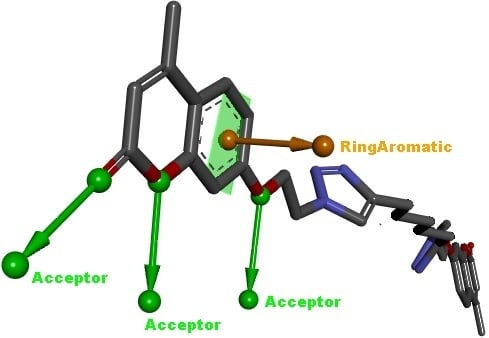
1-[2-(4-Methyl-7-coumarinyloxy)ethyl]-4-(5-{1-[2-(4-methyl-7-coumarinyloxy)ethyl]-1H-1,2,3-triazol-4-yl}pentyl)-1H-1,2,3-triazole
Abstract
:1. Introduction
2. Experimental Section
2.1. General Information
2.2. Synthesis of 1-[2-(4-Methyl-7-coumarinyloxy)ethyl]-4-(5-{1-[2-(4-methyl-7-coumarinyloxy)ethyl]-1H-1,2,3-triazol-4-yl}pentyl)-1H-1,2,3-triazole (3)
Supplementary Materials
Supplementary File 1Supplementary File 2Supplementary File 3Supplementary File 4Acknowledgments
Author Contributions
Conflicts of Interest
References
- Contreras, J.M.; Sippl, W. Homo and heterodimer ligands: The twin drug approach. In The Practice of Medicinal Chemistry, 3nd ed.; Wermuth, C.G., Ed.; Academic Press: London, UK, 2008; pp. 380–414. [Google Scholar]
- Gómez-Outes, A.; Suárez-Gea, M.L.; Calvo-Rojas, G.; Lecumberri, R.; Rocha, E.; Pozo-Hernández, C.; Terleira-Fernández, A.I.; Vargas-Castrillón, E. Discovery of anticoagulant drugs: A historical perspective. Curr. Drug Discov. Technol. 2012, 9, 83–104. [Google Scholar] [CrossRef] [PubMed]
- Matos, M.J.; Viña, D.; Vazquez-Rodriguez, S.; Uriarte, E.; Santana, L. Focusing on new monoamine oxidase inhibitors: Differently substituted coumarins as an interesting scaffold. Curr. Top. Med. Chem. 2012, 12, 2210–2239. [Google Scholar] [CrossRef] [PubMed]
- Vianna, D.R.; Bubols, G.; Meirelles, G.; Silva, B.; Rocha, A.; Lanznaster, M.; Monserrat, J.S.; Garcia, S.C.; von Poser, G.; Eifler-Lima, V.L. Evaluation of the antioxidant capacity of synthesized coumarins. Int. J. Mol. Sci. 2012, 13, 7260–7270. [Google Scholar] [CrossRef] [PubMed]
- Vianna, D.; Hamerski, L.; Figueiró, F.; Bernardi, A.; Visentin, L.; Teixeira, H.; Pires, E.; Eifler-Lima, V.L.; Salbego, C.; Batasttini, A.M.; et al. Selective cytotoxicity and apoptosis induction in glioma cell lines by 5-oxygenated-6,7-methylenedioxycoumarins from Pterocaulon species. Eur. J. Med. Chem. 2012, 57, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Vianna, D.R.; Ruschel, L.; Dietrich, F.; Figueiró, F.; Morrone, F.B.; Canto, R.F.S.; Corvello, F.; Velho, A.; Crestani, A.; Teixeira, H.; et al. 4-Methylcoumarins with cytotoxic activity against T24 and RT4 human bladder câncer cell lines. MedChemComm 2015, 6, 905–911. [Google Scholar] [CrossRef]
- Matos, M.J.; Vazquez-Rodriguez, S.; Santana, L.; Uriarte, E.; Fuentes-Edfuf, C.; Santos, Y.; Muñoz-Crego, A. Looking for new targets: Simple coumarins as antibacterial agents. Med. Chem. 2012, 8, 1140–1145. [Google Scholar] [PubMed]
- Torres, F.C.; Brucker, N.; Andrade, S.F.; Kawano, D.F.; Garcia, S.C.; Poser, G.L.V.; Eifler-Lima, V.L. New insights into the chemistry and antioxidant activity of coumarins. Curr. Top. Med. Chem. 2014, 14, 2600–2623. [Google Scholar] [CrossRef] [PubMed]
- Aragão-Leoneti, V.; Campo, V.L.; Gome, A.S.; Field, R.A.; Carvalho, I. Application of copper(I)-catalysed azide/alkyne cycloaddition (CuAAC) “click chemistry” in carbohydrate drug and neoglycopolymer synthesis. Tetrahedron 2010, 66, 9475–9492. [Google Scholar] [CrossRef]
- Torres, F.C.; Goncalves, G.A.; Vanzolini, K.L.; Merlo, A.A.; Gauer, B.; Garcia, S.C.; Carvalho, I.; Poser, G.L.V.; Kawano, D.F.; Eifler-Lima, V.L.; et al. Combining the pharmacophore features of coumarins and 1,4-substituted 1,2,3-triazoles to design new acetylcholinesterase inhibitors: Fast and easy generation of 4-methylcoumarins/1,2,3-triazoles conjugates via click chemistry. J. Braz. Chem. Soc. 2016, in press. Available online: http://dx.doi.org/10.5935/0103-5053.20160033 (accessed on 8 February 2016). [Google Scholar]
- Armarego, W.L.F.; Chai, C.L.L. Purification of Laboratory Chemicals, 5th ed.; Butterworth-Heinemann: Burlington, VT, USA, 2003. [Google Scholar]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres, F.C.; De Azambuja, G.O.; Gonçalves, I.L.; Gonçalves, G.A.; Von Poser, G.L.; Kawano, D.F.; Eifler-Lima, V.L. 1-[2-(4-Methyl-7-coumarinyloxy)ethyl]-4-(5-{1-[2-(4-methyl-7-coumarinyloxy)ethyl]-1H-1,2,3-triazol-4-yl}pentyl)-1H-1,2,3-triazole. Molbank 2016, 2016, M894. https://doi.org/10.3390/M894
Torres FC, De Azambuja GO, Gonçalves IL, Gonçalves GA, Von Poser GL, Kawano DF, Eifler-Lima VL. 1-[2-(4-Methyl-7-coumarinyloxy)ethyl]-4-(5-{1-[2-(4-methyl-7-coumarinyloxy)ethyl]-1H-1,2,3-triazol-4-yl}pentyl)-1H-1,2,3-triazole. Molbank. 2016; 2016(2):M894. https://doi.org/10.3390/M894
Chicago/Turabian StyleTorres, Fernando Cidade, Gabriel Oliveira De Azambuja, Itamar Luís Gonçalves, Guilherme Arraché Gonçalves, Gilsane Lino Von Poser, Daniel Fábio Kawano, and Vera Lucia Eifler-Lima. 2016. "1-[2-(4-Methyl-7-coumarinyloxy)ethyl]-4-(5-{1-[2-(4-methyl-7-coumarinyloxy)ethyl]-1H-1,2,3-triazol-4-yl}pentyl)-1H-1,2,3-triazole" Molbank 2016, no. 2: M894. https://doi.org/10.3390/M894
APA StyleTorres, F. C., De Azambuja, G. O., Gonçalves, I. L., Gonçalves, G. A., Von Poser, G. L., Kawano, D. F., & Eifler-Lima, V. L. (2016). 1-[2-(4-Methyl-7-coumarinyloxy)ethyl]-4-(5-{1-[2-(4-methyl-7-coumarinyloxy)ethyl]-1H-1,2,3-triazol-4-yl}pentyl)-1H-1,2,3-triazole. Molbank, 2016(2), M894. https://doi.org/10.3390/M894