2,3,4,5,6-Pentabromobenzoic Acid
Abstract
1. Introduction
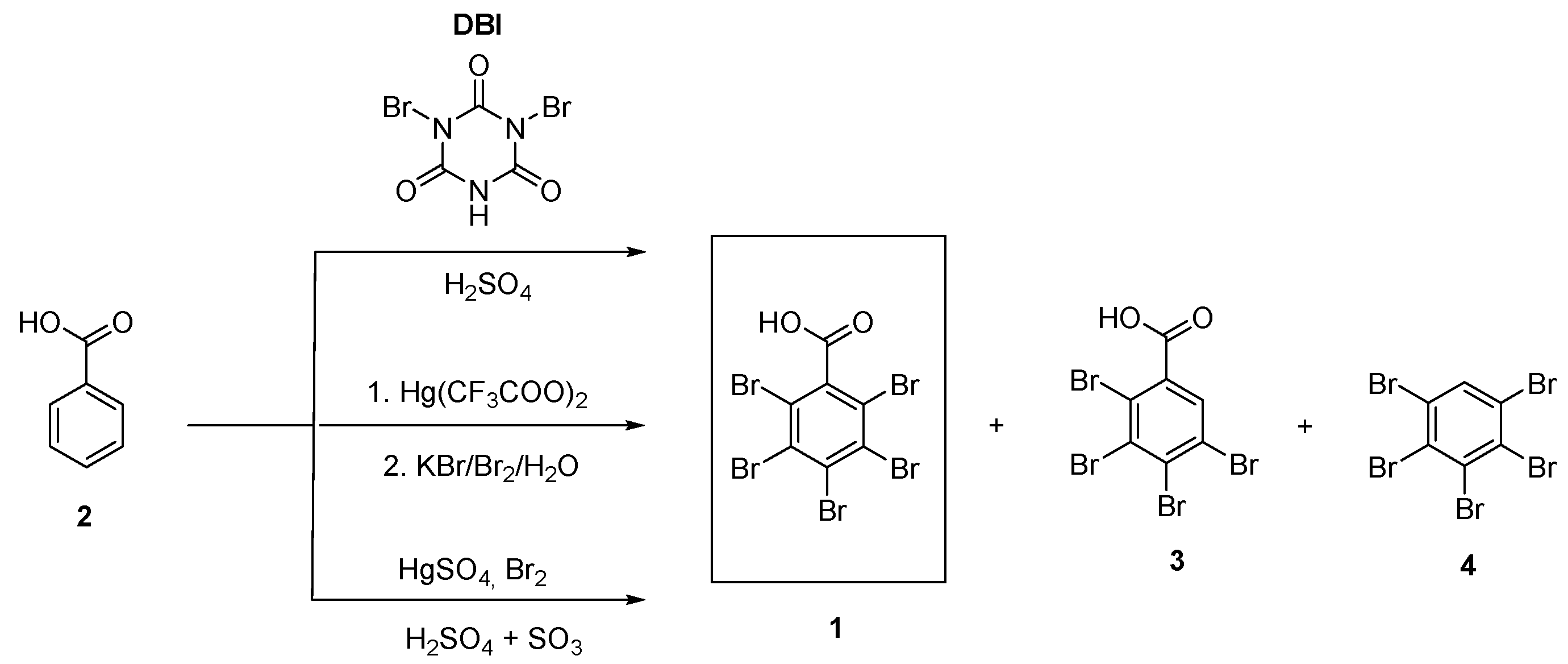
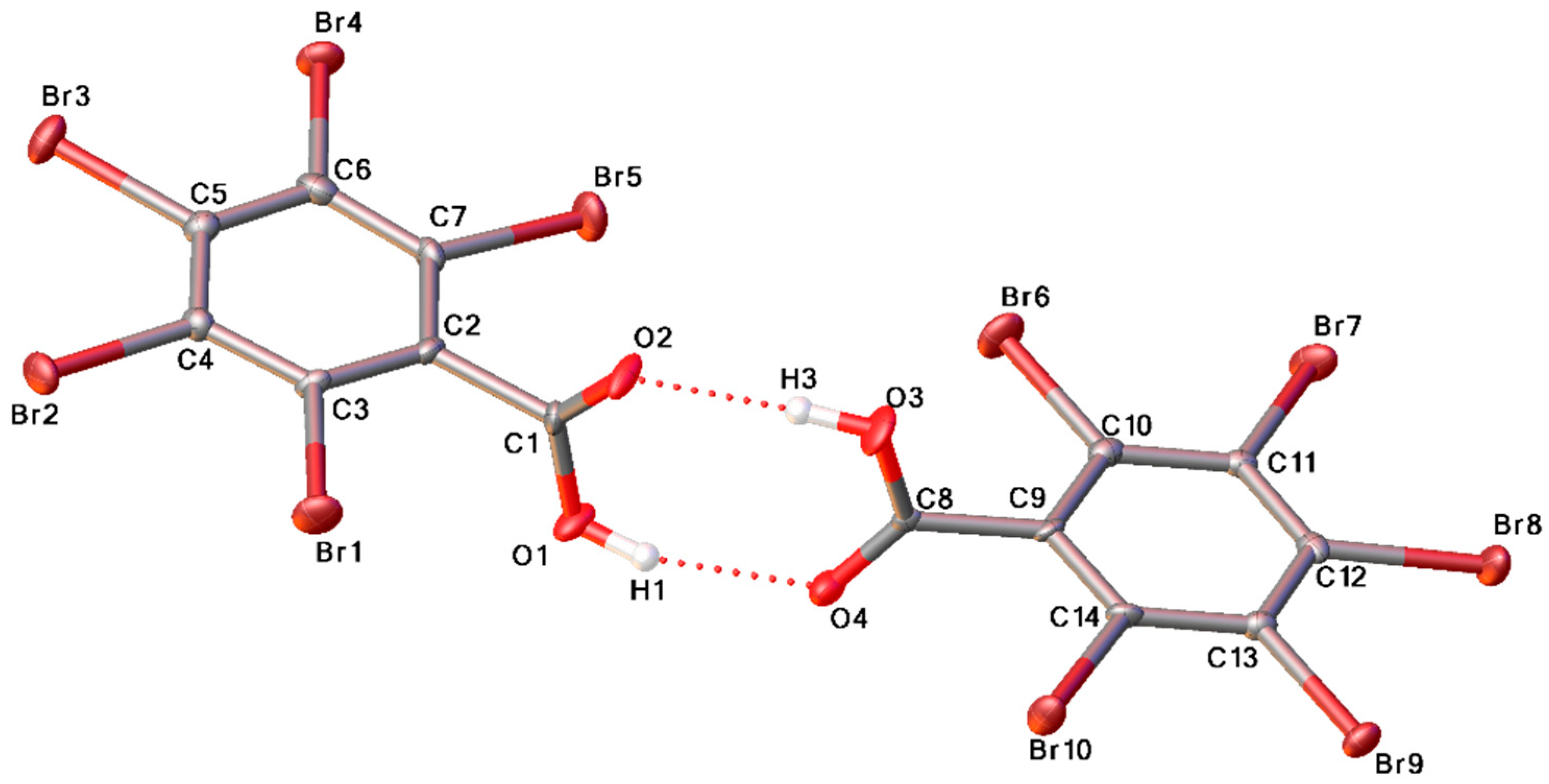
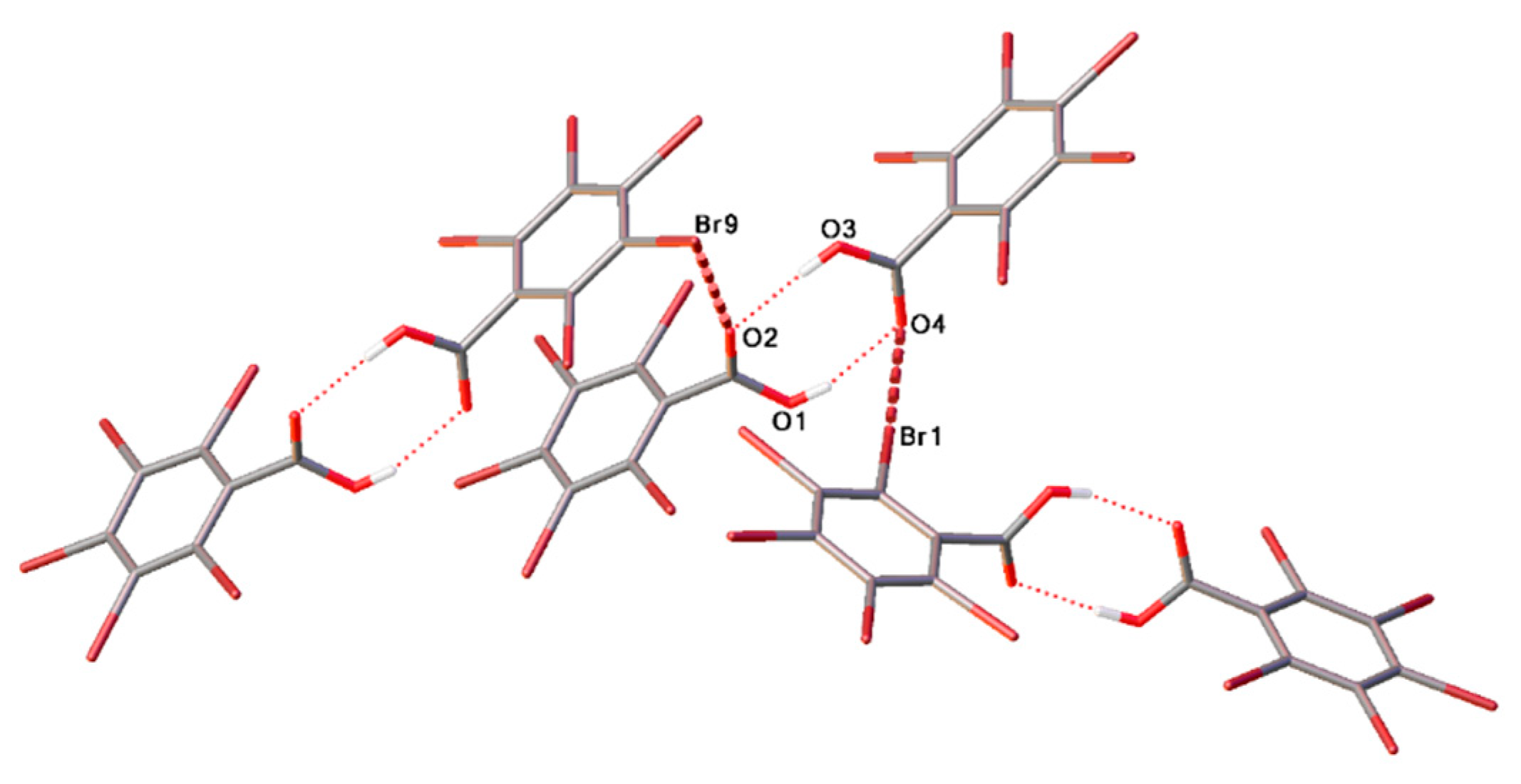
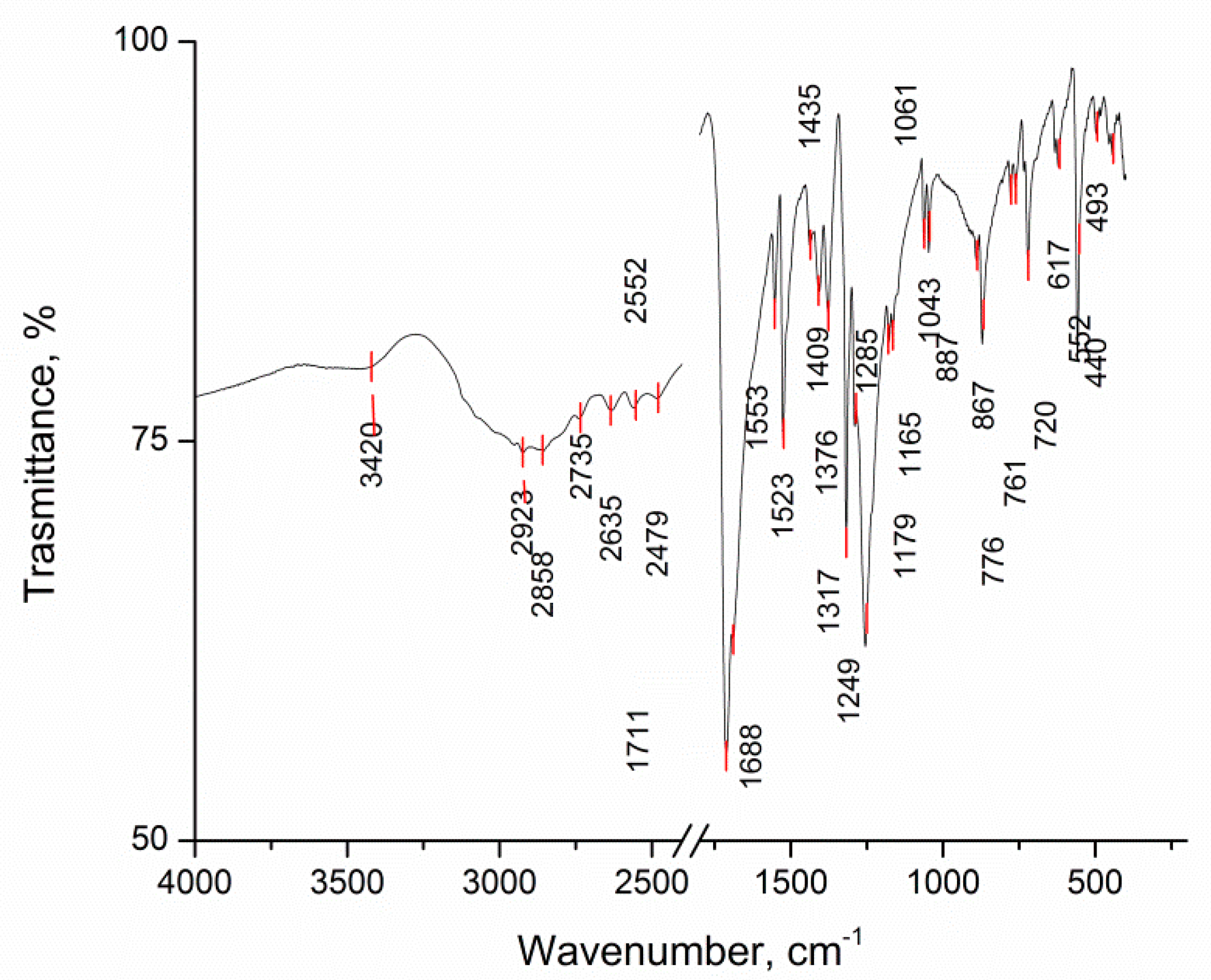
2. Results and Discussion
3. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Han, L.-J.; Kong, Y.-J.; Xu, Y.-Y.; Huang, M.-M. A Zn-Based Coordination Compound for Fluorescence Detection of Fe3+, Cu2+, Ni2+ and CrO42− Ions. Polyhedron 2021, 193, 114868. [Google Scholar] [CrossRef]
- Shmelev, M.A.; Gogoleva, N.V.; Kuznetsova, G.N.; Kiskin, M.A.; Voronina, Y.K.; Yakushev, I.A.; Ivanova, T.M.; Nelyubina, Y.V.; Sidorov, A.A.; Eremenko, I.L. Cd(II) and Cd(II)–Eu(III) Complexes with Pentafluorobenzoic Acid Anions and N-Donor Ligands: Synthesis and Structures. Russ. J. Coord. Chem. 2020, 46, 557–572. [Google Scholar] [CrossRef]
- Cox, B.; Duffy, J.; Zdorichenko, V.; Bellanger, C.; Hurcum, J.; Laleu, B.; Booker-Milburn, K.I.; Elliott, L.D.; Robertson-Ralph, M.; Swain, C.J.; et al. Escaping from Flatland: Antimalarial Activity of Sp3-Rich Bridged Pyrrolidine Derivatives. ACS Med. Chem. Lett. 2020, 11, 2497–2503. [Google Scholar] [CrossRef]
- Černý, I.; Buděšínský, M.; Pouzar, V.; Vyklický, V.; Krausová, B.; Vyklický, L. Neuroactive Steroids with Perfluorobenzoyl Group. Steroids 2012, 77, 1233–1241. [Google Scholar] [CrossRef]
- Shmelev, M.A.; Kiskin, M.A.; Voronina, J.K.; Babeshkin, K.A.; Efimov, N.N.; Varaksina, E.A.; Korshunov, V.M.; Taydakov, I.V.; Gogoleva, N.V.; Sidorov, A.A.; et al. Molecular and Polymer Ln2M2 (Ln = Eu, Gd, Tb, Dy; M = Zn, Cd) Complexes with Pentafluorobenzoate Anions: The Role of Temperature and Stacking Effects in the Structure; Magnetic and Luminescent Properties. Materials 2020, 13, 5689. [Google Scholar] [CrossRef]
- Zhang, H.; Xiao, H.; Liu, F.; Huo, F.; He, Y.; Chen, Z.; Liu, X.; Bo, S.; Qiu, L.; Zhen, Z. Synthesis of Novel Nonlinear Optical Chromophores: Achieving Enhanced Electro-Optic Activity and Thermal Stability by Introducing Rigid Steric Hindrance Groups into the Julolidine Donor. J. Mater. Chem. C 2017, 5, 1675–1684. [Google Scholar] [CrossRef]
- Wu, W.; Huang, Q.; Zhong, C.; Ye, C.; Qin, J.; Li, Z. Second-Order Nonlinear Optical (NLO) Polymers Containing Perfluoroaromatic Rings as Isolation Groups with Ar/ArF Self-Assembly Effect: Enhanced NLO Coefficient and Stability. Polymer 2013, 54, 5655–5664. [Google Scholar] [CrossRef]
- Shang, R.; Fu, Y.; Wang, Y.; Xu, Q.; Yu, H.-Z.; Liu, L. Copper-Catalyzed Decarboxylative Cross-Coupling of Potassium Polyfluorobenzoates with Aryl Iodides and Bromides. Angew. Chem. Int. Ed. 2009, 48, 9350–9354. [Google Scholar] [CrossRef]
- Mattern, D.L. Direct Aromatic Periodination. J. Org. Chem. 1984, 49, 3051–3053. [Google Scholar] [CrossRef]
- Pearson, D.E.; Cowan, D.; Beckler, J.D. A Study of the Entrainment Method for Making Grignard Reagents. J. Org. Chem. 1959, 24, 504–509. [Google Scholar] [CrossRef]
- Taydakov, I.V.; Kiskin, M.A. On the Hydrolysis of Diethyl 2-(Perfluorophenyl)Malonate. Beilstein J. Org. Chem. 2020, 16, 1863–1868. [Google Scholar] [CrossRef] [PubMed]
- Lutoshkin, M.A.; Taydakov, I.V. Selenoyl-Trifluoroacetone: Synthesis, Properties, and Complexation Ability towards Trivalent Rare-Earth Ions. Polyhedron 2021, 207, 115383. [Google Scholar] [CrossRef]
- Gusev, A.; Kiskin, M.; Lutsenko, I.; Svetogorov, R.; Veber, S.; Minakova, O.; Korshunov, V.; Taydakov, I.; Linert, W. Triazole-Based Lanthanide(III) Adducts: Photo- and Thermochromic Luminescence. J. Lumin. 2021, 238, 118305. [Google Scholar] [CrossRef]
- Belousov, Y.A.; Drozdov, A.A.; Taydakov, I.V.; Marchetti, F.; Pettinari, R.; Pettinari, C. Lanthanide Azolecarboxylate Compounds: Structure, Luminescent Properties and Applications. Coord. Chem. Rev. 2021, 445, 214084. [Google Scholar] [CrossRef]
- Gottardi, W. Über Bromierungen mit Dibromoisocyanursaüre unter ionischen Bedingungen, 2. Mitt.: Perbromierungen. Mon. Chem. 1969, 100, 42–50. [Google Scholar] [CrossRef]
- Reinecke, A. Ueber Mono-, Tri-Und Pentabrombenzoesäure. Z. Fur Chem. 1869, 5, 109–111. [Google Scholar]
- Gottardi, W. Über Bromierungen mit Dibromoisocyanursäure unter ionischen Bedingungen, 1. Mitt.: Monobromierungen. Mon. Chem. 1968, 99, 815–822. [Google Scholar] [CrossRef]
- Deacon, G.B.; Farquharson, G.J. Permercurated Arenes. J. Organomet. Chem. 1974, 67, C1–C3. [Google Scholar] [CrossRef]
- Deacon, G.; Farquharson, G. Synthesis and Bromodemercuration of Some Permercurated Arenes. Aust. J. Chem. 1976, 29, 627. [Google Scholar] [CrossRef]
- Shishkin, V.N.; Bolusheva, I.Y.; Lapin, K.K.; Tanaseichuk, B.S. Polybrominated Aromatic Compounds VI. Exhaustive Bromination in the Ring of Deactivated Aromatic Compounds. J. Org. Chem. USSR 1991, 27, 1303–1308. [Google Scholar]
- Mongin, F.; Marzi, E.; Schlosser, M. Extensive Halogen Scrambling and Buttressing Effects Encountered upon Treatment of Oligobromoarenes with Bases. Eur. J. Org. Chem. 2001, 2001, 2771–2777. [Google Scholar] [CrossRef]
- Kumagai, H.; Kawata, S. 4,4′-Bipyridine-1,1′-Diium 2,3,5,6-Tetrabromoterephthalate Dihydrate. Acta Crystallogr. Sect. E Struct. Rep. Online 2011, 67, o2636. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. SHELXT—Integrated Space-Group and Crystal-Structure Determination. Acta Crystallogr. Sect. A Found. Adv. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A Complete Structure Solution, Refinement and Analysis Program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Demko, Z.P.; Bartsch, M.; Sharpless, K.B. Primary Amides. A General Nitrogen Source for Catalytic Asymmetric Aminohydroxylation of Olefins. Org. Lett. 2000, 2, 2221–2223. [Google Scholar] [CrossRef]
- Gottardi, W. Über die Reaktion von Brom mit Alkalicyanuraten. Mon. Chem. 1967, 98, 507–512. [Google Scholar] [CrossRef]
- Petterson, R.C.; Grzeskowiak, U.; Jules, L.H. N-Halogen Compounds. II. 1,2 The N—Cl Stretching Band in Some N-Chloroamides. The Structure of Trichloroisocyanuric Acid. J. Org. Chem. 1960, 25, 1595–1598. [Google Scholar] [CrossRef]
- Gottardi, W. Zur Chemie der Bromisocyanursäuren: Eigenschaften und Reaktionen der Dibromisocyanursäure. Mon. Chem. 1977, 108, 1067–1084. [Google Scholar] [CrossRef]

| Bond | Length, Å | Bond | Length, Å |
|---|---|---|---|
| Br1–C3 | 1.877 (8) | Br10–C14 | 1.871 (8) |
| Br2–C4 | 1.875 (10) | Br9–C13 | 1.883 (9) |
| Br3–C5 | 1.892 (9) | Br8–C12 | 1.904 (9) |
| Br4–C6 | 1.865 (9) | Br7–C11 | 1.874 (9) |
| Br5–C7 | 1.889 (10) | Br6–C10 | 1.870 (9) |
| O1–C1 | 1.316 (11) | O3–C8 | 1.319 (12) |
| O2–C1 | 1.189 (11) | O4–C8 | 1.215 (11) |
| C1–C2 | 1.525 (13) | C8–C9 | 1.486 (13) |
| C2–C3 | 1.389 (12) | C9–C14 | 1.381 (12) |
| C2–C7 | 1.389 (13) | C9–C10 | 1.404 (12) |
| C3–C4 | 1.388 (13) | C14–C13 | 1.407 (13) |
| C4–C5 | 1.376 (13) | C13–C12 | 1.395 (12) |
| C5–C6 | 1.401 (14) | C12–C11 | 1.366 (13) |
| C6–C7 | 1.375 (14) | C11–C10 | 1.408 (13) |
| Bond | O…O, Å | ∠O–H…O, o |
|---|---|---|
| O1-H1…O4 | 2.707 | 158.9 |
| O3-H3…O2 | 2.678 | 176.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taydakov, I.V.; Vlasova, T.S.; Belousov, Y.A.; Zanizdra, S.R.; Gontcharenko, V.E. 2,3,4,5,6-Pentabromobenzoic Acid. Molbank 2022, 2022, M1455. https://doi.org/10.3390/M1455
Taydakov IV, Vlasova TS, Belousov YA, Zanizdra SR, Gontcharenko VE. 2,3,4,5,6-Pentabromobenzoic Acid. Molbank. 2022; 2022(4):M1455. https://doi.org/10.3390/M1455
Chicago/Turabian StyleTaydakov, Ilya V., Tatiana S. Vlasova, Yury A. Belousov, Sergey R. Zanizdra, and Victoria E. Gontcharenko. 2022. "2,3,4,5,6-Pentabromobenzoic Acid" Molbank 2022, no. 4: M1455. https://doi.org/10.3390/M1455
APA StyleTaydakov, I. V., Vlasova, T. S., Belousov, Y. A., Zanizdra, S. R., & Gontcharenko, V. E. (2022). 2,3,4,5,6-Pentabromobenzoic Acid. Molbank, 2022(4), M1455. https://doi.org/10.3390/M1455






