Combustion Analysis of Different Olive Residues
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
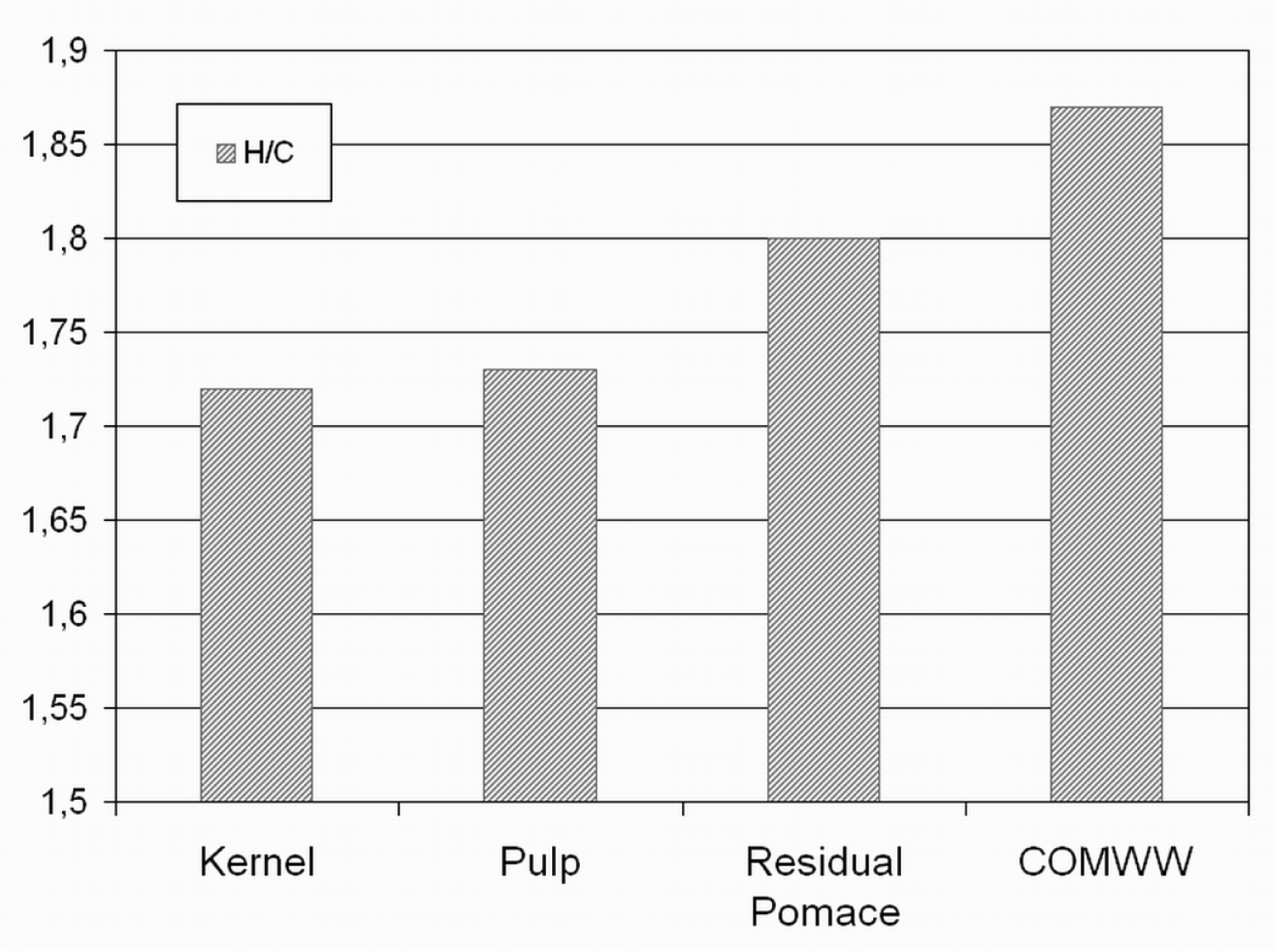
3.1. Physical and Chemical Characteristics
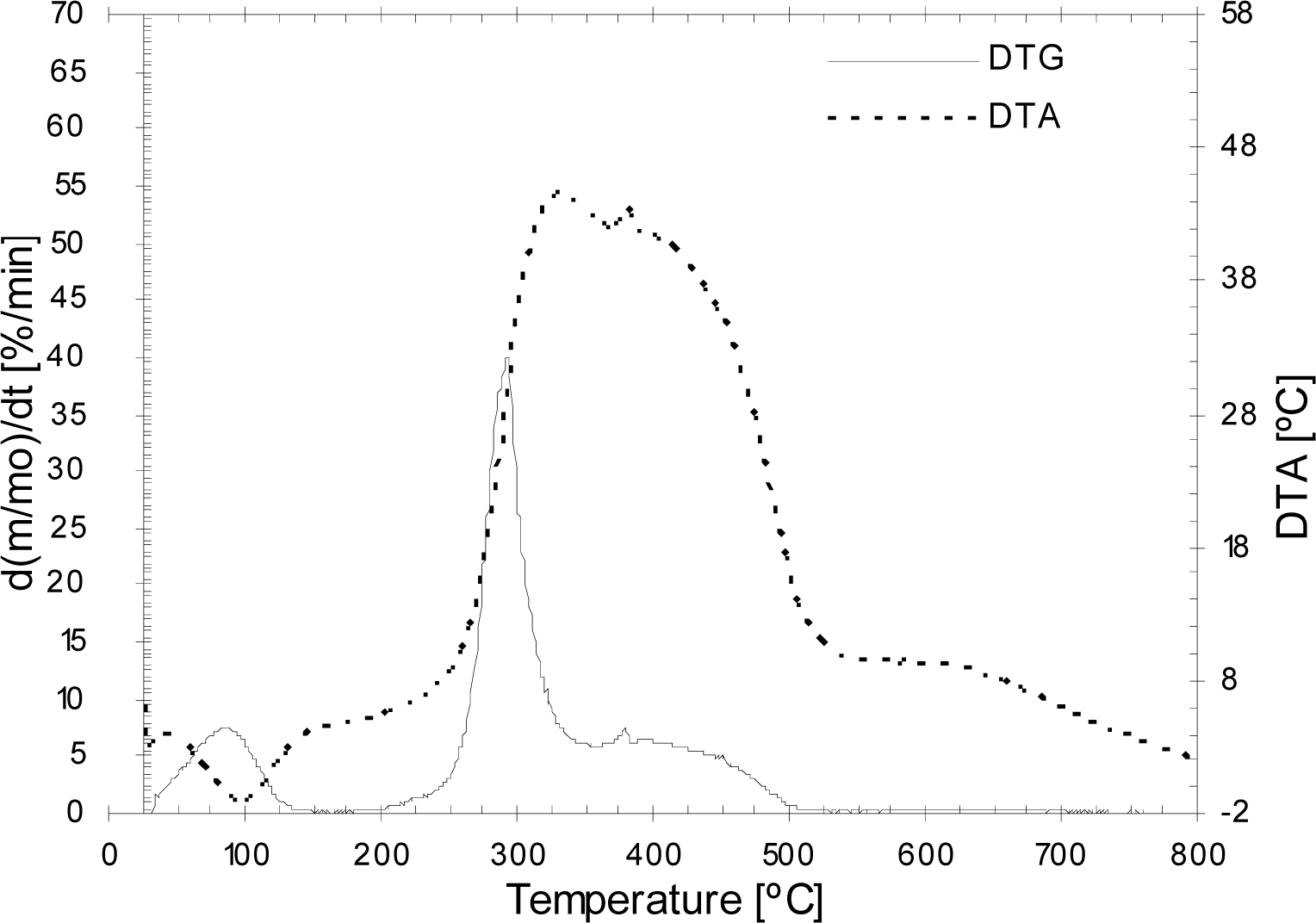
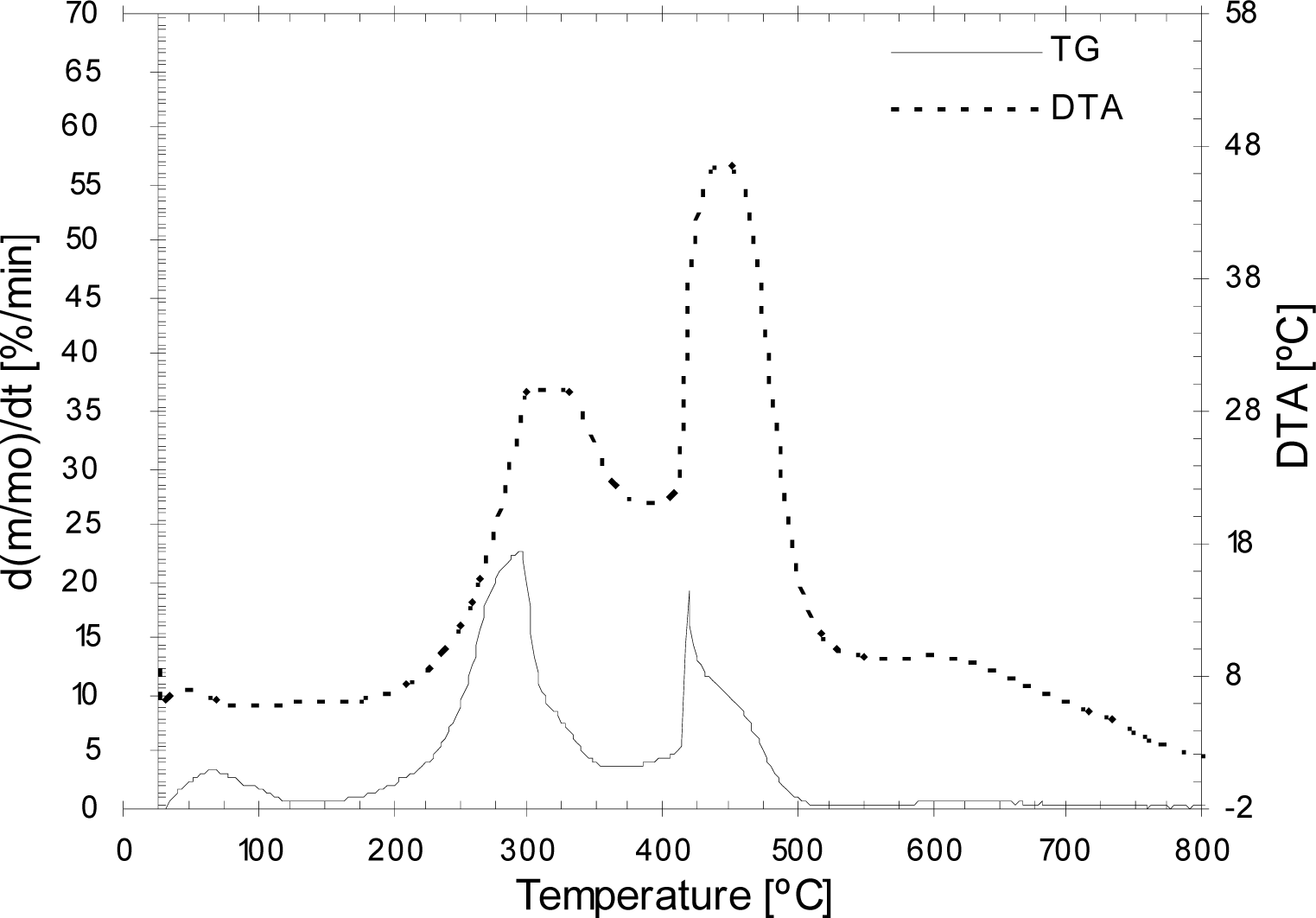
3.2. Thermogravimetric analysis of the olive pit, pulp and residual olive cake
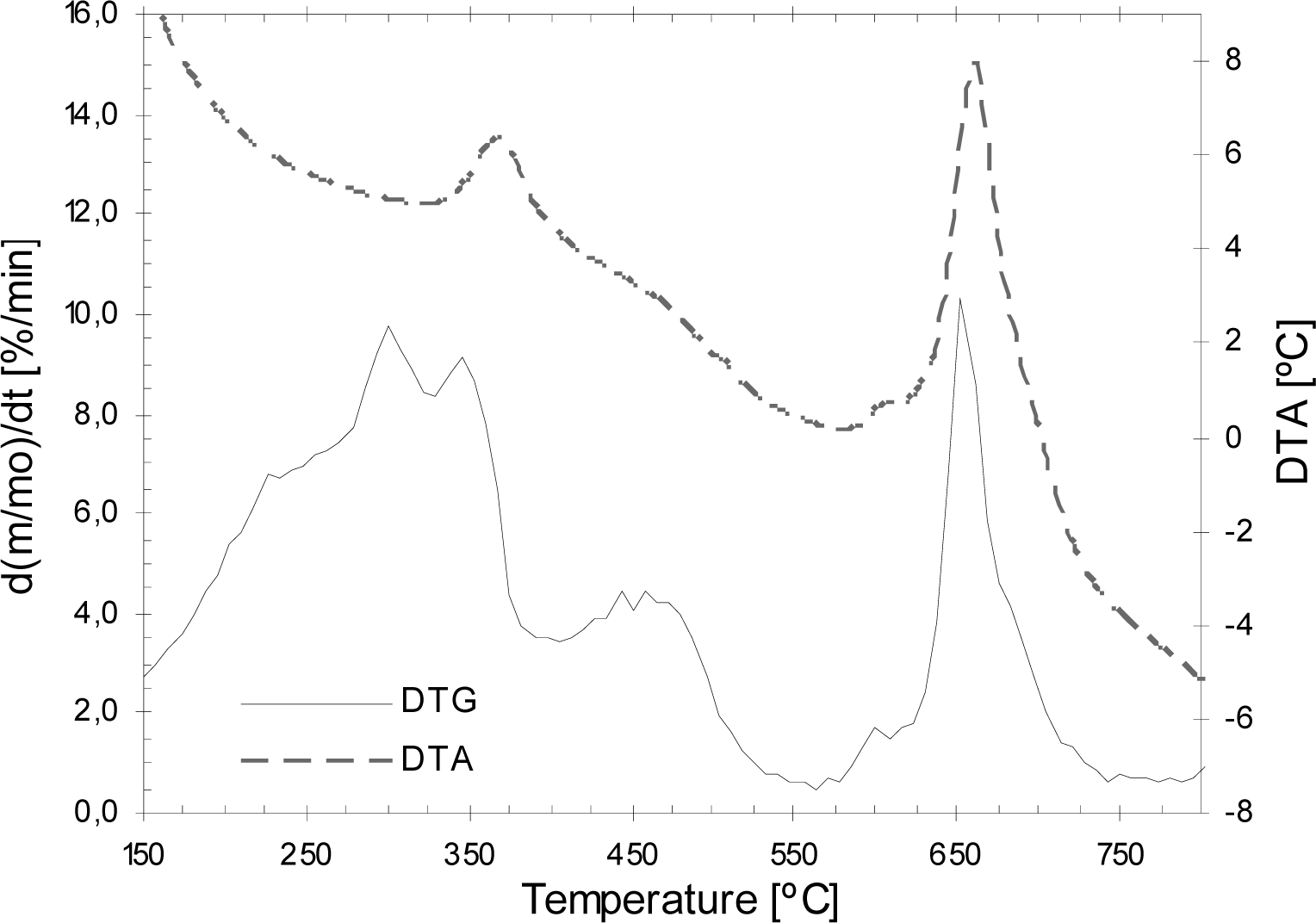
3.3. Thermogravimetric analysis of the COMWW
4. Conclusions
Acknowledgments
References and Notes
- Leung, D; Wang, C. Kinetic study of scrap tyre pyrolysis and combustion. Journal of Analytical and Applied Pyrolysis 1998, 45, 153–169. [Google Scholar]
- Demirbas, A. Combustion characteristics of different biomass fuels. Progress in Energy and Combustion Science 2004, 30, 219–230. [Google Scholar]
- Helsen, L; Van den Bulck, E. Kinetics of the low-temperature pyrolysis of chromated copper arsenate-treated wood. Journal of Analytical and Applied Pyrolysis 2000, 53, 51–79. [Google Scholar]
- Vamvuka, D; Kastanaki, E; Lasithiotakis, M; Papanicolaou, C. Combustion behaviour of xylite/lignite mixtures. Carbon 2004, 42, 351–359. [Google Scholar]
- Ergüdenler, A; Ghaly, A. Determination of reaction kinetics of wheat straw using thermogravimetric analysis. Applied Biochemistry and Biotechnology 1992, 34–35(1), 75–91. [Google Scholar]
- Bining, A; Jenkins, B. Thermochemical reaction kinetics for rice straw from an aproximate integral technique; ASAE, St. Joseph: Michigan, Tech. Rep. 92–6029; 1992. [Google Scholar]
- Antal, M, Jr; Varhegyi, G. Cellulose pyrolysis kinetics: the current state of knowledge. Industrial and Engineering Chemistry Research 1995, 34(3), 703–717. [Google Scholar]
- Nassar, M. Kinetic studies on thermal degradation on nonwood plants. Wood and Fiber Science 1985, 17(2), 266–273. [Google Scholar]
- Haykiri-Açma, H. Combustion Characteristic of different biomass materials. Energy Conversion Management 2003, 44, 155–162. [Google Scholar]
- Russell, N; Beeley, T; Man, C; Gibbins, J; Williamson, J. Development of TG measurements of intrinsic char combustion reactivity for industrial and research purposes. Fuel Processing Technology 1998, 57, 113–130. [Google Scholar]
- Cumming, JW; McLaughlin, J. The termogravimetric behaviour of coal. Themochemical Acta 1982, 87, 253–272. [Google Scholar]
- Smith, SE; Neavel, RC; Hippo, EJ; Miller, RN. DTGA combustion of coals in the Exxon coal library. Fuel 1981, 60, 458–462. [Google Scholar]
- Antal, J; Friedman, HL; Rogers, F. Kinetics of cellulose pyrolysis in nitrogen and steam. Combustion Science and Technology 1980, 21(2), 141–149. [Google Scholar]
- Conesa, J; Font, R; Fullana, A; Caballero, J. Kinetic model for the combustion of tyre wastes. Fuel 1998, 77(13), 1469–1475. [Google Scholar]
- Williams, P; Besler, S. Thermogravimetric analysis of the components of biomass. In Advances in Thermochemical Biomass Conversion; Blaxkie Academic: London, UK, 1994; pp. 771–783. [Google Scholar]
- Caballero, J; Font, R; Marcilla, A. Comparative study of the pyrolysis of almond shells and their fractions, holocellulose and lignin. Product yields and kinetics. Termochimica Acta 1996, 276, 57–77. [Google Scholar]
- Wendlandt, WW. Thermal methods of analysis, 2nd Ed ed; John Wiley Sons: New York, USA, 1974. [Google Scholar]
- Ghaly, A; Ergüdenler, A. Determination of the kinetic of barley straw using thermogravimetric analysis. Journal of Applied Biochemistry and Biotechnology 1994, 45–46(1), 35–50. [Google Scholar]
- Maiti, S; Purakayastha, S; Ghosh, B. Thermal characterization of mustard straw and stalk in nitrogen at different heating rates. Fuel 2007, 86, 1513–1518. [Google Scholar]
- Lester, E; Gong, M; Thompson, A. A method for source apportionment in biomass/coal blends using thermogravimetric analysis. Anal Appl Pyrolysis 2007, 80, 111–117. [Google Scholar]
- García Ibañez, P; Sánchez, M; Cabanillas, A. Thermogravimetric analysis of olive-oil residue in air atmosphere. Fuel Processing Technology 2006, 87, 103–107. [Google Scholar]
- Lapuerta, M; Hernández, JJ; Rodríguez, J. Kinetics of devolatilisation of forestry wastes from thermogravimetric analysis. Biomass and Bioenergy 2004, 27(4), 385–391. [Google Scholar]
- Van Loo, S; Koppejan, J. Handbook of Biomass Combustion and Cofiring; Twente University Press: Enschede, The Netherlands, 2003. [Google Scholar]
- Demirbas, A. Relationships between lignin contents and heating values of biomass. Energy Conversion and Management 2001, 42, 183–188. [Google Scholar]
- Department of the Army, Solid waste incineration, U.S. Army Corps of Engineers, Tech. Rep., 1998.
- Wilen, C; Stahlberg, P; Sipila, K; Ahokas, J. Energy from biomass and wastes; Elsevier Applied Science: London, UK, 1987; Chapter: Pelletization and combustion of straw; pp. 469–83. [Google Scholar]
- Obernberger, I. Decentralized biomass combustion: state of the art and future development. Biomass and Bioenergy 1998, 14(1), 33–56. [Google Scholar]
- Ross, AB; Jones, JM; Kubacki, ML; Bridgeman, T. Classification of macroalgae as fuel and its thermochemical behaviour. Bioresource Technology 2008, (in press). [Google Scholar]
- García Ibáñez, P. Estudio del tratamiento de orujillo de aceituna mediante el proceso de gasificación en un reactor de lecho fluidizado; Ph.D. Thesis; Facultad de Ciencias Químicas, Universidad Complutense de Madrid, 2004. [Google Scholar]
- Alonso, M; Borrego, A; Alvarez, D; Kalkreuth, W; Menédez, R. Physicochemical transformations of coal particles during pyrolysis and combustion. Fuel 2001, 80, 1857–1870. [Google Scholar]
- Milne, T. Biomass gasification principles and technology; Noyes Data Corporation: New Jersey, USA, 1981; pp. 91–118. [Google Scholar]
- Huang, M; Li, X. Thermal degradation cellulose and cellulose esters. Journal of Polymer Science 1998, 68, 293–304. [Google Scholar]
- Benfell, K; Beamish, B; Rodgers, K. Aspects of combustion behaviour of coals from some New Aeland lignite-coal regions determined by thermogravimetry. Thermochemical Acta 1997, 297, 79–84. [Google Scholar]
- Ghetti, P; Ricca, L; Angelini, L. Thermal analysis of biomass and corresponding pyrolysis products. Fuel 1996, 75, 565–573. [Google Scholar]
- Zheng, G; Kozinski, J. Thermal events occurring during the combustion of biomass residue. Fuel 2000, 79, 181–192. [Google Scholar]
- Carpenter, AM. Coal blending for power stations; IEA Coal Research; Tech. Rep. IEA/81; 1995. [Google Scholar]
- Su, S; Pohl, JH; Holcombe, D; Hart, J. Techniques to determine ignition, flame stability and burnout of blended coals in P.F. power station boilers. Progress in Energy and Combustion Science 2001, 27, 75–98. [Google Scholar]
- Rhéna, C; Öhmanb, M; Grefa, R; Wästerlunda, I. Effect of raw material composition in woody biomass pellets on combustion characteristics. Biomass and Bioenergy 2007, 31, 66–72. [Google Scholar]
- Rubiera, F; Arenillas, A; Pevida, C; García, R; Pis, JJ; Steel, KM; Patrick, JW. Coal structure and reactivity changes induced by chemical demineralisation. Fuel Processing Technology 2002, 79, 273–279. [Google Scholar]
- Vamvuka, D; Troulinos, S; Kastanaki, E. The effect of mineral matter on the physical and chemical activation of low rank coal and biomass materials. Fuel 2006, 85, 1763–1771. [Google Scholar]


| Pit | Pulp | Residual Olive cake | COMWW | |
|---|---|---|---|---|
| Ultimate Analysis (% dry basis) | ||||
| Carbon | 52.270 | 55.205 | 54.895 | 50.075 |
| Hydrogen | 7.485 | 7.960 | 8.215 | 7.795 |
| Nitrogen | 0.060 | 1.995 | 2.220 | 2.125 |
| Oxygen | 40.097 | 34.042 | 34.386 | 39.752 |
| Sulfur | <0.1 | <0.1 | <0.1 | <0.1 |
| Chlorine | 0.088 | 0.798 | 0.284 | 0.253 |
| Proximate Analysis (% dry basis) | ||||
| Volatile | 80.94 | 79.10 | 77.77 | 69.29 |
| Ash | 0.56 | 5.60 | 4.31 | 18.82 |
| Fixed Carbon | 18.50 | 15.30 | 17.92 | 11.89 |
| Moisture (% wet basis) | 9–10 | 6–6.5 | 5.5–6 | 70–73 |
| Chemical Analysis (% dry-extractive free basis) | ||||
| Cellulose | 18.6 | 12.1 | 12.4 | 0.6 |
| Hemicellulose | 25.1 | 12.2 | 14.4 | 0.6 |
| Lignin | 39.3 | 43.3 | 42.8 | 51.3 |
| Higher heating value (MJ/kg, dry basis) | 20.61 | 23.39 | 22.42 | 21.36 |
| Higher heating value (MJ/kg, dry-ash free basis) | 20.70 | 24.35 | 23.27 | 26.29 |
| Lower heating value (MJ/kg, dry basis) | 18.96 | 21.64 | 20.61 | 19.64 |
| Pit | Pulp | Residual Olive cake | COMWW | |
|---|---|---|---|---|
| Initial (ºC) | 1165 | 1185 | 1165 | 1040 |
| Deformation temperature (ºC) | 1320 | 1195 | 1185 | 1050 |
| Hemisphere temperature (ºC) | 1335 | 1235 | 1195 | 1055 |
| Flow temperature (ºC) | 1340 | 1350 | 1330 | 1215 |
| Na2O | K2O | CaO | MgO | SiO2 | P2O5 | Fe2O3 | MnO | TiO | Al2O3 | |
|---|---|---|---|---|---|---|---|---|---|---|
| Pit (%) | 0.95 | 42.88 | 8.44 | 1.51 | 12.98 | 3.43 | 0.91 | 0.05 | 0.07 | 0.90 |
| Pulp (%) | 0.38 | 39.61 | 7.52 | 2.04 | 18.51 | 6.22 | 1.37 | 0.04 | 0.11 | 1.95 |
| R. O. cake (%) | 0.51 | 34.62 | 8.50 | 1.87 | 24.59 | 5.64 | 1.34 | 0.04 | 0.13 | 2.44 |
| COMWW (%) | 0.94 | 57.29 | 2.84 | 2.49 | 1.14 | 7.84 | 0.37 | 0.03 | 0.04 | 0.33 |
| Pit | Pulp | Residual Olive Cake | COMWW | |
|---|---|---|---|---|
| Initial temperature (ºC) | 220 | 183 | 181 | 161 |
| Maximum combustion rate (%/min) | 39.82 | 63.01 | 22.81 | 10.27 |
| Peak temperature (ºC) | 292 | 267 | 295 | 653 |
| Burnout temperature (ºC) | 530 | 509 | 519 | 743 |
| Rm (% min−1K−1) | 7.05 | 11.66 | 4.01 | 1.11 |
Share and Cite
Miranda, T.; Esteban, A.; Rojas, S.; Montero, I.; Ruiz, A. Combustion Analysis of Different Olive Residues. Int. J. Mol. Sci. 2008, 9, 512-525. https://doi.org/10.3390/ijms9040512
Miranda T, Esteban A, Rojas S, Montero I, Ruiz A. Combustion Analysis of Different Olive Residues. International Journal of Molecular Sciences. 2008; 9(4):512-525. https://doi.org/10.3390/ijms9040512
Chicago/Turabian StyleMiranda, Teresa, Alberto Esteban, Sebastián Rojas, Irene Montero, and Antonio Ruiz. 2008. "Combustion Analysis of Different Olive Residues" International Journal of Molecular Sciences 9, no. 4: 512-525. https://doi.org/10.3390/ijms9040512
APA StyleMiranda, T., Esteban, A., Rojas, S., Montero, I., & Ruiz, A. (2008). Combustion Analysis of Different Olive Residues. International Journal of Molecular Sciences, 9(4), 512-525. https://doi.org/10.3390/ijms9040512





