Copper Dipyridine Dichloride: An Efficient and Convenient Catalyst for the Synthesis of Bis (Indolyl) Methanes
Abstract
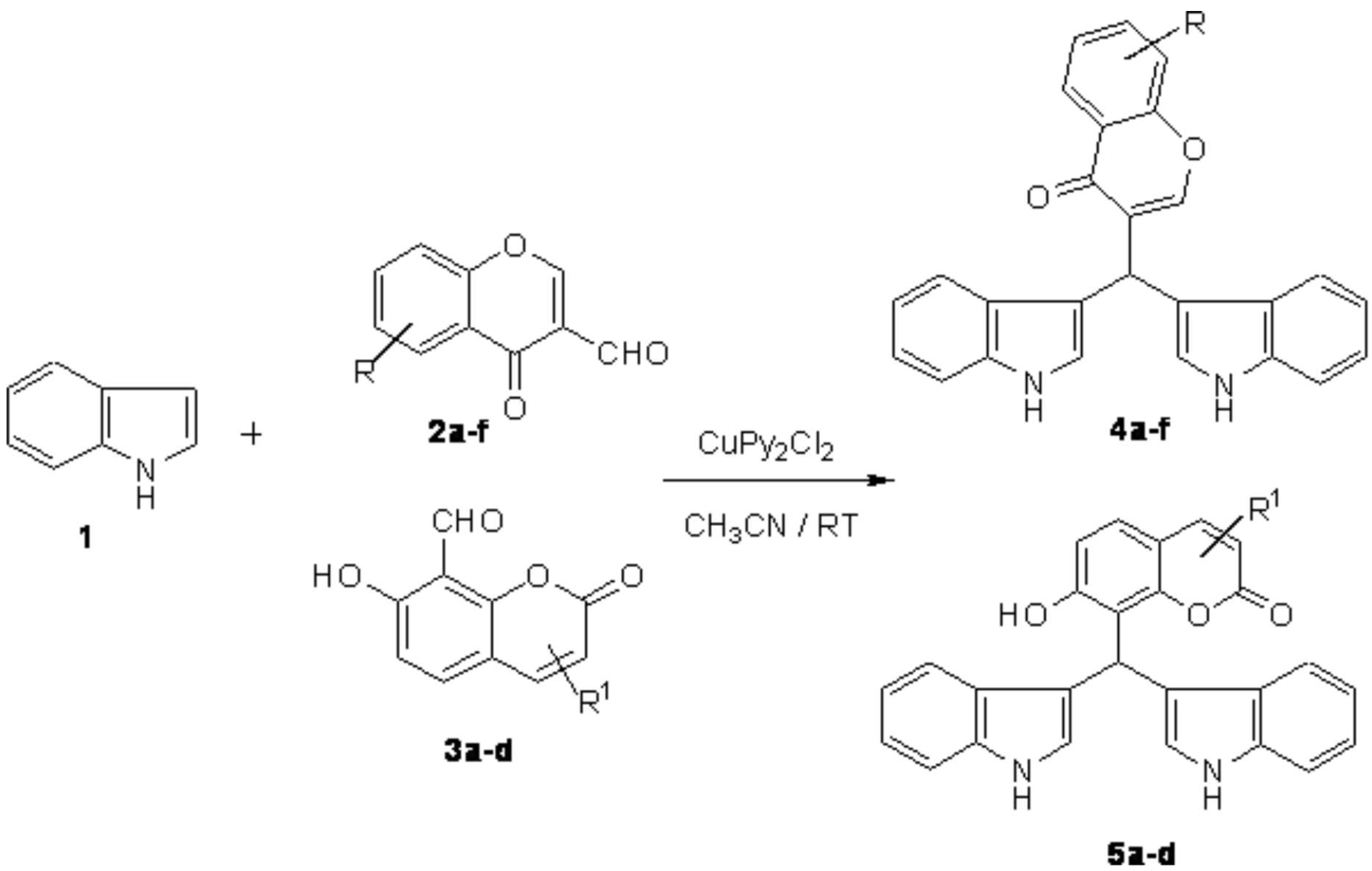
:Introduction
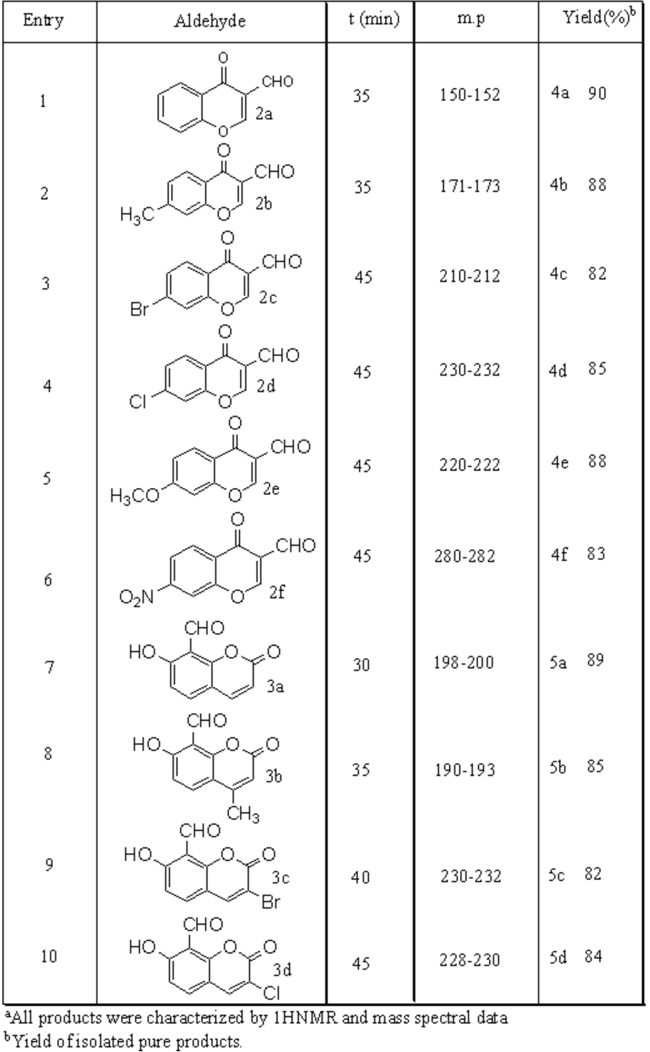
General Procedure
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Supplementary File 4Supplementary File 5Supplementary File 6Acknowledgement
References
- Undberg, R.J. The Chemistry of Indoles; Academic Press: New York, 1996. [Google Scholar]
- (a) Porter, J.K.; Bacon, C.W.; Robins, J.D.; Himmelsbach, D.S.; Higman, H.C. Indole alkaloids from Balansia epichloe. J. Agric .Food Chem. 1977, 25, 88–93. [Google Scholar] [CrossRef] (b) Osawa, T.; Namiki, M. Structure elucidation of streptindole, a novel genotoxic metabolite isolated from intestinal bacteria. Tetrahedron Lett. 1983, 24, 4719–4722. [Google Scholar] [CrossRef] (c) Fahy, E.; Potts, B.C.M.; Faulkner, D.J. 6-Bromotryptamine Derivatives from the Gulf of California Tunicate Didemnum candidum. J. Nat.Prod. 1991, 54, 564–569. [Google Scholar] [CrossRef] (d) Bifulco, G.; Bruno, I.; Riccio, R.; Lavayre, J.; Bourdy, G.J. Further Brominated Bis- and Tris-Indole Alkaloids from the Deep-Water New Caledonian Marine Sponge Orina sp. J. Nat. Prod. 1995, 58, 1254–1260. [Google Scholar] [CrossRef] [PubMed]
- Morgan, L.; Schinior, R. The Chemistry of Neuraminic Acids. I. The Ehrlich Reaction. J. Org.Chem. 1962, 27, 3696–3697. [Google Scholar] [CrossRef]
- Woland, W.; Venkiteswaren, M.; Richards, C. Cyclizative Condensations. I. 2-Methylindole with Acetone and Methyl Ethyl Ketone. J. Org. Chem. 1961, 26, 4241–4248. [Google Scholar]
- Roomi, M.; MacDonald, S. Reductive C-alkylation. II. Can. J. Chem. 1970, 48, 139–143. [Google Scholar] [CrossRef]
- Banerji, J.; Chatterjee, A.; Manna, S.; Pascard, C.; Prange, T.; Shoolery, J. Lewis Acid Induced Electrophilic Substitution of Indole. Heterocycles 1981, 15, 325–336. [Google Scholar] [CrossRef]
- Chatterjee, A.; Manna, S.; Banerji, J.; Pascard, C.; Prange, T.; Shoolery, J. Lewis-acid-induced electrophilic substitution in indoles with acetone. J. Chem. Soc., Perkin Trans. 1980, 553–555. [Google Scholar] [CrossRef]
- Gregorovich, B.; Liang, K.; Clugston, D.; Mac-Donald, S. Reductive C-alkylation. Can. J. Chem. 1968, 46, 3291–3300. [Google Scholar] [CrossRef]
- Chen, D.; Yu, L.; Wang, P.G. Lewis acid-catalyzed reactions in protic media. Lanthanide-catalyzed reactions of indoles with aldehydes or ketones. Tetrahedron Lett. 1996, 37, 4467–4470. [Google Scholar] [CrossRef]
- (a) Nagarajan, R.; Perumal, P.T. InCl3 and In(OTf)3 catalyzed reactions: synthesis of 3-acetyl indoles, bis-indolylmethane and indolylquinoline derivatives. Tetrahedron 2002, 58, 1229–1232. [Google Scholar] [CrossRef] (b) Nagarajan, R.; Perumal, P.T. Electrophilic substitution of indoles catalyzed by triphenyl phosphonium perchlorate: synthesis of 3-acetyl indoles and bis-indolylmethane derivatives. Synth. Commun. 2002, 32, 105–109. [Google Scholar] [CrossRef] (c) Bandgar, B.P.; Shaikh, K.A. Molecular iodine-catalyzed efficient and highly rapid synthesis of bis(indolyl)methanes under mild conditions. Tetrahedron Lett. 2003, 44, 1959–1961. [Google Scholar] [CrossRef] (d) Vijender Reddy, A.; Ravinder, K.; Niranjan Reddy, V.L.; Venkateswer Goud, T.; Ravikanth, V.; Venkateswarlu, V. Zeolite Catalyzed Synthesis of bis(Indolyl) Methanes. Synth. Commun. 2003, 33, 3687–3694. [Google Scholar] [CrossRef] (e) Nagarajan, R.; Permal, P.T. Chem. Lett. 2004, 33, 288–289.
- Duntitz, J. D. The crystal structures of copper dipyridine dichloride and the violet form of cobalt dipyridine dichloride. Acta Cryst. 1957, 10, 307–313. [Google Scholar] [CrossRef]
- Naveen Kumar, V.; Someshwar, P.; Narasimha Reddy, P.; Thirupathi Reddy, Y.; Rajitha, B. Copper dipyridine dichloride as a mild and efficient catalyst for the one pot condensation of biginelli reaction. J. Heterocyclic. Chem. 2005, 42, 1017–1019. [Google Scholar] [CrossRef]
 |
© 2007 by MDPI (http://www.mdpi.org/). Reproduction is permitted for noncommercial purposes.
Share and Cite
Rajitha, B.; Someshwar, P.; Kumar, V.N.; Madhav, J.V. Copper Dipyridine Dichloride: An Efficient and Convenient Catalyst for the Synthesis of Bis (Indolyl) Methanes. Molbank 2007, 2007, M553. https://doi.org/10.3390/M553
Rajitha B, Someshwar P, Kumar VN, Madhav JV. Copper Dipyridine Dichloride: An Efficient and Convenient Catalyst for the Synthesis of Bis (Indolyl) Methanes. Molbank. 2007; 2007(3):M553. https://doi.org/10.3390/M553
Chicago/Turabian StyleRajitha, Bavanthula, Pola Someshwar, Vanam Naveen Kumar, and Janganati Venu Madhav. 2007. "Copper Dipyridine Dichloride: An Efficient and Convenient Catalyst for the Synthesis of Bis (Indolyl) Methanes" Molbank 2007, no. 3: M553. https://doi.org/10.3390/M553
APA StyleRajitha, B., Someshwar, P., Kumar, V. N., & Madhav, J. V. (2007). Copper Dipyridine Dichloride: An Efficient and Convenient Catalyst for the Synthesis of Bis (Indolyl) Methanes. Molbank, 2007(3), M553. https://doi.org/10.3390/M553




