Abstract
Dihalogenated derivatives of 1,2,5-chalcogenadiazoles fused with benzene or heterocyclic rings are of interest as starting compounds for photovoltaic materials. The 1,2,5-chalcogenadiazole ring in these heterocycles was most commonly prepared from the corresponding ortho-diamine moiety. In this communication, 3,6-dibromopyridazine-4,5-diamine was prepared via the reaction of 4,7-dibromo[1,2,5]thiadiazolo[3,4-d]pyridazine with sodium methoxide in THF by heating at reflux for four hours. The structure of the newly synthesized compound was established by means of high resolution mass-spectrometry, 1H, 13C-NMR and IR spectroscopy, and mass-spectrometry.
1. Introduction
1,2,5-Chalcogenadiazoles fused with benzene or heterocyclic rings are intensively used in the construction of various photovoltaic materials such as organic solar cells (OSCs) and organic light emitting diodes (OLEDs) [1,2,3,4,5,6,7]. 4,7-Dibromo-2,1,3-benzothia/selenadiazoles, [1,2,5]thia/selena[3,4-c]pyridines are the most convenient precursors for the synthesis of photoactive materials [8]. 1,2,5-Thia(selena, tellura)diazole cycle can be easily formed from the ortho-diamine moiety of 3,6-dibromobenzene-1,2-diamine and 2,5-dibromopyridine-3,4-diamine [9]. Recently the synthesis of 4,7-dibromo[1,2,5]thiadiazolo[3,4-d]pyridazine 1 has been reported [10] with the formation of the thiadiazole ring in the early steps. Meanwhile, other 4,7-dibromo[1,2,5]chalcogenadiazolo[3,4-d]pyridazines are still unknown due to unavailability of 3,6-dibromopyridazine-4,5-diamine precursor. Although the homologous 3,6-dichloropyridazine-4,5-diamine is described in the literature [11], its synthesis is multistage and the yields were not given. Herein, we report the synthesis of 3,6-dibromopyridazine-4,5-diamine 2.
2. Results and Discussion
Recently, we have shown that 4,7-dibromo[1,2,5]thiadiazolo[3,4-d]pyridazine 1 can be safely and efficiently prepared from the commercial diaminomaleonitrile [12]. Herein, we examined the reduction of this compound (Scheme 1).
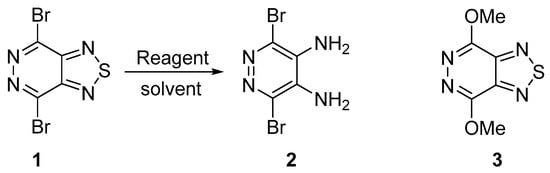
Scheme 1.
Synthesis of 3,6-dibromopyridazine-4,5-diamine 2.
Sodium borohydride (NaBH4) or lithium aluminum hydride (LAH) were commonly used in the reductive cleavage of the 1,2,5-thiadiazole ring to ortho-diamine moiety [13]. We found that the treatment of pyridazine 1 with NaBH4 in EtOH in the presence of catalytic CoCl2·6H2O or with LAH in THF at reflux led to full decomposition of the starting material. Apparently, pyridazine 1 was found to be very sensitive to strong reducing agents. During our investigation of the nucleophilic substitution in 4,7-dibromo[1,2,5]thiadiazolo[3,4-d]pyridazine 1 by alkoxide anions we observed the formation of the new compound as a side product which was proved by mass-spectrometry to be 3,6-dibromopyridazine-4,5-diamine 2. We showed that the treatment of 1 with sodium methoxide in MeOH led to the formation of bis(methoxy) derivative 3 [12] with a traces of diamine 2 (Entry 3). To develop the synthetic approach to the target compound 2 we investigated this reaction in other solvents. The best yield was achieved after refluxing in THF for 3 h (Entry 4). The use of acetonitrile with higher (Entry 5) boiling point did not improve the yield of the target diamine 2. The results are summarized in Table 1.

Table 1.
Reaction of 4,7-dibromo[1,2,5]thiadiazolo[3,4-d]pyridazine 1 with reducing agents.
The structure of diamine 2 was strictly confirmed by means of high resolution mass-spectrometry, 1H, 13C-NMR and IR spectroscopy, and mass-spectrometry.
In conclusion, unexpected synthesis of 3,6-dibromopyridazine-4,5-diamine 2 from the corresponding fused 1,2,5-thiadiazole derivative 1 was discovered. This method may open new possibilities for reductive cleavage of the 1,2,5-thiadiazole ring fused with electron-accepting rings and containing functional derivatives.
3. Experimental Section
3.1. General Information
4,7-Dibromo[1,2,5]thiadiazolo[3,4-d]pyridazine 1 was prepared according to the published method [12]. Melting point was determined on a Kofler hot-stage apparatus and is uncorrected. 1H and 13C-NMR spectra were taken with a Bruker AM-300 machine (Bruker AXS Handheld Inc., Kennewick, WA, USA) (at frequencies of 300.1 and 75.5 MHz, respectively) in acetone-d6 solution, with TMS as the standard. MS spectrum (EI, 70 eV) was obtained with a Finnigan MAT INCOS 50 instrument (Hazlet, NJ, USA). IR spectrum was measured with a Specord M-80 instrument (Carl Zeiss, Jena, Germany) in KBr pellet. High-resolution MS spectrum was measured on a Bruker micrOTOF II instrument (Bruker Daltonik Gmbh, Bremen, Germany) using electrospray ionization (ESI). The measurement was performed in a positive ion mode (interface capillary voltage −4500 V) or in a negative ion mode (3200 V); mass range was from m/z 50 to m/z 3000 Da; external or internal calibration was done with Electrospray Calibrant Solution (Fluka). Syringe injection was used for solutions in acetonitrile, methanol, or water (flow rate 3 L·min−1). Nitrogen was applied as a dry gas; interface temperature was set at 180 °C.
3.2. Synthesis of 3,6-Dibromopyridazine-4,5-diamine 2
4,7-Dibromo[1,2,5]thiadiazolo[3,4-d]pyridazine 1 (40 mg, 0.13 mmol) was added to a solution of MeONa (18 mg, 0.32 mmol) in dry THF (3 mL) with stirring. The reaction mixture was stirred under reflux for 4 h. On completion (monitored by TLC), the mixture was poured into water and extracted with EtOAc (3 × 100 mL). The combined organic layers were washed with brine, dried over MgSO4, filtered, and concentrated under reduced pressure. The residue was subjected to column chromatography on silica gel (Silica gel Merck 60, eluent: EtOAc–CH2Cl2, 1:5, v/v). Yield 22 mg (60%), white solid, mp > 250 °C, Rf = 0.5 (CH2Cl2/EtOAc, 2:1, v/v). IR spectrum, ν, cm−1: 3426, 3329, 3258, 3195, 3124, 1655, 1639, 1619, 1543, 1516, 1349, 1285, 1086, 907, 678, 669, 600. 1H-NMR (ppm): δ 5.60 (s, 4H). 13C-NMR (ppm): δ 136.0, 139.8. HRMS (ESI-TOF), m/z: calcd for C4H579Br2N4 [M + H]+, 266.8875, found, 266.8869. MS (EI, 70eV), m/z (I, %): 270 ([M + 2]+, 48), 269 ([M + 1]+, 30), 268 (M+, 98), 267 ([M − 1]+, 50), 108 (20), 81 (85), 53 (100).
Supplementary Materials
The following are available online, 1H, 13C-NMR, IR and mass-spectra for the compound 2 are available online.
Author Contributions
All authors contributed equally to this work.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Carella, A.; Borbone, F.; Centore, R. Research Progress on Photosensitizers for DSSC. Front. Chem. 2018, 6, 481. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.P.; Li, C.T.; Ho, K.C. Use of organic materials in dye-sensitized solar cells. Mater. Today 2017, 20, 267–283. [Google Scholar] [CrossRef]
- Knyazeva, E.A.; Rakitin, O.A. Influence of structural factors on the photovoltaic properties of dye-sensitized solar cells. Russ. Chem. Rev. 2016, 85, 1146–1183. [Google Scholar] [CrossRef]
- Konstantinova, L.S.; Knyazeva, E.A.; Rakitin, O.A. Recent Developments in the Synthesis and Applications of 1,2,5-Thia- and Selenadiazoles. A Review. Org. Prep. Proc. Int. 2014, 46, 475–544. [Google Scholar] [CrossRef]
- Chulanova, E.A.; Semenov, N.A.; Pushkarevsky, N.A.; Gritsan, N.P.; Zibarev, A.V. Charge-transfer chemistry of chalcogen-nitrogen π-heterocycles. Mendeleev Commun. 2018, 28, 453–460. [Google Scholar] [CrossRef]
- Lonchakov, A.V.; Rakitin, O.A.; Gritsan, N.P.; Zibarev, A.V. Breathing some new life into an old topic: chalcogen-nitrogen π-heterocycles as electron acceptors. Molecules 2013, 18, 9850–9900. [Google Scholar] [CrossRef] [PubMed]
- Gritsan, N.P.; Zibarev, A.V. Chalcogen-nitrogen π-heterocyclic radical anion salts: the synthesis and properties. Russ. Chem. Bull. 2011, 60, 2131–2140. [Google Scholar] [CrossRef]
- Rakitin, O.A.; Zibarev, A.V. Recent Progress in Synthesis and Applications of 5-Membered Chalcogen-Nitrogen π-Heterocycles with Three Heteroatoms. Asian J. Org. Chem. 2018, 7, 2397–2416. [Google Scholar] [CrossRef]
- Knyazeva, E.A.; Rakitin, O.A. 4,7-Dibromo-substituted 2,1,3-benzothia(selena, oxa)diazoles and [1,2,5]thia(selena)diazolo[3,4-c]pyridine as building blocks in solar cells components (microreview). Chem. Heterocycl. Comp. 2017, 53, 855–857. [Google Scholar] [CrossRef]
- Chmovzh, T.N.; Knyazeva, E.A.; Mikhalchenko, L.V.; Golovanov, I.S.; Amelichev, S.A.; Rakitin, O.A. Synthesis of 4,7-dibromo derivative of ultrahigh electron-deficient [1,2,5]thiadiazolo[3,4-d]pyridazine heterocycle and its cross-coupling reactions. Eur. J. Org. Chem. 2018. [Google Scholar] [CrossRef]
- Yanai, M.; Kinoshita, T.; Takeda, S.; Sadaki, H.; Watanabe, H. Synthesis of pyridazine derivatives. XII. Synthesis of 4,5-diaminopyridazine derivatives. Chem. Pharm. Bull. 1970, 18, 1680–1685. [Google Scholar] [CrossRef]
- Chmovzh, T.N.; Knyazeva, E.A.; Lyssenko, K.A.; Popov, V.V.; Rakitin, O.A. Safe synthesis of 4,7-dibromo[1,2,5]thiadiazolo[3,4-d]pyridazine and its SNAr reactions. Molecules 2018, 23, 2576. [Google Scholar] [CrossRef] [PubMed]
- Koutentis, P.A. 1,2,5-Thiadiazoles. In Comprehensive Heterocyclic Chemistry III; Katritzky, A.R., Ramsden, C.A., Scriven, E.F.V., Taylor, R.J.K., Eds.; Elsevier: Oxford, UK, 2008; Volume 5, pp. 516–564. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).