C14DM Ablation Leads to Reduced Tolerance to Plasma Membrane Stress and Increased Drug Sensitivity in Leishmania major
Abstract
1. Introduction
2. Results
2.1. C14dm− Mutants Are Hypersensitive to Triton X-100, Dimethyl Sulfoxide (DMSO), and Osmotic Changes
2.2. C14dm− Mutants Show Hypersensitivity to Pseudomonas aeruginosa Spent Medium and Pyocyanin
2.3. C14dm− Mutants Show Autophagy Defects
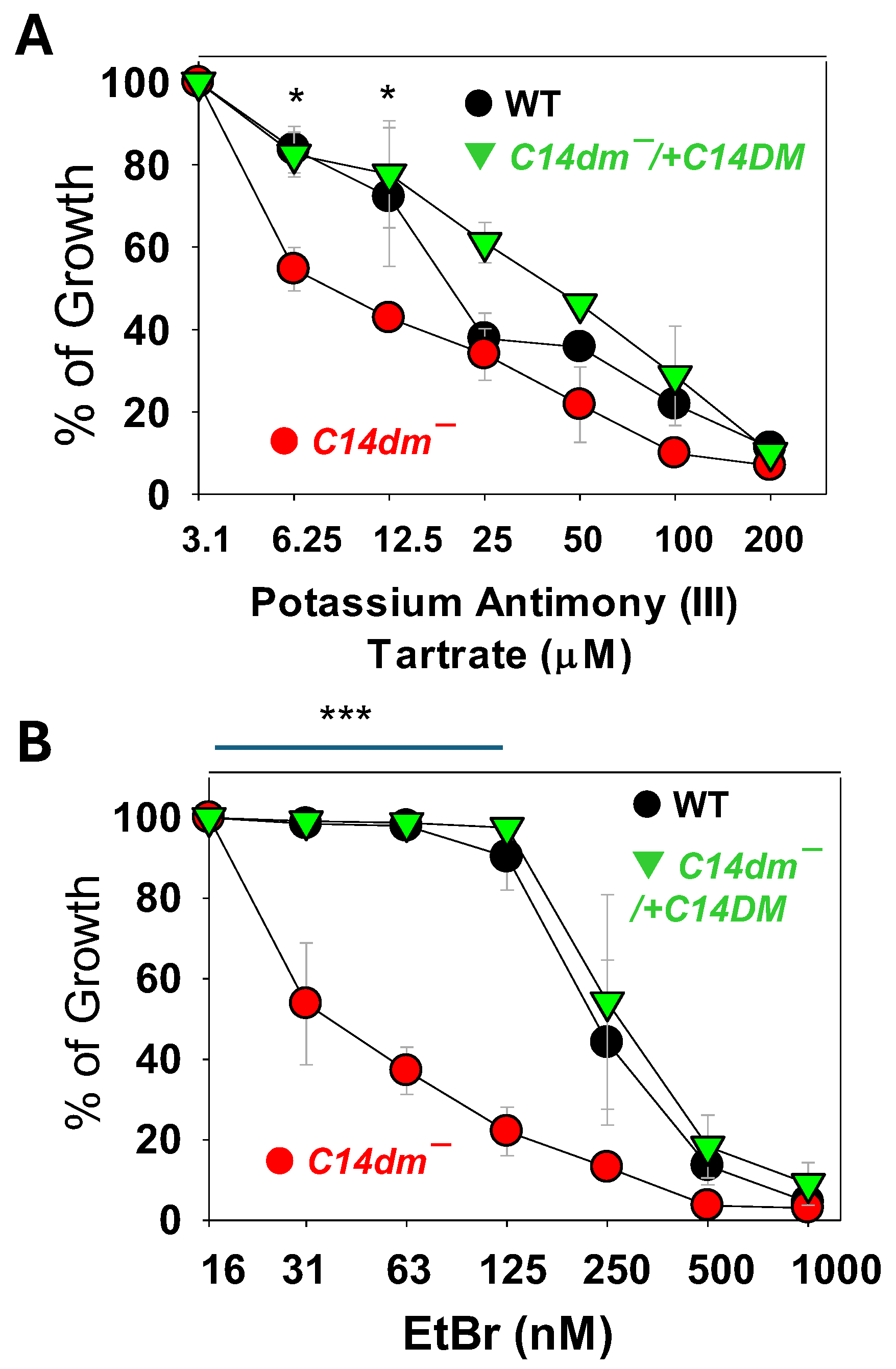
2.4. C14dm− Parasites Show Increased Sensitivity to Antimony and Ethidium Bromide (EtBr)
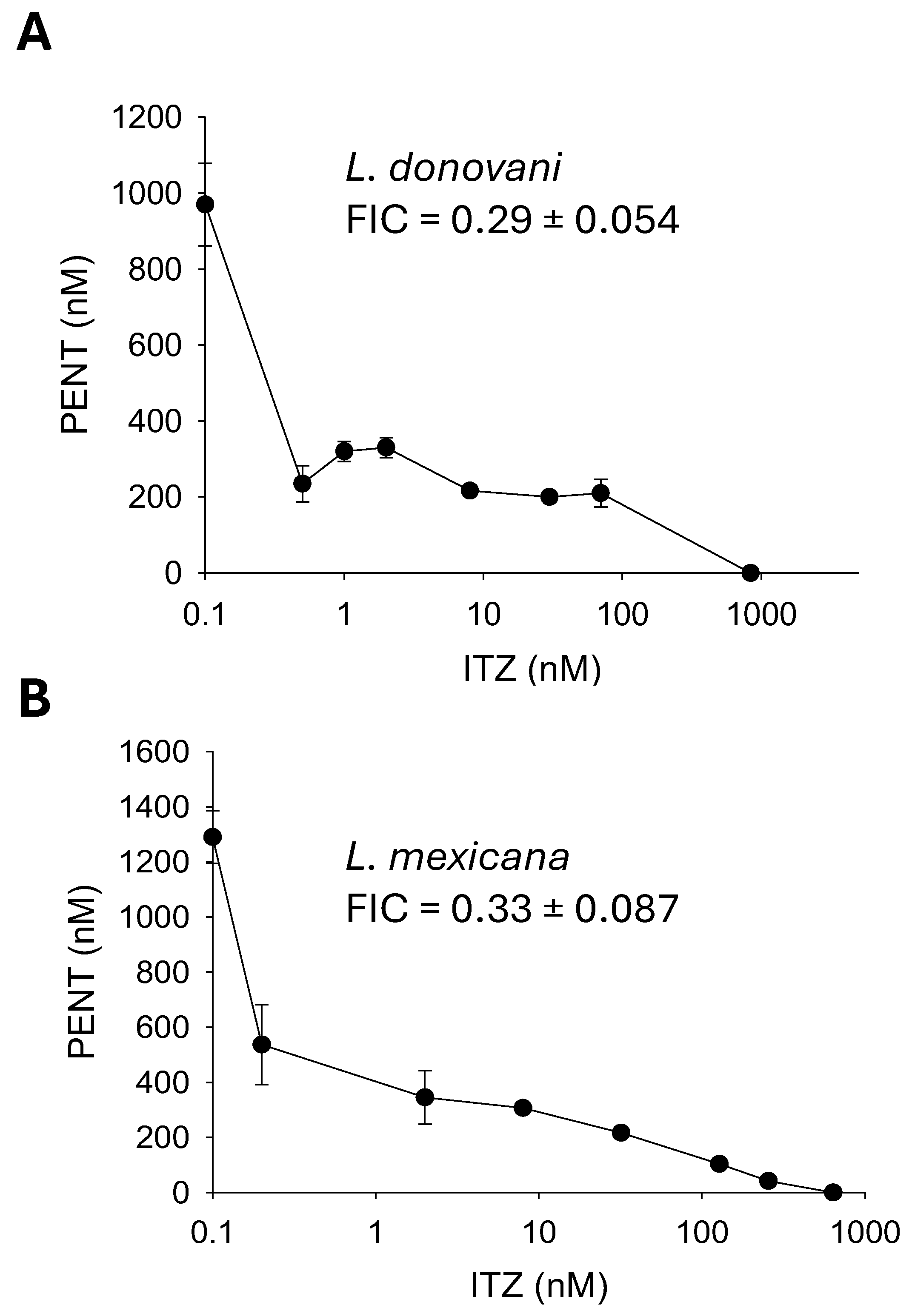
2.5. Synergistic Inhibition of Leishmania mexicana and Leishmania donovani with Pentamidine (PENT) and Itraconazole (ITZ)
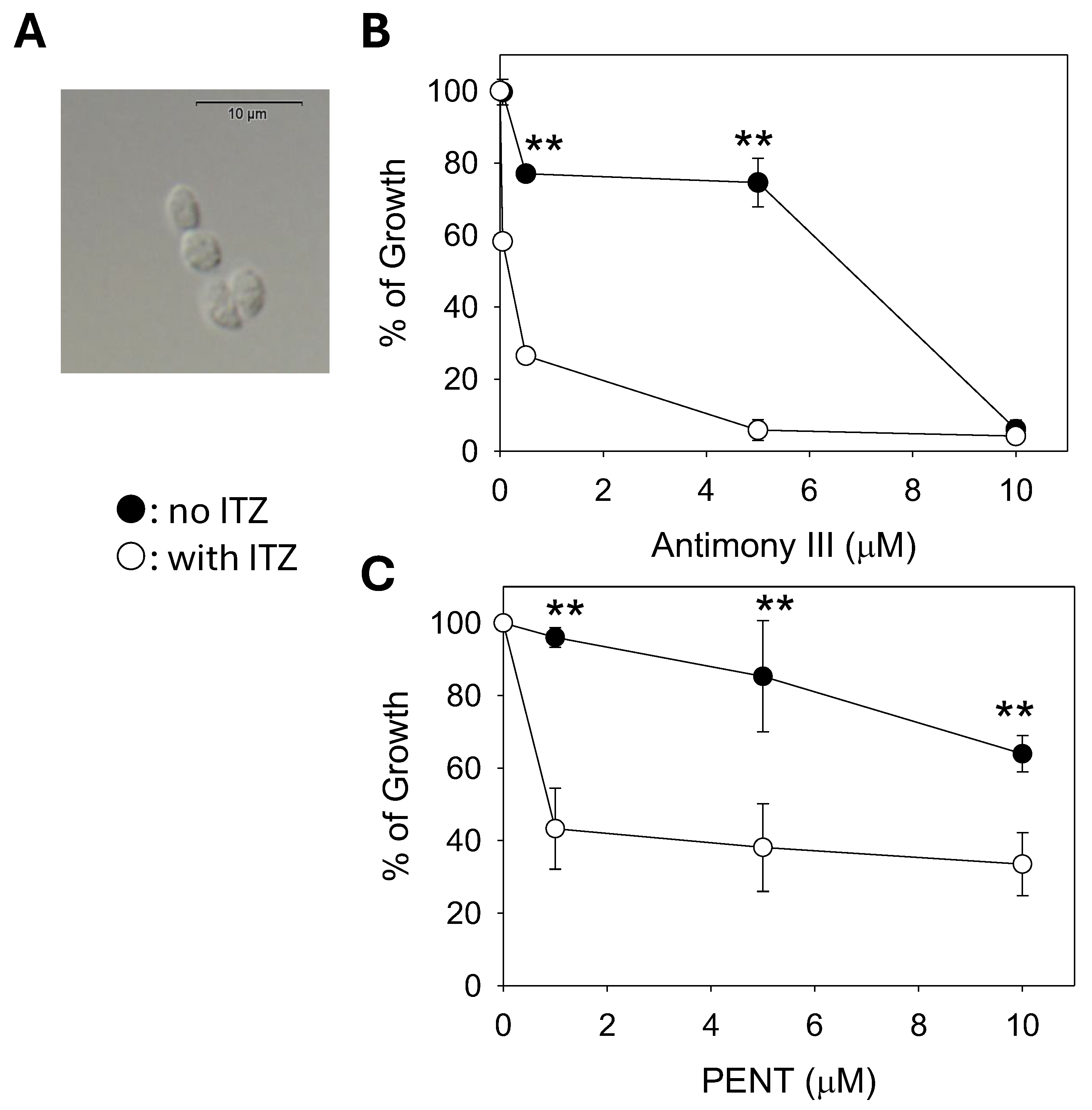
2.6. C14DM Inhibition Enhances Drug Sensitivity in L. mexicana Axenic Amastigotes
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Leishmania Culture
4.3. To Determine the Effects of Membrane-Perturbing Agents, P. aeruginosa PA14-Conditioned Medium, Pyocyanin, and Chemical Inhibitors
4.4. Synergy Calculations
4.5. Response to Osmotic Stress
4.6. Autophagy Assays and Microscopy
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pace, D. Leishmaniasis. J. Infect. 2014, 69 (Suppl. 1), S10–S18. [Google Scholar] [CrossRef]
- Uliana, S.R.B.; Trinconi, C.T.; Coelho, A.C. Chemotherapy of leishmaniasis: Present challenges. Parasitology 2018, 145, 464–480. [Google Scholar] [CrossRef]
- de Souza, W.; Rodrigues, J.C. Sterol Biosynthesis Pathway as Target for Anti-trypanosomatid Drugs. Interdiscip. Perspect. Infect. Dis. 2009, 2009, 642502. [Google Scholar] [CrossRef]
- de Macedo-Silva, S.T.; de Souza, W.; Rodrigues, J.C. Sterol Biosynthesis Pathway as an Alternative for the Anti-Protozoan Parasite Chemotherapy. Curr. Med. Chem. 2015, 22, 2186–2198. [Google Scholar] [CrossRef] [PubMed]
- Tanwar, S.; Kalra, S.; Bari, V.K. Insights into the role of sterol metabolism in antifungal drug resistance: A mini-review. Front. Microbiol. 2024, 15, 1409085. [Google Scholar] [CrossRef]
- Xu, W.; Hsu, F.F.; Baykal, E.; Huang, J.; Zhang, K. Sterol Biosynthesis Is Required for Heat Resistance but Not Extracellular Survival in Leishmania. PLoS Pathog. 2014, 10, e1004427. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Moitra, S.; Xu, W.; Hernandez, V.; Zhang, K. Sterol 14-alpha-demethylase is vital for mitochondrial functions and stress tolerance in Leishmania major. PLoS Pathog. 2020, 16, e1008810. [Google Scholar] [CrossRef] [PubMed]
- Karamysheva, Z.N.; Moitra, S.; Perez, A.; Mukherjee, S.; Tikhonova, E.B.; Karamyshev, A.L.; Zhang, K. Unexpected Role of Sterol Synthesis in RNA Stability and Translation in Leishmania. Biomedicines 2021, 9, 696. [Google Scholar] [CrossRef]
- McCall, L.I.; El Aroussi, A.; Choi, J.Y.; Vieira, D.F.; De Muylder, G.; Johnston, J.B.; Chen, S.; Kellar, D.; Siqueira-Neto, J.L.; Roush, W.R.; et al. Targeting Ergosterol biosynthesis in Leishmania donovani: Essentiality of sterol 14 alpha-demethylase. PLoS Neglected Trop. Dis. 2015, 9, e0003588. [Google Scholar] [CrossRef]
- Tulloch, L.B.; Tinti, M.; Wall, R.J.; Weidt, S.K.; Corpas-Lopez, V.; Dey, G.; Smith, T.K.; Fairlamb, A.H.; Barrett, M.P.; Wyllie, S. Sterol 14-alpha demethylase (CYP51) activity in Leishmania donovani is likely dependent upon cytochrome P450 reductase 1. PLoS Pathog. 2024, 20, e1012382. [Google Scholar] [CrossRef]
- Francesconi, V.A.; Francesconi, F.; Ramasawmy, R.; Romero, G.A.S.; Alecrim, M. Failure of fluconazole in treating cutaneous leishmaniasis caused by Leishmania guyanensis in the Brazilian Amazon: An open, nonrandomized phase 2 trial. PLoS Neglected Trop. Dis. 2018, 12, e0006225. [Google Scholar] [CrossRef]
- Beach, D.H.; Goad, L.J.; Holz, G.G., Jr. Effects of antimycotic azoles on growth and sterol biosynthesis of Leishmania promastigotes. Mol. Biochem. Parasitol. 1988, 31, 149–162. [Google Scholar] [CrossRef]
- Yamamoto, E.S.; Jesus, J.A.; Bezerra-Souza, A.; Laurenti, M.D.; Ribeiro, S.P.; Passero, L.F.D. Activity of Fenticonazole, Tioconazole and Nystatin on New World Leishmania Species. Curr. Top. Med. Chem. 2018, 18, 2338–2346. [Google Scholar] [CrossRef]
- Fernandez, O.L.; Rosales-Chilama, M.; Quintero, N.; Travi, B.L.; Wetzel, D.M.; Gomez, M.A.; Saravia, N.G. Potency and Preclinical Evidence of Synergy of Oral Azole Drugs and Miltefosine in an Ex Vivo Model of Leishmania (Viannia) panamensis Infection. Antimicrob. Agents Chemother. 2022, 66, e01425-21. [Google Scholar] [CrossRef]
- Joice, A.C.; Yang, S.; Farahat, A.A.; Meeds, H.; Feng, M.; Li, J.; Boykin, D.W.; Wang, M.Z.; Werbovetz, K.A. Antileishmanial Efficacy and Pharmacokinetics of DB766-Azole Combinations. Antimicrob. Agents Chemother. 2018, 62, 10-1128. [Google Scholar] [CrossRef] [PubMed]
- Koley, D.; Bard, A.J. Triton X-100 concentration effects on membrane permeability of a single HeLa cell by scanning electrochemical microscopy (SECM). Proc. Natl. Acad. Sci. USA 2010, 107, 16783–16787. [Google Scholar] [CrossRef]
- Kapler, G.M.; Coburn, C.M.; Beverley, S.M. Stable transfection of the human parasite Leishmania major delineates a 30-kilobase region sufficient for extrachromosomal replication and expression. Mol. Cell. Biol. 1990, 10, 1084–1094. [Google Scholar] [PubMed]
- Dagger, F.; Valdivieso, E.; Marcano, A.K.; Ayesta, C. Regulatory volume decrease in Leishmania mexicana: Effect of anti-microtubule drugs. Mem. Inst. Oswaldo Cruz 2013, 108, 84–90. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tom, A.; Kumar, N.P.; Kumar, A.; Saini, P. Interactions between Leishmania parasite and sandfly: A review. Parasitol. Res. 2023, 123, 6. [Google Scholar] [CrossRef]
- Campolina, T.B.; Villegas, L.E.M.; Monteiro, C.C.; Pimenta, P.F.P.; Secundino, N.F.C. Tripartite interactions: Leishmania, microbiota and Lutzomyia longipalpis. PLoS Neglected Trop. Dis. 2020, 14, e0008666. [Google Scholar] [CrossRef]
- Borbon, T.Y.; Scorza, B.M.; Clay, G.M.; Lima Nobre de Queiroz, F.; Sariol, A.J.; Bowen, J.L.; Chen, Y.; Zhanbolat, B.; Parlet, C.P.; Valadares, D.G.; et al. Coinfection with Leishmania major and Staphylococcus aureus enhances the pathologic responses to both microbes through a pathway involving IL-17A. PLoS Neglected Trop. Dis. 2019, 13, e0007247. [Google Scholar] [CrossRef]
- Al-Alousy, N.W.; Al-Nasiri, F.S. Bacterial infections associated with cutaneous leishmaniasis in Salah Al-Din province, Iraq. Microb. Pathog. 2025, 198, 107144. [Google Scholar] [CrossRef] [PubMed]
- Karimian, F.; Koosha, M.; Choubdar, N.; Oshaghi, M.A. Comparative analysis of the gut microbiota of sand fly vectors of zoonotic visceral leishmaniasis (ZVL) in Iran; host-environment interplay shapes diversity. PLoS Neglected Trop. Dis. 2022, 16, e0010609. [Google Scholar] [CrossRef] [PubMed]
- Ligero-Lopez, J.; Villagrasa-Boli, P.; Bularca, E.; Portillo, A.; Oteo, J.A.; Lopez-Alonso, B.; Anton-Berenguer, V.; Beltran-Rosel, A. Emerging challenges: An imported case of Leishmania mexicana with Pseudomonas aeruginosa superinfection. Diagn. Microbiol. Infect. Dis. 2024, 110, 116431. [Google Scholar] [CrossRef] [PubMed]
- Van Der Vliet, D.; Le Guern, A.S.; Freitag, S.; Gounod, N.; Therby, A.; Darie, H.; Buffet, P.A. Pseudomonas aeruginosa otochondritis complicating localized cutaneous leishmaniasis: Prevention of mutilation by early antibiotic therapy. Am. J. Trop. Med. Hyg. 2006, 75, 270–272. [Google Scholar] [CrossRef]
- Wood, S.J.; Goldufsky, J.W.; Seu, M.Y.; Dorafshar, A.H.; Shafikhani, S.H. Pseudomonas aeruginosa Cytotoxins: Mechanisms of Cytotoxicity and Impact on Inflammatory Responses. Cells 2023, 12, 195. [Google Scholar] [CrossRef]
- Deng, Q.; Zhang, Y.; Barbieri, J.T. Intracellular trafficking of PseudomonasExoS, a type III cytotoxin. Traffic 2007, 8, 1331–1345. [Google Scholar] [CrossRef]
- Abdelaziz, A.A.; Kamer, A.M.A.; Al-Monofy, K.B.; Al-Madboly, L.A. Pseudomonas aeruginosa’s greenish-blue pigment pyocyanin: Its production and biological activities. Microb. Cell Fact. 2023, 22, 110. [Google Scholar] [CrossRef]
- Goncalves, T.; Vasconcelos, U. Colour Me Blue: The History and the Biotechnological Potential of Pyocyanin. Molecules 2021, 26, 927. [Google Scholar] [CrossRef]
- Williams, R.A.; Smith, T.K.; Cull, B.; Mottram, J.C.; Coombs, G.H. ATG5 is essential for ATG8-dependent autophagy and mitochondrial homeostasis in Leishmania major. PLoS Pathog. 2012, 8, e1002695. [Google Scholar] [CrossRef]
- Williams, R.A.; Woods, K.L.; Juliano, L.; Mottram, J.C.; Coombs, G.H. Characterization of unusual families of ATG8-like proteins and ATG12 in the protozoan parasite Leishmania major. Autophagy 2009, 5, 159–172. [Google Scholar] [CrossRef]
- Besteiro, S.; Williams, R.A.; Morrison, L.S.; Coombs, G.H.; Mottram, J.C. Endosome sorting and autophagy are essential for differentiation and virulence of Leishmania major. J. Biol. Chem. 2006, 281, 11384–11396. [Google Scholar] [CrossRef]
- J, B.; M, B.M.; Chanda, K. An Overview on the Therapeutics of Neglected Infectious Diseases-Leishmaniasis and Chagas Diseases. Front. Chem. 2021, 9, 622286. [Google Scholar] [CrossRef]
- Riou, G.; Delain, E. Abnormal circular DNA molecules induced by ethidium bromide in the kinetoplast of Trypanosoma cruzi. Proc. Natl. Acad. Sci. USA 1969, 64, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Roy Chowdhury, A.; Bakshi, R.; Wang, J.; Yildirir, G.; Liu, B.; Pappas-Brown, V.; Tolun, G.; Griffith, J.D.; Shapiro, T.A.; Jensen, R.E.; et al. The killing of African trypanosomes by ethidium bromide. PLoS Pathog. 2010, 6, e1001226. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.; Padmanabhan, P.K.; Sahani, M.H.; Barrett, M.P.; Madhubala, R. Roles for mitochondria in pentamidine susceptibility and resistance in Leishmania donovani. Mol. Biochem. Parasitol. 2006, 145, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Hallander, H.O.; Dornbusch, K.; Gezelius, L.; Jacobson, K.; Karlsson, I. Synergism between aminoglycosides and cephalosporins with antipseudomonal activity: Interaction index and killing curve method. Antimicrob. Agents Chemother. 1982, 22, 743–752. [Google Scholar] [CrossRef]
- Gupta, N.; Goyal, N.; Rastogi, A.K. In vitro cultivation and characterization of axenic amastigotes of Leishmania. Trends Parasitol. 2001, 17, 150–153. [Google Scholar] [CrossRef]
- Tabbabi, A.; Mizushima, D.; Yamamoto, D.S.; Zhioua, E.; Kato, H. Comparative analysis of the microbiota of sand fly vectors of Leishmania major and L. tropica in a mixed focus of cutaneous leishmaniasis in southeast Tunisia; ecotype shapes the bacterial community structure. PLoS Neglected Trop. Dis. 2024, 18, e0012458. [Google Scholar] [CrossRef]
- Fraihi, W.; Fares, W.; Perrin, P.; Dorkeld, F.; Sereno, D.; Barhoumi, W.; Sbissi, I.; Cherni, S.; Chelbi, I.; Durvasula, R.; et al. An integrated overview of the midgut bacterial flora composition of Phlebotomus perniciosus, a vector of zoonotic visceral leishmaniasis in the Western Mediterranean Basin. PLoS Neglected Trop. Dis. 2017, 11, e0005484. [Google Scholar] [CrossRef]
- Cecilio, P.; Rogerio, L.A.; Serafim, T.D.; Tang, K.; Willen, L.; Iniguez, E.; Meneses, C.; Chaves, L.F.; Zhang, Y.; dos Santos Felix, L.; et al. Leishmania sand fly-transmission is disrupted by Delftia tsuruhatensis TC1 bacteria. Nat. Commun. 2025, 16, 3571. [Google Scholar] [CrossRef]
- Sant’Anna, M.R.; Diaz-Albiter, H.; Aguiar-Martins, K.; Al Salem, W.S.; Cavalcante, R.R.; Dillon, V.M.; Bates, P.A.; Genta, F.A.; Dillon, R.J. Colonisation resistance in the sand fly gut: Leishmania protects Lutzomyia longipalpis from bacterial infection. Parasites Vectors 2014, 7, 329. [Google Scholar] [CrossRef]
- Kelly, P.H.; Bahr, S.M.; Serafim, T.D.; Ajami, N.J.; Petrosino, J.F.; Meneses, C.; Kirby, J.R.; Valenzuela, J.G.; Kamhawi, S.; Wilson, M.E. The Gut Microbiome of the Vector Lutzomyia longipalpis Is Essential for Survival of Leishmania infantum. mBio 2017, 8, 10-1128. [Google Scholar] [CrossRef]
- Gallo-Francisco, P.H.; Brocchi, M.; Giorgio, S. Leishmania and its relationships with bacteria. Future Microbiol. 2022, 17, 199–218. [Google Scholar] [CrossRef]
- Giri, S.; Shaha, C. Leishmania donovani parasite requires Atg8 protein for infectivity and survival under stress. Cell Death Dis. 2019, 10, 808. [Google Scholar] [CrossRef] [PubMed]
- Haldar, A.K.; Sen, P.; Roy, S. Use of antimony in the treatment of leishmaniasis: Current status and future directions. Mol. Biol. Int. 2011, 2011, 571242. [Google Scholar] [CrossRef]
- Wyllie, S.; Cunningham, M.L.; Fairlamb, A.H. Dual action of antimonial drugs on thiol redox metabolism in the human pathogen Leishmania donovani. J. Biol. Chem. 2004, 279, 39925–39932. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Yan, R.; Liu, Y.; Yu, M.; He, Z.; Xiao, J.; Li, K.; Liu, G.; Ning, Q.; Li, Y. Progress in antileishmanial drugs: Mechanisms, challenges, and prospects. PLoS Neglected Trop. Dis. 2025, 19, e0012735. [Google Scholar] [CrossRef] [PubMed]
- de Macedo-Silva, S.T.; Visbal, G.; Urbina, J.A.; de Souza, W.; Rodrigues, J.C. Potent In Vitro Antiproliferative Synergism of Combinations of Ergosterol Biosynthesis Inhibitors against Leishmania amazonensis. Antimicrob. Agents Chemother. 2015, 59, 6402–6418. [Google Scholar] [CrossRef]
- Sakyi, P.O.; Amewu, R.K.; Devine, R.; Bienibuor, A.K.; Miller, W.A., 3rd; Kwofie, S.K. Unravelling the myth surrounding sterol biosynthesis as plausible target for drug design against leishmaniasis. J. Parasit. Dis. 2021, 45, 1152–1171. [Google Scholar] [CrossRef]
- Bates, P.A. Axenic culture of Leishmania amastigotes. Parasitol. Today (Personal. Ed.) 1993, 9, 143–146. [Google Scholar] [CrossRef]
- Ha, D.S.; Schwarz, J.K.; Turco, S.J.; Beverley, S.M. Use of the green fluorescent protein as a marker in transfected Leishmania. Mol. Biochem. Parasitol. 1996, 77, 57–64. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moitra, S.; Mukherjee, S.; Hernandez, V.L.; Zhang, K. C14DM Ablation Leads to Reduced Tolerance to Plasma Membrane Stress and Increased Drug Sensitivity in Leishmania major. Int. J. Mol. Sci. 2025, 26, 8473. https://doi.org/10.3390/ijms26178473
Moitra S, Mukherjee S, Hernandez VL, Zhang K. C14DM Ablation Leads to Reduced Tolerance to Plasma Membrane Stress and Increased Drug Sensitivity in Leishmania major. International Journal of Molecular Sciences. 2025; 26(17):8473. https://doi.org/10.3390/ijms26178473
Chicago/Turabian StyleMoitra, Samrat, Sumit Mukherjee, Veronica L. Hernandez, and Kai Zhang. 2025. "C14DM Ablation Leads to Reduced Tolerance to Plasma Membrane Stress and Increased Drug Sensitivity in Leishmania major" International Journal of Molecular Sciences 26, no. 17: 8473. https://doi.org/10.3390/ijms26178473
APA StyleMoitra, S., Mukherjee, S., Hernandez, V. L., & Zhang, K. (2025). C14DM Ablation Leads to Reduced Tolerance to Plasma Membrane Stress and Increased Drug Sensitivity in Leishmania major. International Journal of Molecular Sciences, 26(17), 8473. https://doi.org/10.3390/ijms26178473





