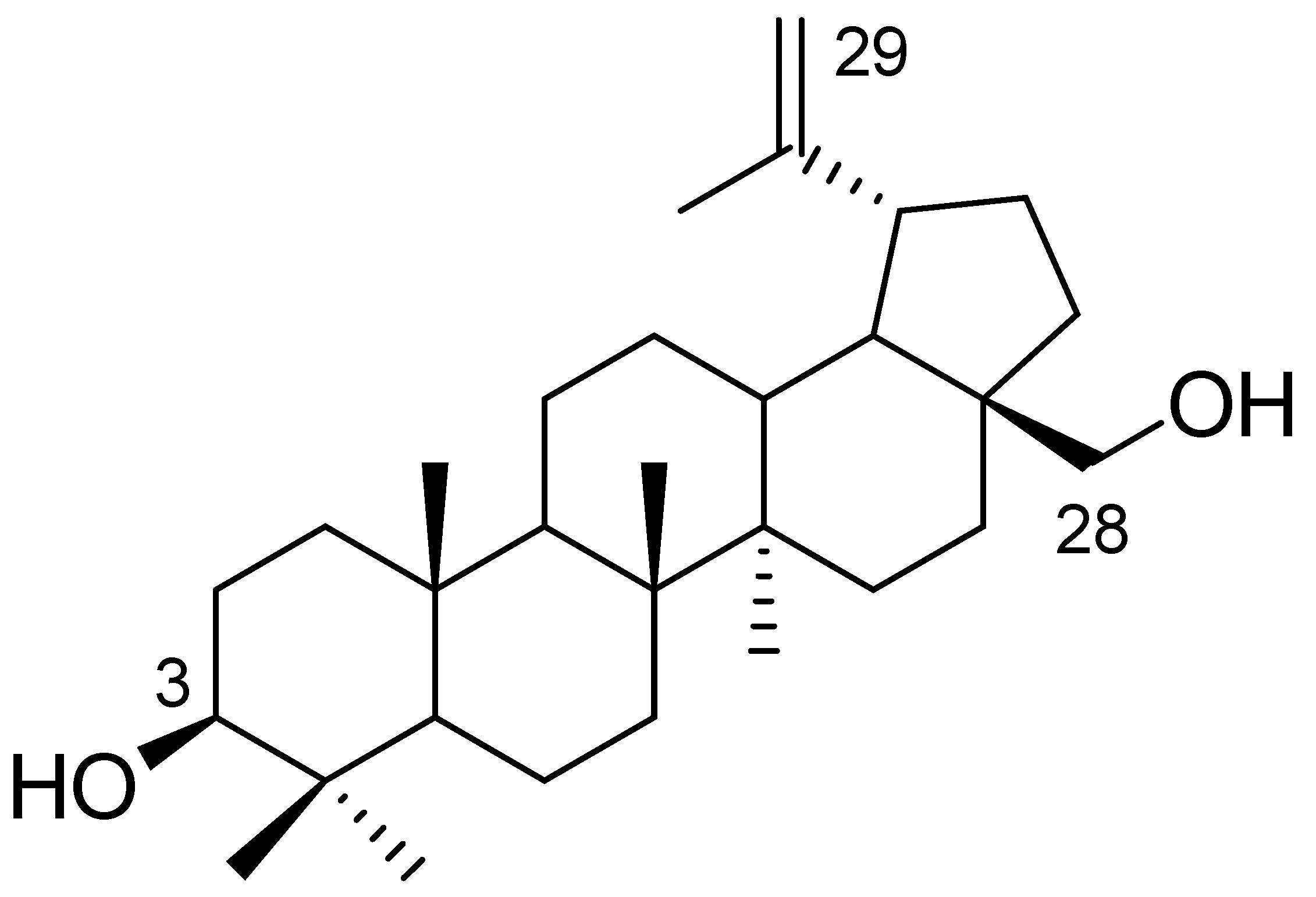
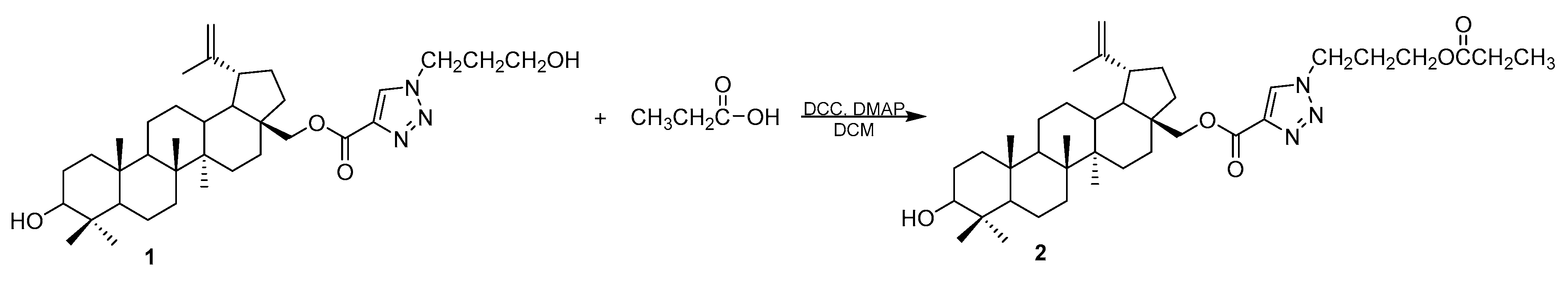
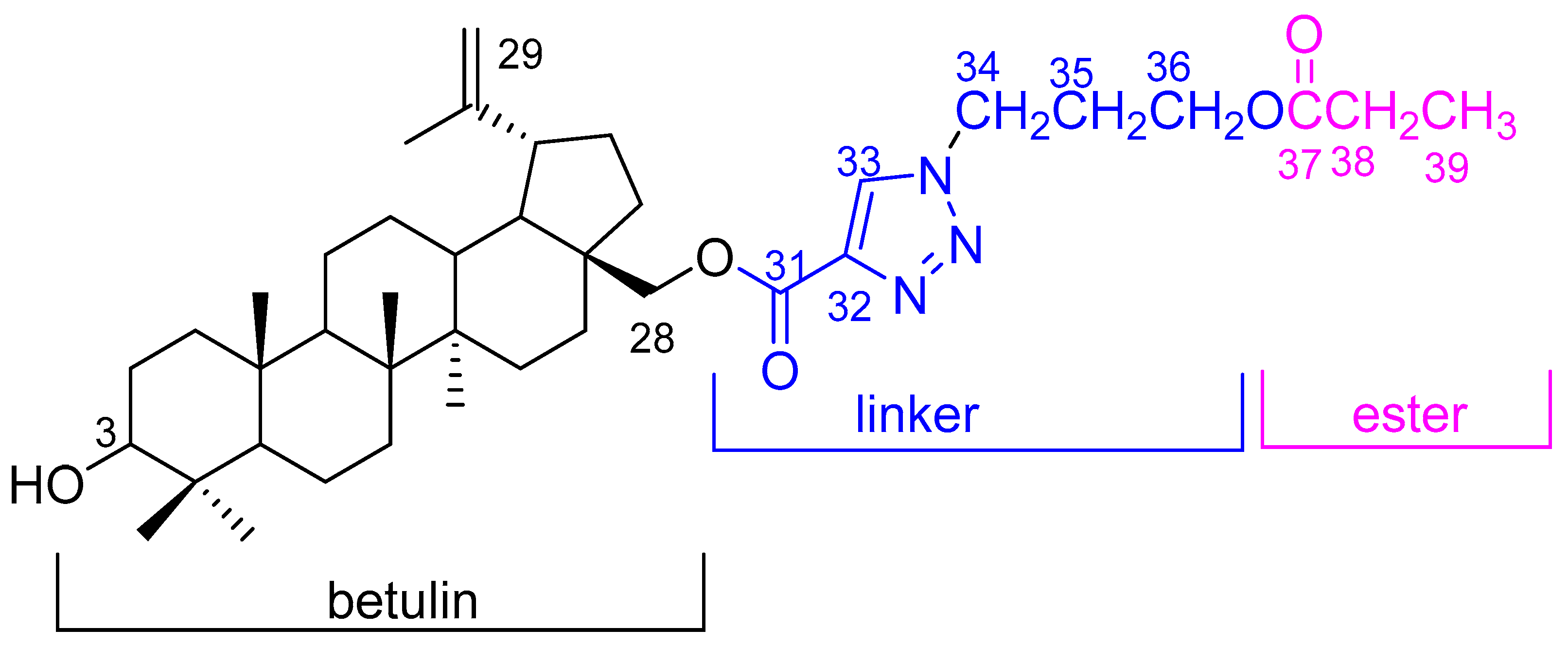
28-[1-(3-(Propionyloxy)propyl)-1H-1,2,3-triazol-4-yl]carbonylbetulin
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. General Method
3.2. Synthesis of 28-[1-(3-(Propionyloxy)propyl)-1H-1,2,3-triazol-4-yl]carbonylbetulin 2
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kaur, P.; Arora, S.; Singh, R. Isolation, characterization and biological activities of betulin from Acacia nilotica bark. Sci. Rep. 2022, 12, 9370. [Google Scholar] [CrossRef]
- Sułkowska-Ziaja, K.; Szewczyk, A.; Galanty, A.; Gdula-Argasińska, J.; Muszyńska, B. Chemical composition and biological activity of extracts from fruiting bodies and mycelial cultures of Fomitopsis betulina. Mol. Biol. Rep. 2018, 45, 2535–2544. [Google Scholar] [CrossRef] [PubMed]
- Hordyjewska, A.; Ostapiuk, A.; Horecka, A.; Kurzepa, J. Betulin and betulinic acid: Triterpenoids derivatives with a powerful biological potential. Phytochem. Rev. 2019, 18, 929–951. [Google Scholar] [CrossRef]
- Pavlova, N.I.; Savinova, O.V.; Nikolaeva, S.N.; Boreko, E.; Flekhterb, I.B. Antiviral activity of betulin, betulinic and betulonic acids against some enveloped and non-enveloped viruses. Fitoterapia 2003, 75, 489–492. [Google Scholar] [CrossRef]
- Oloyede, O.H.B.; Ajiboye, H.O.; Salawu, M.; Ajiboye, O.T. Influence of oxidative stress on the antibacterial activity of betulin, betulinic acid and ursolic acid. Microb. Pathog. 2017, 111, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Myz, S.A.; Mikhailovskaya, A.V.; Mikhailenko, M.A.; Bulina, M.; Kuznetsova, S.; Shakhtshneider, T.P. New crystalline betulin-based materials: Improving betulin solubility via cocrystal formation. Mater. Today: Proc. 2019, 12, 82–85. [Google Scholar] [CrossRef]
- Grigoreva, A.; Kolobova, E.; Pakrieva, E.; Mäki-Arvela, P.; Kuznetsova, S.; Carabineiro, S.A.C.; Bogdanchikova, N.; Pestryakov, A.; Murzin, D.Y. Liquid-phase oxidation of betulin over supported Ag NPs catalysts: Kinetic regularities, catalyst deactivation and reactivation. Mol. Catal. 2022, 528, 112461. [Google Scholar] [CrossRef]
- Asmaa, M.F.; Amr, M.; Medhat, A.I. Experimental and theoretical studies of some propiolate esters derivatives. J. Mol. Struct. 2021, 1236, 130281. [Google Scholar]
- Boryczka, S.; Bębenek, E.; Wietrzyk, J.; Kempińska, K.; Jastrzębska, M.; Kusz, J.; Nowak, M. Synthesis, structure and cytotoxic activity of new acetylenic derivatives of betulin. Molecules 2013, 18, 4526–4543. [Google Scholar] [CrossRef]
- Abdelli, A.; Azzouni, S.; Plais, R.; Gaucher, A.; Efrit, M.L.; Prim, D. Recent advances in the chemistry of 1,2,4-triazoles: Synthesis, reactivity and biological activities. Tetrahedron Lett. 2021, 86, 153518. [Google Scholar] [CrossRef]
- Xu, X.; Zhao, S.J.; Liu, Y. 1,2,3-Triazole-containing hybrids as potential anticancer agents:Current developments, action mechanisms and structure-activity relationships. Eur. J. Med. Chem. 2019, 183, 111700. [Google Scholar] [CrossRef] [PubMed]
- Kuczynska, K.; Bończak, B.; Rárová, L.; Kvasnicová, M.; Strnad, M.; Pakulski, Z.; Cmoch, P.; Fiałkowski, M. Synthesis and cytotoxic activity of 1,2,3-triazoles derived from 2,3-seco-dihydrobetulin via a click chemistry approach. J. Mol. Struct. 2022, 1250, 131751. [Google Scholar] [CrossRef]
- Kadela-Tomanek, M.; Jastrzębska, M.; Marciniec, K.; Chrobak, E.; Bębenek, E.; Latocha, M.; Kuśmierz, D.; Boryczka, S. Design, synthesis and biological activity of 1,4-quinone moiety attached to betulin derivatives as potent DT-diaphorase substrate. Bioorg. Chem. 2021, 106, 104478. [Google Scholar] [CrossRef]
- Chrobak, E.; Kadela-Tomanek, M.; Bębenek, E.; Marciniec, K.; Wietrzyk, J.; Trynda, J.; Pawełczak, B.; Kusz, J.; Kasperczyk, J.; Chodurek, E.; et al. New phosphate derivatives of betulin as anticancer agents: Synthesis, crystal structure, and molecular docking study. Bioorg. Chem. 2019, 87, 613–628. [Google Scholar] [CrossRef]
- Bębenek, E.; Kadela-Tomanek, M.; Chrobak, E.; Latocha, M.; Boryczka, S. Novel triazoles of 3-acetylbetulin and betulone as anticancer agents. Med. Chem. Res. 2018, 27, 2051–2061. [Google Scholar] [CrossRef]
- Bębenek, E.; Kadela-Tomanek, M.; Chrobak, E.; Wietrzyk, J.; Sadowska, J.; Boryczka, S. New acetylenic derivatives of betulin and betulone, synthesis and cytotoxic activity. Med. Chem. Res. 2017, 26, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bębenek, E.; Jastrzębska, M.; Kadela-Tomanek, M.; Chrobak, E.; Orzechowska, B.; Zwolińska, K.; Latocha, M.; Mertas, A.; Czuba, Z.; Boryczka, S. Novel triazole hybrids of betulin: Synthesis and biological activity profile. Molecules 2017, 22, 1876–1898. [Google Scholar] [CrossRef]
- Wang, R.; Li, Y.; Hu, H.; Persoons, L.; Daelemans, D.; De Jonghe, S.; Luyten, W.; Krasniqi, B.; Dehaen, W. Antibacterial and antitumoral properties of 1,2,3-triazolo fused triterpenes and their mechanism of inhibiting the proliferation of HL-60 cells. Eur. J. Med. Chem. 2021, 224, 113727. [Google Scholar] [CrossRef]
- Wang, H.; Xu, R.; Shi, Y.; Si, L.; Jiao, P.; Fan, Z.; Han, H.; Wu, X.; Zhou, X.; Yu, F.; et al. Design, synthesis and biological evaluation of novel l-ascorbic acid-conjugated pentacyclic triterpene derivatives as potential influenza virus entry inhibitors. Eur. J. Med. Chem. 2016, 110, 376–388. [Google Scholar] [CrossRef]
- Gonzalez, G.; Hodoň, J.; Kazakova, A.; D’Acunto, C.; Kaňovský, P.; Urban, M.; Strnad, M. Novel pentacyclic triterpenes exhibiting strong neuroprotective activity in SH-SY5Y cells in salsolinol- and glutamate-induced neurodegeneration models. Eur. J. Med. Chem. 2021, 213, 113168. [Google Scholar] [CrossRef]
- Liao, C.R.; Kuo, Y.H.; Ho, Y.L.; Wang, C.Y.; Yang, C.S.; Lin, C.W.; Chang, Y.S. Studies on cytotoxic constituents from the leaves of Elaeagnus oldhamii Maxim. in non-small cell lung cancer A549 cells. Molecules 2014, 19, 9515–9534. [Google Scholar] [CrossRef] [PubMed]
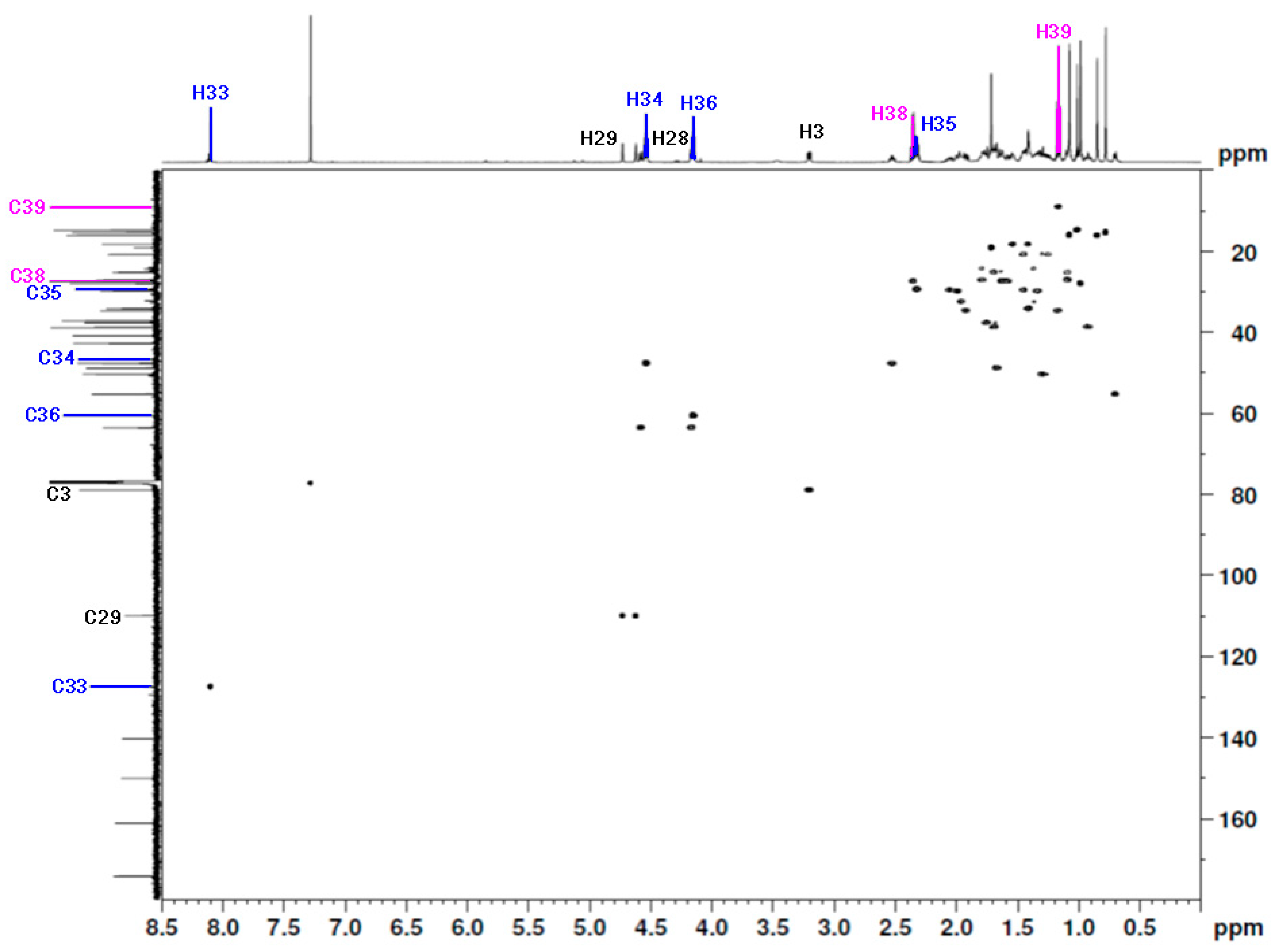
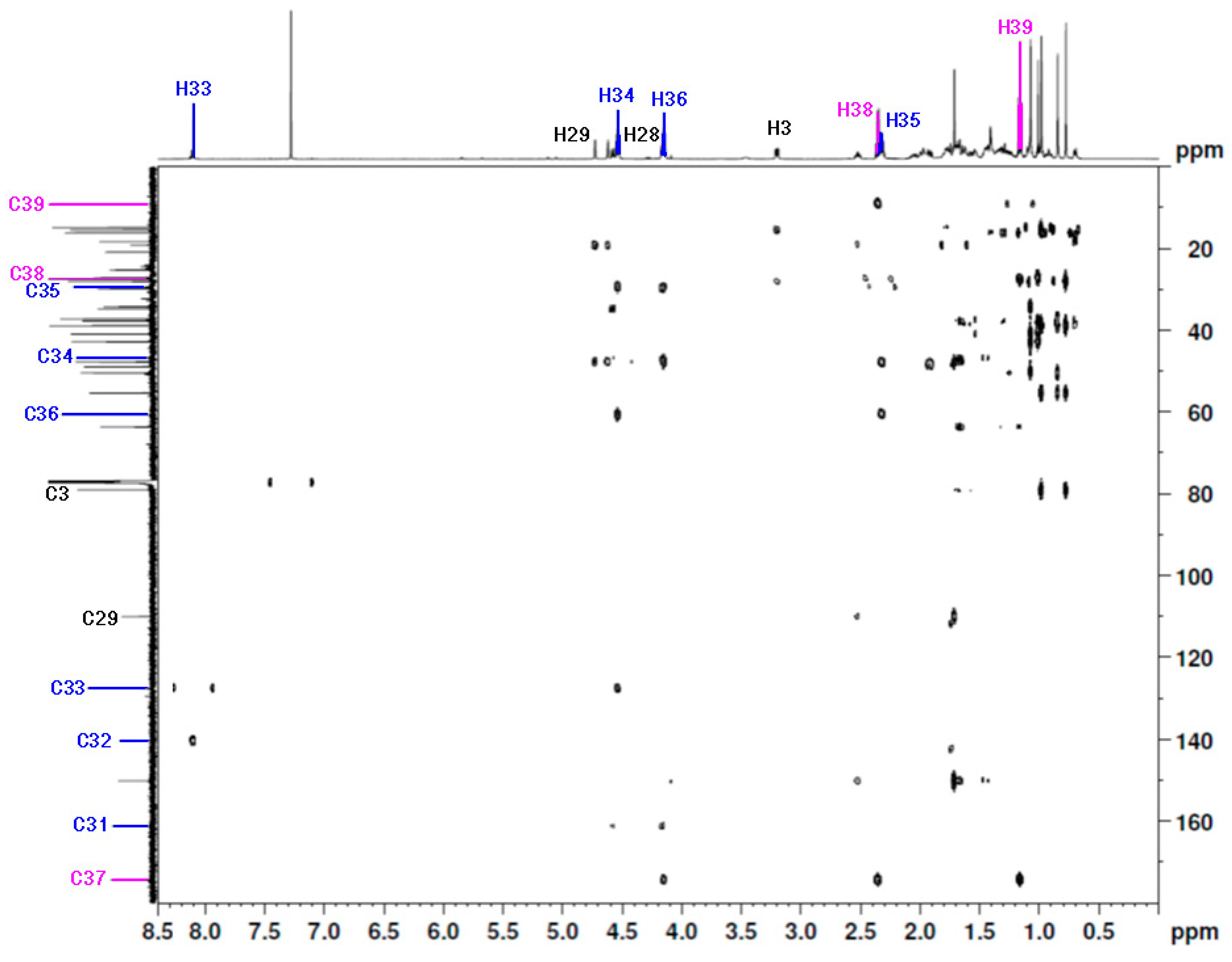
| Proton | 1H NMR δ [ppm] | HSQC | Carbon | 13C NMR δ [ppm] | HMBC |
|---|---|---|---|---|---|
| H39 | 1.16 | H39(1.16)-C39(9.1) | C39 | 9.1 | H39(1.16)-C38(27.4) H39(1.16)-C37(174.2) |
| H38 | 2.36 | H38(2.36)-C38(27.4) | C38 | 27.4 | H38(2.36)-C39(9.1) H38(2.36)-C37(174.2) |
| - | - | - | C37 | 174.2 | C37(174.2)-H39(1.16) C37(174.2)-H38(2.36) C37(174.2)-H36(4.15) |
| H36 | 4.15 | H36(4.15)-C36(60.6) | C36 | 60.6 | H36(4.15)-C37(174.2) H36(4.15)-C35(29.4) |
| H35 | 2.32 | H35(2.32)-C35(29.4) | C35 | 29.4 | H35(2.32)-C34(47.7) H35(2.32)-C36(60.6) |
| H34 | 4.54 | H34(4.54)-C34(47.7) | C34 | 47.7 | H34(4.54)-C33(127.5) H34(4.54)-C36(60.6) H34(4.54)-C35(29.4) |
| H33 | 8.10 | H33(8.10)-C33(127.5) | C33 | 127.5 | H33(8.10)-C32(140.3) |
| - | - | - | C32 | 140.3 | C32(140.3)-H33(8.10) |
| - | - | - | C31 | 161.1 | C31(161.1)-H28b(4.59) C31(161.1)-H28a(4.15) |
| H29a | 4.73 | H29a(4.73)-C29(110.0) | C29 | 110.0 | H29a(4.73)-Cbet(47.7) H29a(4.73)-Cbet(19.1) |
| H29b | 4.62 | H29a(4.62)-C29(110.0) | C29 | 110.0 | H29b(4.62)-Cbet(47.7) H29b(4.62)-Cbet(19.1) |
| H28a | 4.59 | H28a(4.59)-C28(63.6) | C28 | 63.6 | H28a(4.59)-C31(161.1) |
| H28b | 4.15 | H28a(4.16)-C28(63.6) | C28 | 63.6 | H28b(4.15)-C31(161.1) |
| H3 | 3.21 | H3(3.21)-C3(79.0) | C3 | 79.0 | H3(3.21)-Cbet(28.0) H3(3.21)-Cbet(15.4) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kadela-Tomanek, M.; Chrobak, E.; Bębenek, E. 28-[1-(3-(Propionyloxy)propyl)-1H-1,2,3-triazol-4-yl]carbonylbetulin. Molbank 2022, 2022, M1487. https://doi.org/10.3390/M1487
Kadela-Tomanek M, Chrobak E, Bębenek E. 28-[1-(3-(Propionyloxy)propyl)-1H-1,2,3-triazol-4-yl]carbonylbetulin. Molbank. 2022; 2022(4):M1487. https://doi.org/10.3390/M1487
Chicago/Turabian StyleKadela-Tomanek, Monika, Elwira Chrobak, and Ewa Bębenek. 2022. "28-[1-(3-(Propionyloxy)propyl)-1H-1,2,3-triazol-4-yl]carbonylbetulin" Molbank 2022, no. 4: M1487. https://doi.org/10.3390/M1487
APA StyleKadela-Tomanek, M., Chrobak, E., & Bębenek, E. (2022). 28-[1-(3-(Propionyloxy)propyl)-1H-1,2,3-triazol-4-yl]carbonylbetulin. Molbank, 2022(4), M1487. https://doi.org/10.3390/M1487






