Polyphenolic Profile and Antioxidant Activities of Oolong Tea Infusion under Various Steeping Conditions
Abstract
:1. Introduction
2. Results and Discussion
2.1 Extraction yield of oolong tea
2.2 DPPH radical scavenging activity of oolong tea infusion
2.3 Superoxide radical scavenging activity of oolong tea infusion
2.4 Effects of various infusion conditions on total phenolic content
2.5 Major catechins in oolong tea infusion
2.6 Relationship of polyphenolic profile and antioxidant activity of AEOT
3. Conclusions
4. Experimental Section
4. 1Plant materials
4.2 Reagents
4.3 Sample preparation
4.4 Scavenging DPPH· radical activity
4.5 Superoxide radical scavenging activity
4.6 Total phenolic content
4.7 High performance liquid chromatography
4.8 Data analysis
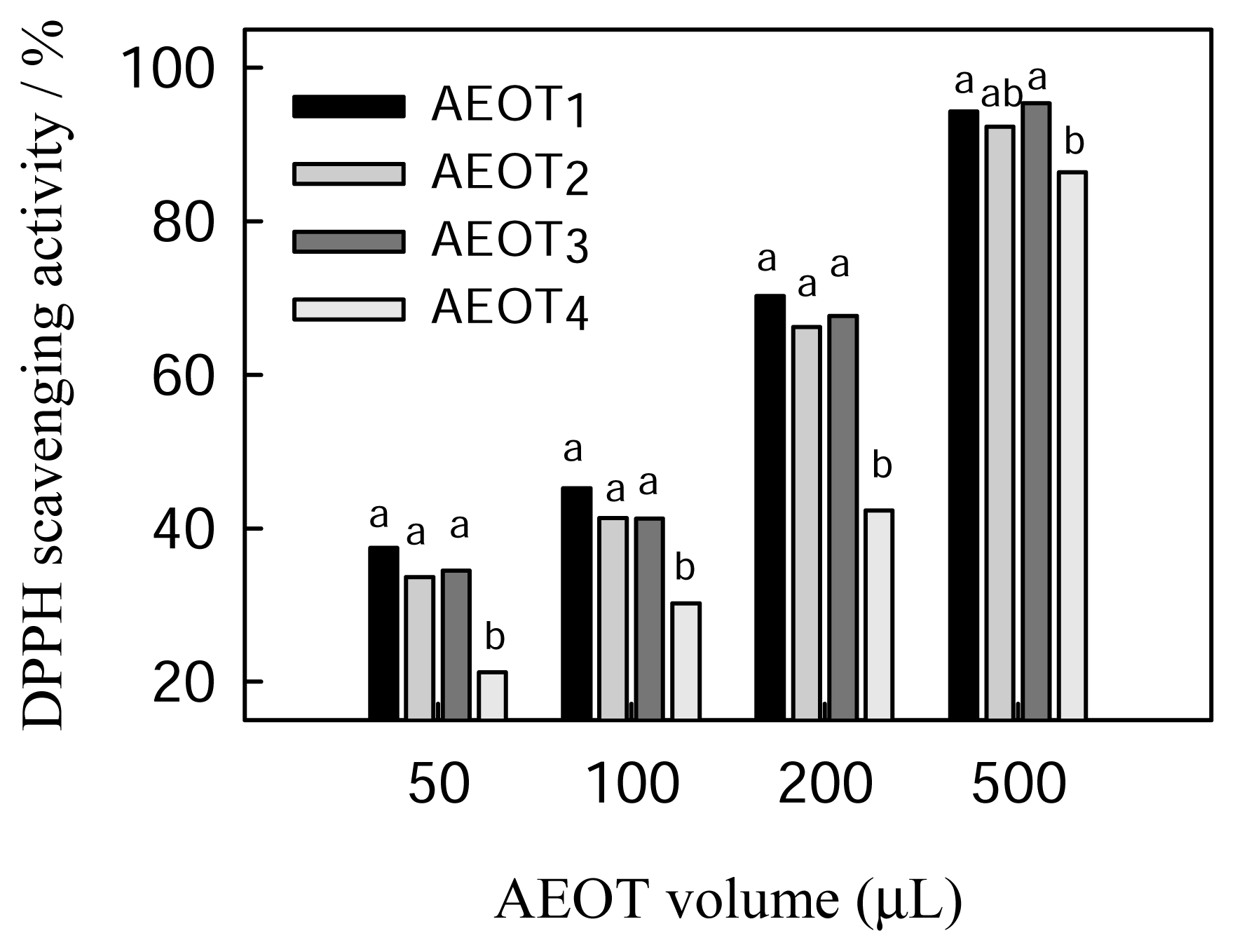
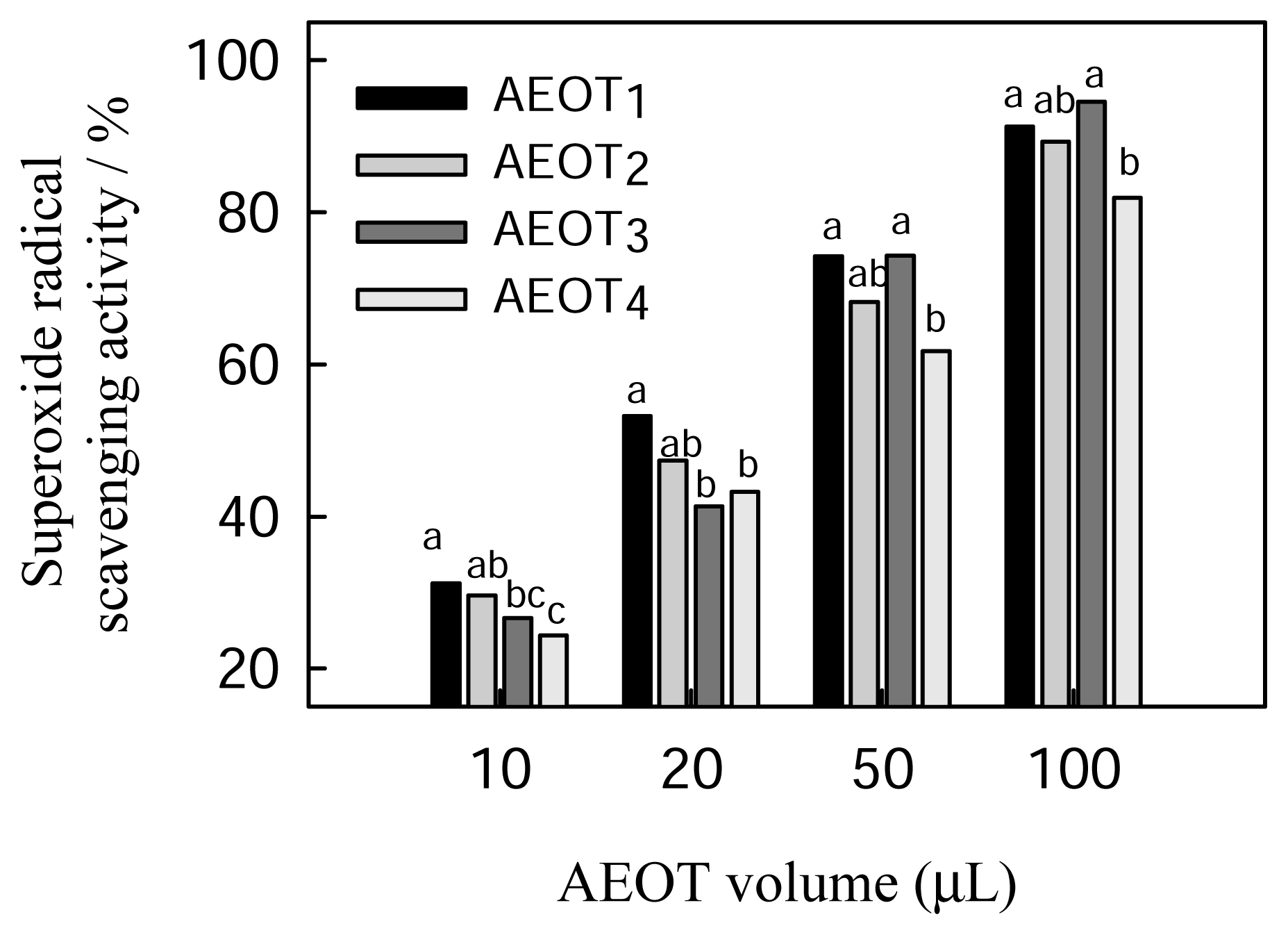
| Sample | Polyphenolic content (mg of GAE/mL) a | DPPH radical scavenging activity (EC50 μL) b | Superoxide radical scavenging activity (EC50 μL) c |
|---|---|---|---|
| AEOT 1 | 2.83 ab | 119.1 b | 24.4 c |
| AEOT 2 | 2.78 abc | 134.8 b | 31.1 ab |
| AEOT 3 | 2.93 a | 115.4 b | 21.2 c |
| AEOT 4 | 2.65 c | 251.0 a | 33.5 a |
| Vc d | 91.0 | 17.4 |
| Sample | EGCG | EGC | C | EC | ECG |
|---|---|---|---|---|---|
| AEOT1 | 18.12 a | 28.44 a | 0.6 a | 2.6 b | 7.32 b |
| AEOT2 | 15.79 ab | 26.11 a | 0.8 a | 3.4 ab | 10.36 b |
| AEOT3 | 17.52 a | 29.89 a | 0.9 a | 4.4 a | 14.33 a |
| AEOT4 | 12.46 b | 21.44 b | 0.7 a | 3.1 ab | 14.85 a |
| Correlation coefficients (r)a | ||
|---|---|---|
| DPPH radical scavenging activity (EC50) | Superoxide radical scavenging activity (EC50) | |
| EGCG | 0.9592 | 0.8718 |
| EGC | 0.9486 | 0.9327 |
| C | 0.2646 | 0.2828 |
| EC | 0.2646 | 0.4359 |
| ECG | 0.5558 | 0.1732 |
Acknowledgements
References
- Bloknina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: a Review. Ann. Bot.-London 2003, 91, 179–194. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M.C. Free radicals in biology and medicine, 3rd ed; Oxford University Press Inc: New York, 1999. [Google Scholar]
- Moskovitz, J.; Yim, K.A.; Choke, P.B. Free radicals and disease. Arch. Biochem. Biophys 2002, 397, 354–359. [Google Scholar]
- Zhao, B.L. Natural antioxidants for neurodegenerative diseases. Mol. Neurobiol 2005, 31, 283–293. [Google Scholar]
- Podsedek, A. Natural antioxidants and antioxidant capacity of Brassica vegetables: A Review. Food Sci. Technol.-LEB 2007, 40, 1–11. [Google Scholar]
- Zhu, Q.Y.; Hackman, R.M.; Ensunsa, J.L.; Holt, R.R.; Keen, C.L. Antioxidative activities of oolong tea. J. Agr. Food Chem 2002, 50, 6929–6934. [Google Scholar]
- Buschman, J.L. Green tea and cancer in humans: a review of the literature. Nutr. Cancer 1998, 31, 51–57. [Google Scholar]
- Yang, C.S. Tea and health. Nutrition 1999, 15, 946–949. [Google Scholar]
- Zuo, Y.; Chen, H.; Deng, Y. Simultaneous determination of catechins, caffeine and gallic acids in green, Oolong, black and puerh teas using HPLC with a photodiode array detector. Talanta 2002, 57, 307–316. [Google Scholar]
- Harbowy, M.E.; Balentine, D.A. Tea chemistry. Crit. Rev. Plant Sci 1997, 16, 415–480. [Google Scholar]
- Yen, G.C.; Chen, H.Y. Comparison of antimutagenic effect of various tea extracts (green, oolong, pouching and black tea). J. Food Prot 1994, 57, 54–58. [Google Scholar]
- Ohe, T.; Marutani, K.; Nakase, S. Catechins are not major components responsible for antigenotoxic effects of tea extracts against nitroarenes. Mutat. Res 2001, 496, 75–81. [Google Scholar]
- Gong, S.Y.; Gu, Z.L. Comparison on aroma and taste of oolong tea between two organoleptic evaluation methods. J. Tea Sci 2001, 21, 166–169. (in Chinese).. [Google Scholar]
- Astill, C.; Birch, M.R.; Dacombe, C.; Humphrey, P.G.; Martin, P.T. Factors affecting the caffeine and polyphenol contents of black and green tea infusions. J. Agr. Food Chem 2001, 49, 5340–5347. [Google Scholar]
- Khokhar, S.; Magnusdottir, S.G.M. Total phenol, catechin, and caffeine contents of teas commonly consumed in the United Kingdom. J. Agr. Food Chem 2002, 50, 565–570. [Google Scholar]
- Robinson, E.E.; Maxwell, S.R.J.; Thorpe, G.H.G. An investigation of the antioxidant activity of black tea using enhanced chemiluminescence. Free Radical Res 1997, 26, 291–302. [Google Scholar]
- Su, X.G.; Duan, J.; Jiang, Y.M.; Shi, J.; Kakuda, Y. Effects of soaking conditions on the antioxidant potentials of oolong tea. J. Food Compos. Anal 2006, 19, 348–353. [Google Scholar]
- Rice-Evans, C.; Miller, N.J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci 1997, 2, 152–159. [Google Scholar]
- Sanchez-Moreno, C. Methods used to evaluate the free radical scavenging activity in foods and biological systems. Food Sci. Technol. Int 2002, 8, 121–137. [Google Scholar]
- Hibasami, H.; Jin, Z.X.; Hasegawa, M.; Urakawa, K.; Nakagawa, M.; Ishii, Y.; Yoshioka, K. Oolong tea polyphenol extract induces apoptosis in human stomach cancer cells. Anticancer Res 2000, 20, 4403–4406. [Google Scholar]
- Standley, L.; Winterton, P.; Marnewick, J.L.; Gelderblom, W.C.A.; Joubert, E.; Britz, T.J. Influence of processing stages on antimutagenic and antioxidant potentials of Rooibos tea. J. Agr. Food Chem 2001, 49, 114–117. [Google Scholar]
- Fernandez, P.L.; Pablos, F.; Martin, M.J.; Gonzalez, A.G. Study of catechin and xanthine tea profiles as geographical tracers. J. Agr. Food Chem 2002, 50, 1833–1839. [Google Scholar]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: food sources and bioavailability. Am. J. Clin. Nutr 2004, 79, 727–747. [Google Scholar]
- Larrauri, J.A.; Sanchez-Moreno, C.; Saura-Calixto, F. Effect of temperature on the free radical scavenging capacity of extracts from red and white grape pomace peels. J. Agr. Food Chem 1998, 46, 2694–2697. [Google Scholar]
- Siddhurajua, P.; Mohanb, P.S.; Bechera, K. Studies on the antioxidant activity of Indian Laburnum (Cassia fistula L.): a preliminary assessment of crude extracts from stem bark, leaves, flowers and fruit pulp. Food Chem 2002, 79, 61–67. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Amer. J. Enol. Viticult 1965, 16, 144–158. [Google Scholar]
© 2007 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Su, X.; Duan, J.; Jiang, Y.; Duan, X.; Chen, F. Polyphenolic Profile and Antioxidant Activities of Oolong Tea Infusion under Various Steeping Conditions. Int. J. Mol. Sci. 2007, 8, 1196-1205. https://doi.org/10.3390/i8121196
Su X, Duan J, Jiang Y, Duan X, Chen F. Polyphenolic Profile and Antioxidant Activities of Oolong Tea Infusion under Various Steeping Conditions. International Journal of Molecular Sciences. 2007; 8(12):1196-1205. https://doi.org/10.3390/i8121196
Chicago/Turabian StyleSu, Xinguo, Jun Duan, Yueming Jiang, Xuewu Duan, and Feng Chen. 2007. "Polyphenolic Profile and Antioxidant Activities of Oolong Tea Infusion under Various Steeping Conditions" International Journal of Molecular Sciences 8, no. 12: 1196-1205. https://doi.org/10.3390/i8121196
APA StyleSu, X., Duan, J., Jiang, Y., Duan, X., & Chen, F. (2007). Polyphenolic Profile and Antioxidant Activities of Oolong Tea Infusion under Various Steeping Conditions. International Journal of Molecular Sciences, 8(12), 1196-1205. https://doi.org/10.3390/i8121196





