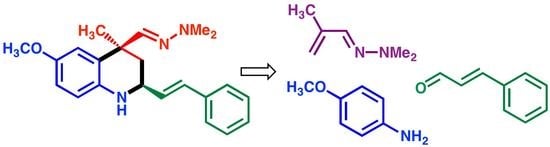
(2S*,4S*)-4-[(E)-(2,2-Dimethylhydrazono)methyl]-6-methoxy-4-methyl-2-[(E)-styryl]-1,2,3,4-tetrahydroquinoline
Abstract
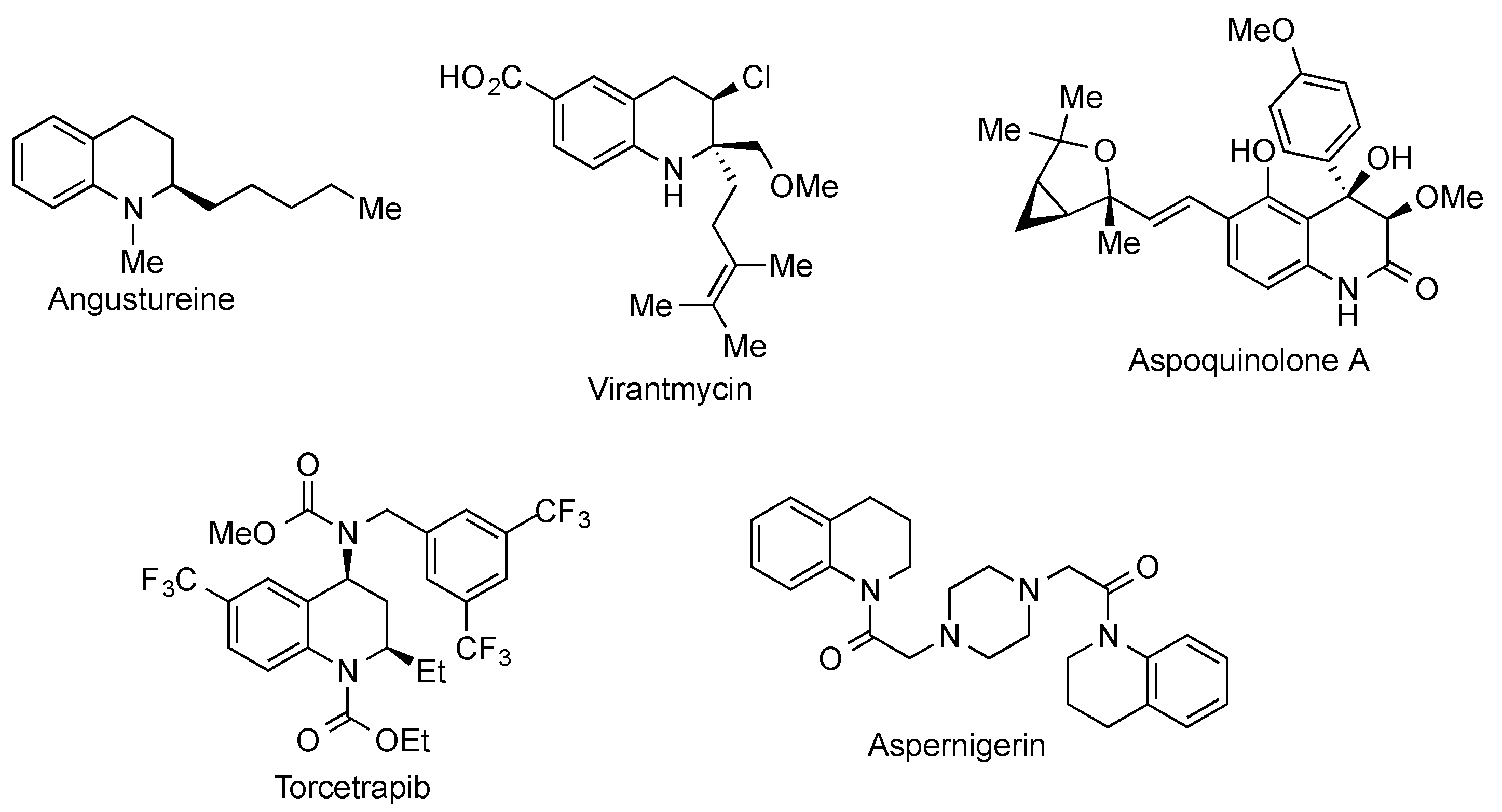
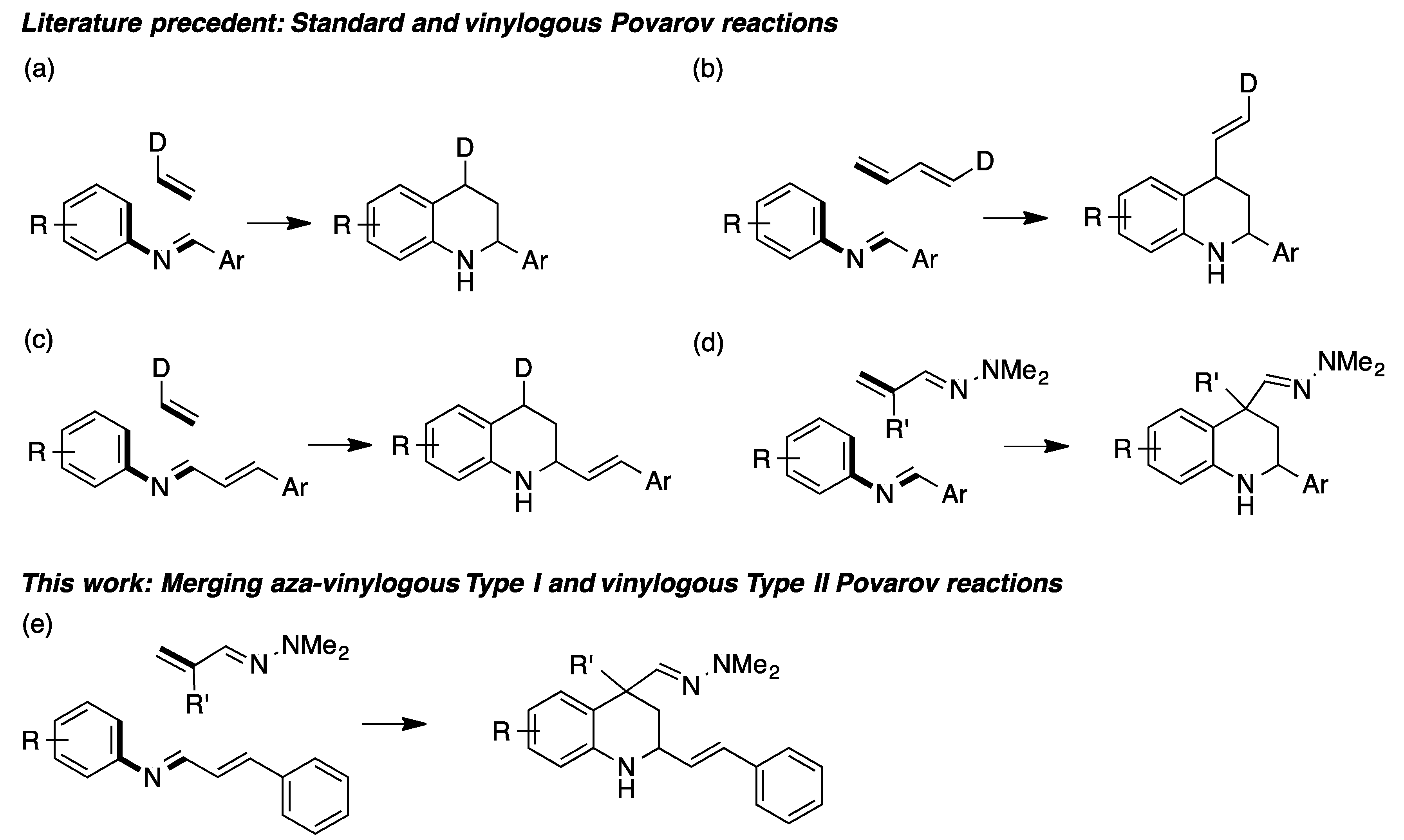
:1. Introduction
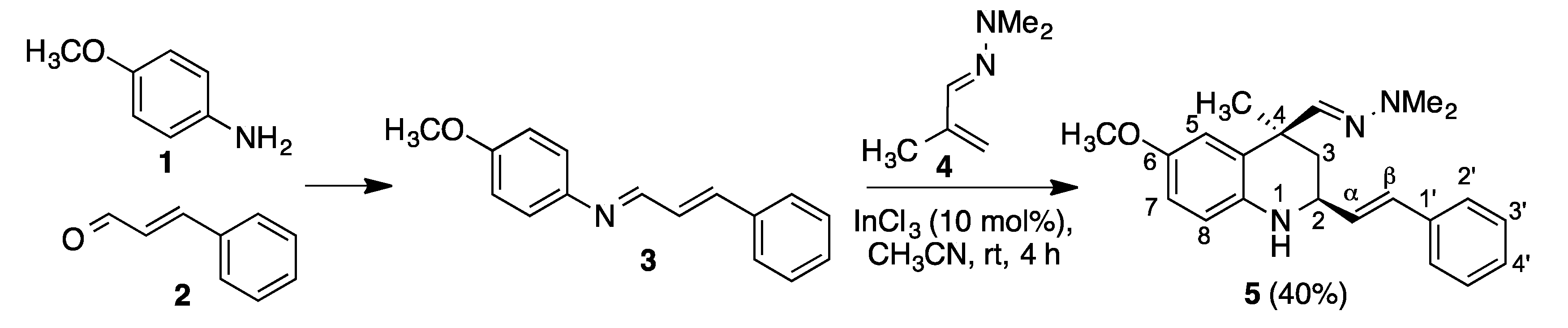
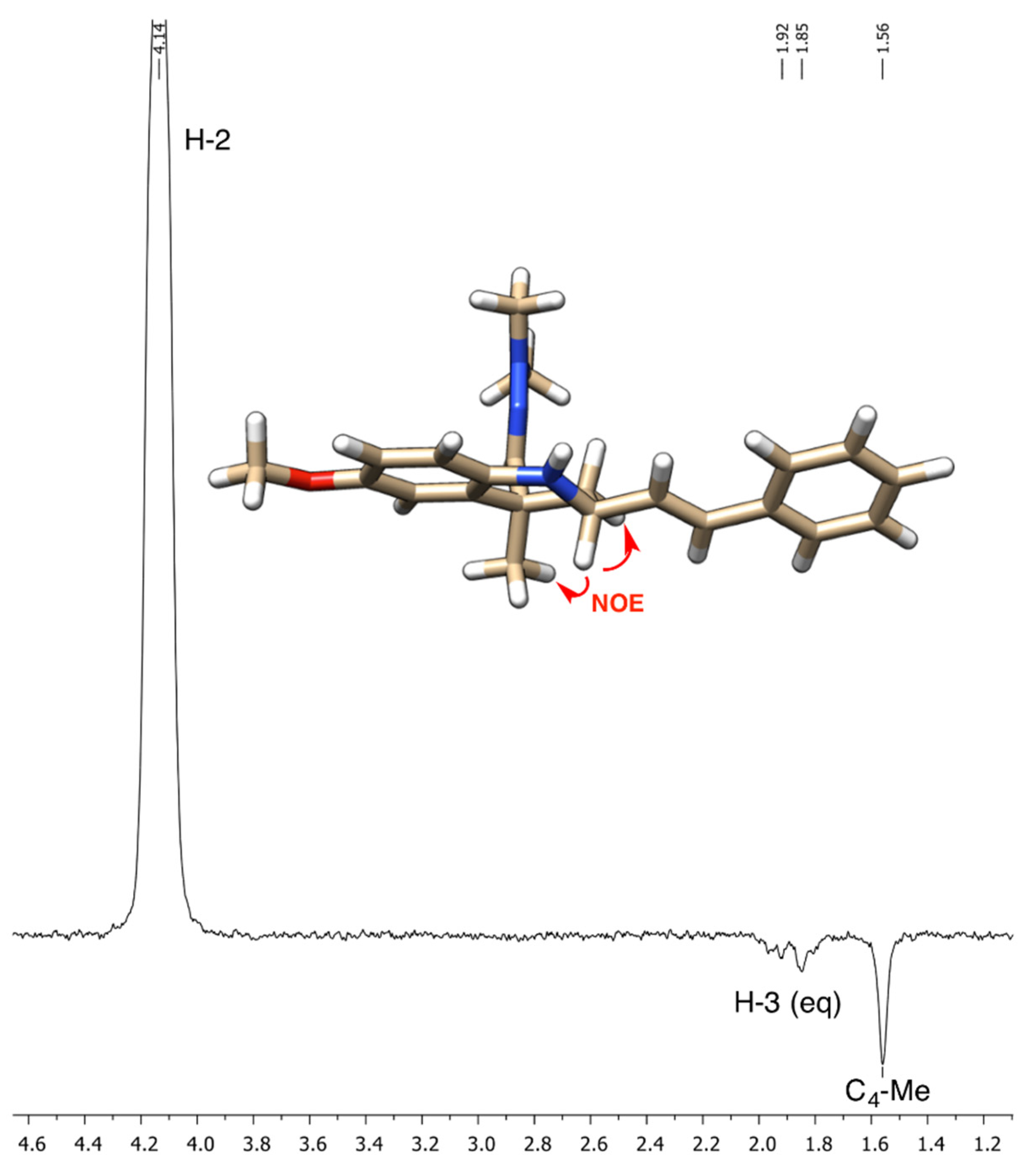
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sridharan, V.; Suryavanshi, P.; Menéndez, J.C. Advances in the chemistry of tetrahydroquinolines. Chem. Rev. 2011, 111, 7157–7259. [Google Scholar] [CrossRef] [PubMed]
- Muthukrishnan, I.; Sridharan, V.; Menéndez, J.C. Progress in the chemistry of tetrahydroquinolines. Chem. Rev. 2019, 119, 5057–5191. [Google Scholar] [CrossRef] [PubMed]
- Katritzky, A.R.; Rachwal, S.; Rachwal, B. Recent progress in the synthesis of 1,2,3,4-tetrahydroquinolines. Tetrahedron 1996, 52, 15031–15070. [Google Scholar] [CrossRef]
- Kouznetsov, V.V. Recent synthetic developments in a powerful imino Diels–Alder reaction (Povarov reaction): Application to the synthesis of N-polyheterocycles and related alkaloids. Tetrahedron 2009, 65, 2721–2750. [Google Scholar] [CrossRef]
- Ghashghaei, O.; Masdeu, C.; Alonso, C.; Palacios, F.; Lavilla, R. Recent advances of the Povarov reaction in medicinal chemistry. Drug Discov. Today Technol. 2018, 29, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Sridharan, V.; Perumal, P.T.; Avendaño, C.; Menéndez, J.C. The first aza Diels–Alder reaction involving an α,β-unsaturated hydrazone as the dienophile: Stereoselective synthesis of C-4 functionalized 1,2,3,4-tetrahydroquinolines containing a quaternary stereocenter. Org. Biomol. Chem. 2007, 5, 1351–1353. [Google Scholar] [CrossRef] [PubMed]
- Sridharan, V.; Ribelles, P.; Estévez, V.; Villacampa, M.; Ramos, M.T.; Perumal, P.T.; Menéndez, J.C. New types of reactivity of α,β-unsaturated N,N-dimethylhydrazones: Chemodivergent, diastereoselective synthesis of functionalized tetrahydroquinolines and hexahydropyrrolo[3,2-b]indoles. Chem. Eur. J. 2012, 18, 5056–5063. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clerigué, J.; Ramos, M.T.; Menéndez, J.C. (2S*,4S*)-4-[(E)-(2,2-Dimethylhydrazono)methyl]-6-methoxy-4-methyl-2-[(E)-styryl]-1,2,3,4-tetrahydroquinoline. Molbank 2021, 2021, M1220. https://doi.org/10.3390/M1220
Clerigué J, Ramos MT, Menéndez JC. (2S*,4S*)-4-[(E)-(2,2-Dimethylhydrazono)methyl]-6-methoxy-4-methyl-2-[(E)-styryl]-1,2,3,4-tetrahydroquinoline. Molbank. 2021; 2021(2):M1220. https://doi.org/10.3390/M1220
Chicago/Turabian StyleClerigué, José, M. Teresa Ramos, and J. Carlos Menéndez. 2021. "(2S*,4S*)-4-[(E)-(2,2-Dimethylhydrazono)methyl]-6-methoxy-4-methyl-2-[(E)-styryl]-1,2,3,4-tetrahydroquinoline" Molbank 2021, no. 2: M1220. https://doi.org/10.3390/M1220
APA StyleClerigué, J., Ramos, M. T., & Menéndez, J. C. (2021). (2S*,4S*)-4-[(E)-(2,2-Dimethylhydrazono)methyl]-6-methoxy-4-methyl-2-[(E)-styryl]-1,2,3,4-tetrahydroquinoline. Molbank, 2021(2), M1220. https://doi.org/10.3390/M1220