Release and Cytocompatibility Study of New Hybrid Materials Based on Ferulic Acid for Biomedical Use
Abstract
1. Introduction
2. Results
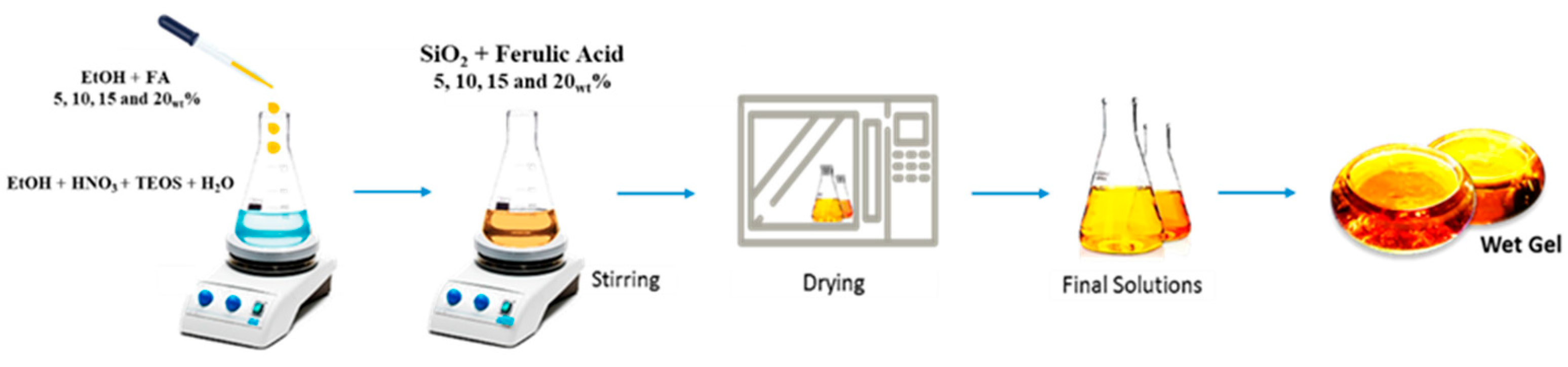
2.1. Sol–Gel Synthesis
2.2. Interactions Between SiO2 and SiO2/FA
2.3. Kinetics Release
2.4. Biocompatibility
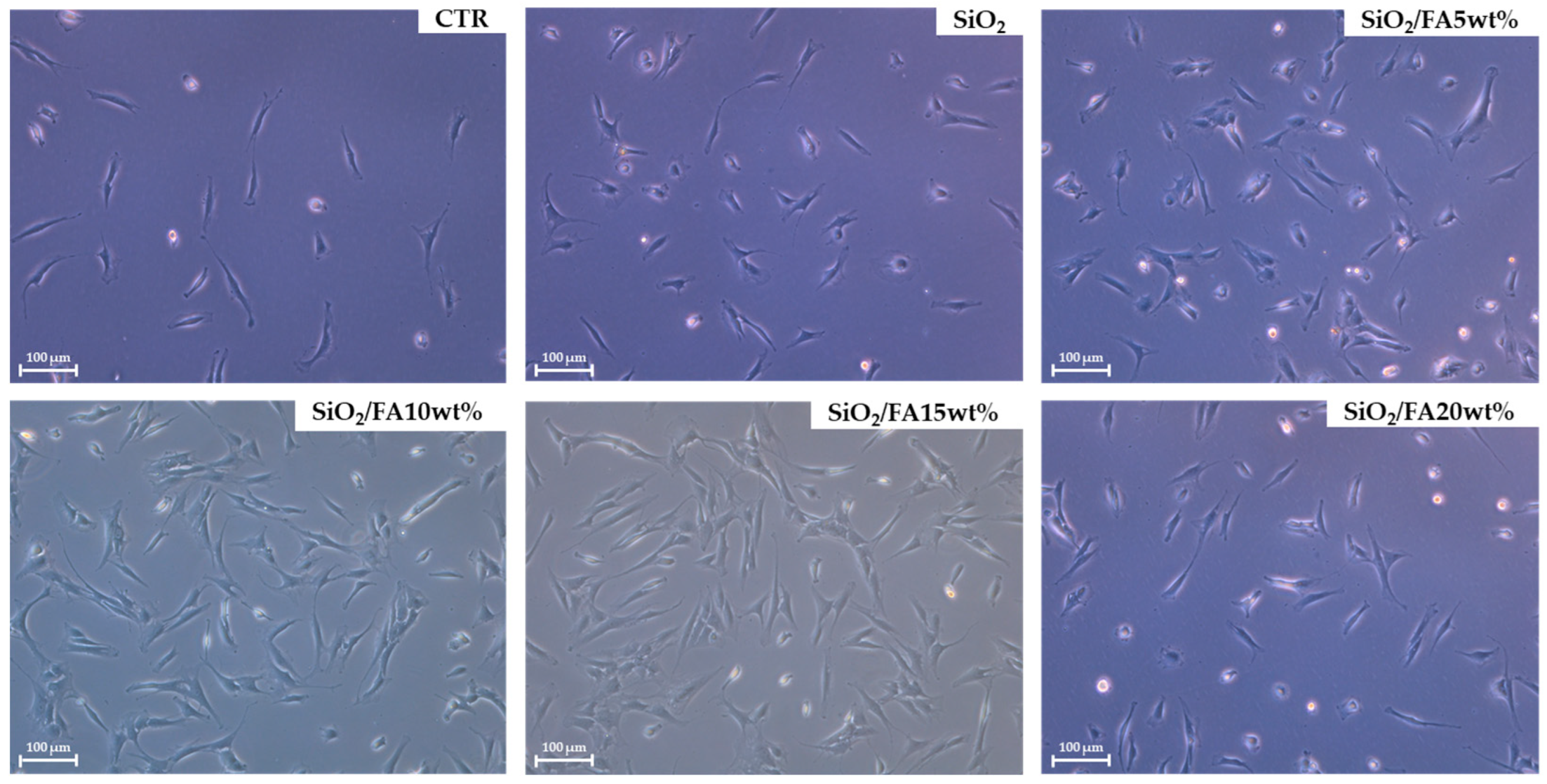
2.4.1. Cytocompatibility
2.4.2. Gene Expression
3. Discussion
4. Materials and Methods
4.1. Synthesis by Sol–Gel Technique
4.2. FTR-ATR Analysis
4.3. Drug Release
4.3.1. PBS Preparation
4.3.2. SBF Preparation
4.3.3. Calibration Curve
4.4. Cytotoxicity Assay
4.5. Real-Time Quantitative PCR
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| FA | Ferulic acid |
| TEOS | Tetraethyl orthosilicate |
| FTIR | Fourier transform infrared spectroscopy |
| ATR | Attenuated total reflectance |
| MTT | (3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide) |
| ACAN | Aggrecan |
| IL-6 | Interleukin 6 |
| COL II | Type II collagen |
References
- Silva, F. Regenerative medicine. J. Transl. Med. 2024, 22, 721. [Google Scholar] [CrossRef]
- Stupp, S.I. Biomaterials for regenerative medicine. MRS Bull. 2025, 30, 546–553. [Google Scholar] [CrossRef]
- Barrino, F. Hybrid organic–inorganic materials prepared by sol–gel and sol–gel-coating method for biomedical use: Study and synthetic review of synthesis and properties. Coatings 2024, 14, 425. [Google Scholar] [CrossRef]
- Ishikawa, K.; Garskaite, E.; Kareiva, A. Sol–gel synthesis of calcium phosphate-based biomaterials—A review of environmentally benign, simple, and effective synthesis routes. J. Sol-Gel Sci. Technol. 2020, 94, 551–572. [Google Scholar] [CrossRef]
- Munir, T.; Mahmood, A.; Peter, N.; Rafaqat, N.; Imran, M.; Ali, H.E. Structural, morphological and optical properties at various concentration of Ag doped SiO2-NPs via sol gel method for antibacterial and anticancer activities. Surf. Interfaces 2023, 38, 102759. [Google Scholar] [CrossRef]
- Hammadi, A.H.; Aysa, N.H.; Gassim, F.A.Z.G. Synthesis Characterization and Adsorption of doxorubicin hydrochloride on SiO2 nanoparticle for drug delivery. Baghdad Sci. J. 2024, 22, 472–485. [Google Scholar] [CrossRef]
- Perumal, S.; Kumar Ramadass, S.; Madhan, B. Sol–gel processed mupirocin silica microspheres loaded collagen scaffold: A synergistic bio-composite for wound healing. Eur. J. Pharm. Sci. 2014, 52, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Marcato, D.C.; Spagnol, C.M.; Salgado, H.R.N.; Isaac, V.L.B.; Corrêa, M.A. New and potential properties, characteristics, and analytical methods of ferulic acid: A review. Braz. J. Pharm. Sci. 2022, 58, e18747. [Google Scholar] [CrossRef]
- Jin, C.; Chen, D.; Zhu, T.; Chen, S.; Du, J.; Zhang, H.; Dong, W. Poly (ferulic acid)-hybrid nanofibers for reducing thrombosis and restraining intimal hyperplasia in vascular tissue engineering. Biomater. Adv. 2023, 146, 213278. [Google Scholar] [CrossRef]
- Druzian, D.M.; Da Silva, M.D.C.; Da Silva, W.L.; Santos, J.H.Z.D. Sol-Gel Technologies for Drug Delivery in Potential Cancer Treatments. In Handbook of Cancer and Immunology; Springer International Publishing: Cham, Switzerland, 2024; pp. 1–22. [Google Scholar]
- Khan, H.M.; Liao, X.; Sheikh, B.A.; Wang, Y.; Su, Z.; Guo, C.; Kong, Q. Smart biomaterials and their potential applications in tissue engineering. J. Mater. Chem. B 2022, 10, 6859–6895. [Google Scholar] [CrossRef]
- Lv, Y.; Li, W.; Liao, W.; Jiang, H.; Liu, Y.; Cao, J.; Lu, W.; Feng, Y. Nano-drug delivery systems based on natural products. Int. J. Nanomed. 2024, 19, 541–569. [Google Scholar] [CrossRef] [PubMed]
- Elshazly, N.; Nasr, F.E.; Hamdy, A.; Saied, S.; Elshazly, M. Advances in clinical applications of bioceramics in the new regenerative medicine era. World J. Clin. Cases 2024, 12, 1863. [Google Scholar] [CrossRef]
- Pacifico, S.; Piccolella, S.; Barrino, F.; Catauro, M. Biomaterials containing the natural antioxidant quercetin: Synthesis and health benefits. In Macromolecular Symposia; Wiley: Hoboken, NJ, USA, 2020; Volume 389, No. 1; p. 1900060. [Google Scholar]
- Abdelhamid, M.A.; Khalifa, H.O.; Ki, M.R.; Pack, S.P. Nanoengineered silica-based biomaterials for regenerative medicine. Int. J. Mol. Sci. 2024, 25, 6125. [Google Scholar] [CrossRef]
- Song, X.; Segura-Egea, J.J.; Díaz-Cuenca, A. Sol–Gel technologies to obtain advanced bioceramics for dental therapeutics. Molecules 2023, 28, 6967. [Google Scholar] [CrossRef]
- Baskaran, K.; Ali, M.; Gingrich, K.; Porter, D.L.; Chong, S.; Riley, B.J.; Peak, W.C.; Naleway, S.E.; Zharov, I.; Carlson, K. Sol-gel derived silica: A review of polymer-tailored properties for energy and environmental applications. Microporous Mesoporous Mater. 2022, 336, 111874. [Google Scholar] [CrossRef]
- Beganskienė, A.; Sirutkaitis, V.; Kurtinaitienė, M.; Juškėnas, R.; Kareiva, A. FTIR, TEM and NMR investigations of Stöber silica nanoparticles. Mater. Sci. (Medžiagotyra) 2024, 10, 287–290. [Google Scholar]
- Ciprioti, S.V.; Naviglio, D.; Gallo, M.; Barrino, F.; Catauro, M. Spectroscopic, Thermal Analysis and Bioactivity Study of New Ferrous Citrate Based Materials Prepared by Sol–Gel Method. In Macromolecular Symposia; Wiley: Hoboken, NJ, USA, 2020; Volume 389, No. 1; p. 1900084. [Google Scholar]
- He, X.; Liu, X.; Nie, B.; Song, D. FTIR and Raman spectroscopy characterization of functional groups in various rank coals. Fuel 2017, 206, 555–563. [Google Scholar] [CrossRef]
- Catauro, M.; Tranquillo, E.; Naviglio, D.; Barrino, F. The influence of polymer on Fe (II) Citrate Release from Hybrid Materials. Macromol. Symp 2020, 389, 1900057. [Google Scholar] [CrossRef]
- Filip, N.; Radu, I.; Veliceasa, B.; Filip, C.; Pertea, M.; Clim, A.; Serban, I.L. Biomaterials in orthopedic devices: Current issues and future perspectives. Coatings 2022, 12, 1544. [Google Scholar] [CrossRef]
- Kuo, S.H.; Shen, C.J.; Shen, C.F.; Cheng, C.M. Role of pH value in clinically relevant diagnosis. Diagnostics 2020, 10, 107. [Google Scholar] [CrossRef]
- Rezaie, J.; Feghhi, M.; Etemadi, T. A review on exosomes application in clinical trials: Perspective, questions, and challenges. Cell Commun. Signal. 2022, 20, 145. [Google Scholar] [CrossRef]
- De Gaetano, F.; Ambrosio, L.; Raucci, M.G.; Marotta, A.; Catauro, M. Sol-gel processing of drug delivery materials and release kinetics. J. Mater. Sci. Mater. Med. 2025, 16, 261–265. [Google Scholar] [CrossRef]
- Kim, K.J.; Hwang, M.J.; Shim, W.G.; Youn, Y.N.; Yoon, S.D. Sustained drug release behavior of captopril-incorporated chitosan/carboxymethyl cellulose biomaterials for antihypertensive therapy. Int. J. Biol. Macromol. 2024, 255, 128087. [Google Scholar] [CrossRef] [PubMed]
- Naseri, E.; Ahmadi, A. A review on wound dressings: Antimicrobial agents, biomaterials, fabrication techniques, and stimuli-responsive drug release. Eur. Polym. J. 2022, 173, 111293. [Google Scholar] [CrossRef]
- Barrino, F.; Vassallo, V.; Cammarota, M.; Lepore, M.; Portaccio, M.; Schiraldi, C.; La Gatta, A. A comprehensive in vitro characterization of non-crosslinked, diverse tissue-derived collagen-based membranes intended for assisting bone regeneration. PLoS ONE 2024, 19, e0298280. [Google Scholar] [CrossRef] [PubMed]
- Vassallo, V.; Stellavato, A.; Russo, R.; Cimini, D.; Valletta, M.; Alfano, A.; Schiraldi, C. Molecular fingerprint of human pathological synoviocytes in response to extractive sulfated and biofermentative unsulfated chondroitins. Int. J. Mol. Sci. 2022, 23, 15865. [Google Scholar] [CrossRef]
- La Gatta, A.; Aschettino, M.; Stellavato, A.; D’Agostino, A.; Vassallo, V.; Bedini, E.; Schiraldi, C. Hyaluronan hydrogels for injection in superficial dermal layers: An in vitro characterization to compare performance and unravel the scientific basis of their indication. Int. J. Mol. Sci. 2021, 22, 6005. [Google Scholar] [CrossRef]
- Catauro, M.; Barrino, F.; Pacifico, S.; Piccolella, S.; Lancellotti, I.; Leonelli, C. Synthesis of WEEE-based geopolymers and their cytotoxicity. Mater. Today Proc. 2021, 34, 121–124. [Google Scholar] [CrossRef]
- Meganathan, I.; Pachaiyappan, M.; Aarthy, M.; Radhakrishnan, J.; Mukherjee, S.; Shanmugam, G.; You, J.; Ayyadurai, N. Recombinant and genetic code expanded collagen-like protein as a tailorable biomaterial. Mater. Horiz. 2022, 9, 2698–2721. [Google Scholar] [CrossRef]
- Martyniak, K.; Lokshina, A.; Cruz, M.A.; Karimzadeh, M.; Kemp, R.; Kean, T.J. Biomaterial composition and stiffness as decisive properties of 3D bioprinted constructs for type II collagen stimulation. Acta Biomater. 2022, 152, 221–234. [Google Scholar] [CrossRef]
- Lin, D.Y.; Huang, W.T.; Lin, Y.C.; Hung, H.H.; Ou, S.C.; Chang, C.W.; Huang, S.T. Prescription system to calculate precise doses of Chinese herbal medicine to avoid toxic effects. Heliyon 2023, 9, e16612. [Google Scholar] [CrossRef]
- Meng, F.; Liao, Z.; Xing, C.; Yuan, Z.; Zhang, R.; Zhu, H.; Li, J. Preparation of SnO2/SiO2 nanocomposites by sol-gel method for enhancing the gas sensing performance to triethylamine. J. Alloys Compd. 2022, 893, 162189. [Google Scholar] [CrossRef]
- Kokubo, T.; Yamaguchi, S. Simulated body fluid and the novel bioactive materials derived from it. J. Biomed. Mater. Res.-A 2019, 107, 968–977. [Google Scholar] [CrossRef] [PubMed]
- Catauro, M.; Barrino, F.; Scolaro, C.; Visco, A. Surface modifications induced in UHMWPE based nanocomposites during the ageing in simulated synovial fluid. In Macromolecular Symposia; Wiley: Hoboken, NJ, USA, 2020; Volume 389, No. 1; p. 1900055. [Google Scholar]
- Calamia, V.; Lourido, L.; Fernández-Puente, P.; Mateos, J.; Rocha, B.; Montell, E.; Blanco, F.J. Secretome analysis of chondroitin sulfate-treated chondrocytes reveals anti-angiogenic, anti-inflammatory and anti-catabolic properties. J. Biomed. Mater. Res. 2012, 14, R202. [Google Scholar] [CrossRef] [PubMed]

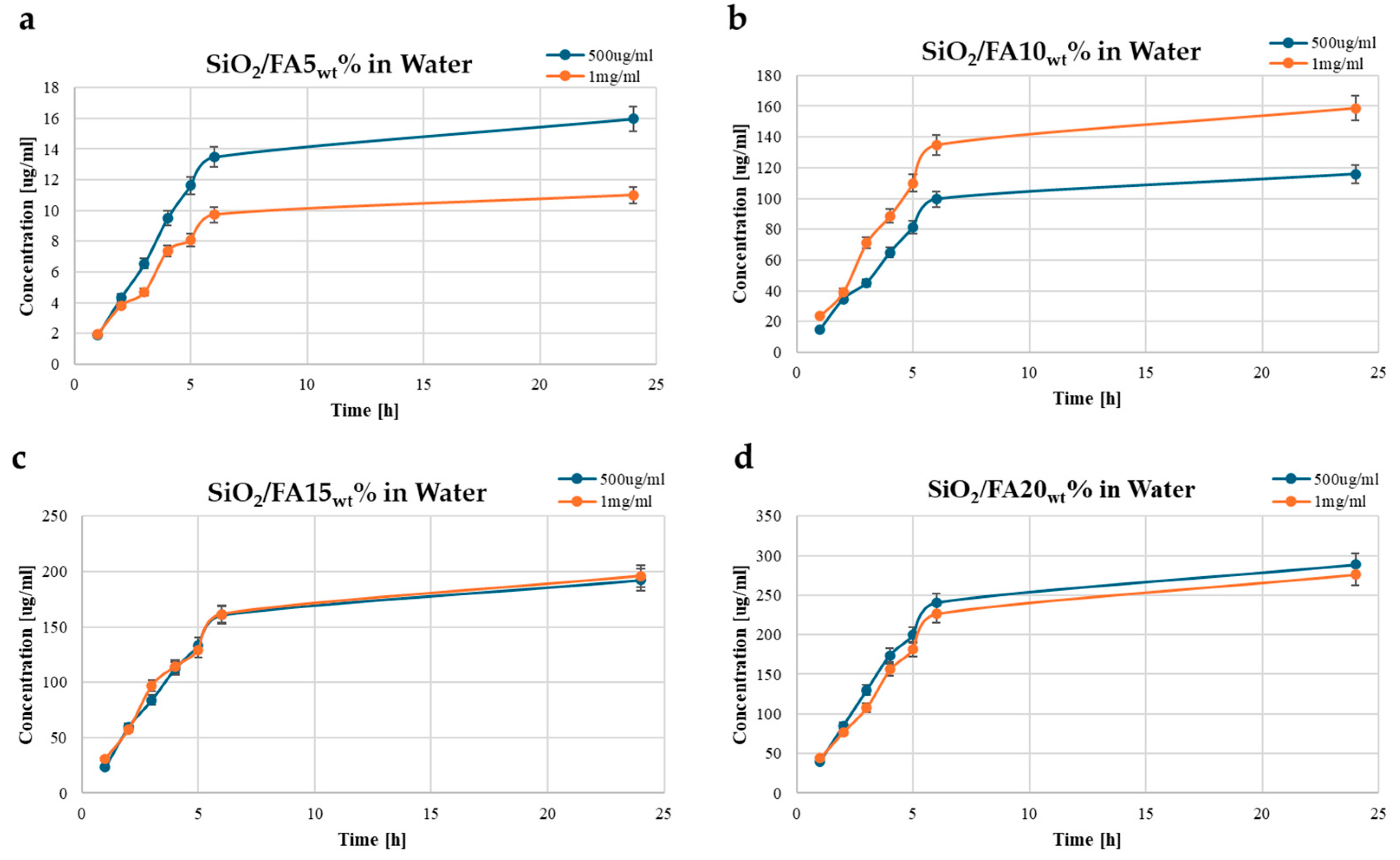
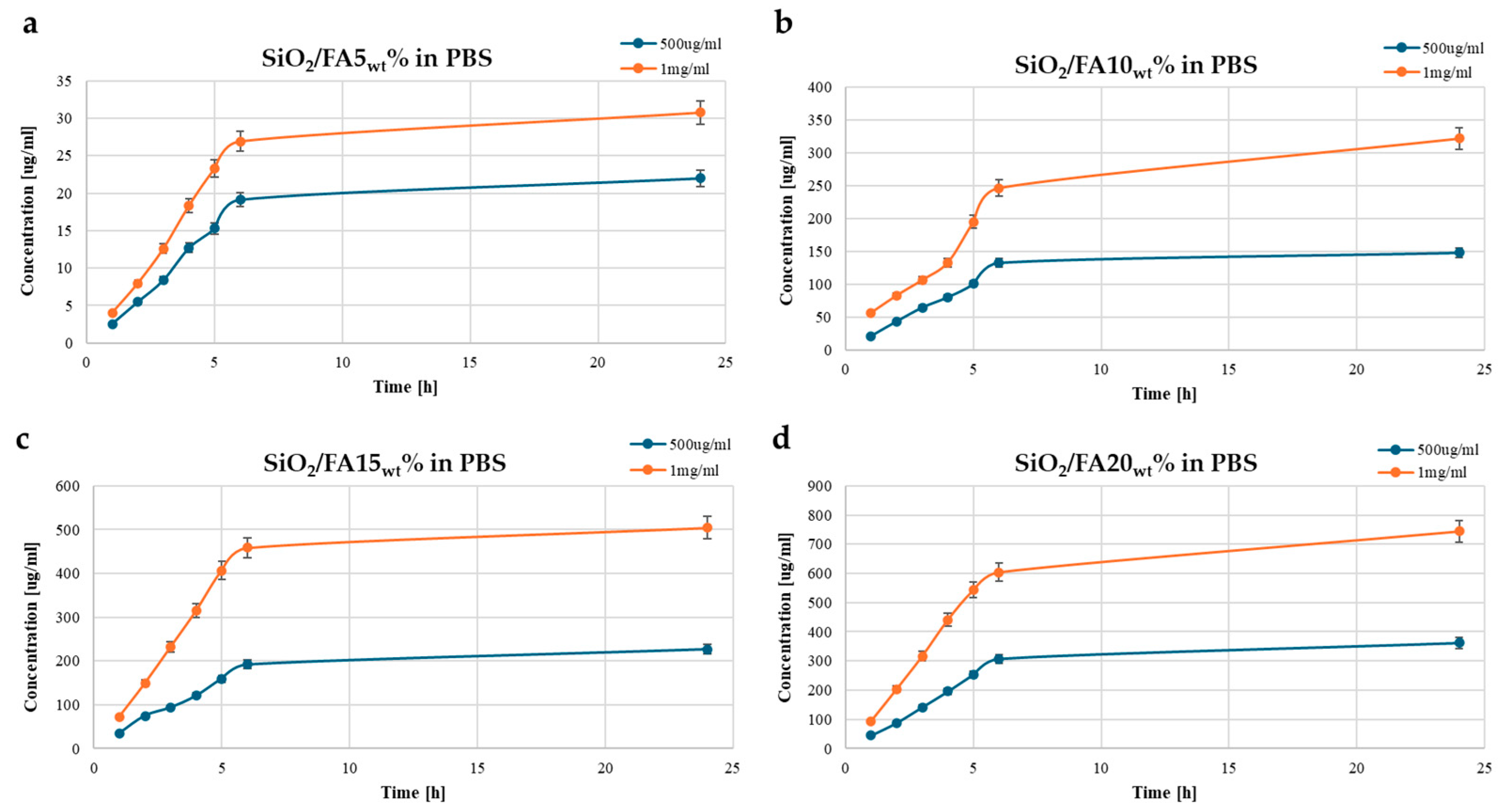
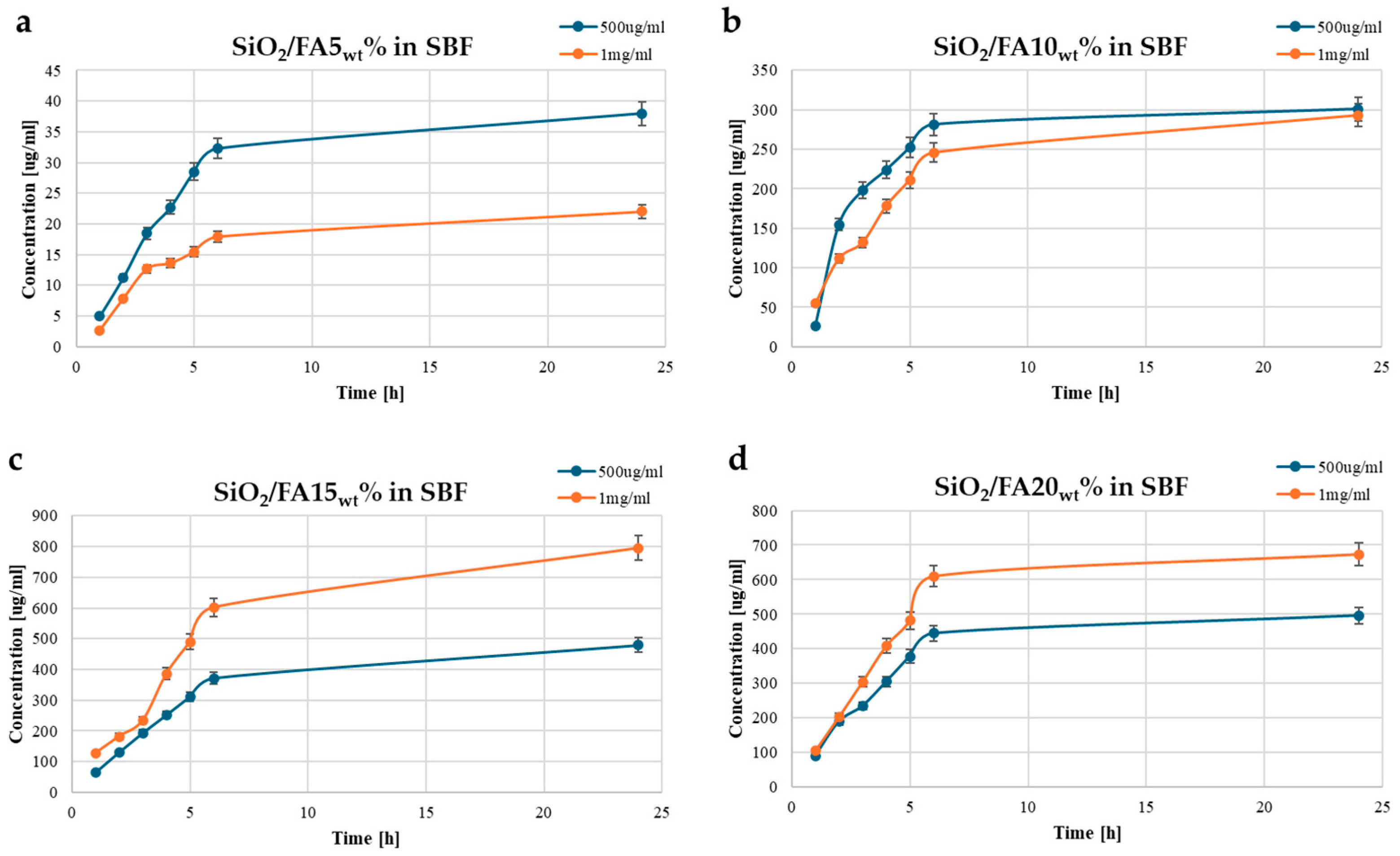
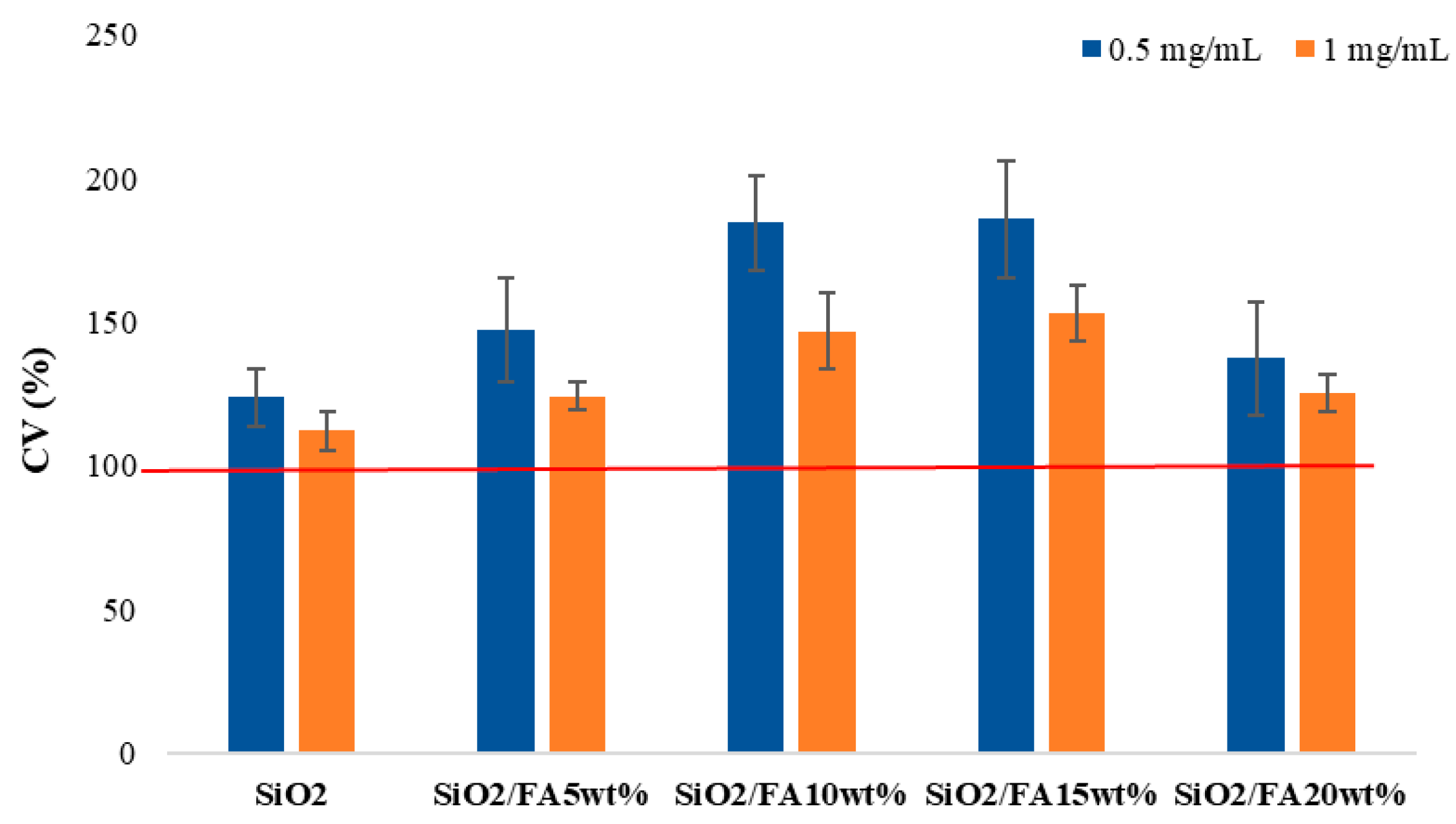

| Label | System Composition | |
|---|---|---|
| Inorganic Matrix SiO2 (wt%) | Organic Matrix FA (wt%) | |
| SiO2 | 100 | --- |
| SiO2/FA5wt% | 95 | 5 |
| SiO2/FA10wt% | 90 | 10 |
| SiO2/FA15wt% | 85 | 15 |
| SiO2/FA20wt% | 80 | 20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barrino, F.; Giuliano, F.; Dispenza, C. Release and Cytocompatibility Study of New Hybrid Materials Based on Ferulic Acid for Biomedical Use. Int. J. Mol. Sci. 2025, 26, 8450. https://doi.org/10.3390/ijms26178450
Barrino F, Giuliano F, Dispenza C. Release and Cytocompatibility Study of New Hybrid Materials Based on Ferulic Acid for Biomedical Use. International Journal of Molecular Sciences. 2025; 26(17):8450. https://doi.org/10.3390/ijms26178450
Chicago/Turabian StyleBarrino, Federico, Federica Giuliano, and Clelia Dispenza. 2025. "Release and Cytocompatibility Study of New Hybrid Materials Based on Ferulic Acid for Biomedical Use" International Journal of Molecular Sciences 26, no. 17: 8450. https://doi.org/10.3390/ijms26178450
APA StyleBarrino, F., Giuliano, F., & Dispenza, C. (2025). Release and Cytocompatibility Study of New Hybrid Materials Based on Ferulic Acid for Biomedical Use. International Journal of Molecular Sciences, 26(17), 8450. https://doi.org/10.3390/ijms26178450







