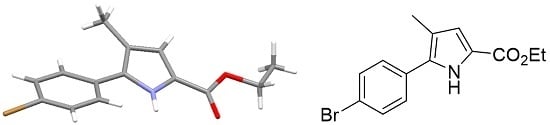
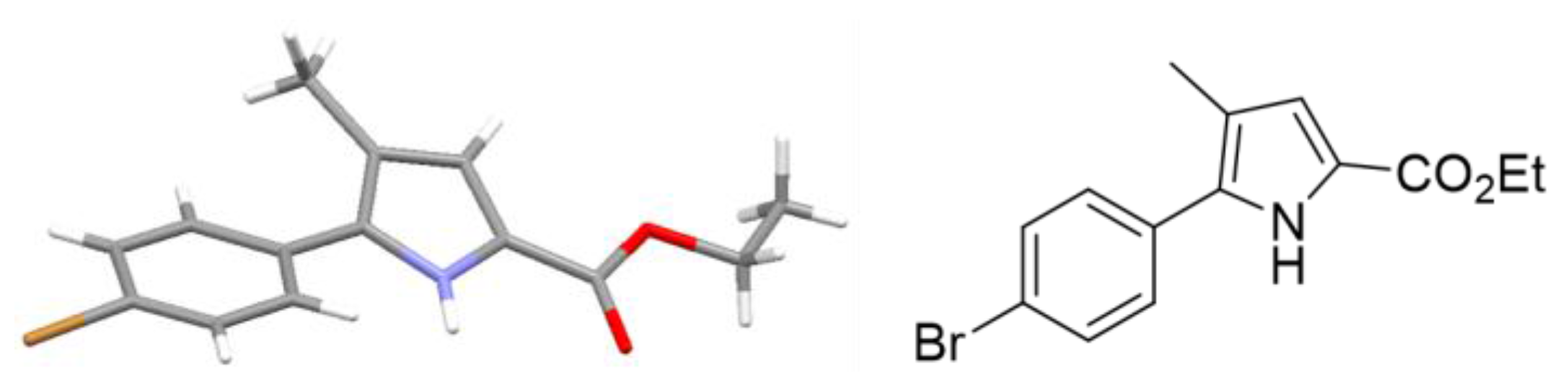
Ethyl 5-(4-Bromophenyl)-4-methyl-1H-pyrrole-2-carboxylate
Abstract
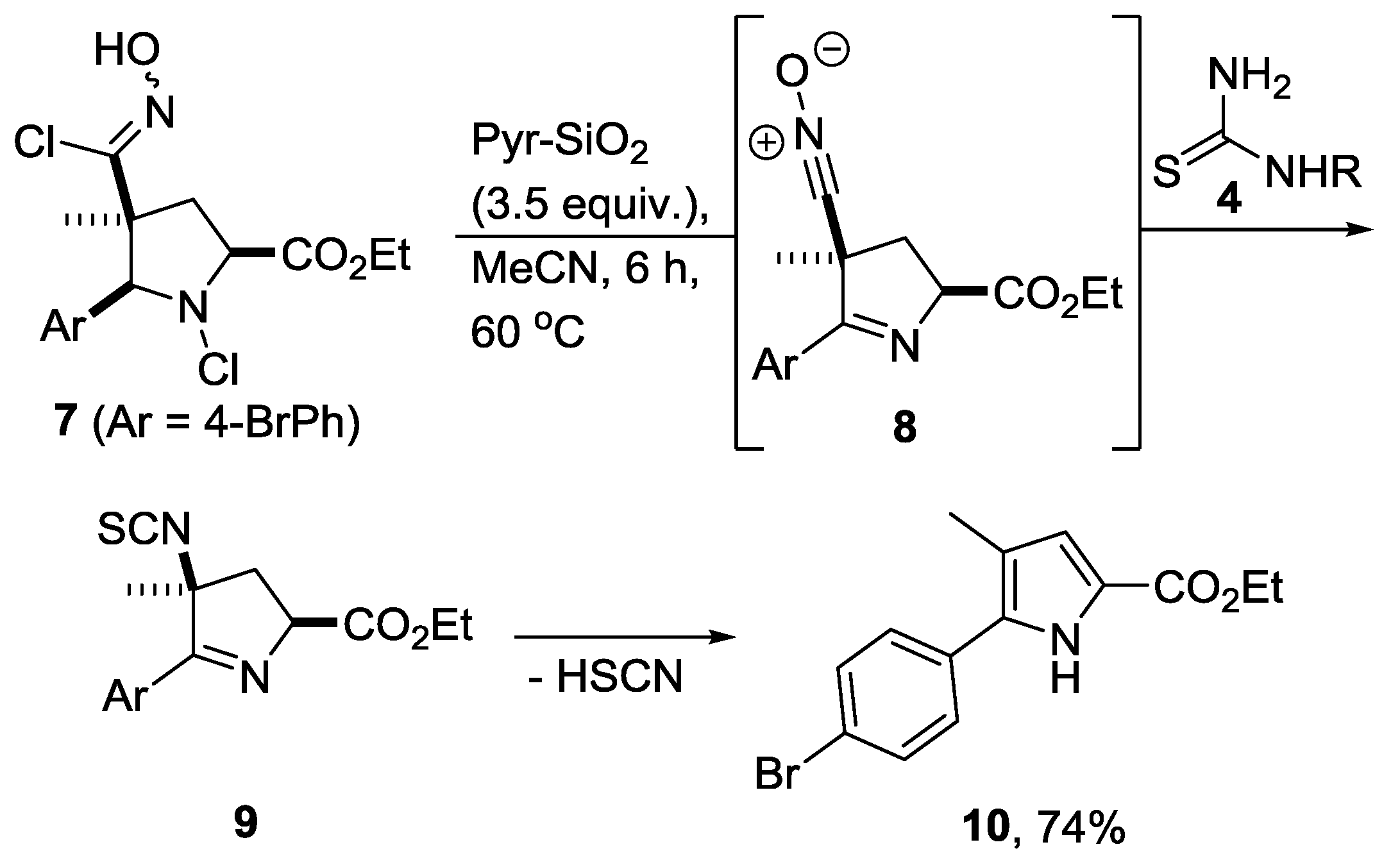
1. Introduction
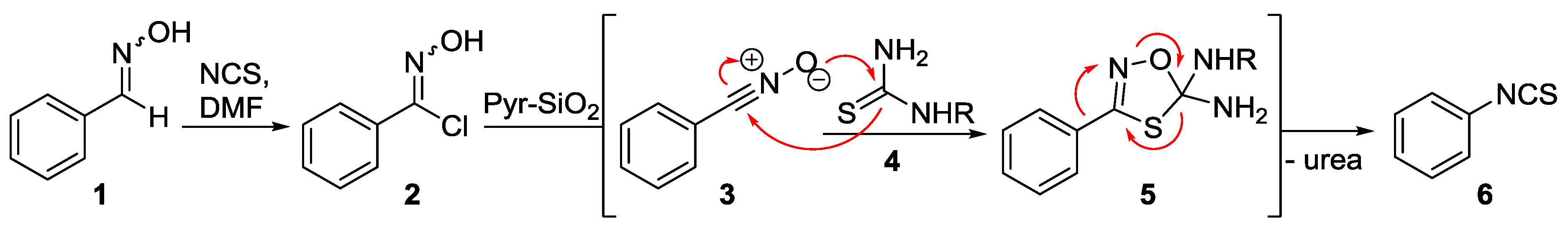
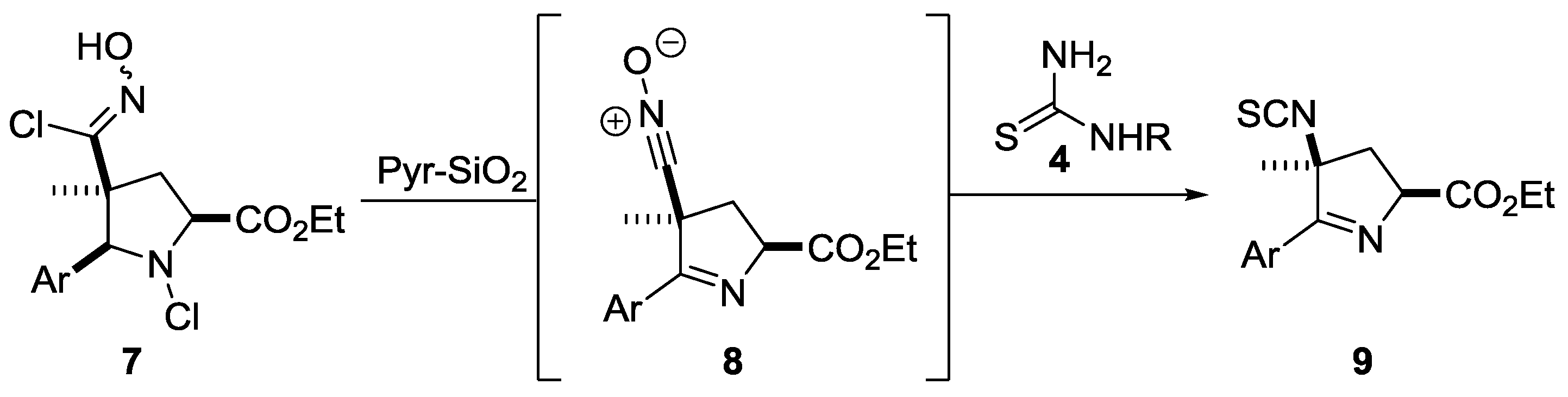
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Rasmussen, S.C.; Evenson, S.J. Dithieno [3,2-b:2′,3′-d]pyrrole-based materials: Synthesis and application to organic electronics. Prog. Polym. Sci. 2013, 38, 1773–1804. [Google Scholar] [CrossRef]
- Baumann, M.; Baxendale, I.R.; Ley, S.V.; Nikbin, N. An overview of the key routes to the best selling 5-membered ring heterocyclic pharmaceuticals. Beilstein J. Org. Chem. 2011, 7, 442–495. [Google Scholar] [CrossRef] [PubMed]
- Young, I.S.; Thornton, P.D.; Thompson, A. Synthesis of natural products containing the pyrrolic ring. Nat. Prod. Rep. 2010, 27, 1801–1839. [Google Scholar] [CrossRef] [PubMed]
- Estevez, V.; Villacampa, M.; Menendez, J.C. Multicomponent reactions for the synthesis of pyrroles. Chem. Soc. Rev. 2010, 39, 4402–4421. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.-B.; Selander, N. Divergent iron-catalyzed coupling of o-acyloximes with silyl enol ethers. Chem. Eur. J. 2017, 23, 1779–1783. [Google Scholar] [CrossRef] [PubMed]
- Baumann, M.; Baxendale, I.R. The rapid generation of isothiocyanates in flow. Beilstein J. Org. Chem. 2013, 9, 1613–1619. [Google Scholar] [CrossRef] [PubMed]
- These pyrrolidines are conveniently prepared by dipolar cycloaddition reactions between azomethine ylides and methyl methacrylate, giving the endo-diastereomer in preference over the exo-diastereomer. See reference [5] for further details.
- The X-ray structure of pyrrole 10 has been deposited with the Cambridge Crystallographic Data Centre as CCDC 1562557.
- Silica-supported pyridine (Pyr-SiO2, 40–63 μm, 1.39 mmol/g) is commercially available from Silicycle.
- QuadraPure™ Thiourea resin (QP-SA, loading 4.0–5.5 mmol/g) is commercially available from Johnson-Matthey.
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baumann, M.; Baxendale, I.R. Ethyl 5-(4-Bromophenyl)-4-methyl-1H-pyrrole-2-carboxylate. Molbank 2017, 2017, M951. https://doi.org/10.3390/M951
Baumann M, Baxendale IR. Ethyl 5-(4-Bromophenyl)-4-methyl-1H-pyrrole-2-carboxylate. Molbank. 2017; 2017(3):M951. https://doi.org/10.3390/M951
Chicago/Turabian StyleBaumann, Marcus, and Ian R. Baxendale. 2017. "Ethyl 5-(4-Bromophenyl)-4-methyl-1H-pyrrole-2-carboxylate" Molbank 2017, no. 3: M951. https://doi.org/10.3390/M951
APA StyleBaumann, M., & Baxendale, I. R. (2017). Ethyl 5-(4-Bromophenyl)-4-methyl-1H-pyrrole-2-carboxylate. Molbank, 2017(3), M951. https://doi.org/10.3390/M951