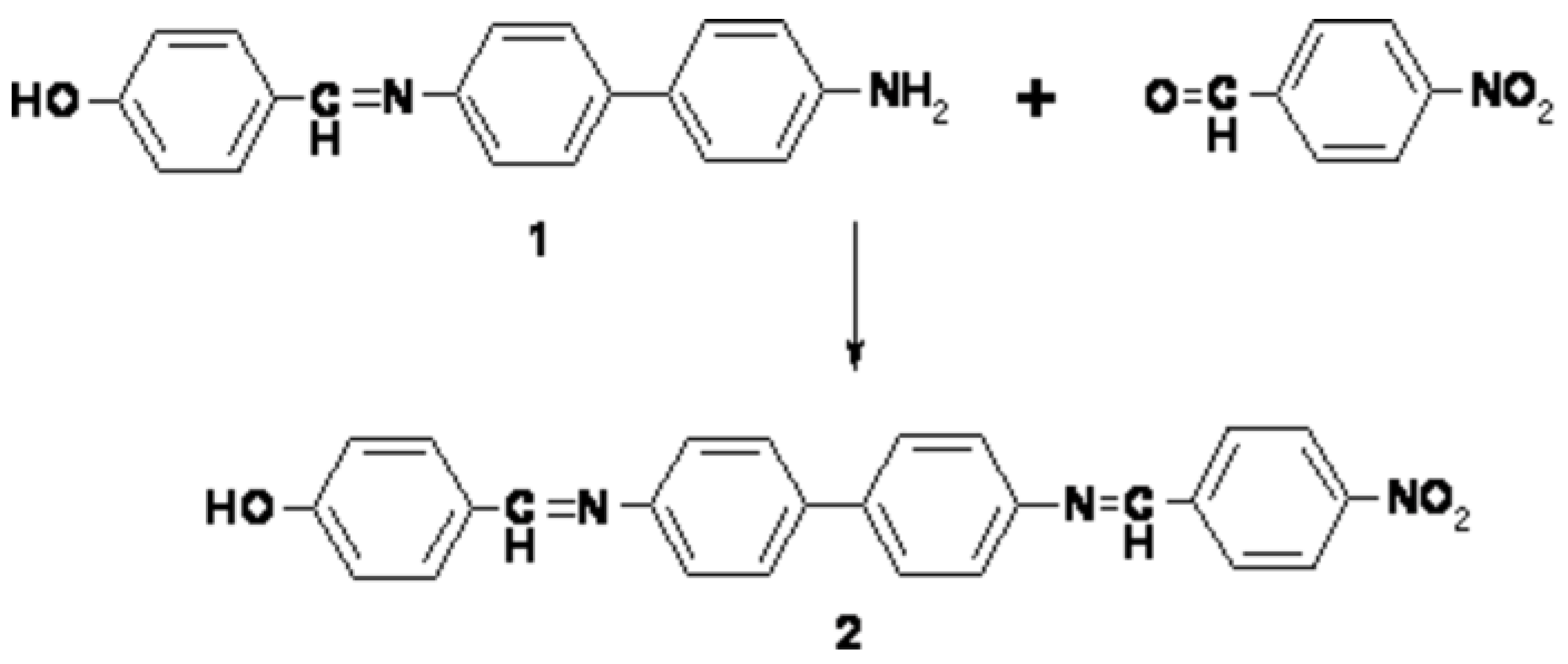
Synthesis of 4-[4-(4-nitrobenzylideneiminophenylene)phenyleneimino methylidene] phenol
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References:
- Cozan, V.; Avram, E. Eur. Polym. J. 2003, 39, 107–114.
- Racles, C.; Cozan, V. High Performance Polymers 2002, 14, 169–181.
© 2006 MDPI. All rights reserved.
Share and Cite
Marin, L.; Cozan, V. Synthesis of 4-[4-(4-nitrobenzylideneiminophenylene)phenyleneimino methylidene] phenol. Molbank 2006, 2006, M488. https://doi.org/10.3390/M488
Marin L, Cozan V. Synthesis of 4-[4-(4-nitrobenzylideneiminophenylene)phenyleneimino methylidene] phenol. Molbank. 2006; 2006(5):M488. https://doi.org/10.3390/M488
Chicago/Turabian StyleMarin, L., and V. Cozan. 2006. "Synthesis of 4-[4-(4-nitrobenzylideneiminophenylene)phenyleneimino methylidene] phenol" Molbank 2006, no. 5: M488. https://doi.org/10.3390/M488
APA StyleMarin, L., & Cozan, V. (2006). Synthesis of 4-[4-(4-nitrobenzylideneiminophenylene)phenyleneimino methylidene] phenol. Molbank, 2006(5), M488. https://doi.org/10.3390/M488




