2,4,6-Tris(4-Iodophenyl)-1,3,5-trimethylbenzene
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
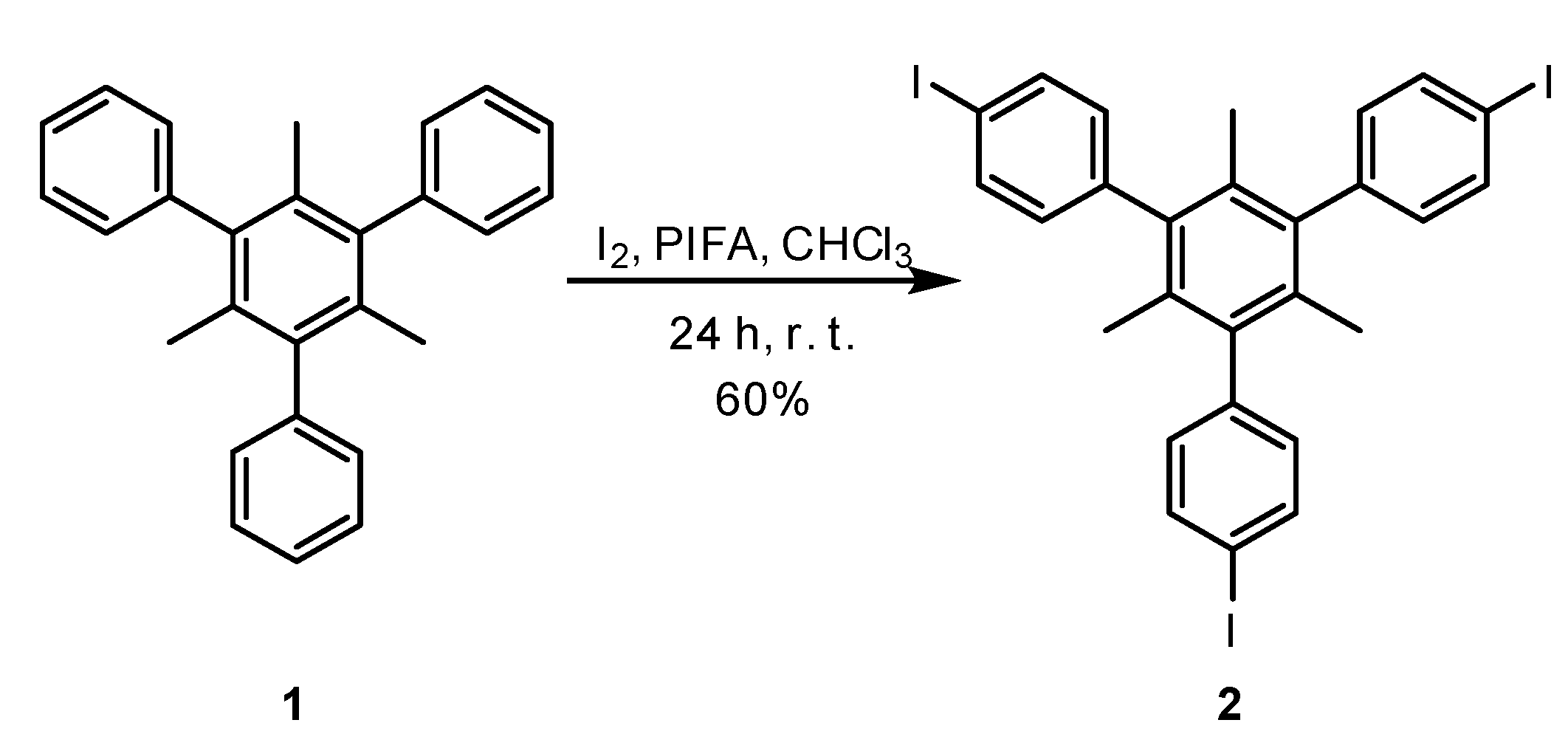
Synthesis of 2,4,6-Tris(4-Iodophenyl)-1,3,5-trimethylbenzene (2)
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Morisaki, Y.; Gon, M.; Tsuji, Y.; Kajiwara, Y.; Chujo, Y. Synthesis and Characterization of [2.2]Paracyclophane-Containing Conjugated Microporous Polymers. Macromol. Chem. Phys. 2012, 213, 572–579. [Google Scholar] [CrossRef]
- Jiao, T.; Chen, L.; Yang, D.; Li, X.; Wu, G.; Zeng, P.; Zhou, A.; Yin, Q.; Pan, Y.; Wu, B.; et al. Trapping White Phosphorus within a Purely Organic Molecular Container Produced by Imine Condensation. Angew. Chem. Int. Ed. 2017, 56, 14545–14550. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yan, X.; Li, T.; Zhang, W.D.; Fu, Q.T.; Lu, H.S.; Wang, X.; Gu, Z.G. Three-dimensional porphyrin-based covalent organic frameworks with tetrahedral building blocks for single-site catalysis. New J. Chem. 2019, 43, 16907–16914. [Google Scholar] [CrossRef]
- Bahrin, L.G.; Bejan, D.; Shova, S.; Gdaniec, M.; Fronc, M.; Lozan, V.; Janiak, C. Alkali- and alkaline-earth metal–organic networks based on a tetra(4-carboxyphenyl)bimesitylene-linker. Polyhedron 2019, 173, 114128. [Google Scholar] [CrossRef]
- Bumstead, A.M.; Cordes, D.B.; Dawson, D.M.; Chakarova, K.K.; Mihaylov, M.Y.; Hobday, C.L.; Duren, T.; Hadjiivanov, K.I.; Slawin, A.M.Z.; Ashbrook, S.E.; et al. Modulator-Controlled Synthesis of Microporous STA-26, an interpenetrated 8,3-Connected Zirconium MOF with the the-iTopology, and its Reversible Lattice Shift. Chem. Eur. J. 2018, 24, 6115–6126. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.L.; Feng, D.; Liu, T.F.; Li, J.R.; Zhou, H.C. Pore Surface Engineering with Controlled Loadings of Functional Groups via Click Chemistry in Highly Stable Metal–Organic Frameworks. J. Am. Chem. Soc. 2012, 134, 14690–14693. [Google Scholar] [CrossRef] [PubMed]
- Bejan, D.; Bahrin, L.G.; Shova, S.; Sardaru, M.; Clima, L.; Nicolescu, A.; Marangoci, N.; Lozan, V.; Janiak, C. Spontaneous resolution of non-centrosymmetric coordination polymers of zinc(II) with achiral imidazole-biphenyl-carboxylate ligands. Inorg. Chim. Acta 2018, 482, 275–283. [Google Scholar] [CrossRef]
- Kuramochi, Y.; Satake, A.; Kobuke, Y. Light-Harvesting Macroring Accommodating a Tetrapodal Ligand Based on Complementary and Cooperative Coordinations. J. Am. Chem. Soc. 2004, 126, 8668–8669. [Google Scholar] [CrossRef] [PubMed]
- Kuramochi, Y.; Satake, A.; Sandanayaka, A.S.D.; Araki, Y.; Ito, O.; Kobuke, Y. Fullerene- and Pyromellitdiimide-Appended Tripodal Ligands Embedded in Light-Harvesting Porphyrin Macrorings. Inorg. Chem. 2011, 50, 10249–10258. [Google Scholar] [CrossRef] [PubMed]
- Constable, E.C.; Eich, O.; Housecroft, C.E. High-nuclearity cobaltadendrimers. J. Chem. Soc. Dalton Trans. 1999, 1363–1364. [Google Scholar] [CrossRef]
- Felix, L.; Sezer, U.; Arndt, M.; Mayor, M. Synthesis of Highly Fluoroalkyl-Functionalized Oligoporphyrin Systems. Eur. J. Org. Chem. 2014, 6884–6895. [Google Scholar] [CrossRef]
- Ohshiro, N.; Takei, F.; Onitsuka, K.; Takahashi, S. Synthesis of organometallic dendrimers with a backbone composed of platinum-acetylide units. J. Organomet. Chem. 1998, 569, 195–202. [Google Scholar] [CrossRef]
- Boudjada, A.; Hernandez, O.; Meinnel, J.; Mani, M.; Paulus, W. 1,3,5-Triiodo-2,4,6-trimethylbenzene at 293 K. Acta Cryst. 2001, C57, 1106–1108. [Google Scholar] [CrossRef] [PubMed]
- da Silva Correa, C.M.M.; Oliviera, M.A.B.C.S. Reaction of arenesulphonyl halides with free radicals. Part 2. J. Chem. Soc. Perkin Trans. 2 1983, 711–715. [Google Scholar] [CrossRef]
- Krasnokutskaya, E.A.; Semenischeva, N.I.; Filimonov, V.D.; Knochel, P. A New, One-Step, Effective Protocol for the Iodination of Aromatic and Heterocyclic Compounds via Aprotic Diazotization of Amines. Synthesis 2007, 81–84. [Google Scholar] [CrossRef]
- Heering, C.; Francis, B.; Nateghi, B.; Makhloufi, G.; Lüdekeb, S.; Janiak, C. Syntheses, structures and properties of group 12 element (Zn, Cd, Hg) coordination polymers with a mixed-functional phosphonate-biphenylcarboxylate linker. CrystEngComm. 2016, 18, 5209–5223. [Google Scholar] [CrossRef]
- Moorthy, J.N.; Natarajan, R.; Venugopalan, P. Three-Dimensional Four-Connecting Organic Scaffolds with a Twist: Synthesis and Self-Assembly. J. Org. Chem. 2005, 70, 8568–8571. [Google Scholar] [CrossRef] [PubMed]
- Łapok, Ł.; Gut, A.; Nowakowska, M. Synthesis and spectroscopic propertiesof 5-tert-butyl-3-(trifluoromethyl)phthalonitrile: A novel precursorfor the synthesis of phthalocyanines. Tetrahedron Lett. 2013, 54, 4388–4391. [Google Scholar] [CrossRef]
- He, H.M.; Fanwick, P.E.; Wood, K.; Cushman, M. A Novel 1,3 O .fwdarw. C Silyl Shift and Deacylation Reaction Mediated by Tetra-n-butylammonium Fluoride in an Aromatic System. J. Org. Chem. 1995, 60, 5905–5909. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bejan, D.; Marangoci, N.L.; Rotaru, A.; Trandabat, A.F.; Bahrin, L.G. 2,4,6-Tris(4-Iodophenyl)-1,3,5-trimethylbenzene. Molbank 2020, 2020, M1121. https://doi.org/10.3390/M1121
Bejan D, Marangoci NL, Rotaru A, Trandabat AF, Bahrin LG. 2,4,6-Tris(4-Iodophenyl)-1,3,5-trimethylbenzene. Molbank. 2020; 2020(1):M1121. https://doi.org/10.3390/M1121
Chicago/Turabian StyleBejan, Dana, Narcisa Laura Marangoci, Alexandru Rotaru, Alexandru Florentin Trandabat, and Lucian Gabriel Bahrin. 2020. "2,4,6-Tris(4-Iodophenyl)-1,3,5-trimethylbenzene" Molbank 2020, no. 1: M1121. https://doi.org/10.3390/M1121
APA StyleBejan, D., Marangoci, N. L., Rotaru, A., Trandabat, A. F., & Bahrin, L. G. (2020). 2,4,6-Tris(4-Iodophenyl)-1,3,5-trimethylbenzene. Molbank, 2020(1), M1121. https://doi.org/10.3390/M1121




