8,18-Dithia-1,4,11,14-tetraazapentacyclo[11.7.0.03,11.05,9.015,19]icosa-3,5(9),6,13,15(19),16-hexaene-10,20-dione
Abstract
1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. General Information
3.2. Synthesis of 8,18-Dithia-1,4,11,14-tetraazapentacyclo[11.7.0.03,11.05,9.015,19]icosa-3,5(9),6,13,15(19),16-hexaene-10,20-dione (4)
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kim, J.; Kwon, J.; Lee, D.; Jo, S.; Park, D.-S.; Choi, J.; Park, E.; Hwang, J.Y.; Ko, Y.; Choi, I.; et al. Serendipitous discovery of 2-((phenylsulfonyl)methyl)-thieno[3,2-d]pyrimidine derivatives as novel HIV-1 replication inhibitors. Bioorg. Med. Chem. Lett. 2014, 24, 5473–5477. [Google Scholar] [CrossRef] [PubMed]
- Bogolubsky, A.V.; Moroz, Y.S.; Savych, O.; Pipko, S.; Konovets, A.; Platonov, M.O.; Vasylchenko, O.V.; Hurmach, V.V.; Grygorenko, O.O. An Old Story in the Parallel Synthesis World: An Approach to Hydantoin Libraries. ACS Comb. Sci. 2018, 20, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Vasan, M.; Neres, J.; Williams, J.; Wilson, D.J.; Teitelbaum, A.M.; Remmel, R.P.; Aldrich, C.C. Inhibitors of the Salicylate Synthase (MbtI) from Mycobacterium tuberculosis Discovered by High-Throughput Screening. ChemMedChem 2010, 5, 2079–2087. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.H.T.; Chin, N.C.; Dipietro, L.V.; Fan, J.; Palermo, M.G.; Shultz, M.D.; Toure, B.B. Piperidinyl compounds for use as tankyrase inhibitors. U.S. Patent WO2013/008217, 17 January 2013. [Google Scholar]
- Kurasawa, O.; Homma, M.; Oguro, Y.; Miyazaki, T.; Mori, K.; Uchiyama, N.; Iwai, K.; Ohashi, A.; Hara, H.; Yoshida, S.; et al. 2-Aminomethylthieno[3,2-d]pyrimidin-4(3H)-ones bearing 3-methylpyrazole hinge binding moiety: Highly potent, selective, and time-dependent inhibitors of Cdc7 kinase. Bioorg. Med. Chem. 2017, 25, 3658–3670. [Google Scholar] [CrossRef] [PubMed]
- Borchardt, A.J.; Gonzalez, J.; Li, H.; Linton, M.A.; Tatlock, J.H. Inhibitors of hepatitis C virus RNA-dependent RNA polymerase, and compositions and treatments using the same. U.S. Patent WO2004/074270, 2 September 2004. [Google Scholar]
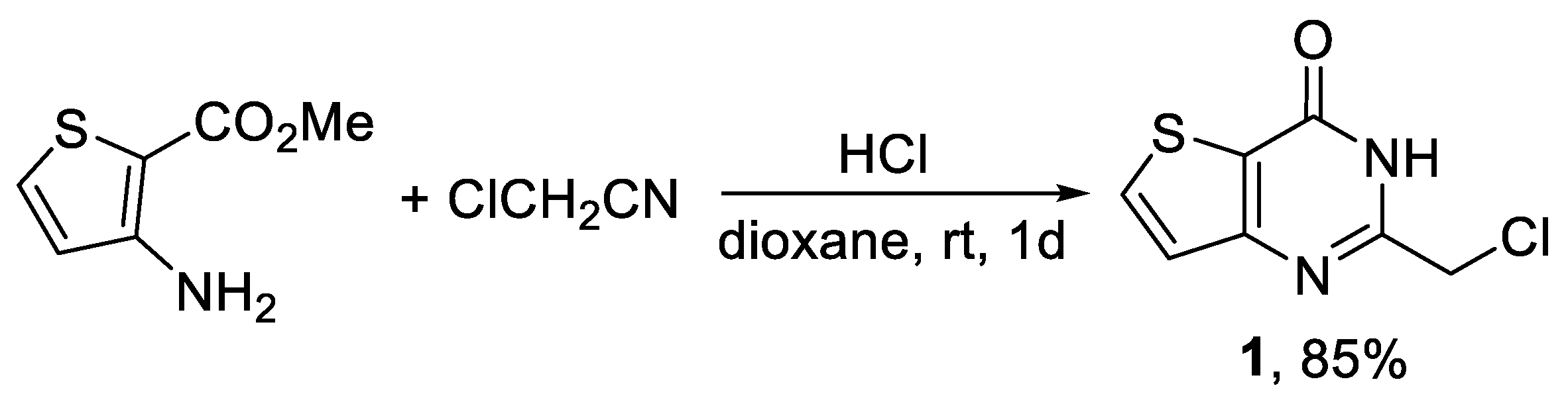
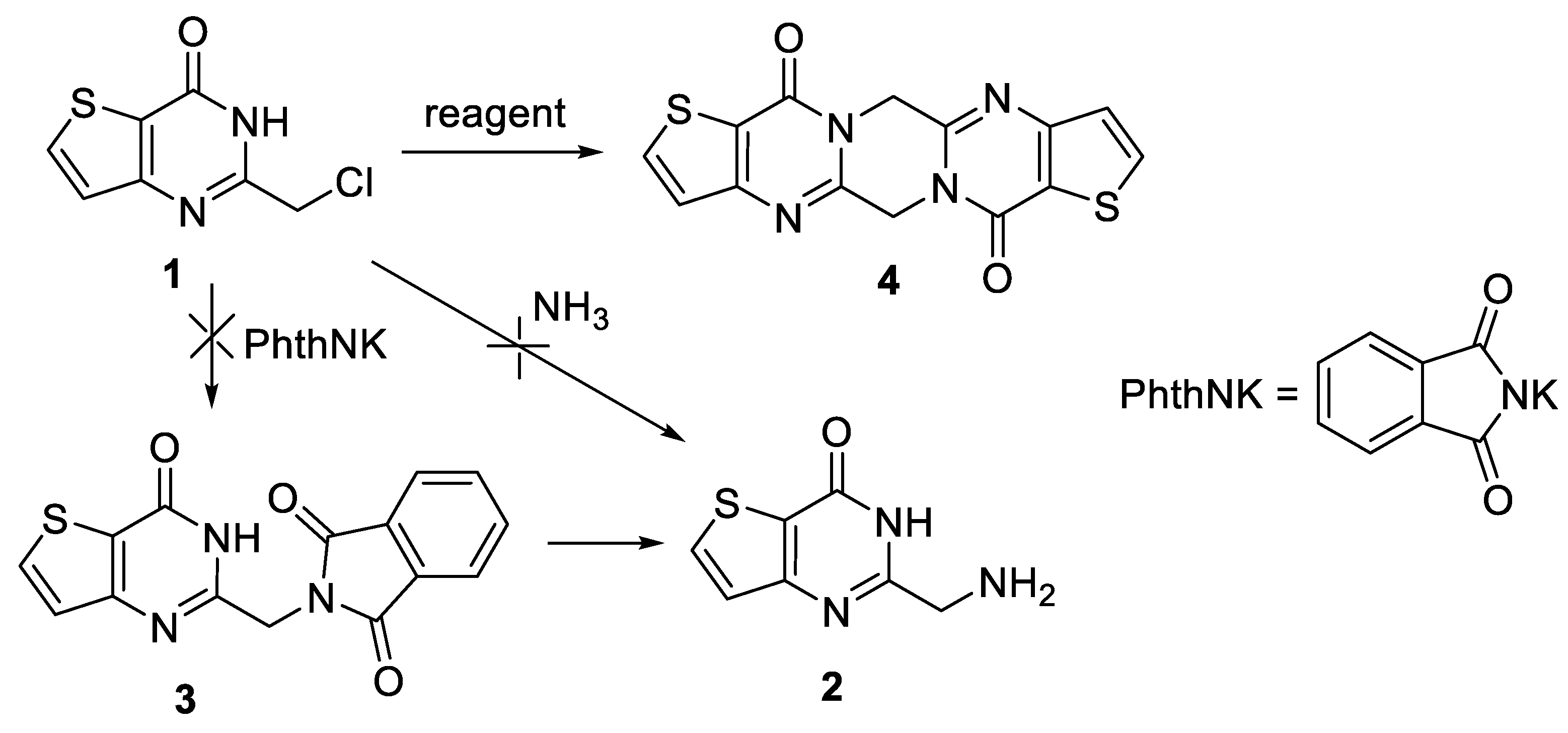
| Entry | Solvent | Reagent | Temperature (°C) | Time (h) | Yield (%) | |
|---|---|---|---|---|---|---|
| 4 | 1 | |||||
| 1 | THF | PhthNK | 66 | 5 | 0 | 95 |
| 2 | acetone | PhthNK | 56 | 5 | 15 | 83 |
| 3 | acetone | PhthNK | 56 | 10 | 15 | 79 |
| 4 | EtOH | PhthNK | 78 | 5 | 24 | 0 |
| 5 | DMSO | PhthNK | 110 | 5 | 39 | 0 |
| 6 | DMF | PhthNK | 110 | 5 | 80 | 0 |
| 7 | DMF | NaH | rt | 10 | 0 | 0 |
| 8 | THF | NaH | 66 | 5 | 0 | 0 |
| 9 | EtOH | KOH | 78 | 10 | 4 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogurtsov, V.A.; Rakitin, O.A. 8,18-Dithia-1,4,11,14-tetraazapentacyclo[11.7.0.03,11.05,9.015,19]icosa-3,5(9),6,13,15(19),16-hexaene-10,20-dione. Molbank 2019, 2019, M1056. https://doi.org/10.3390/M1056
Ogurtsov VA, Rakitin OA. 8,18-Dithia-1,4,11,14-tetraazapentacyclo[11.7.0.03,11.05,9.015,19]icosa-3,5(9),6,13,15(19),16-hexaene-10,20-dione. Molbank. 2019; 2019(2):M1056. https://doi.org/10.3390/M1056
Chicago/Turabian StyleOgurtsov, Vladimir A., and Oleg A. Rakitin. 2019. "8,18-Dithia-1,4,11,14-tetraazapentacyclo[11.7.0.03,11.05,9.015,19]icosa-3,5(9),6,13,15(19),16-hexaene-10,20-dione" Molbank 2019, no. 2: M1056. https://doi.org/10.3390/M1056
APA StyleOgurtsov, V. A., & Rakitin, O. A. (2019). 8,18-Dithia-1,4,11,14-tetraazapentacyclo[11.7.0.03,11.05,9.015,19]icosa-3,5(9),6,13,15(19),16-hexaene-10,20-dione. Molbank, 2019(2), M1056. https://doi.org/10.3390/M1056







