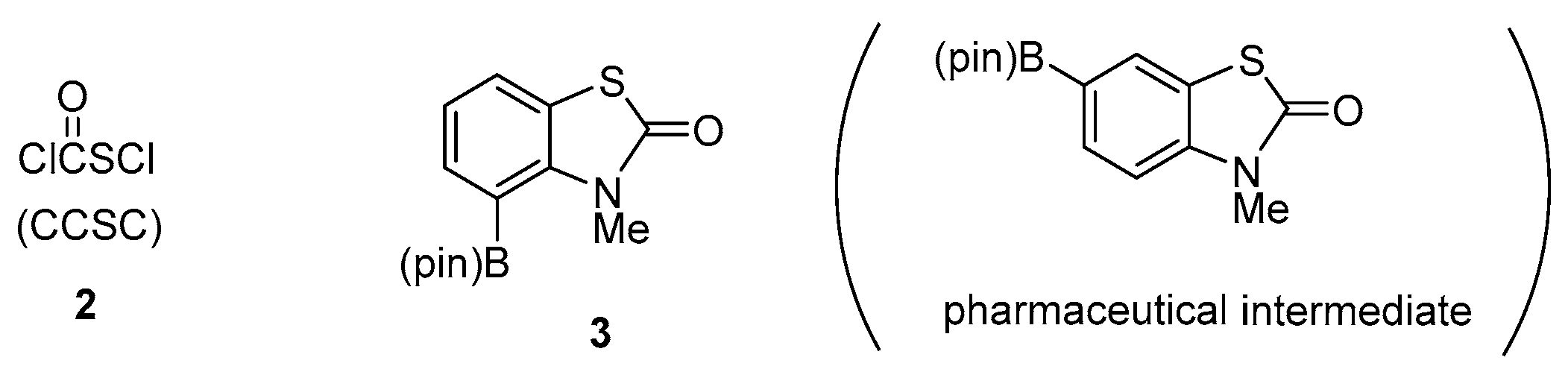
Straightforward Synthesis of N-Methyl-4-(pin)B-2(3H)-benzothiazol-2-one: A Promising Cross-Coupling Reagent
Abstract
:1. Introduction
2. Experimental Section
General
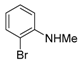
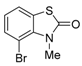
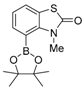
3. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Elderfield, R.C. Heterocyclic Compounds; John Wiley & Sons: New York, NY, USA, 1957; p. 484. [Google Scholar]
- Folco, G.C.; Vigano, T.; Sautebin, L.; Malandrinno, S.; Omini, C.; Berti, F. The mode of action of tiaramide hydrochloride: A new anti-asthmaticdrug. Pharmacol. Res. Commun. 1979, 11, 703–718. [Google Scholar] [CrossRef]
- Godson, D.H.; Leafe, E.L.; Lush, G.B.; Stevenson, H.A. Herbicidal 2-benzothiazolinone derivatives. GB862226, 8 March 1961. [Google Scholar]
- Uematsu, T.; Inoue, S.; Yamashita, N. Benzazole Derivatives. Jpn. Patent 54,041,870, 3 April 1979. [Google Scholar]
- Inoue, S.; Uematsu, T.; Kato, T.; Ueda, K. New melanin biosynthesis inhibitors and their structural similarities. Pest Manag. Sci. 1985, 16, 589–598. [Google Scholar] [CrossRef]
- Shin-ya, K.; Umeda, Y.; Chijiwa, S.; Furihata, K.; Furihata, K.; Hayakawa, Y.; Seto, H. Mevashuntin, a novel metabolite produced by inhibition of the mevalonate pathway in Streptomyces prunicolor. Tetrahedron Lett. 2005, 46, 1273. [Google Scholar] [CrossRef]
- Nawrat, C.C.; Moody, C.J. Total Synthesis of Mevashuntin. Org. Lett. 2012, 14, 1484–1487. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.W.; Terefenko, E.A.; Wrobel, J.; Zhang, Z.M.; Zhu, Y.; Cohen, J.; Marschke, K.B.; Mais, D. Synthesis and progesterone receptor antagonist activities of 6-aryl benzimidazolones and benzothiazolones. Bioorg. Med. Chem. Lett. 2001, 11, 2747–2750. [Google Scholar] [CrossRef]
- Ucar, H.; Van derpoorten, K.; Cacciaguerra, S.; Spampinato, S.; Stables, J.P.; Depovere, P.; Isa, M.; Masereel, B.; Delarge, J.; Poupaert, J.H. Synthesis and anticonvulsant activity of 2(3H)-benzoxazolone and 2(3H)-benzothiazolone derivatives. J. Med. Chem. 1998, 41, 1138–1145. [Google Scholar] [CrossRef] [PubMed]
- Yous, S.; Wallez, V.; Belloir, M.; Caignard, D.H.; McCurdy, C.R.; Poupaert, J.H. Novel 2(3H)-benzothiazolones as highly potent and selective sigma-1 receptor ligands. Med. Chem. Res. 2005, 14, 158–168. [Google Scholar] [CrossRef]
- Weng, J.Q.; Liu, X.H.; Huang, H.; Tan, C.X.; Chen, J. Synthesis, structure and antifungal activity of new 3-[(5-aryl-1,3,4-oxadiazol-2-yl)methyl]benzo[d]thiazol-2(3H)-ones. Molecules 2012, 17, 989–1001. [Google Scholar] [CrossRef] [PubMed]
- Muehlbauer, E.; Weiss, W. Process for the Preparation of Substituted 1,3-Oxathiol-2-ones. German Patent 1,233,882, 18 March 1965. [Google Scholar]
- Zumack, V.G.; Kühle, E. Chlorosulfenylated carbonic acid derivatives. Angew. Chem. Int. Ed. 1970, 82, 54–63. [Google Scholar] [CrossRef]
- Tanabe, Y.; Okabe, T.; Kakimizu, A.; Ohno, N.; Yoshioka, H. An improved method for preparation of N-alkyl-2(3H)-benzothiazolone. Bull. Chem. Soc. Jpn. 1983, 56, 1255–1256. [Google Scholar] [CrossRef]
- Tanabe, Y.; Sanemitsu, Y. A covenient synthesis of 3-chloromethyl-2(3H)-benzothiazolone. Synthesis 1988, 482–484. [Google Scholar] [CrossRef]
- Sanemitsu, Y.; Kawamura, S.; Tanabe, Y. Regioselective α-methoxycarbonyl-sulfenylation of ketones and aldehydes: A versatile method for preparation of thiazolones, thiadiazinones, and 3-indolethiols. J. Org. Chem. 1992, 57, 1053–1056. [Google Scholar] [CrossRef]
- Tanabe, Y.; Mori, K.; Nishii, Y. Regioselective cyclocondensations of chlorocarbonylsulfenyl chloride with hydrazones: Effective synthesis of a class of sulfur and nitrogen containing heterocycles with -COS- linkage. Heterocycles 1996, 43, 141–149. [Google Scholar] [CrossRef]
- Ashida, Y.; Sato, Y.; Suzuki, T.; Ueno, K.; Kai, K.; Nakatsuji, H.; Tanabe, Y. (E)-, (Z)-parallel preparative methods for stereodefined β,β-Diaryl- and α,β-diaryl-α,β-unsaturated esters: Application to stereocomplementary concise synthesis of zimelidine. Chem. Eur. J. 2015, 21, 5934–5945. [Google Scholar] [CrossRef] [PubMed]
- Nakatsuji, H.; Ashida, Y.; Hori, H.; Sato, Y.; Honda, A.; Taira, M.; Tanabe, Y. (E)- and (Z)-stereodefined enol phosphonates derived from β-ketoesters: Stereocomplementary synthesis of fully-substituted α,β-unsaturated esters. Org. Biomol. Chem. 2015, 13, 8205–8210. [Google Scholar] [CrossRef] [PubMed]
- Ashida, Y.; Nakatsuji, H.; Tanabe, Y. (Z)-Enol p-Tosylate Derived from Methyl Acetoacetate: A Useful Cross-coupling Partner for the Synthesis of Methyl (Z)-3-Phenyl (or Aryl)-2-butenoate. Org. Synth. 2017, 94, 93–108. [Google Scholar] [CrossRef]
- Ashida, Y.; Honda, A.; Sato, Y.; Nakatsuji, H.; Tanabe, Y. Divergent synthetic access to (E)- and (Z)-stereodefined all-carbon (fully)-substituted olefin scaffolds: Application to parallel synthesis of both (E)- and (Z)-tamoxifens. ChemistryOpen 2017, 6, 73–89. [Google Scholar] [CrossRef] [PubMed]
- Nakatsuji, H.; Kamada, R.; Kitaguchi, H.; Tanabe, Y. Dehydration-type Ti-claisen condensation (carbonhomologation) of α-heteroatom-substituted acetates with alkyl formates: Utilization as (Z)-stereodefined cross-coupling partners and application to concise synthesis of strobilurin A. Adv. Synth. Catal. 2017, 359, 3865–3879. [Google Scholar] [CrossRef]
- Neya, M.; Yamazaki, H.; Ohne, K.; Sawada, Y.; Mizutani, T.; Imamura, Y.; Mukai, N. Thiazepinyl Hydroxamic Acid Derivatives as Matrix Metalloproteinase Inhibitors. PCT Int. Appl. WO2001060808, 20 February 2001. [Google Scholar]
- Oku, T.; Hirayama, Y.; Yamagami, K.; Ohkubo, Y.; Matsuoka, H. Preparation of Substituted 2-(1,1-Dioxoperhydro-1,4-thiazepin-7-yl)acetamides for Treating Inflammatory Respiratory Diseases. PCT Int. Appl. WO2003018019, 6 March 2003. [Google Scholar]
- Adams, C.M.; Chamoin, S.; Hu, Q.Y.; Zhang, C. Preparation of 5-(Pyridin-3-yl)-1,3-dihydroindol-2-one Derivatives as Modulators of Aldosterone Synthase and/or CYP11B1. PCT Int. Appl. WO2010130794, 18 November 2010. [Google Scholar]
- Kaneko, S.; Sato, K.; Shikanai, D.; Yamada, R.; Sakurada, K. Preparation of 8-Substituted Isoquinoline Derivatives as IKKβ Kinase Inhibitors. PCT Int. Appl. WO2010038465, 8 April 2010. [Google Scholar]
- Fananas, F.J.; Granados, A.; Sanz, R.; Ignacio, J.M.; Barluenga, J. Synthesis of functionalized pyrrole and indole derivatives through carbometallation of lithiated double bonds. Chem. Eur. J. 2001, 7, 2896–2907. [Google Scholar] [CrossRef]
- Ishiyama, T.; Miyaura, N. Metal-catalyzed reactions of diborons for synthesis of organoboron compounds. Chem. Rec. 2004, 3, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, R.; Sheinin, E.B.; Bell, C.L.; Bauer, L. N-(9-o-carbazolyl)-haloanilines from the decompositions of o-carboxybenzenediazonium halides involving benzyne intermediates. J. Heterocycl. Chem. 1975, 12, 203–206. [Google Scholar] [CrossRef]
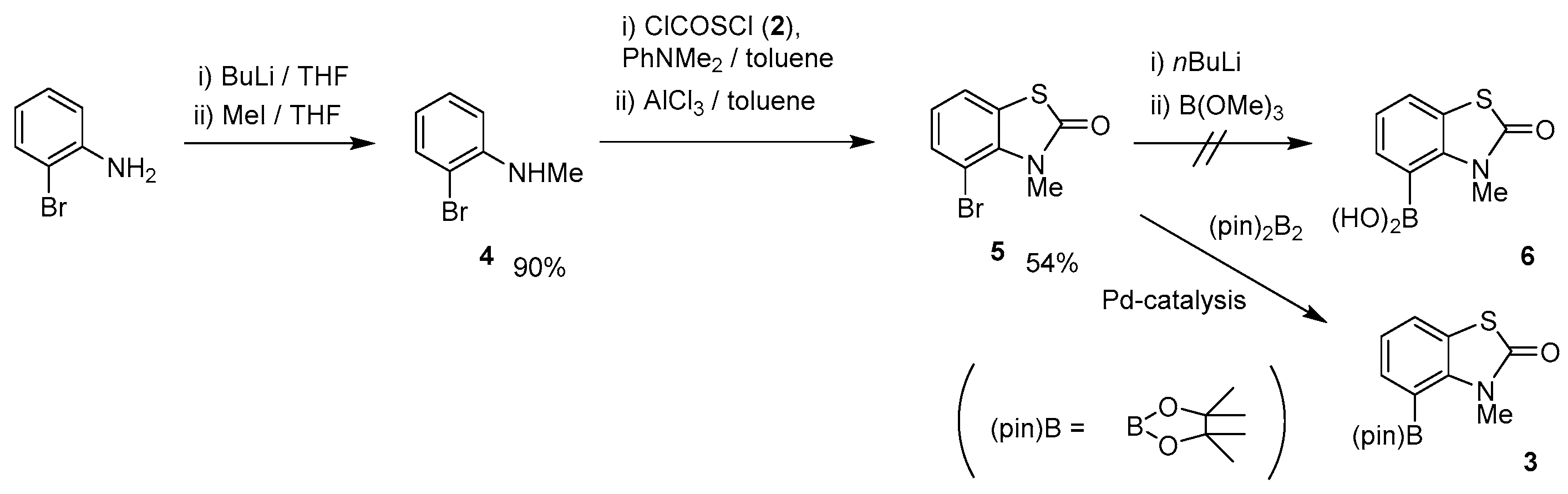
 | |||||||
|---|---|---|---|---|---|---|---|
| Entry | Catalyst | Equivalent (AcOK) | Solvent | Temp./°C | Yield 3/% a | Yield 7/% a | Recovery 5/% a |
| 1 | PdCl2(dppf) | 1.5 | MeOH | 60 | 6 | 7 | 52 |
| 2 | 2.0 | Toluene | 110 | 7 | 44 | 0 | |
| 3 | 3.0 | DMSO | trace | 30 | 0 | ||
| 4 | 1,4-dioxane | 100 | 20 | 78 | 0 | ||
| 5 | 1.0 | 7 | 46 | 0 | |||
| 6 | 1.5 | 30 | 45 | 0 | |||
| 7 | PdCl2(dppe) | 18 | 18 | 38 | |||
| 8 | PdCl2(dppb) | 40 | 27 | 0 | 0 | ||
| 9 | Pd2(dba)3 | 0 | 0 | 0 | |||
| 10 | PdCl2(PPh3)2 | 47 | 32 | 0 | |||
| 11 | DME | 80 | 20 | 13 | 24 | ||
| 12 | MTBE | 55 | 22 | 14 | 33 | ||
| 13 | CPME | 100 | 60 (51) b | 15 | 0 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Izawa, S.; Nakatsuji, H.; Tanabe, Y. Straightforward Synthesis of N-Methyl-4-(pin)B-2(3H)-benzothiazol-2-one: A Promising Cross-Coupling Reagent. Molbank 2018, 2018, M976. https://doi.org/10.3390/M976
Izawa S, Nakatsuji H, Tanabe Y. Straightforward Synthesis of N-Methyl-4-(pin)B-2(3H)-benzothiazol-2-one: A Promising Cross-Coupling Reagent. Molbank. 2018; 2018(1):M976. https://doi.org/10.3390/M976
Chicago/Turabian StyleIzawa, Shotaro, Hidefumi Nakatsuji, and Yoo Tanabe. 2018. "Straightforward Synthesis of N-Methyl-4-(pin)B-2(3H)-benzothiazol-2-one: A Promising Cross-Coupling Reagent" Molbank 2018, no. 1: M976. https://doi.org/10.3390/M976
APA StyleIzawa, S., Nakatsuji, H., & Tanabe, Y. (2018). Straightforward Synthesis of N-Methyl-4-(pin)B-2(3H)-benzothiazol-2-one: A Promising Cross-Coupling Reagent. Molbank, 2018(1), M976. https://doi.org/10.3390/M976






