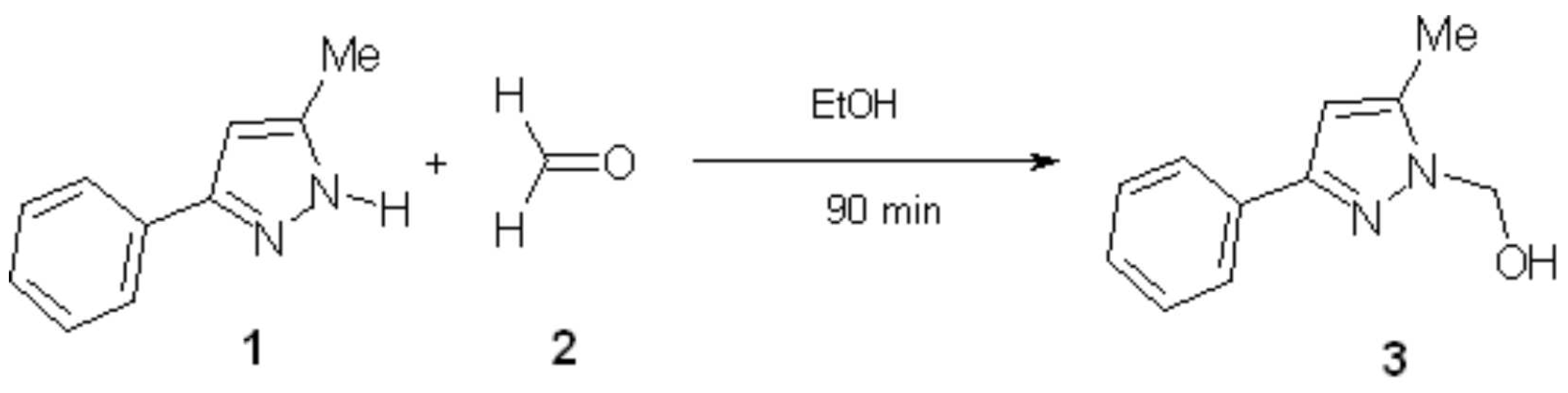
1-Hydroxymethyl-3-phenyl-5-methylpyrazole
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References and Notes:
- Ali, S. S.; Ashraf, C. M.; Younas, M.; Ehsan, A. Pak. J. Sci. Ind. Res. 1993, 36, 502–510.
- Driessen, W. J. R.. Neth. Chem. Soc. 1982, 101, 441.
- Bouabdallah, I.; Zidane, I; Ramdani, A. Rapport de DESA; Faculty of Sciences: Oujda, Maroc, 2001. [Google Scholar]
- Sample Availability: Available from MDPI.
© 2005 MDPI. All rights reserved.
Share and Cite
Bouabdallah, I.; Ramdani, A.; Zidane, I.; Touzani, R. 1-Hydroxymethyl-3-phenyl-5-methylpyrazole. Molbank 2005, 2005, M427. https://doi.org/10.3390/M427
Bouabdallah I, Ramdani A, Zidane I, Touzani R. 1-Hydroxymethyl-3-phenyl-5-methylpyrazole. Molbank. 2005; 2005(3):M427. https://doi.org/10.3390/M427
Chicago/Turabian StyleBouabdallah, Ibrahim, Abdelkrim Ramdani, Ismail Zidane, and Rachid Touzani. 2005. "1-Hydroxymethyl-3-phenyl-5-methylpyrazole" Molbank 2005, no. 3: M427. https://doi.org/10.3390/M427
APA StyleBouabdallah, I., Ramdani, A., Zidane, I., & Touzani, R. (2005). 1-Hydroxymethyl-3-phenyl-5-methylpyrazole. Molbank, 2005(3), M427. https://doi.org/10.3390/M427



