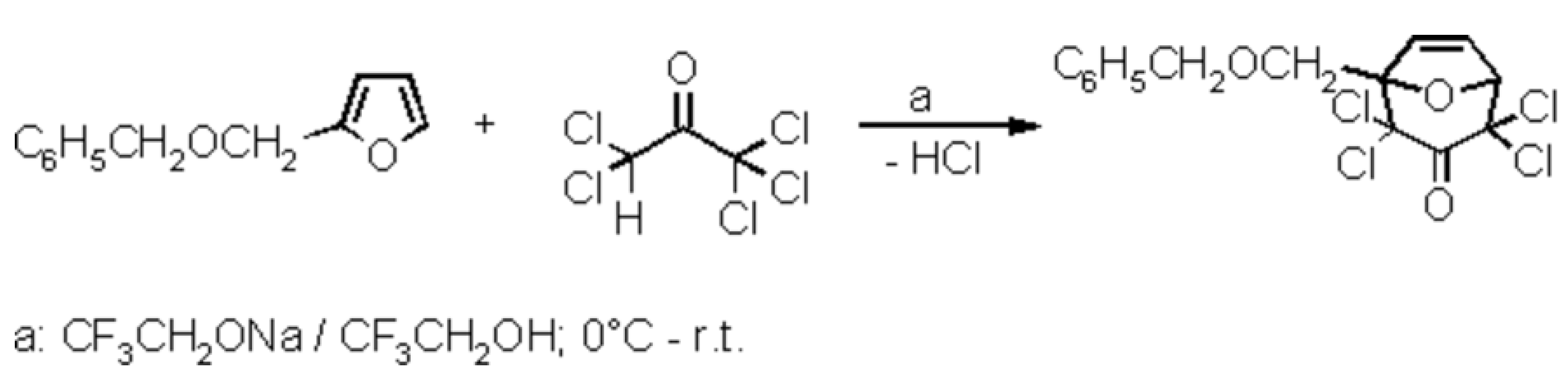
1-Benzyloxymethyl-2,2,4,4-tetrachloro-8-oxabicyclo[3.2.1]oct-6-en-3-one
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References and Notes
- Achmatowicz, O.; Burzynska, M. H. Tetrahedron 1982, 38, 3507–3513.
- Bugrova, L. V.; Rudnev, G. K.; Kristich, A. I.; Radchenko, V. I.; Mishchenko, L. F. Zh. Prikl. Khim. (Leningrad) 1973, 46, 1529–1533, J. Appl. Chem. USSR 1973, 2, 1627–1631.
- Föhlisch, B.; Gehrlach, E.; Herter, R. Angew. Chem. 1982, 94, 144, Angew. Chem. Suppl. 1982, 94, 241; Angew. Chem., Int. Ed. Engl. 1982, 21, 137.Sendelbach, S.; Schwetzler-Raschke, R.; Radl, A.; Kaiser, R.; Henle, G. H.; Korfant, H.; Reiner, S.; Föhlisch, B. J. Org. Chem. 1999, 64, 3398–3408.
- This work-up, i.e. precipitation by careful addition of water, was also used for the preparation of oxabicyclic compounds from pentachloroacetone and furan (71% yield, m.p. 90–91 °C), 2-methylfuran (63%, m.p. 56–57 °C), 3-methylfuran (88%, m.p. 89–90 °C), 2,5-dimethylfuran (89%, 112–113 °C), and 2-(3-butenyl)-3-methylfuran (86%, m.p. 59–60 °C)[3]. If the products do not crystallize immediately, the oily precipitate becomes solid on scratching with a glass rod. The compounds are pure enough for dechlorination, but may be purified by sublimation in vacuo.
© 2004 MDPI. All rights reserved.
Share and Cite
Kreiselmeier, G.; Föhlisch, B. 1-Benzyloxymethyl-2,2,4,4-tetrachloro-8-oxabicyclo[3.2.1]oct-6-en-3-one. Molbank 2004, 2004, M390. https://doi.org/10.3390/M390
Kreiselmeier G, Föhlisch B. 1-Benzyloxymethyl-2,2,4,4-tetrachloro-8-oxabicyclo[3.2.1]oct-6-en-3-one. Molbank. 2004; 2004(1):M390. https://doi.org/10.3390/M390
Chicago/Turabian StyleKreiselmeier, Günter, and Baldur Föhlisch. 2004. "1-Benzyloxymethyl-2,2,4,4-tetrachloro-8-oxabicyclo[3.2.1]oct-6-en-3-one" Molbank 2004, no. 1: M390. https://doi.org/10.3390/M390
APA StyleKreiselmeier, G., & Föhlisch, B. (2004). 1-Benzyloxymethyl-2,2,4,4-tetrachloro-8-oxabicyclo[3.2.1]oct-6-en-3-one. Molbank, 2004(1), M390. https://doi.org/10.3390/M390



