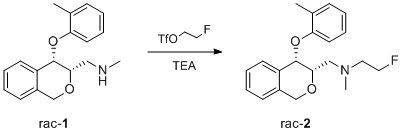
2-Fluoro-N-methyl-N-{[(3S*,4S*)-4-(2-methylphenoxy)-3,4-dihydro-1H-isochromen-3-yl]methyl}ethanamine
1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. General Information
3.2. 2-Fluoro-N-methyl-N-{[(3S*,4S*)-4-(2-methylphenoxy)-3,4-dihydro-1H-isochromen-3-yl]methyl}ethanamine (FE@PHOXI1)
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Author Contributions
Conflicts of Interest
References
- Sung, U.; Apparsundram, S.; Galli, A.; Kahlig, K.M.; Savchenko, V.; Schroeter, S.; Quick, M.W.; Blakely, R.D. A regulated interaction of syntaxin 1A with the antidepressant-sensitive norepinephrine transporter establishes catecholamine clearance capacity. J. Neurosci. 2003, 23, 1697–1709. [Google Scholar] [PubMed]
- Kim, C.H.; Hahn, M.K.; Joung, Y.; Anderson, S.L.; Steele, A.H.; Mazei-Robinson, M.S.; Gizer, I.; Teicher, M.H.; Cohen, B.M.; Robertson, D.; et al. A polymorphism in the norepinephrine transporter gene alters promoter activity and is associated with attention-deficit hyperactivity disorder. Proc. Natl. Acad. Sci. USA 2006, 103, 19164–19169. [Google Scholar] [CrossRef] [PubMed]
- Hahn, M.K.; Robertson, D.; Blakely, R.D. A mutation in the human norepinephrine transporter gene (SLC6A2) associated with orthostatic intolerance disrupts surface expression of mutant and wild-type transporters. J. Neurosci. 2003, 23, 4470–4478. [Google Scholar] [PubMed]
- Mirbolooki, M.R.; Upadhyay, S.K.; Constantinescu, C.C.; Pan, M.L.; Mukherjee, J. Adrenergic pathway activation enhances brown adipose tissue metabolism: A [18F]FDG PET/CT study in mice. Nucl. Med. Biol. 2014, 41, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.L.; Fan, X.; Yeckel, C.W.; Weinzimmer, D.; Mulnix, T.; Gallezot, J.D.; Carson, R.E.; Sherwin, R.S.; Ding, Y.S. Ex vivo and in vivo Evaluation of the Norepinephrine Transporter Ligand [11C]MRB for Brown Adipose Tissue Imaging. Nucl. Med. Biol. 2012, 39, 1081–1086. [Google Scholar] [CrossRef] [PubMed]
- Neudorfer, C. Development of Novel Precursors and Reference Compounds for PET Based Investigations Targeting the Norepinephrine Transporter and Monoamine Oxidase B. Ph.D. Dissertation, University of Vienna, Vienna, Austria, 18 December 2014. [Google Scholar]
- Hudson, S.; Kiankarimi, M.; Eccles, W.; Dwight, W.; Mostofi, Y.S.; Genicot, M.J.; Fleck, B.A.; Gogas, K.; Aparicio, A.; Wang, H.; et al. Structure-activity relationships of chiral selective norepinephrine reuptake inhibitors (sNRI) with increased oxidative stability. Bioorg. Med. Chem. Lett. 2008, 18, 4491–4494. [Google Scholar] [CrossRef] [PubMed]
- Hudson, S.; Kiankarimi, M.; Eccles, W.; Mostofi, Y.S.; Genicot, M.J.; Dwight, W.; Fleck, B.A.; Gogas, K.; Wade, W.S. Synthesis and structure-activity relationships of selective norepinephrine reuptake inhibitors (sNRI) with a heterocyclic ring constraint. Bioorg. Med. Chem. Lett. 2008, 18, 4495–4498. [Google Scholar] [CrossRef] [PubMed]
- Kiankarimi, M.; Hudson, S.; Dwight, W.J.; Wesley, J.; Wade, W.S. Monoamine Re-Uptake Inhibitors and Methdos Relating Thereto. U.S. Pat. Appl. Publ. US 20060252818 A1, November 2006. [Google Scholar]
- Claremon, D.A.; Zhuang, L.; Leftheris, K.; Tice, C.M.; Xu, Z.; Ye, Y.; Singh, S.B.; Cacatian, S.; Zhao, W.; Himmelsbach, F.; et al. Cyclic Inhibitors of 11beta-Hydroxysteroid Dehydrogenase 1. PCT Int. Appl. WO 2009134392 A1, November 2009. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neudorfer, C.; Shanab, K.; Holzer, W.; Rami-Mark, C.; Mitterhauser, M.; Wadsak, W.; Spreitzer, H. 2-Fluoro-N-methyl-N-{[(3S*,4S*)-4-(2-methylphenoxy)-3,4-dihydro-1H-isochromen-3-yl]methyl}ethanamine. Molbank 2015, 2015, M862. https://doi.org/10.3390/M862
Neudorfer C, Shanab K, Holzer W, Rami-Mark C, Mitterhauser M, Wadsak W, Spreitzer H. 2-Fluoro-N-methyl-N-{[(3S*,4S*)-4-(2-methylphenoxy)-3,4-dihydro-1H-isochromen-3-yl]methyl}ethanamine. Molbank. 2015; 2015(2):M862. https://doi.org/10.3390/M862
Chicago/Turabian StyleNeudorfer, Catharina, Karem Shanab, Wolfgang Holzer, Christina Rami-Mark, Markus Mitterhauser, Wolfgang Wadsak, and Helmut Spreitzer. 2015. "2-Fluoro-N-methyl-N-{[(3S*,4S*)-4-(2-methylphenoxy)-3,4-dihydro-1H-isochromen-3-yl]methyl}ethanamine" Molbank 2015, no. 2: M862. https://doi.org/10.3390/M862
APA StyleNeudorfer, C., Shanab, K., Holzer, W., Rami-Mark, C., Mitterhauser, M., Wadsak, W., & Spreitzer, H. (2015). 2-Fluoro-N-methyl-N-{[(3S*,4S*)-4-(2-methylphenoxy)-3,4-dihydro-1H-isochromen-3-yl]methyl}ethanamine. Molbank, 2015(2), M862. https://doi.org/10.3390/M862