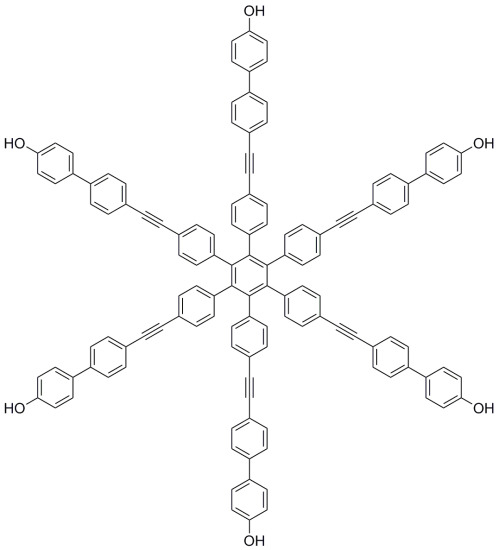
Hexakis{4-[(4′-hydroxybiphenyl-4-yl)ethynyl]phenyl}benzene
Abstract
:Introduction
Results and Discussion
Experimental Section
General Methods
Hexakis{4-[(4′-hydroxybiphenyl-4-yl)ethynyl]phenyl}benzene (2)
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Supplementary File 4Acknowledgments
Author Contributions
Conflicts of Interest
References
- Milov, A.D.; Salikhov, K.M.; Shirov, M.D. Application of the double resonance method to electron spin echo in a study of the spatial distribution of paramagnetic centres in solids. Sov. Phys. Solid State 1981, 23, 565–569. [Google Scholar]
- Pannier, M.; Veit, S.; Godt, A.; Jeschke, G.; Spiess, H.W. Dead-time free measurement of dipole–dipole interactions between electron spins. J. Magn. Reson. 2000, 142, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Jeschke, G. DEER distance measurements on proteins. Annu. Rev. Phys. Chem. 2012, 63, 419–446. [Google Scholar] [CrossRef] [PubMed]
- Shigemori, K.; Hara, H.; Kawamori, A.; Akabori, K. Determination of distances from tyrosine D to QA and chlorophyllZ in photosystem II studied by `2+1' pulsed EPR. BBA-Bioenergetics 1998, 1363, 187–198. [Google Scholar] [CrossRef]
- Ward, R.; Pliotas, C.; Branigan, E.; Hacker, C.; Rasmussen, A.; Hagelueken, G.; Booth, I.R.; Miller, S.; Lucocq, J.; Naismith, J.H.; et al. Probing the structure of the mechanosensitive channel of small conductance in lipid bilayers with pulsed electron-electron double resonance. Biophys. J. 2014, 106, 834–842. [Google Scholar] [CrossRef] [PubMed]
- Krstić, I.; Endeward, B.; Margraf, D.; Marko, A.; Prisner, T. Structure and dynamics of nucleic acids. In EPR Spectroscopy; Drescher, M., Jeschke, G., Eds.; Springer: Berlin, Germany; Heidelberg, Germany, 2012; Volume 321, pp. 159–198. [Google Scholar]
- Jeschke, G.; Sajid, M.; Schulte, M.; Godt, A. Three-spin correlations in double electron-electron resonance. Phys. Chem. Chem. Phys. 2009, 11, 6580–6591. [Google Scholar] [CrossRef] [PubMed]
- Giannoulis, A.; Ward, R.; Branigan, E.; Naismith, J.H.; Bode, B.E. PELDOR in rotationally symmetric homo-oligomers. Mol. Phys. 2013, 111, 2845–2854. [Google Scholar] [CrossRef] [PubMed]
- Valera, S.; Taylor, J.E.; Daniels, D.S.B.; Dawson, D.M.; Athukorala Arachchige, K.S.; Ashbrook, S.E.; Slawin, A.M.Z.; Bode, B.E. A modular approach for the synthesis of nanometer-sized polynitroxide multi-spin systems. J. Org. Chem. 2014, 79, 8313–8323. [Google Scholar] [CrossRef] [PubMed]
- Hyatt, J.A. Synthesis of a hexaalkynylhexaphenylbenzene. Org. Prep. Proced. Int. 1991, 23, 460–463. [Google Scholar] [CrossRef]
- Kobayashi, K.; Kobayashi, N.; Ikuta, M.; Therrien, B.; Sakamoto, S.; Yamaguchi, K. Syntheses of hexakis(4-functionalized-phenyl)benzenes and hexakis[4-(4ʹ-functionalized- phenylethynyl)phenyl]benzenes directed to host molecules for guest-inclusion networks. J. Org. Chem. 2005, 70, 749–752. [Google Scholar] [CrossRef] [PubMed]
- Mohamed Ahmed, M.S.; Mori, A. Sonogashira coupling with aqueous ammonia directed to the synthesis of azotolane derivatives. Tetrahedron 2004, 60, 9977–9982. [Google Scholar] [CrossRef]
- FT-IR (ATR): 1487 (m), 1138 (m), 1058 (m), 1003 (s), 823 (m). 1H NMR (300 MHz, Chloroform-d) δ 7.23 (s, 12H), 6.54–6.39 (m, 12H).
- ESI and EI mass spectrometry methods did not give the wanted ions. Low resolution MALDI techniques gave the wanted mass peak, however high resolution analysis using MALDI could not be achieved due to the lack of an appropriate standard. Microanalysis for 2 did not give acceptable results. We attribute this to the hygroscopic nature of the compound. Similar behaviour with respect to microanalysis and mass spectrometry was found for similar compounds bearing several phenol groups (see Ref. 9).

© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valera, S.; Bode, B.E. Hexakis{4-[(4′-hydroxybiphenyl-4-yl)ethynyl]phenyl}benzene. Molbank 2015, 2015, M865. https://doi.org/10.3390/M865
Valera S, Bode BE. Hexakis{4-[(4′-hydroxybiphenyl-4-yl)ethynyl]phenyl}benzene. Molbank. 2015; 2015(3):M865. https://doi.org/10.3390/M865
Chicago/Turabian StyleValera, Silvia, and Bela E. Bode. 2015. "Hexakis{4-[(4′-hydroxybiphenyl-4-yl)ethynyl]phenyl}benzene" Molbank 2015, no. 3: M865. https://doi.org/10.3390/M865
APA StyleValera, S., & Bode, B. E. (2015). Hexakis{4-[(4′-hydroxybiphenyl-4-yl)ethynyl]phenyl}benzene. Molbank, 2015(3), M865. https://doi.org/10.3390/M865