The Role and Mechanism of Nrf2 in Ameliorating Oxidative Stress and Inflammation in IR Mice by Aerobic Exercise
Abstract
1. Introduction
2. Result
2.1. Establish IR Model Mice by High-Fat Diet Feeding
2.2. Aerobic Exercise Reduces the Blood Lipid, Blood Glucose Levels and HOMA-IR in IR Mice
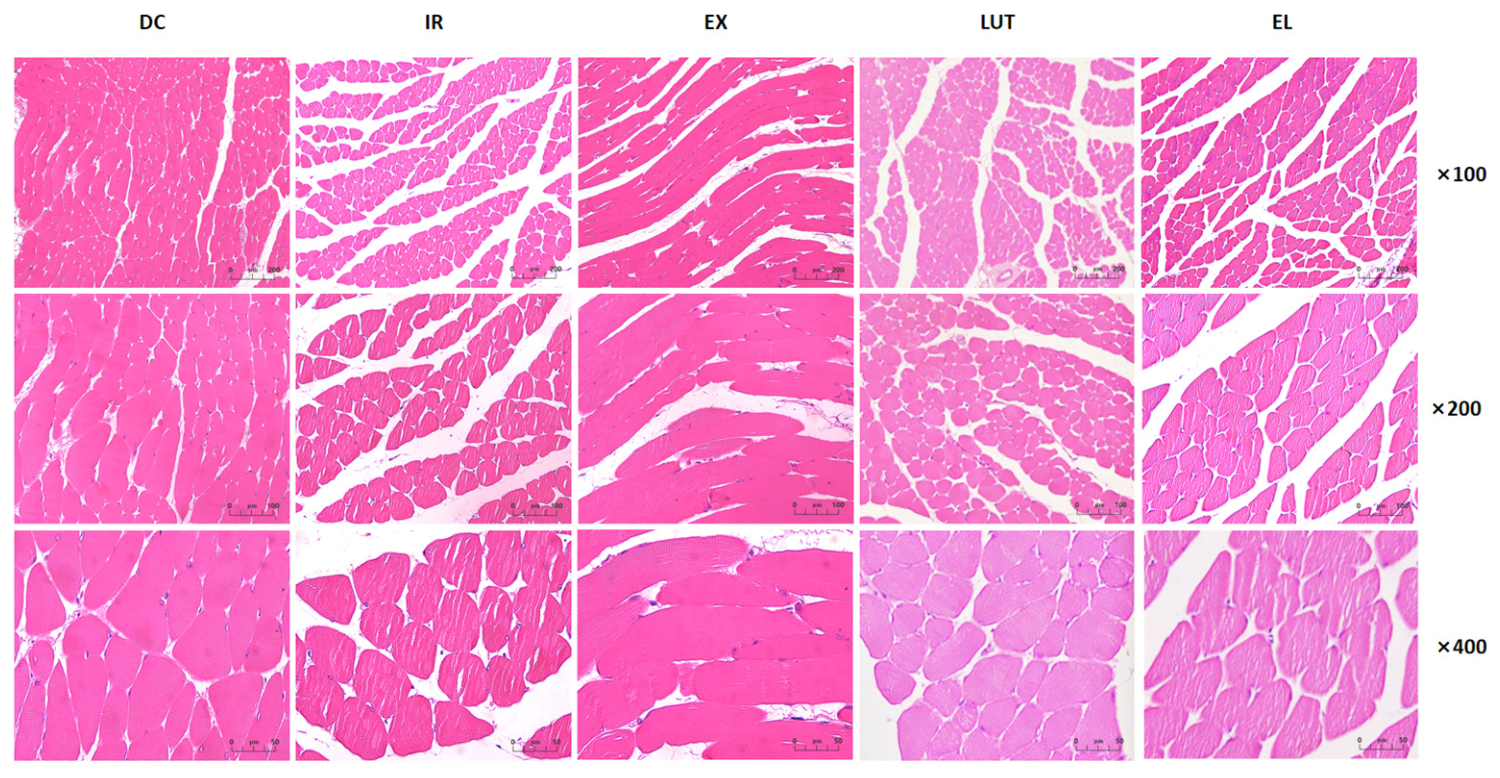
2.3. Aerobic Exercise Improves the Muscle Tissue Morphology of IR Mice
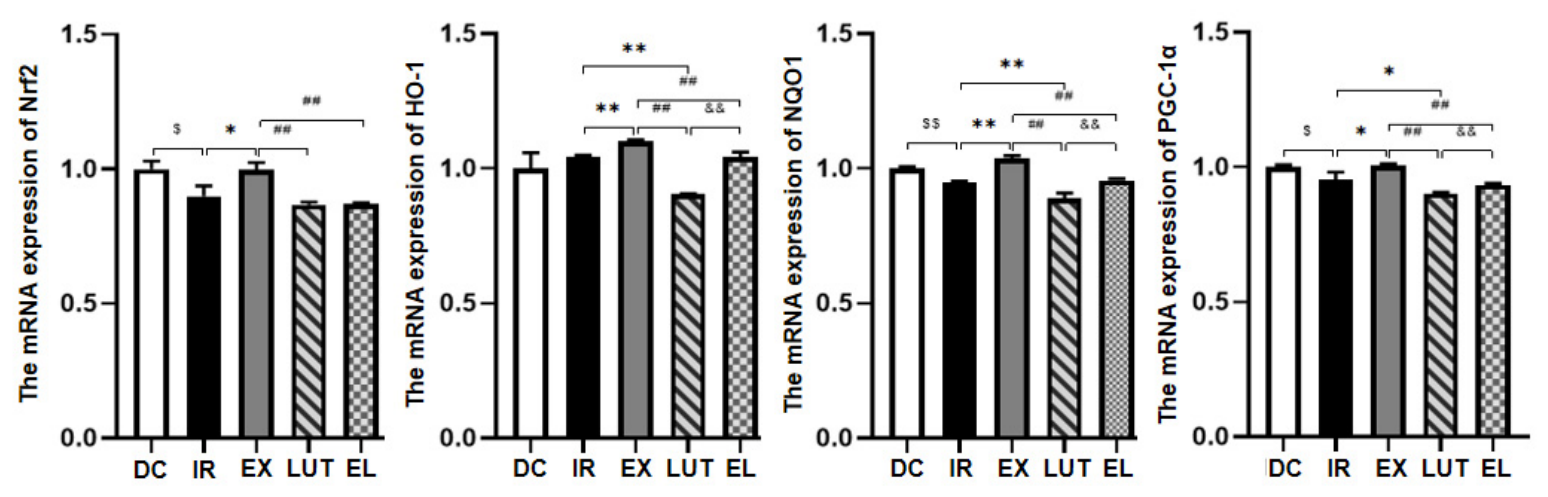
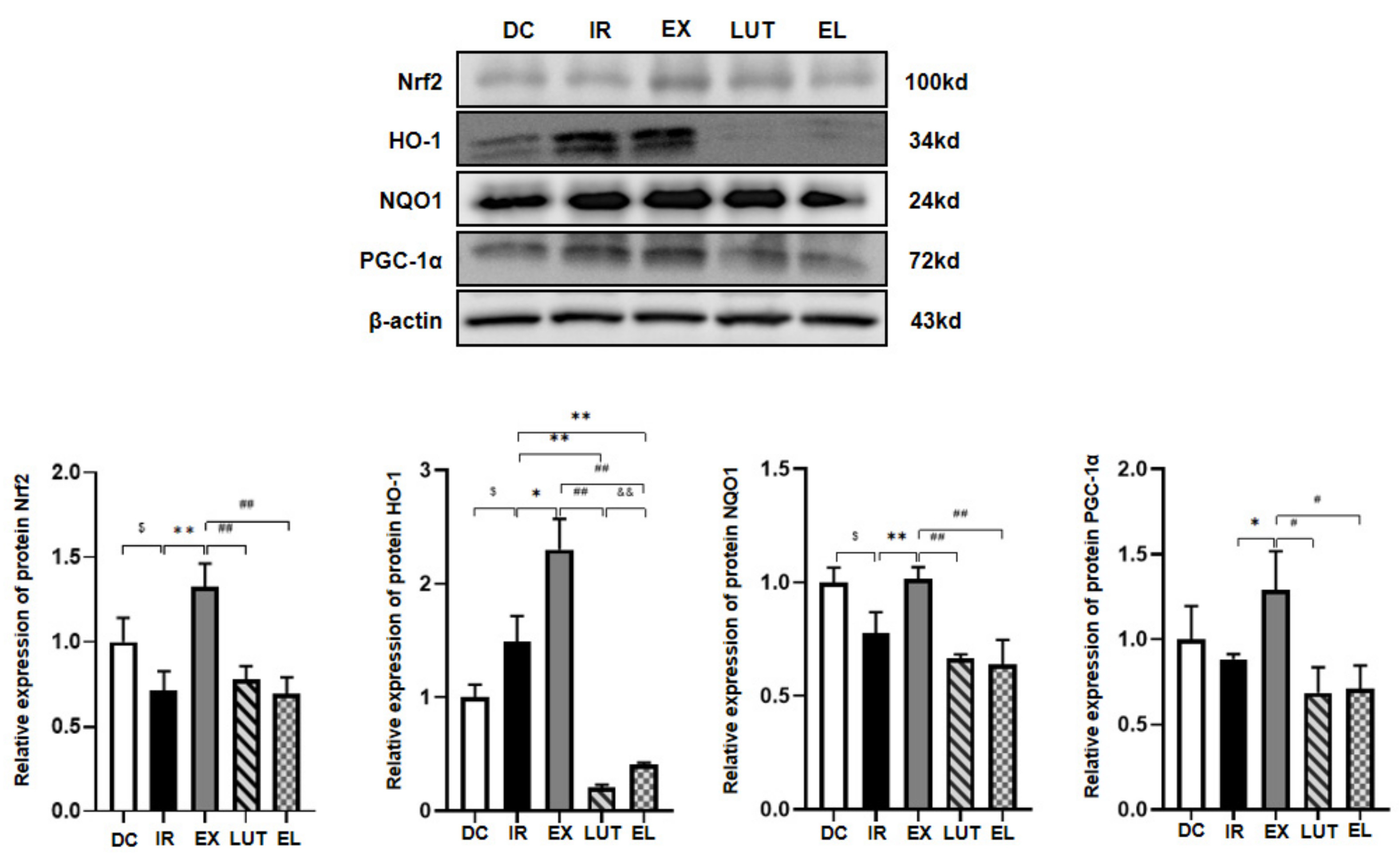
2.4. Aerobic Exercise Modulates the Expression of Nrf2 and Its Downstream Genes
2.5. Aerobic Exercise Regulates the Expression of NLRP3 and Related Inflammatory Factors
2.6. The Association Between Nrf2 Activity and the Inflammatory Response
3. Discussion
4. Materials and Methods
4.1. General Protocol
4.2. Experimental Animals
4.3. Establishment of IR Model Mice
4.4. Intervention Plan
4.5. Organizing the Collection and Processing of Materials
4.5.1. Blood Lipid and Blood Sugar Tests, Glucose Tolerance Test, and Calculation of Insulin Resistance Index
4.5.2. Hematoxylin and Eosin (H&E) Staining
4.5.3. Immunofluorescence Staining Analysis
4.5.4. Real-Time Fluorescence Quantitative PCR
4.5.5. Western Blotting Analysis
4.6. Main Observation Indicators
- The changes in related indices such as blood glucose, blood lipids, and insulin resistance in mice from each group after 6 weeks of exercise and luteolin intraperitoneal injection intervention.
- Morphological changes in the skeletal muscle tissue of mice from each group.
- Protein and mRNA expression of oxidative stress markers such as Nrf2, HO-1, and NQO1 in the skeletal muscle of mice from each group.
- Protein expression levels of inflammatory factors such as NLRP3, IL-1β, and IL-18 in the skeletal muscle of mice from each group.
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| The following abbreviations are used in this manuscript | . |
| IR | Insulin Resistance |
| HOMA-IR | Homeostasis model assessment-Insulin Resistance |
| FINS | Fasting Insulin |
| IRS | Insulin receptor substrate |
| PI3K | Phosphatidylinositol 3-Kinase |
| Akt | Protein Kinase B |
| Nrf2 | Nuclear factor E2-related factor 2 |
| ROS | Reactive Oxygen Species |
| HO-1 | Heme Oxygenase-1 |
| NQO1 | NAD(P)H: Quinone Oxidoreductase 1 |
| PGC-1α | Peroxisome proliferator-activated receptor gamma coactivator 1-alpha |
| HFD | High fat diet |
| TG | Triglyceride |
| TC | Total cholesterol |
| LDL-C | Low-Density Lipoprotein Cholesterol |
| HDL-C | High-Density Lipoprotein Cholesterol |
| NLRP3 | NOD like receptor protein 3 |
| IL-1β | Interleukin-1β |
| IL-18 | Interleukin-18 |
| TXNIP | Thioredoxin Interacting Protein |
| AMPK | AMP-activated protein kinase |
| FBG | Fasting Blood Glucose |
| GTT | Glucose tolerance test |
References
- Marušić, M.; Paić, M.; Knobloch, M.; Liberati Pršo, A.M. NAFLD, Insulin Resistance, and Diabetes Mellitus Type 2. Can. J. Gastroenterol. Hepatol. 2021, 2021, 6613827. [Google Scholar] [CrossRef]
- Kosmas, C.E.; Bousvarou, M.D.; Kostara, C.E.; Papakonstantinou, E.J.; Salamou, E.; Guzman, E. Insulin resistance and cardiovascular disease. J. Int. Med. Res. 2023, 51, 3000605231164548. [Google Scholar] [CrossRef]
- Kasai, S.; Kokubu, D.; Mizukami, H.; Itoh, K. Mitochondrial Reactive Oxygen Species, Insulin Resistance, and Nrf2-Mediated Oxidative Stress Response-Toward an Actionable Strategy for Anti-Aging. Biomolecules 2023, 13, 1544. [Google Scholar] [CrossRef]
- Garg, R.; Agarwal, A.; Katekar, R.; Goand, U.K.; Singh, N.; Yadav, S.; Rathaur, S.; Verma, S.; Maity, D.; Vishwakarma, S.; et al. Pancreastatin Inhibitor PSTi8 Ameliorates Insulin Resistance by Decreasing Fat Accumulation and Oxidative Stress in High-Fat Diet-Fed Mice. Amino Acids. 2023, 55, 1587–1600. [Google Scholar] [CrossRef]
- Sasaki, T.; Sugiyama, M.; Kuno, M.; Miyata, T.; Kobayashi, T.; Yasuda, Y.; Onoue, T.; Takagi, H.; Hagiwara, D.; Iwama, S.; et al. Voluntary Exercise Suppresses Inflammation and Improves Insulin Resistance in the Arcuate Nucleus and Ventral Tegmental Area in Mice on a High-Fat Diet. Physiol. Behav. 2024, 287, 114703. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Peng, Z.; Wang, J. Trelagliptin Alleviates Lipopolysaccharide (LPS)-Induced Inflammation and Oxidative Stress in Acute Lung Injury Mice. Inflammation 2021, 44, 1507–1517. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.B.; Wang, S.Y.; Guo, X.Y.; Guo, X.J.; Hao, L.R. Uric Acid Induces Oxidative Stress and Inflammation in Human Proximal Renal Tubular Epithelial Cells via AIF-1/NF-κB Inflammatory Signaling Pathway. Chin. J. Gerontol. 2025, 45, 3213–3217. [Google Scholar]
- Zhang, Y.; Wu, Q.; Liu, J.; Zhang, Z.; Ma, X.; Zhang, Y.; Zhu, J.; Thring, R.W.; Wu, M.; Gao, Y.; et al. Sulforaphane Alleviates High Fat Diet-Induced Insulin Resistance via AMPK/Nrf2/GPx4 axis. Biomed. Pharmacother. 2022, 152, 113273. [Google Scholar] [CrossRef]
- Zhang, C.; Xie, L.; Sun, W. The effect of Lycium polysaccharides on improving insulin resistance in gestational diabetes rats through regulating the Nrf2/HO-1/GPX4 iron death pathway. Chin. Pat. Med. 2024, 46, 626–630. [Google Scholar]
- Pei, X.L.; Liang, Y.L.; Duan, Y.Q.; Zhu, X.D.; Song, B.; Bai, M.; Zhao, Y.H.; Zhao, S.C.; Liu, Z.T.; Su, B.B. Shenqi Luo Wan Activates the Nrf2/HO-1/NQO1 Signaling Pathway to Improve Oxidative Stress Injury in Skeletal Muscle of Type 2 Diabetes Mice. Chin. J. Exp. Pharmacol. Ther. 2024, 30, 131–139. [Google Scholar]
- Böttcher, N.; Suhr, F.; Pufe, T.; Wruck, C.J.; Fragoulis, A. Luteolin Induces Nrf2 Activity in C2C12 Cells: Implications for Muscle Health. Int. J. Mol. Sci. 2025, 26, 4092. [Google Scholar] [CrossRef]
- Tang, X.; Wang, H.; Fan, L.; Wu, X.; Xin, A.; Ren, H.; Wang, X.J. Luteolin inhibits Nrf2 leading to negative regulation of the Nrf2/ARE pathway and sensitization of human lung carcinoma A549 cells to therapeutic drugs. Free Radic. Biol. Med. 2011, 50, 1599–1609. [Google Scholar] [CrossRef]
- Yang, H.; Liu, B.F.; Xie, F.J.; Yang, W.L.; Cao, N. Luteolin induces mitochondrial apoptosis in HT29 cells by inhibiting the Nrf2/ARE signaling pathway. Exp. Ther. Med. 2020, 19, 2179–2187. [Google Scholar] [CrossRef] [PubMed]
- Tsai, K.J.; Tsai, H.Y.; Tsai, C.C.; Chen, T.Y.; Hsieh, T.H.; Chen, C.L.; Mbuyisa, L.; Huang, Y.B.; Lin, M.W. Luteolin Inhibits Breast Cancer Stemness and Enhances Chemosensitivity through the Nrf2-Mediated Pathway. Molecules 2021, 26, 6452. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Dun, Y.; You, B.; Qiu, L.; Ripley-Gonzalez, J.W.; Cheng, J.; Fu, S.; Li, C.; Liu, S. Trimetazidine and exercise offer analogous improvements to the skeletal muscle insulin resistance of mice through Nrf2 signaling. BMJ Open Diabetes Res. Care 2022, 10, e002699. [Google Scholar] [CrossRef]
- Li, N.; Shi, H.; Guo, Q.; Gan, Y.; Zhang, Y.; Jia, J.; Zhang, L.; Zhou, Y. Aerobic Exercise Prevents Chronic Inflammation and Insulin Resistance in Skeletal Muscle of High-Fat Diet Mice. Nutrients 2022, 14, 3730. [Google Scholar] [CrossRef]
- Zhao, J.F.; Yan, P.R.; Wang, Y.X.; Jia, Y.X.; Zeng, Y. Study on the Effects and Mechanisms of Aerobic Exercise on lnc-ORA in Regulating Fatogenesis and Insulin Resistance in Mice. Chin. J. Sports Med. 2025, 44, 128–138. [Google Scholar]
- Liu, Y.; Yang, W.; Wang, H. The role of the iron homeostasis pathway mediated by Nrf2/FPN1 in preventing hepatic insulin resistance in mice through aerobic exercise. Chin. J. Sports Med. 2023, 42, 294–302. [Google Scholar]
- Che, K.; Lu, M.; Qiu, J. Aerobic Exercise for Preventing Obesity-Associated Insulin Resistance: From the Perspective of Targeting Inflammation. Chin. J. Prev. Control Chronic Dis. 2024, 32, 790–795. [Google Scholar]
- Ping, Y.; Zhang, P.W.; Yuan, X.M.; Xiang, M.Q.; Yang, M.F.; Lin, X.X.; Dong, S.R.; Liu, Y.T.; Zhang, Y. Study on the Effect of Exercise on Modulating the Nrf2/HO-1 Pathway to Improve Oxidative Stress in Hepatocytes Induced by HFFC Diet. Chin. J. Exp. Anim. Sci. 2024, 32, 566–575. [Google Scholar]
- Chen, X.Y.; Li, J.H.; Zhao, L.H.; Zhao, J.L.; Yan, S.S.; Lin, Z.W.; Gao, L.S. The mechanism of aerobic exercise in improving cognitive function of elderly rats with sleep disorders through the Keap1-Nrf2-HO-1 pathway. Med. Res. Trauma Treat. 2024, 37, 125–129. [Google Scholar]
- Li, J.; Wang, S.B.; Xu, G.Q.; Ji, L.L.; Liu, Y.P. Effects and Mechanisms of Aerobic Exercise and Metformin on Vascular Inflammation in Type 2 Diabetic Rats. J. Shanghai Univ. Sport 2019, 43, 69–76. [Google Scholar]
- Hua, K.X.; Pang, W.Q.; Hou, X.R.; Mu, S.M.; Kou, X.J.; Li, C.Y.; Qian, S.W. Effects of BRD4 inhibitor JQ1 and/or endurance exercise intervention on glucose and lipid metabolism disorders in insulin-resistant mice. J. Huazhong Univ. Sci. Technol. Med. Sci. 2024, 53, 751–758. [Google Scholar]
- Li, Z.; Yu, D.P.; Luo, W.; Tao, T.; Chen, H.; Wang, N. Impacts of FABP4 silencing on endoplasmic reticulum stress and insulin resistance in gestational diabetes mellitus rats by regulating PERK/eIF2α/ATF4/CHOP signaling pathway. Chin. J. Immunol. 2024, 40, 705–713. [Google Scholar]
- Wali, J.A.; Jarzebska, N.; Raubenheimer, D.; Simpson, S.J.; Rodionov, R.N.; O’Sullivan, J.F. Cardio-Metabolic Effects of High-Fat Diets and Their Underlying Mechanisms-A Narrative Review. Nutrients 2020, 12, 1505. [Google Scholar] [CrossRef]
- Zhao, S.C.; Liang, Y.L.; Zhu, X.D.; Bai, M.; Zhang, L.L.; An, D.; Pei, X.L. Dahuang Tangluo Pills Regulate RXRA/TNF-α/GLUT4 Pathway to Improve Skeletal Muscle Insulin Resistance in Type 2 Diabetes. Chin. J. Pharmacol. Clin. Pract. 2024, 40, 55–63. [Google Scholar]
- Rasool, S.; Geetha, T.; Broderick, T.L.; Babu, J.R. High Fat with High Sucrose Diet Leads to Obesity and Induces Myodegeneration. Front. Physiol. 2018, 9, 1054. [Google Scholar] [CrossRef]
- Lin, J.; Zhang, X.; Sun, Y.; Xu, H.; Li, N.; Wang, Y.; Tian, X.; Zhao, C.; Wang, B.; Zhu, B.; et al. Exercise Ameliorates Muscular Excessive Mitochondrial Fission, Insulin Resistance and Inflammation in Diabetic Rats via Irisin/AMPK Activation. Sci. Rep. 2024, 14, 19174, Corrected in Sci. Rep. 2024, 14, 10658. [Google Scholar] [CrossRef] [PubMed]
- Yi, M.; Cruz Cisneros, L.; Cho, E.J.; Alexander, M.; Kimelman, F.A.; Swentek, L.; Ferrey, A.; Tantisattamo, E.; Ichii, H. Nrf2 Pathway and Oxidative Stress as a Common Target for Treatment of Diabetes and Its Comorbidities. Int. J. Mol. Sci. 2024, 25, 821. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Liu, Y.; Cao, J.; Wu, C.; Tang, L.; Bian, W.; Chen, Y.; Yu, L.; Wu, Y.; Li, S.; et al. Targeting epigenetic and post-translational modifications of NRF2: Key regulatory factors in disease treatment. Cell Death Discov. 2025, 11, 189. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Chen, C.; Zhuang, W.; Wang, W.; Liu, W.; Zhao, H.; Lv, J.; Xie, D.; Wang, Q.; He, F.; et al. Silencing TXNIP ameliorates high uric acid-induced insulin resistance via the IRS2/AKT and Nrf2/HO-1 pathways in macrophages. Free Radic. Biol. Med. 2022, 178, 42–53. [Google Scholar] [CrossRef]
- Ross, D.; Siegel, D. The diverse functionality of NQO1 and its roles in redox control. Redox Biol. 2021, 41, 101950. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Ao, W.; Huang, T.; Liu, H. Mechanism exploration of yam saponin activating PERK-Nrf2-HO-1 pathway to improve insulin resistance in diabetic mice. Prac. Pharm. Clin. Remed. 2022, 25, 966–971. [Google Scholar]
- Ko, Y.; Hong, M.; Lee, S.; Kumar, M.; Ibrahim, L.; Nutsch, K.; Stanton, C.; Sondermann, P.; Sandoval, B.; Bulos, M.L.; et al. S-lactoyl modification of KEAP1 by a reactive glycolytic metabolite activates NRF2 signaling. Proc. Natl. Acad. Sci. USA 2023, 120, e2300763120. [Google Scholar] [CrossRef]
- Bellezza, I.; Giambanco, I.; Minelli, A.; Donato, R. Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim. Biophys. Acta Mol. Cell Res. 2018, 1865, 721–733. [Google Scholar] [CrossRef]
- Park, S.Y.; Choi, M.H.; Li, M.; Li, K.; Park, G.; Choi, Y.W. AMPK/Nrf2 signaling is involved in the anti-neuroinflammatory action of Petatewalide B from Petasites japonicus against lipopolysaccharides in microglia. Immunopharmacol. Immunotoxicol. 2018, 40, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Chen, Z.; Sun, C.; Yang, D.; Zhou, Z.; Peng, X.; Zheng, L.; Tang, C. Exercise Intervention Mitigates Pathological Liver Changes in NAFLD Zebrafish by Activating SIRT1/AMPK/NRF2 Signaling. Int. J. Mol. Sci. 2021, 22, 10940. [Google Scholar] [CrossRef]
- Mo, Z.W.; Wang, F.; Wei, W.P.; Chen, C.R.; Chen, D.X. Effects of exenatide on renal injury and Nrf2/HO-1 pathway in insulin-resistant rats. Chin. J. Clin. Pharmacol. 2019, 35, 1474–1478. [Google Scholar]
- Jais, A.; Einwallner, E.; Sharif, O.; Gossens, K.; Lu, T.T.; Soyal, S.M.; Medgyesi, D.; Neureiter, D.; Paier-Pourani, J.; Dalgaard, K.; et al. Heme oxygenase-1 drives metaflammation and insulin resistance in mouse and man. Cell 2014, 158, 25–40. [Google Scholar] [CrossRef]
- Medina, M.V.; Sapochnik, D.; Garcia Sola, M.; Coso, O. Regulation of the Expression of Heme Oxygenase-1: Signal Transduction, Gene Promoter Activation, and Beyond. Antioxid. Redox Signal. 2020, 32, 1033–1044. [Google Scholar] [CrossRef]
- Yeligar, S.M.; Machida, K.; Kalra, V.K. Ethanol-induced HO-1 and NQO1 are differentially regulated by HIF-1alpha and Nrf2 to attenuate inflammatory cytokine expression. J. Biol. Chem. 2010, 285, 35359–35373. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, G.; Ding, Q.; Zheng, F.; Shi, X.; Lin, Z.; Liang, Y. The ROS/TXNIP/NLRP3 pathway mediates LPS-induced microglial inflammatory response. Cytokine 2024, 181, 156677. [Google Scholar] [CrossRef] [PubMed]
- Cao, Q.Y.; Wang, Y.; Yang, F.; Chen, J.; Zhou, Y. Effects of Relinqing granules on TXNIP/NLRP3 inflammasome pathway and immune status in chronic urinary tract infection model rats. Chin. J. Immunol. 2023, 39, 1269–1273. [Google Scholar]
- Xu, W.; Huang, Y.; Zhou, R. NLRP3 inflammasome in neuroinflammation and central nervous system diseases. Cell. Mol. Immunol. 2025, 22, 341–355. [Google Scholar] [CrossRef]
- Vandanmagsar, B.; Youm, Y.H.; Ravussin, A.; Galgani, J.E.; Stadler, K.; Mynatt, R.L.; Ravussin, E.; Stephens, J.M.; Dixit, V.D. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat. Med. 2011, 17, 179–188. [Google Scholar] [CrossRef]
- Sun, W.P.; Zhao, D.X.; Wang, P.H.; Wang, S.L.; Li, J.; Kang, Y. Mechanism of TXNIP/NLRP3 signaling pathway in chronic inflammation of ovaries in PCOS-IR rats. Chin. J. Immunol. 2022, 38, 1300–1305. [Google Scholar]
- Bian, X.P.; Ji, R.F.; Liu, B.B.; Hu, J.Y.; Li, M.M.; Xue, X.L.; Lou, S.J. Aerobic exercise reduces the expression of pyroptosis-related proteins and inflammatory factors in hippocampal cells of mice with insulin resistance. Acta Physiol. Sin. 2020, 72, 455–462. [Google Scholar]
- Ji, R.F.; Bian, X.P.; Liu, B.B.; Hu, J.Y.; Xue, X.L.; Lou, S.J. Effects of resistance exercise on pyroptosis-related proteins in the hippocampus of insulin-resistant mice. Chin. J. Appl. Physiol. 2020, 36, 456–461. [Google Scholar]
- Li, J.; Feng, L.; Liu, Y. Effects of aerobic exercise on vascular inflammation and Nrf2/ARE signal pathway in type 2 diabetic rats. Immunol. J. 2019, 35, 179–184. [Google Scholar]
- Lei, B.K.; Li, S.C.; Gao, D.R.; Ke, Z.F.; Wang, R.Y.; Su, Q.S. The role of Nrf2/HO-1 pathway in aerobic exercise intervention on oxidative stress of skeletal muscle in type 2 diabetic rats. Chin. J. Rehabil. Med. 2022, 37, 289–295. [Google Scholar]
- Merry, T.L.; MacRae, C.; Pham, T.; Hedges, C.P.; Ristow, M. Deficiency in ROS-sensing nuclear factor erythroid 2-like 2 causes altered glucose and lipid homeostasis following exercise training. Am. J. Physiol. Cell Physiol. 2020, 318, C337–C345. [Google Scholar] [CrossRef]
- Li, D.; Chen, R.; Chen, J.Q.; Zheng, H.; Zhou, B.C.; Mao, H.F.; Chen, Y.L.; Qu, H.L.; Chen, W.; Peng, Q.; et al. Effects of aerobic exercise and black cumin flavonoids on inflammatory factors in mice with alcoholic liver injury model. Chin. J. Tissue Eng. Res. 2018, 22, 5197–5202. [Google Scholar]









| Target Genes | Genetic Sequences | |
|---|---|---|
| Nrf2 | Forward primer 5′-3′ | AAGACTGCGTTCCTGCTCAAC |
| Reverse primer 5′-3′ | AAAGCCCTACAGCAACTGTCG | |
| HO-1 | Forward primer 5′-3′ | AAGCCGAGAATGCTGAGTTCA |
| Reverse primer 5′-3′ | GCCGTGTAGATATGGTACAAGGA | |
| NQO1 | Forward primer 5′-3′ | GCCGAACACAAGAAGCTGGAAG |
| Reverse primer 5′-3′ | GGCAAATCCTGCTACGAGCACT | |
| PGC-1α | Forward primer 5′-3′ | TATGGAGTGACATAGAGTGTGCT |
| Reverse primer 5′-3′ | CCACTTCAATCCACCCAGAAAG | |
| β-actin | Forward primer 5′-3′ | GCTCTGGCTCCTAGCACCAT |
| Reverse primer 5′-3′ | GCCACCGATCCACACAGAGT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liu, X.; Ding, Y.; Chen, T.; Wu, Z.; Hu, S.; Wang, X. The Role and Mechanism of Nrf2 in Ameliorating Oxidative Stress and Inflammation in IR Mice by Aerobic Exercise. Int. J. Mol. Sci. 2026, 27, 4310. https://doi.org/10.3390/ijms27104310
Liu X, Ding Y, Chen T, Wu Z, Hu S, Wang X. The Role and Mechanism of Nrf2 in Ameliorating Oxidative Stress and Inflammation in IR Mice by Aerobic Exercise. International Journal of Molecular Sciences. 2026; 27(10):4310. https://doi.org/10.3390/ijms27104310
Chicago/Turabian StyleLiu, Xuan, Yuqing Ding, Tao Chen, Zhengkang Wu, Shujuan Hu, and Xianwang Wang. 2026. "The Role and Mechanism of Nrf2 in Ameliorating Oxidative Stress and Inflammation in IR Mice by Aerobic Exercise" International Journal of Molecular Sciences 27, no. 10: 4310. https://doi.org/10.3390/ijms27104310
APA StyleLiu, X., Ding, Y., Chen, T., Wu, Z., Hu, S., & Wang, X. (2026). The Role and Mechanism of Nrf2 in Ameliorating Oxidative Stress and Inflammation in IR Mice by Aerobic Exercise. International Journal of Molecular Sciences, 27(10), 4310. https://doi.org/10.3390/ijms27104310





