Dietary Berries and Ellagic Acid Prevent Oxidative DNA Damage and Modulate Expression of DNA Repair Genes
Abstract
1. Introduction
2. Results
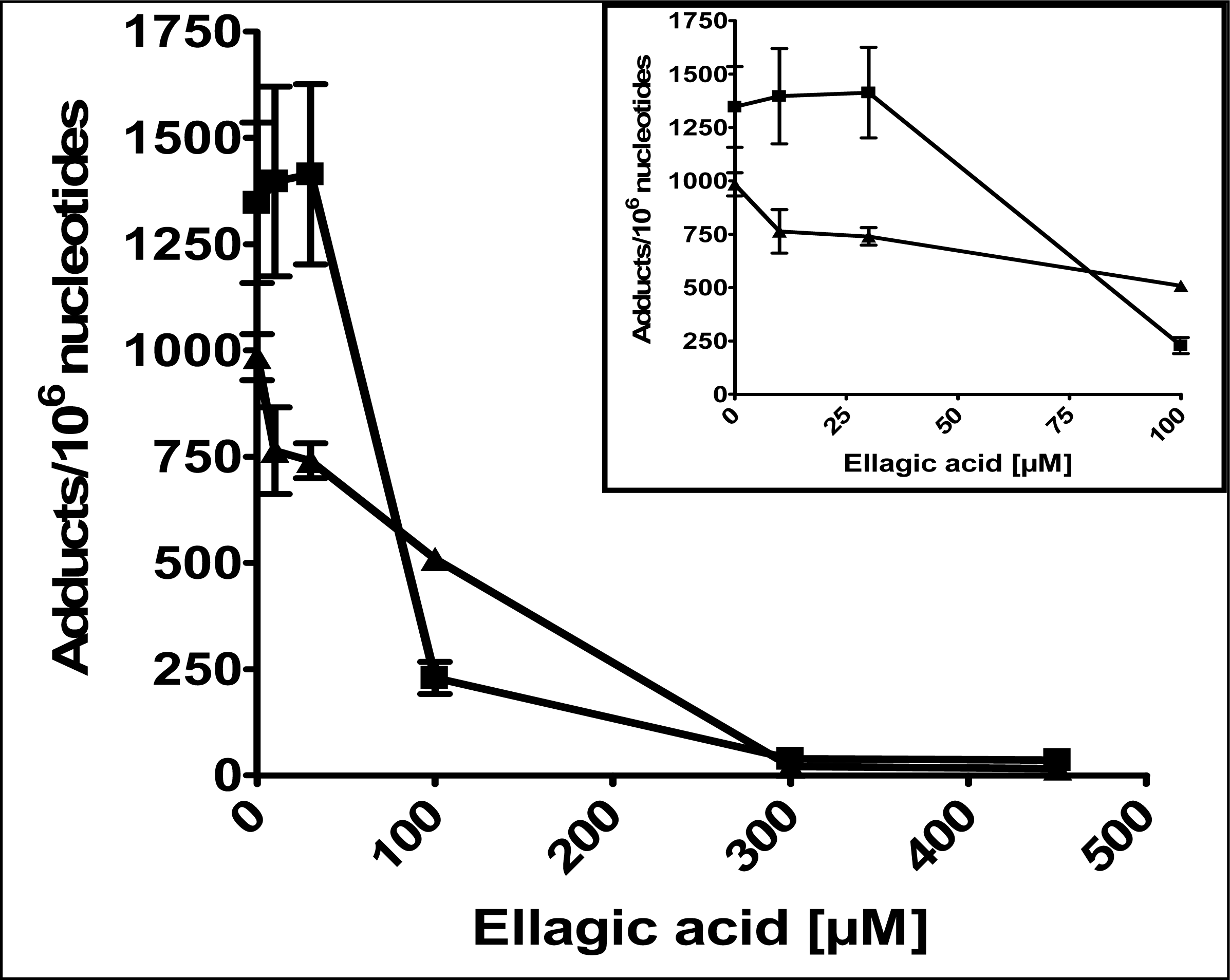
2.1 Modulation of 4E2/CuCl2-induced oxidative DNA adducts by ellagic acid
2.2 Modulation of baseline endogenous oxidative-DNA damage by ellagic acid and berries
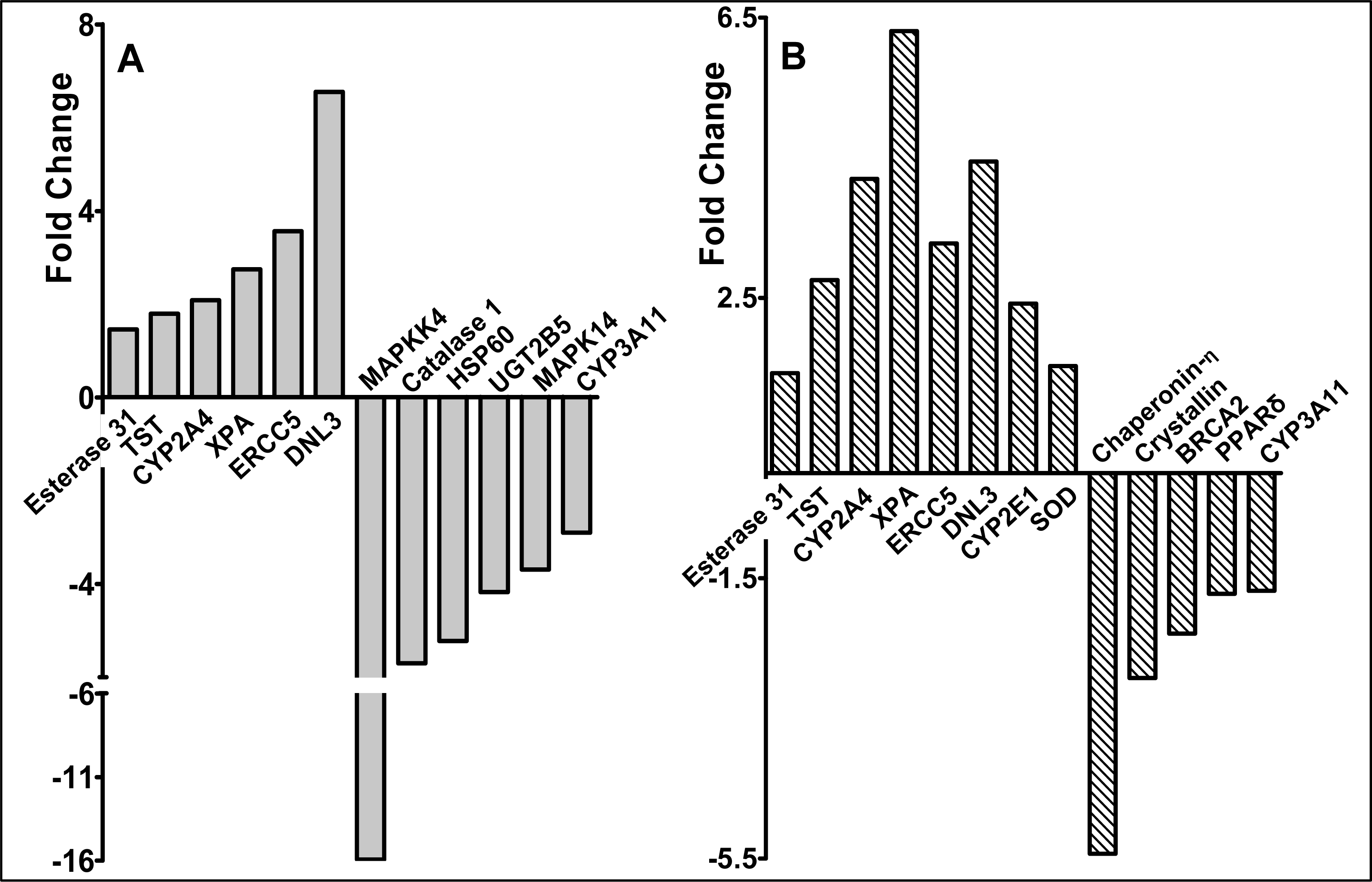
2.3 Modulation of gene expression by red raspberry and ellagic acid diets
3. Discussion and Conclusions
Conclusion
4. Materials and Methods
4.1 Chemicals
4.2 Induction of DNA damage by 4E2/CuCl2
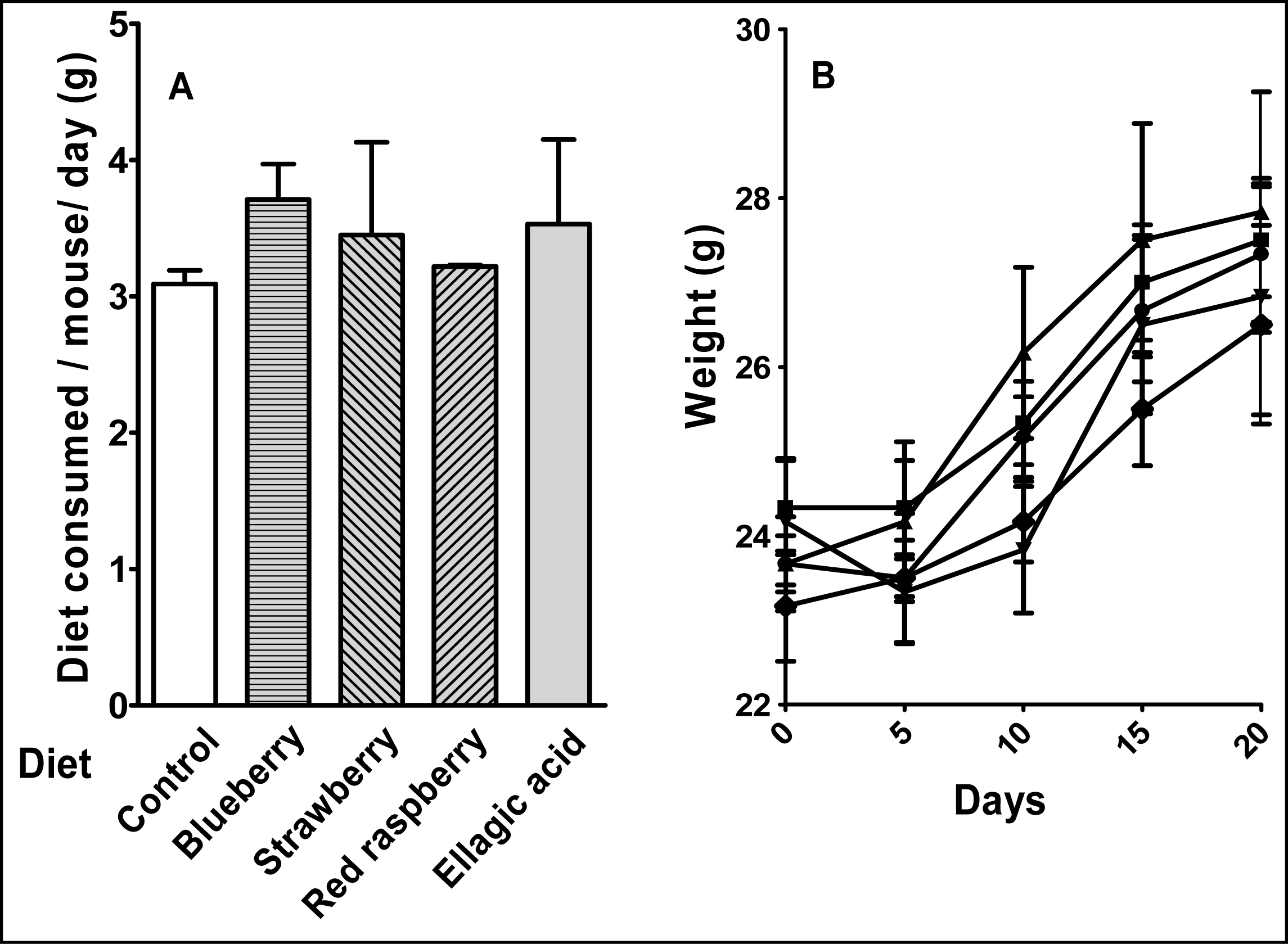
4.3 Animals and diet
4.4 Analysis of polar oxidative DNA adducts by 32P-postlabeling/TLC
4.5 Gene expression analysis
4.6 Statistics
Acknowledgments
References and Notes
- ACS. Cancer Facts and Figures-2007; American Cancer Society: Atlanta, USA, 2007. [Google Scholar]
- Liehr, JG. Is estradiol a genotoxic mutagenic carcinogen? Endocr. Rev. 2000, 21(1), 40–54. [Google Scholar]
- Mailander, PC; Meza, JL; Higginbotham, S; Chakravarti, D. Induction of A.T to G.C mutations by erroneous repair of depurinated DNA following estrogen treatment of the mammary gland of ACI rats. J. Steroid Biochem. Mol. Biol. 2006, 101(4–5), 204–215. [Google Scholar]
- Russo, J; Hu, YF; Yang, X; Russo, IH. Developmental, cellular, and molecular basis of human breast cancer. J. Natl. Cancer Inst. Monogr. 2000, 27, 17–37. [Google Scholar]
- Li, Y; Trush, MA; Yager, JD. DNA damage caused by reactive oxygen species originating from a copper-dependent oxidation of the 2-hydroxy catechol of estradiol. Carcinogenesis 1994, 15(7), 1421–1427. [Google Scholar]
- Yager, JD. Endogenous estrogens as carcinogens through metabolic activation. J. Natl. Cancer Inst. Monogr. 2000, 27, 67–73. [Google Scholar]
- Devanesan, P; Santen, RJ; Bocchinfuso, WP; Korach, KS; Rogan, EG; Cavalieri, E. Catechol estrogen metabolites and conjugates in mammary tumors and hyperplastic tissue from estrogen receptor-alpha knock-out (ERKO)/Wnt-1 mice: implications for initiation of mammary tumors. Carcinogenesis 2001, 22(9), 1573–1576. [Google Scholar]
- Yue, W; Santen, RJ; Wang, JP; Li, Y; Verderame, MF; Bocchinfuso, WP; Korach, KS; Devanesan, P; Todorovic, R; Rogan, EG; Cavalieri, EL. Genotoxic metabolites of estradiol in breast: potential mechanism of estradiol induced carcinogenesis. J. Steroid Biochem. Mol. Biol. 2003, 86(3–5), 477–486. [Google Scholar]
- Dizdaroglu, M; Jaruga, P; Birincioglu, M; Rodriguez, H. Free radical-induced damage to DNA: mechanisms and measurement. Free Radic. Biol. Med. 2002, 32(11), 1102–1115. [Google Scholar]
- Gupta, RC; Arif, JM. An improved (32)P-postlabeling assay for the sensitive detection of 8-oxodeoxyguanosine in tissue DNA. Chem. Res. Toxicol. 2001, 14(8), 951–957. [Google Scholar]
- Gupta, RC; Ravoori, S; Vadhanam, MV. In Adducteomics-Novel subgroups of DNA adducts detected in human tissues. In Proc. Am. Assn. Can. Res, Washington DC, 2003; Washington DC, 2003; p. 1217. [Google Scholar]
- Ravoori, S; Vadhanam, MV; Davey, DD; Srinivasan, C; Nagarajan, B; Gupta, RC. Modulation of novel DNA adducts during human uterine cervix cancer progression. Int. J. Oncol. 2006, 29(6), 1437–43. [Google Scholar]
- Gupta, RC; Ravikuma, MNV; Vadhanam, MV; Arif, JM. In Detection of Oxidatively damaged Polar DNA adducts by 32P-postlabeling; 2001; p. 468. [Google Scholar]
- Smith, WA; Arif, JM; Gupta, RC. 1,2-dithiole-3-thione and its structural analogue oltipraz are potent inhibitors of dibenz. Int. J. Cancer 2001, 91(1), 132–136. [Google Scholar]
- Smith, WA; Freeman, JW; Gupta, RC. Effect of chemopreventive agents on DNA adduction induced by the potent mammary carcinogen dibenzo[a,l]pyrene in the human breast cells MCF-7. Mutat. Res. 2001, 480–481, 97–108. [Google Scholar]
- Smith, WA; Gupta, RC. Determining efficacy of cancer chemopreventive agents using a cell-free system concomitant with DNA adduction. Mutat. Res. 1999, 425(1), 143–152. [Google Scholar]
- Daniel, EM; Krupnick, AS; Heur, Y; Blinzler, JA; Nims, RW; Stoner, GD. Extraction, stability and quantitation of ellagic acid in various fruits and nuts. J. Food Composition and Analysis 1989, 2, 338–349. [Google Scholar]
- Newbold, RR; Liehr, JG. Induction of uterine adenocarcinoma in CD-1 mice by catechol estrogens. Cancer Res. 2000, 60(2), 235–237. [Google Scholar]
- Srinivasan, P; Vadhanam, MV; Arif, JM; Gupta, RC. A rapid screening assay for antioxidant potential of natural and synthetic agents in vitro. Int. J. Oncol. 2002, 20(5), 983–986. [Google Scholar]
- Smart, RC; Huang, MT; Chang, RL; Sayer, JM; Jerina, DM; Conney, AH. Disposition of the naturally occurring antimutagenic plant phenol, ellagic acid, and its synthetic derivatives, 3-O-decylellagic acid and 3,3′-di-O-methylellagic acid in mice. Carcinogenesis 1986, 7(10), 1663–1667. [Google Scholar]
- Hiraku, Y; Yamashita, N; Nishiguchi, M; Kawanishi, S. Catechol estrogens induce oxidative DNA damage and estradiol enhances cell proliferation. Int. J. Cancer 2001, 92(3), 333–337. [Google Scholar]
- Frelon, S; Douki, T; Favier, A; Cadet, J. Hydroxyl radical is not the main reactive species involved in the degradation of DNA bases by copper in the presence of hydrogen peroxide. Chem. Res. Toxicol. 2003, 16(2), 191–197. [Google Scholar]
- Oikawa, S; Hirosawa, I; Hirakawa, K; Kawanishi, S. Site specificity and mechanism of oxidative DNA damage induced by carcinogenic catechol. Carcinogenesis 2001, 22(8), 1239–1245. [Google Scholar]
- Oakley, GG; Devanaboyina, U; Robertson, LW; Gupta, RC. Oxidative DNA damage induced by activation of polychlorinated biphenyls (PCBs): implications for PCB-induced oxidative stress in breast cancer. Chem. Res. Toxicol. 1996, 9(8), 1285–1292. [Google Scholar]
- Aruoma, OI; Halliwell, B; Gajewski, E; Dizdaroglu, M. Copper-ion-dependent damage to the bases in DNA in the presence of hydrogen peroxide. Biochem. J. 1991, 273 Pt 3, 601–604. [Google Scholar]
- Dixit, R; Gold, B. Inhibition of N-methyl-N-nitrosourea-induced mutagenicity and DNA methylation by ellagic acid. Proc. Natl. Acad. Sci. USA 1986, 83(21), 8039–8043. [Google Scholar]
- Teel, RW. Ellagic acid binding to DNA as a possible mechanism for its antimutagenic and anticarcinogenic action. Cancer Lett. 1986, 30(3), 329–336. [Google Scholar]
- Mandal, S; Shivapurkar, NM; Galati, AJ; Stoner, GD. Inhibition of N-nitrosobenzylmethylamine metabolism and DNA binding in cultured rat esophagus by ellagic acid. Carcinogenesis 1988, 9(7), 1313–1316. [Google Scholar]
- Dixit, R; Teel, RW; Daniel, FB; Stoner, GD. Inhibition of benzo(a)pyrene and benzo(a)pyrene-trans-7,8-diol metabolism and DNA binding in mouse lung explants by ellagic acid. Cancer Res. 1985, 45(7), 2951–2956. [Google Scholar]
- Mandal, S; Ahuja, A; Shivapurkar, NM; Cheng, SJ; Groopman, JD; Stoner, GD. Inhibition of aflatoxin B1 mutagenesis in Salmonella typhimurium and DNA damage in cultured rat and human tracheobronchial tissues by ellagic acid. Carcinogenesis 1987, 8(11), 1651–1656. [Google Scholar]
- Wu, X; Beecher, GR; Holden, JM; Haytowitz, DB; Gebhardt, SE; Prior, RL. Concentrations of anthocyanins in common foods in the United States and estimation of normal consumption. J. Agric. Food Chem. 2006, 54(11), 4069–4075. [Google Scholar]
- Ahn, D; Putt, D; Kresty, L; Stoner, GD; Fromm, D; Hollenberg, PF. The effects of dietary ellagic acid on rat hepatic and esophageal mucosal cytochromes P450 and phase II enzymes. Carcinogenesis 1996, 17(4), 821–828. [Google Scholar]
- Mandal, S; Stoner, GD. Inhibition of N-nitrosobenzylmethylamine-induced esophageal tumorigenesis in rats by ellagic acid. Carcinogenesis 1990, 11(1), 55–61. [Google Scholar]
- Carlton, PS; Kresty, LA; Siglin, JC; Morse, MA; Lu, J; Morgan, C; Stoner, GD. Inhibition of N-nitrosomethylbenzylamine-induced tumorigenesis in the rat esophagus by dietary freeze-dried strawberries. Carcinogenesis 2001, 22(3), 441–446. [Google Scholar]
- Aziz, R; Nines, R; Rodrigio, K; Harris, K; Hudson, T; Gupta, A; Morse, MA; Carlton, PS; Stoner, GD. The effect of freeze-dried blueberries on N-nitrosomethylbenzylamine tumorigenesis in the rat esophagus. Pharmaceutical Biology 2002, 40 Supplement 1, 43–49. [Google Scholar]
- Wang, SY; Lin, HS. Antioxidant activity in fruits and leaves of blackberry, raspberry, and strawberry varies with cultivar and developmental stage. J. Agric. Food Chem. 2000, 48(2), 140–146. [Google Scholar]
- Singh, K; Khanna, AK; Chander, R. Hepatoprotective activity of ellagic acid against carbon tetrachloride induced hepatotoxicity in rats. Indian J. Exp. Biol. 1999, 37(10), 1025–1026. [Google Scholar]
- Thresiamma, KC; Kuttan, R. Inhibition of liver fibrosis by ellagic acid. Indian J. Physiol. Pharmacol. 1996, 40(4), 363–6. [Google Scholar]
- Ahmed, S; Rahman, A; Saleem, M; Athar, M; Sultana, S. Ellagic acid ameliorates nickel induced biochemical alterations: diminution of oxidative stress. Hum. Exp. Toxicol. 1999, 18(11), 691–698. [Google Scholar]
- Chakraborty, S; Roy, M; Bhattacharya, RK. Prevention and repair of DNA damage by selected phytochemicals as measured by single cell gel electrophoresis. J. Environ. Pathol. Toxicol. Oncol. 2004, 23(3), 215–226. [Google Scholar]
- Ferguson, LR. Role of plant polyphenols in genomic stability. Mutat. Res. 2001, 475(1–2), 89–111. [Google Scholar]
- Freese, R. Markers of oxidative DNA damage in human interventions with fruit and berries. Nutr. Cancer 2006, 54(1), 143–147. [Google Scholar]
- Gupta, RC. 32P-Postlabeling for Detection of DNA Adducts. In Technologies for Detection of DNA Damage and Mutations; Pfeifer, P, Ed.; Plenum Press: New York, 1996; pp. 45–61. [Google Scholar]
- Aiyer, HS; Srinivasan, C; Gupta, RC. Dietary berries and ellagic acid diminish estrogen-mediated mammary tumors in ACI rats. Nutr. Cancer 2008, (in press). [Google Scholar]
- Reeves, PG; Nielsen, FH; Fahey, GC, Jr. AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J. Nutr. 1993, 123(11), 1939–1951. [Google Scholar]
- Patriotis, PC; Querec, TD; Gruver, BN; Brown, TR; Patriotis, C. ArrayExplorer, a program in Visual Basic for robust and accurate filter cDNA array analysis. Biotechniques 2001, 31(4). [Google Scholar]
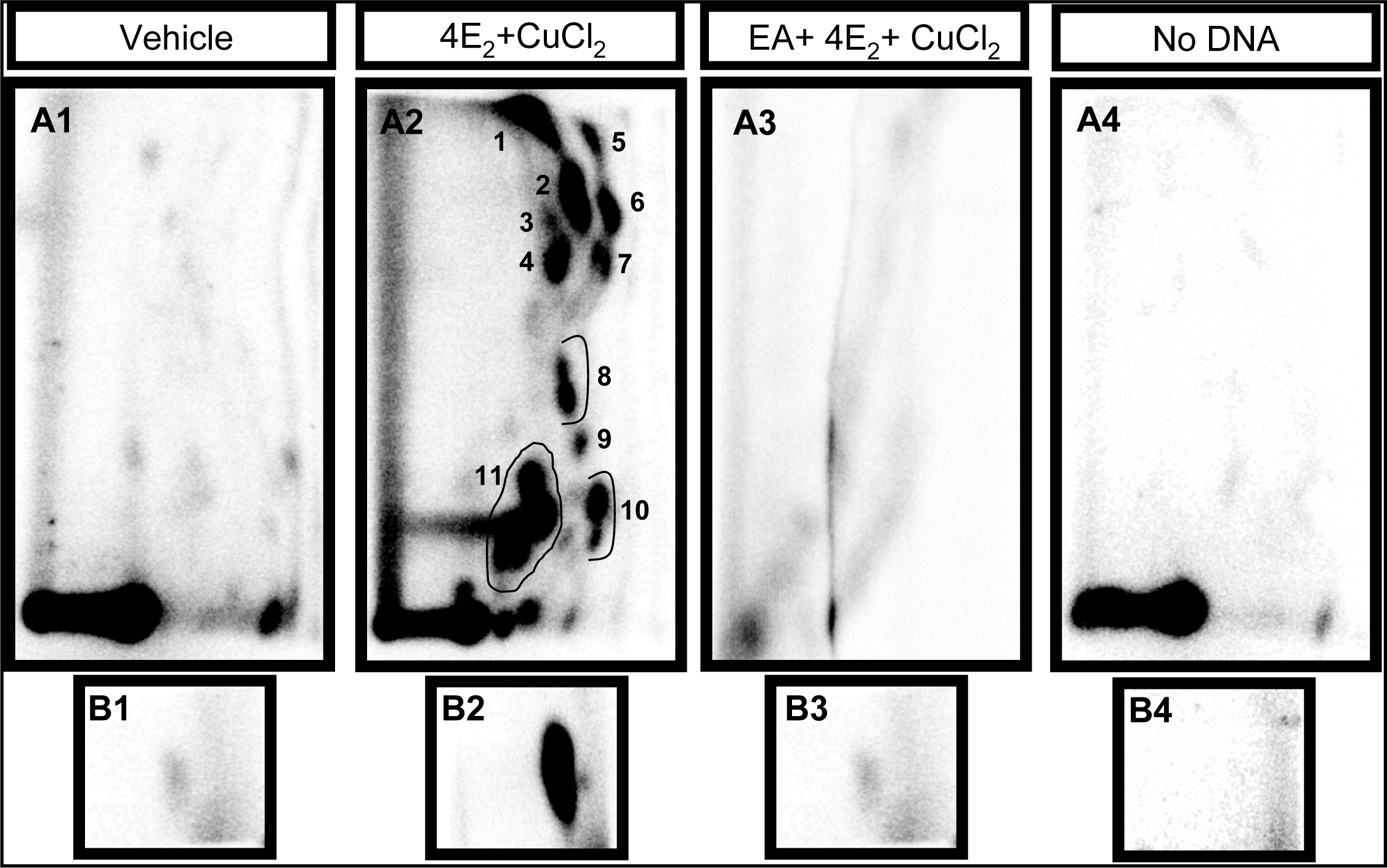
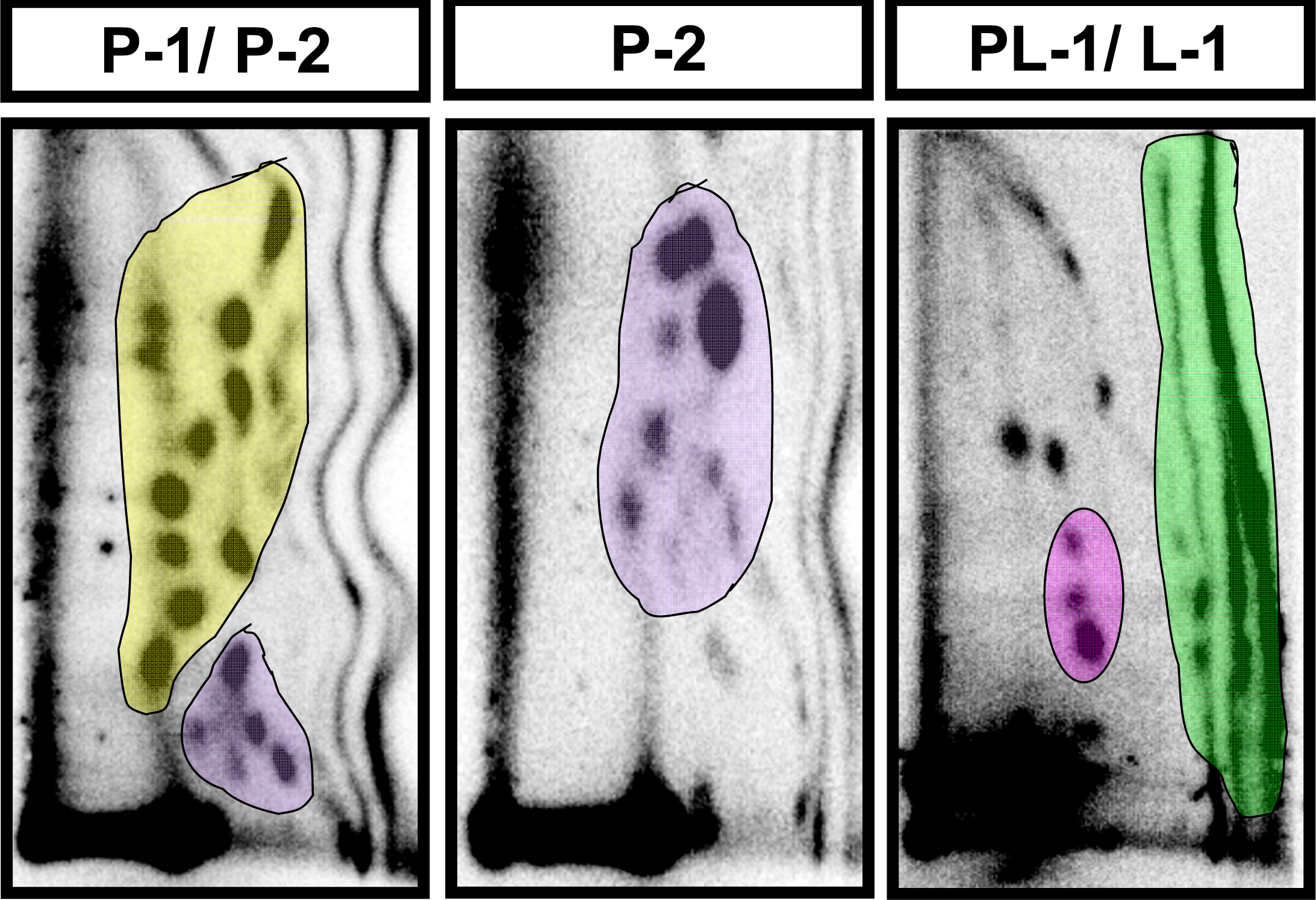
| Adduct subgroup | P-1 | P-2 | PL-1 | L-1 | Total |
|---|---|---|---|---|---|
| Adducts/ 109 nucleotides (mean ± SE) | |||||
| Control (n=4) | 4810 ± 596 | 3398 ± 316 | 197 ± 32 | 3074 ± 697 | 11,479 ± 1128 |
| Strawberry (n=4) | 3726 ± 1053 | 2182 ± 647 | 187 ± 23 | 2404 ± 502 | 8452 ± 1561 |
| Blueberry (n=5) | 4982 ± 513 | 2243 ± 285 | 186 ± 37 | 1870 ± 248 | 8833 ± 668 |
| Red raspberry (n=5) | 1803 ± 239 * | 1090 ± 149 ** | 84 ± 16 | 1790 ± 433 | 4733 ± 333** |
| p value | p < 0.05 | p < 0.001 | NS | NS | p < 0.001 |
| Ellagic acid (n=5) | 2060 ± 306 * | 1207 ± 106 * | 101 ± 8 | 2540 ± 254 | 5908 ± 540 * |
| p value | p < 0.05 | p < 0.01 | NS | NS | p < 0.01 |
| Percent of composition | ||||
|---|---|---|---|---|
| Ingredients | AIN-93M diet | Control diet | Berry diet | Ellagic acid diet |
| Corn starch | 46.6% | 36.03% | 31.03% | 36.03% |
| Dextrose | 15.5% | 36.04% | 36.04% | 36.04% |
| Sucrose | 10% | 0% | 0% | 0% |
| Berries | – | 0% | 5% | 0% |
| Total CHO calories | 72.1% | 72.07% | 72.07% | 72.07% |
| Casien | 14% | |||
| Soy Bean Oil | 4% | |||
| Fiber | 5% | |||
| AIN-93 Vitamin Mix | 1% | |||
| AIN-93 Mineral mix | 3.5% | |||
| L- Cysteine | 0.18% | |||
| Choline Bitartrate | 0.25% | |||
Share and Cite
Aiyer, H.S.; Vadhanam, M.V.; Stoyanova, R.; Caprio, G.D.; Clapper, M.L.; Gupta, R.C. Dietary Berries and Ellagic Acid Prevent Oxidative DNA Damage and Modulate Expression of DNA Repair Genes. Int. J. Mol. Sci. 2008, 9, 327-341. https://doi.org/10.3390/ijms9030327
Aiyer HS, Vadhanam MV, Stoyanova R, Caprio GD, Clapper ML, Gupta RC. Dietary Berries and Ellagic Acid Prevent Oxidative DNA Damage and Modulate Expression of DNA Repair Genes. International Journal of Molecular Sciences. 2008; 9(3):327-341. https://doi.org/10.3390/ijms9030327
Chicago/Turabian StyleAiyer, Harini S., Manicka V. Vadhanam, Radka Stoyanova, Gerard D. Caprio, Margie L. Clapper, and Ramesh C. Gupta. 2008. "Dietary Berries and Ellagic Acid Prevent Oxidative DNA Damage and Modulate Expression of DNA Repair Genes" International Journal of Molecular Sciences 9, no. 3: 327-341. https://doi.org/10.3390/ijms9030327
APA StyleAiyer, H. S., Vadhanam, M. V., Stoyanova, R., Caprio, G. D., Clapper, M. L., & Gupta, R. C. (2008). Dietary Berries and Ellagic Acid Prevent Oxidative DNA Damage and Modulate Expression of DNA Repair Genes. International Journal of Molecular Sciences, 9(3), 327-341. https://doi.org/10.3390/ijms9030327






