4,4′-Di-tert-butyl-2,2′-bipyridinium Trifluoromethanesulfonate
Abstract
:1. Introduction
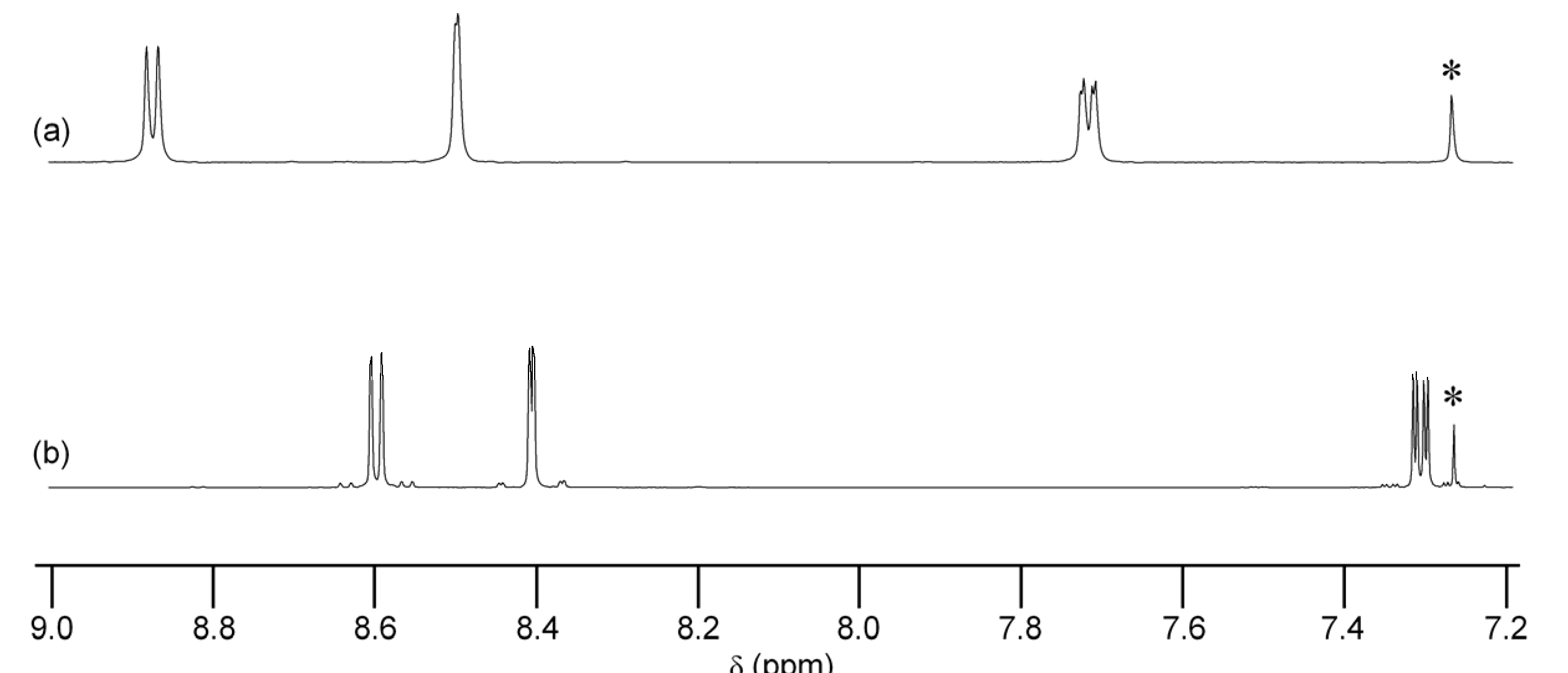
2. Results and Discussion
3. Materials and Methods
3.1. General
3.2. Synthesis of 4,4′-Di-tert-butyl-2,2′-bipyridinium Trifluoromethanesulfonate (2)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Corey, E.J.; Suggs, J.W. Pyridinium chlorochromate. An efficient reagent for oxidation of primary and secondary alcohols to carbonyl compounds. Tetrahedron Lett. 1975, 16, 2647–2650. [Google Scholar] [CrossRef]
- Nugent, B.M.; Yoder, R.A.; Johnston, J.N. Chiral Proton Catalysis: A Catalytic Enantioselective Direct Aza-Henry Reaction. J. Am. Chem. Soc. 2004, 126, 3418–3419. [Google Scholar] [CrossRef] [PubMed]
- Takenaka, N.; Chen, J.; Captain, B.; Sarangthem, R.S.; Chandrakumar, A. Helical Chiral 2-Aminopyridinium Ions: A New Class of Hydrogen Bond Donor Catalysts. J. Am. Chem. Soc. 2010, 132, 4536–4537. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, Y.; Nakano, S.; Tahira, Y.; Terazawa, K.; Yamazaki, K.; Kitamura, C.; Hara, O. Chiral Pyridinium Phosphoramide as a Dual Brønsted Acid Catalyst for Enantioselective Diels–Alder Reaction. Org. Lett. 2016, 18, 2004–2007. [Google Scholar] [CrossRef]
- Ling, R.; Yoshida, M.; Mariano, P.S. Exploratory Investigations Probing a Preparatively Versatile, Pyridinium Salt Photoelectrocyclization−Solvolytic Aziridine Ring Opening Sequence. J. Org. Chem. 1996, 61, 4439–4449. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Meng, Z.; Chen, C.-F. Acid/base controllable complexation of a triptycene-derived macrotricyclic host and protonated 4,4′-bipyridinium/pyridinium salts. Chem. Commun. 2016, 52, 590–593. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Shi, Q.; Han, Y.; Chen, C.-F. Complexation of 2,6-helic[6]arene and its derivatives with 1,1′- dimethyl-4,4′-bipyridinium salts and protonated 4,4’-bipyridinium salts: An acid−base controllable complexation. Beilstein J. Org. Chem. 2019, 15, 1795–1804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pannwitz, A.; Wenger, O.S. Photoinduced Electron Transfer Coupled to Donor Deprotonation and Acceptor Protonation in a Molecular Triad Mimicking Photosystem II. J. Am. Chem. Soc. 2017, 139, 13308–13311. [Google Scholar] [CrossRef]
- Beattie, I.R.; Webster, M. The Base Strengths of 2,2′-Bipyridyl and 1,10-Phenanthroline. J. Phys. Chem. 1962, 66, 115–116. [Google Scholar] [CrossRef]
- Guziec, F.S.; Luzzio, F.A. The Oxidation of Alcohols Using 2,2’-Bipyridinium Chlorochromate. Synthesis 1980, 9, 691–694. [Google Scholar] [CrossRef]
- Luzzio, F.A.; Bobb, R.A. 2,2′-Bipyrldinium Chlorochromate/m-Chloroperbenzoic Acid-Mediated Cleavage of Cyclic Acetals to Hydroxyesters. Tetrahedron Lett. 1997, 38, 1733–1736. [Google Scholar] [CrossRef]
- Adachi, K.; Ohira, Y.; Tomizawa, G.; Ishihara, S.; Oishi, S. Electrophilic fluorination with N,N′-difluoro-2,2′-bipyridinium salt and elemental fluorine. J. Fluorine Chem. 2003, 120, 173–183. [Google Scholar] [CrossRef]
- Milani, B.; Anzilutti, A.; Vicentini, L.; Sessanta o Santi, A.; Zangrando, E.; Geremia, S.; Mestroni, G. Bis-Chelated Palladium(II) Complexes with Nitrogen-Donor Chelating Ligands are Efficient Catalyst Precursors for the CO/Styrene Copolymerization Reaction. Organometallics 1997, 16, 5064–5075. [Google Scholar] [CrossRef]
- Kodama, S.; Hashidate, S.; Nomoto, A.; Yano, S.; Ueshima, M.; Ogawa, A. Vanadium-catalyzed Atmospheric Oxidation of Benzyl Alcohols Using Water as Solvent. Chem. Lett. 2011, 40, 495–497. [Google Scholar] [CrossRef] [Green Version]
- Kodama, S.; Nomoto, A.; Yano, S.; Ueshima, M.; Ogawa, A. Novel Heterotetranuclear V2Mo2 or V2W2 Complexes with 4,4′-Di-tert-butyl-2,2′-bipyridine: Syntheses, Crystal structures, and Catalytic Activities. Inorg. Chem. 2011, 50, 9942–9947. [Google Scholar] [CrossRef] [PubMed]
- Marui, K.; Higashiura, Y.; Kodama, S.; Hashidate, S.; Nomoto, A.; Yano, S.; Ueshima, M.; Ogawa, A. Vanadium-catalyzed green oxidation of benzylic alcohols in water under air atmosphere. Tetrahedron 2014, 70, 2431–2438. [Google Scholar] [CrossRef] [Green Version]
- Kodama, S.; Taya, N.; Ishii, Y. A Novel Octanuclear Vanadium(V) Oxide Cluster Complex Having an Unprecedented Neutral V8O20 Core Functionalized with 4,4’-Di-tert-butyl-2,2’- bipyridine. Inorg. Chem. 2014, 53, 2754–2756. [Google Scholar] [CrossRef] [PubMed]
- Kodama, S.; Taya, N.; Inoue, Y.; Ishii, Y. Synthesis and Interconversion of V4, V7, and V8 Oxide Clusters: Unexpected Formation of Neutral Heptanuclear Oxido(alkoxido)vanadium(V) Clusters [V7O17(OR)(4,4’-tBubpy)3] (R = Et, MeOC2H4). Inorg. Chem. 2016, 55, 6712–6718. [Google Scholar] [CrossRef]
- Kobayashi, D.; Kodama, S.; Ishii, Y. An Oxidovanadium(IV) Complex Having a Perrhenato Ligand: An Efficient Catalyst for Aerobic Oxidation Reactions of Benzylic and Propargylic Alcohols. Tetrahedron Lett. 2017, 58, 3306–3310. [Google Scholar] [CrossRef]
- Inoue, Y.; Kodama, S.; Taya, N.; Sato, H.; Oh-ishi, K.; Ishii, Y. Reductive Formation of a Vanadium(IV/V) Oxide Cluster Complex [V8O19(4,4′-tBubpy)3] Having a C3-Symmetric Propeller-Shaped Nonionic V8O19 Core. Inorg. Chem. 2018, 57, 7491–7494. [Google Scholar] [CrossRef]
- Kodama, S.; Kondo, S.; Nomoto, A.; Ogawa, A. Tris(4,4’-di-tert-butyl-2,2’-bipyridine)(trans-4-tert-butylcyclohexanolato)deca-μ-oxido-heptaoxidoheptavanadium acetonitrile monosolvate including another unknown solvent molecule. IUCrData 2020, 5, x200449. [Google Scholar] [CrossRef]
- Kobayashi, S. Scandium Triflate in Organic Synthesis. Eur. J. Org. Chem. 1999, 1999, 15–27. [Google Scholar] [CrossRef]
- Pellissier, H. Recent developments in enantioselective scandium-catalyzed transformations. Coord. Chem. Rev. 2016, 313, 1–37. [Google Scholar] [CrossRef]
- Wabnitz, T.C.; Yu, J.-Q.; Spencer, J.B. Evidence That Protons Can Be the Active Catalysts in Lewis Acid Mediated HeteroMichael Addition Reactions. Chem. Eur. J. 2004, 10, 484–493. [Google Scholar] [CrossRef] [PubMed]
- Šolić, I.; Seankongsuk, P.; Loh, J.K.; Vilaivan, T.; Bates, R.W. Scandium as a pre-catalyst for the deoxygenative allylation of benzylic alcohols. Org. Biomol. Chem. 2018, 16, 119–123. [Google Scholar] [CrossRef] [PubMed]
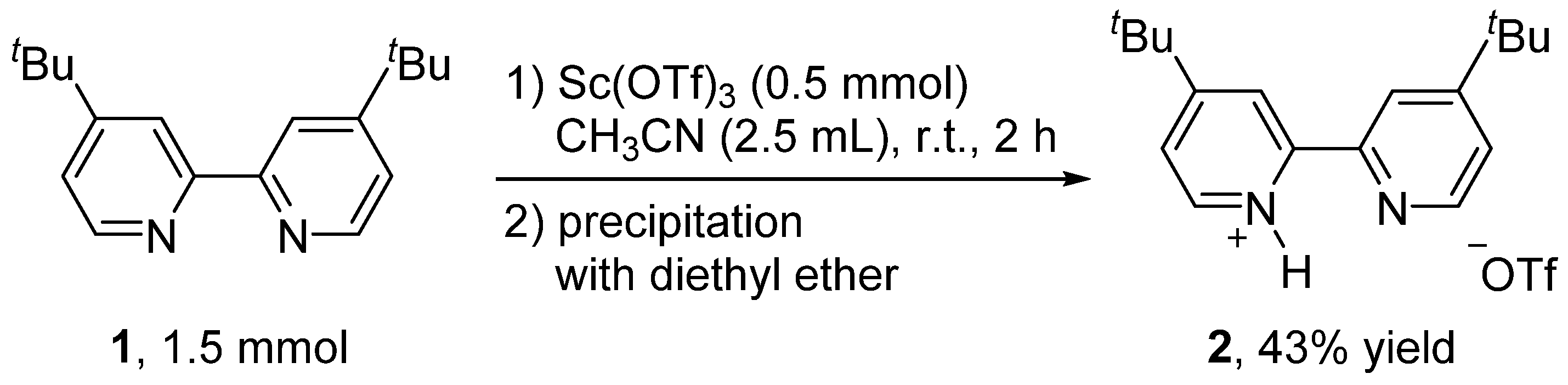
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kodama, S.; Bunno, K.; Nomoto, A.; Ogawa, A. 4,4′-Di-tert-butyl-2,2′-bipyridinium Trifluoromethanesulfonate. Molbank 2021, 2021, M1261. https://doi.org/10.3390/M1261
Kodama S, Bunno K, Nomoto A, Ogawa A. 4,4′-Di-tert-butyl-2,2′-bipyridinium Trifluoromethanesulfonate. Molbank. 2021; 2021(3):M1261. https://doi.org/10.3390/M1261
Chicago/Turabian StyleKodama, Shintaro, Kazuki Bunno, Akihiro Nomoto, and Akiya Ogawa. 2021. "4,4′-Di-tert-butyl-2,2′-bipyridinium Trifluoromethanesulfonate" Molbank 2021, no. 3: M1261. https://doi.org/10.3390/M1261
APA StyleKodama, S., Bunno, K., Nomoto, A., & Ogawa, A. (2021). 4,4′-Di-tert-butyl-2,2′-bipyridinium Trifluoromethanesulfonate. Molbank, 2021(3), M1261. https://doi.org/10.3390/M1261





