The Role of the SLC1A5 Transporter on Glutathione Homeostasis and Enterocyte Apoptosis in Chronic Treatment of Rats with Immunosuppressive Drugs
Abstract
1. Introduction
2. Results
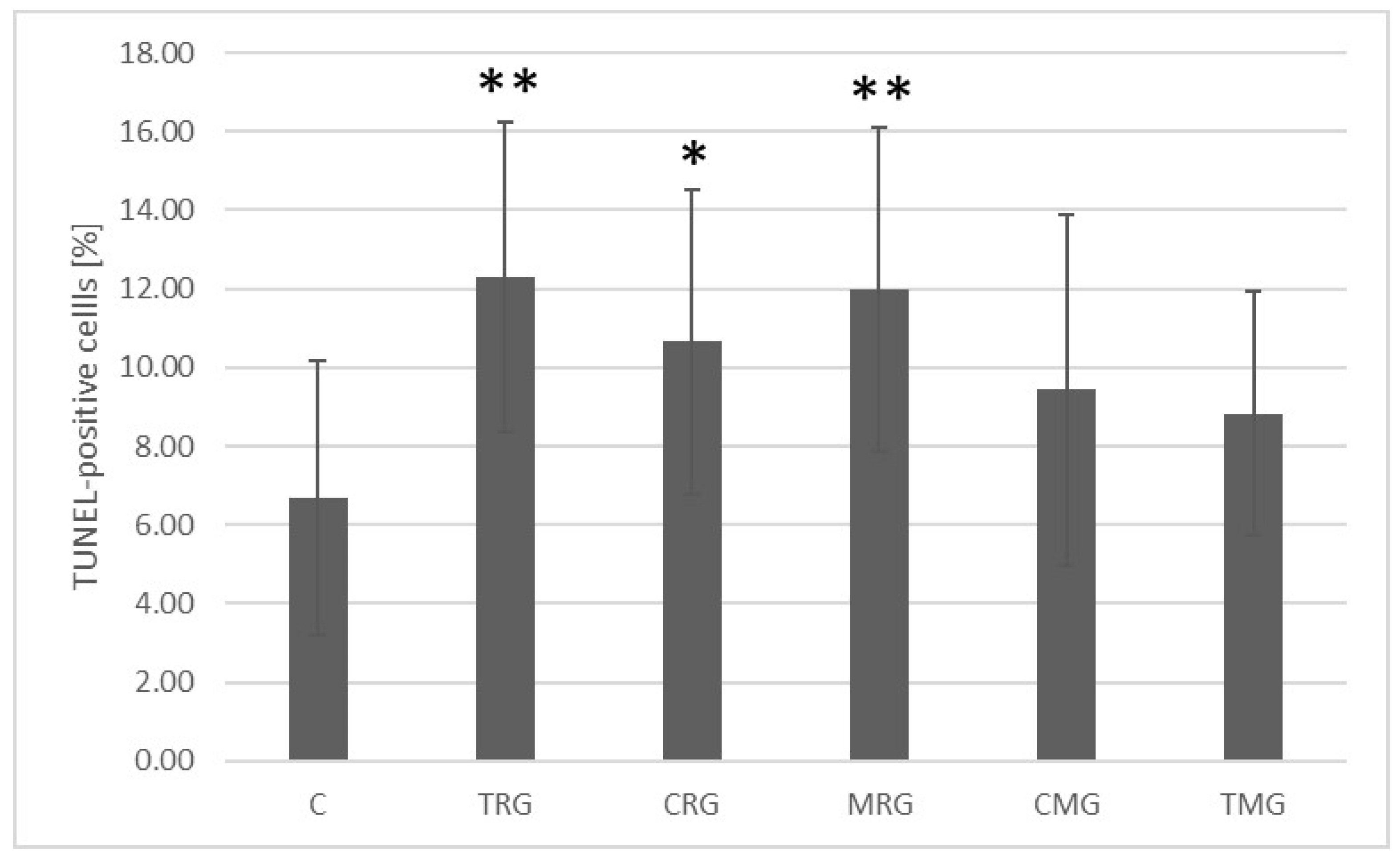
2.1. Quantification of Apoptotic Cells Using the TUNEL Assay
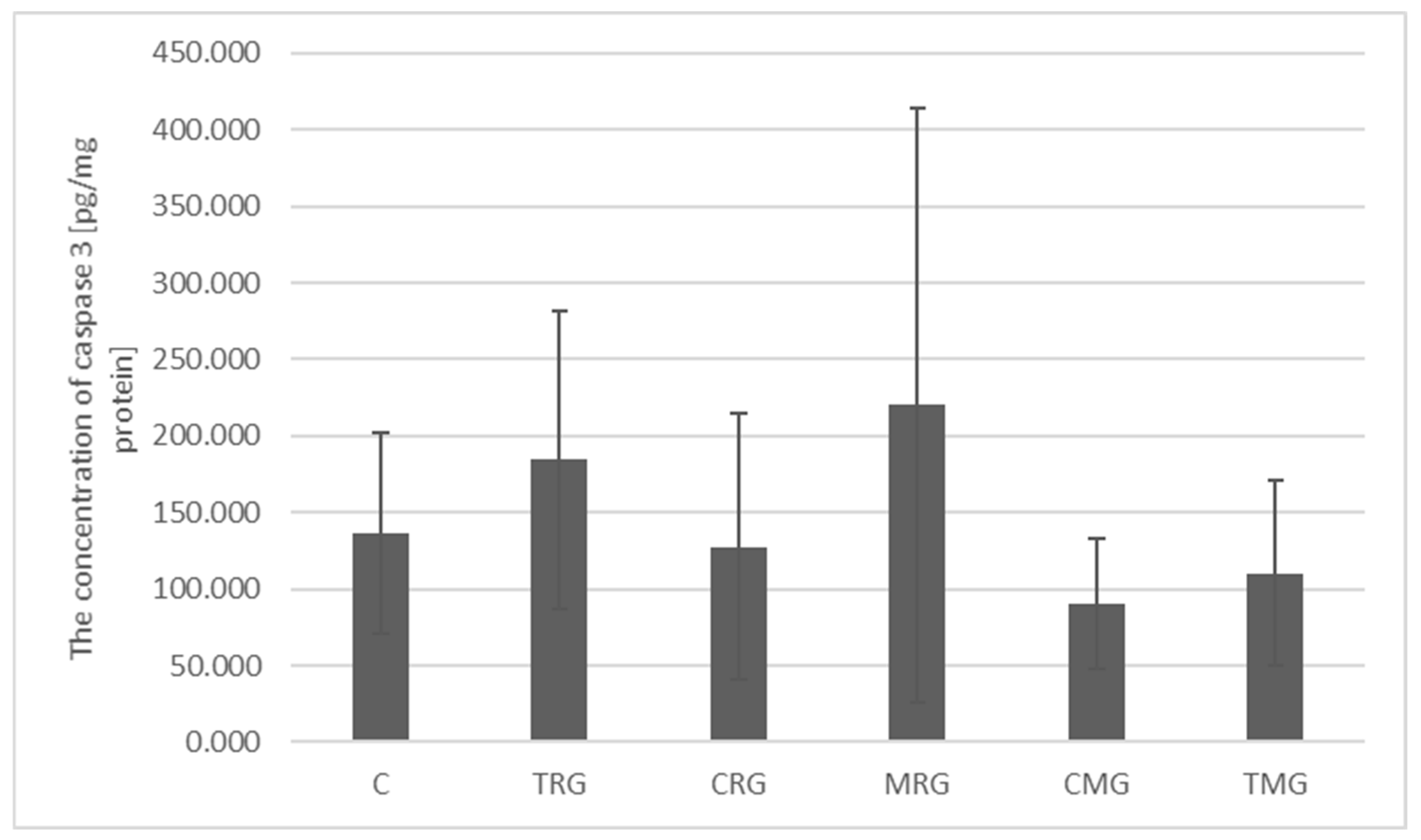
2.2. Quantitative Analysis of Caspase 3 Concentration
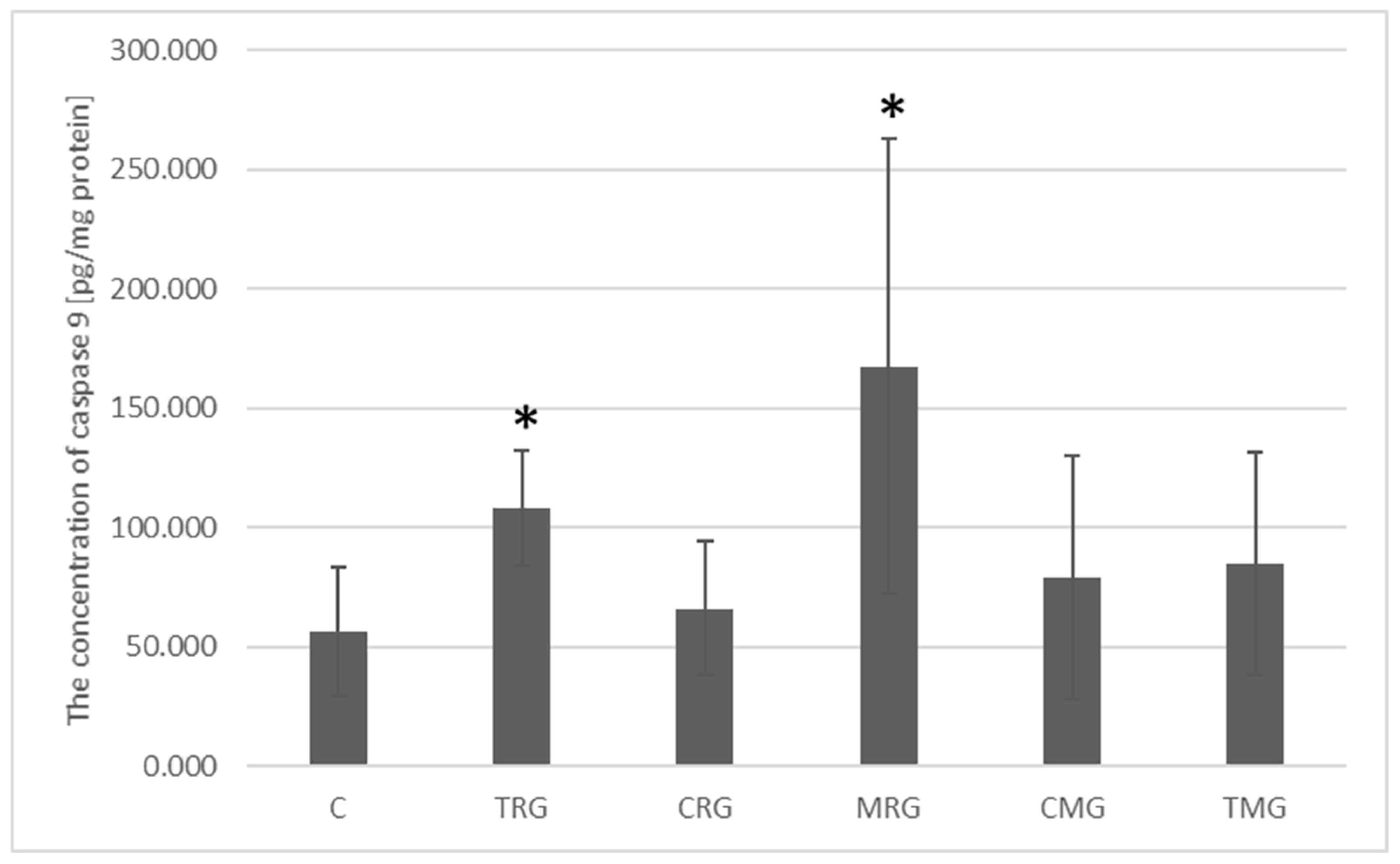
2.3. Quantitative Analysis of Caspase 9 Concentration
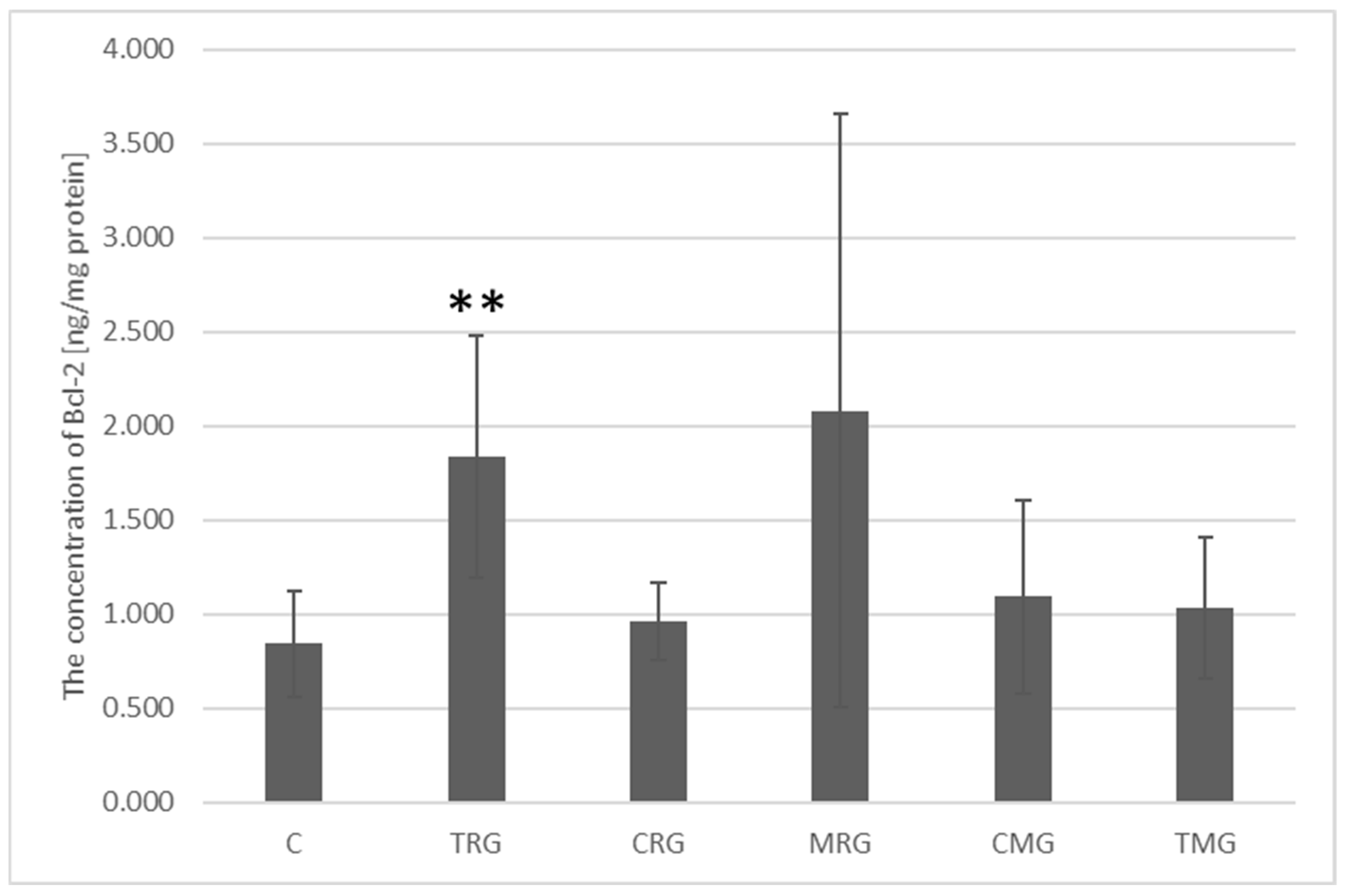
2.4. Quantitative Analysis of Bcl-2 Protein Concentration
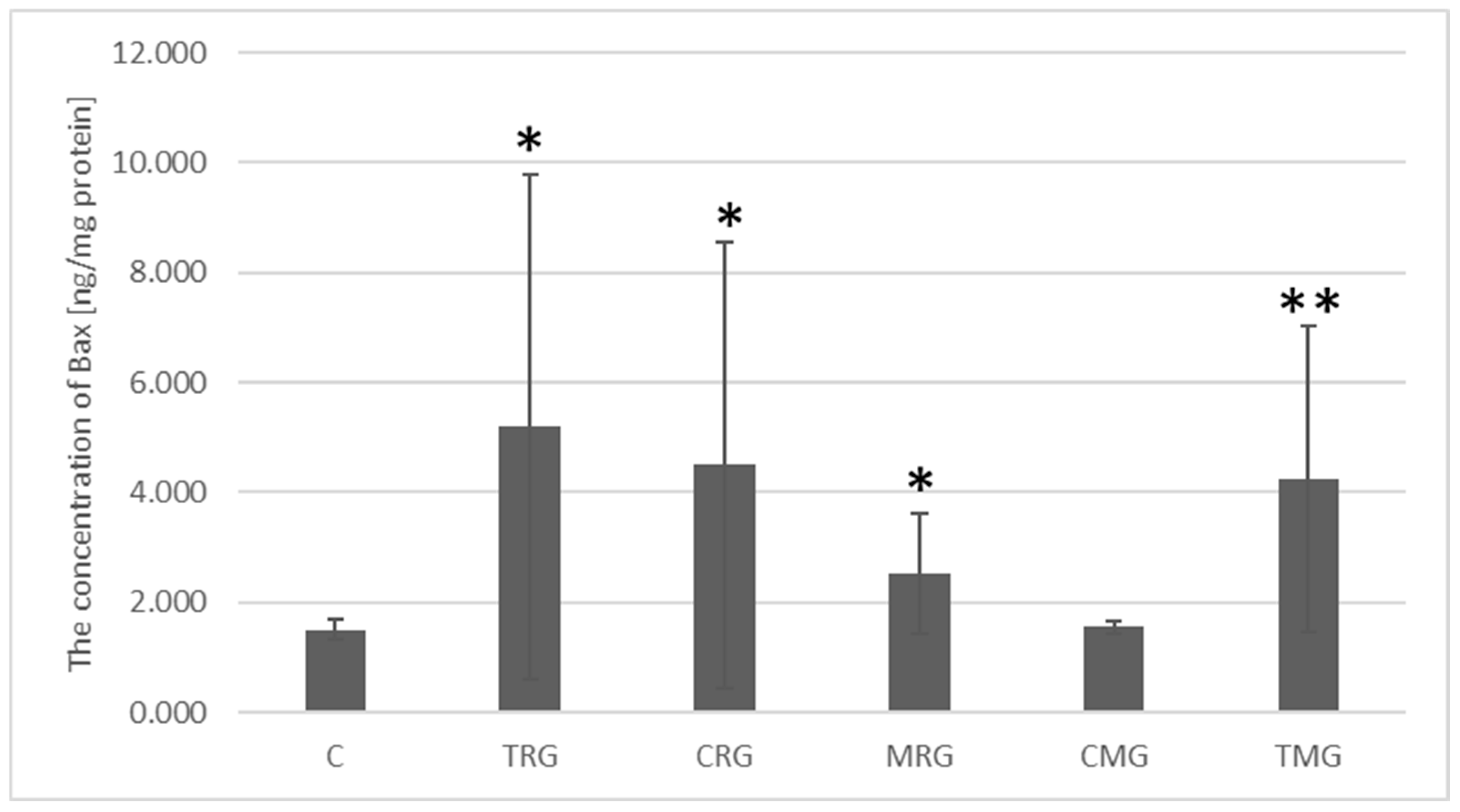
2.5. Quantitative Analysis of Bax Protein Concentration
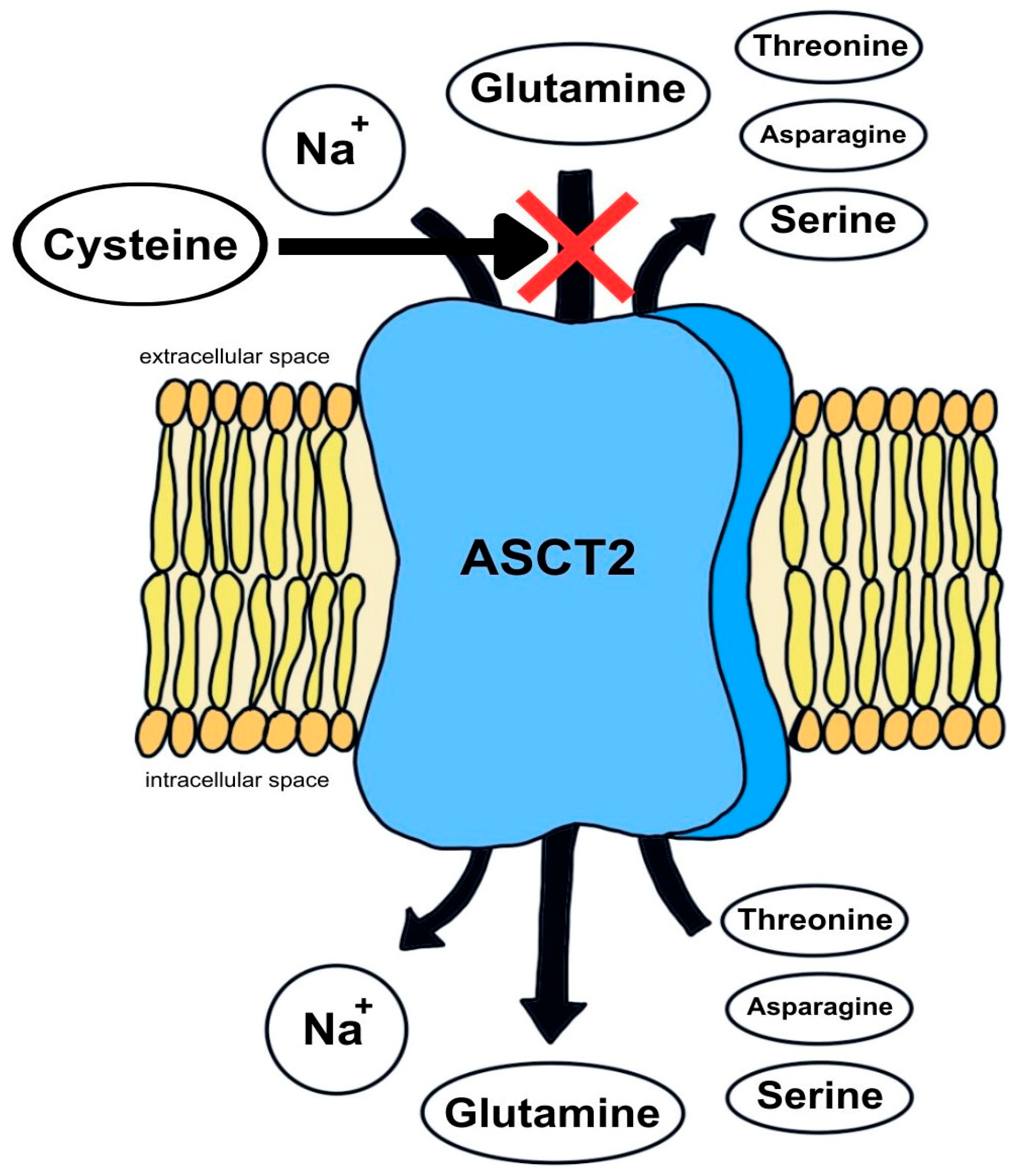
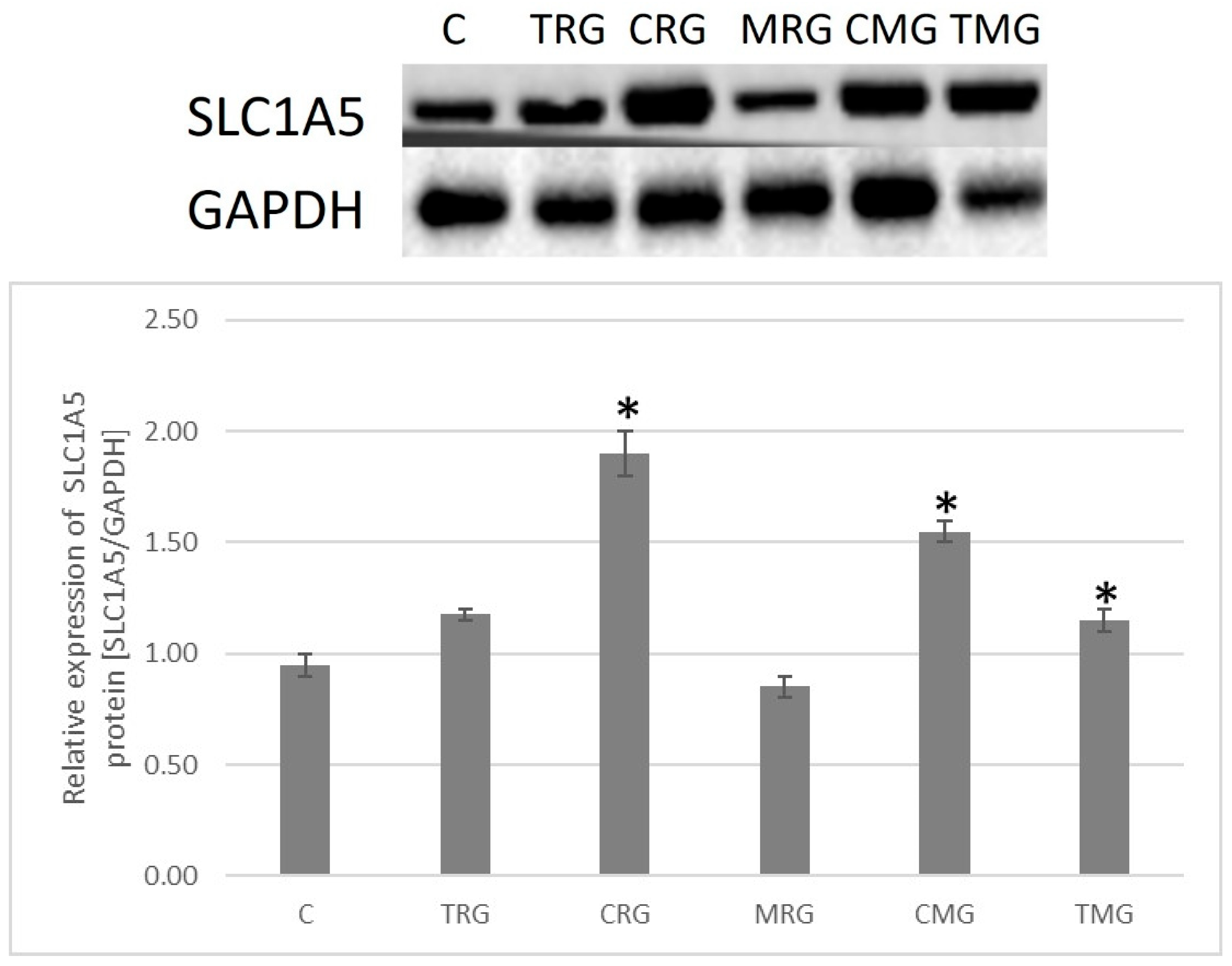
2.6. Quantitative Analysis of SLC1A5 (ASCT2) Transporter Expression
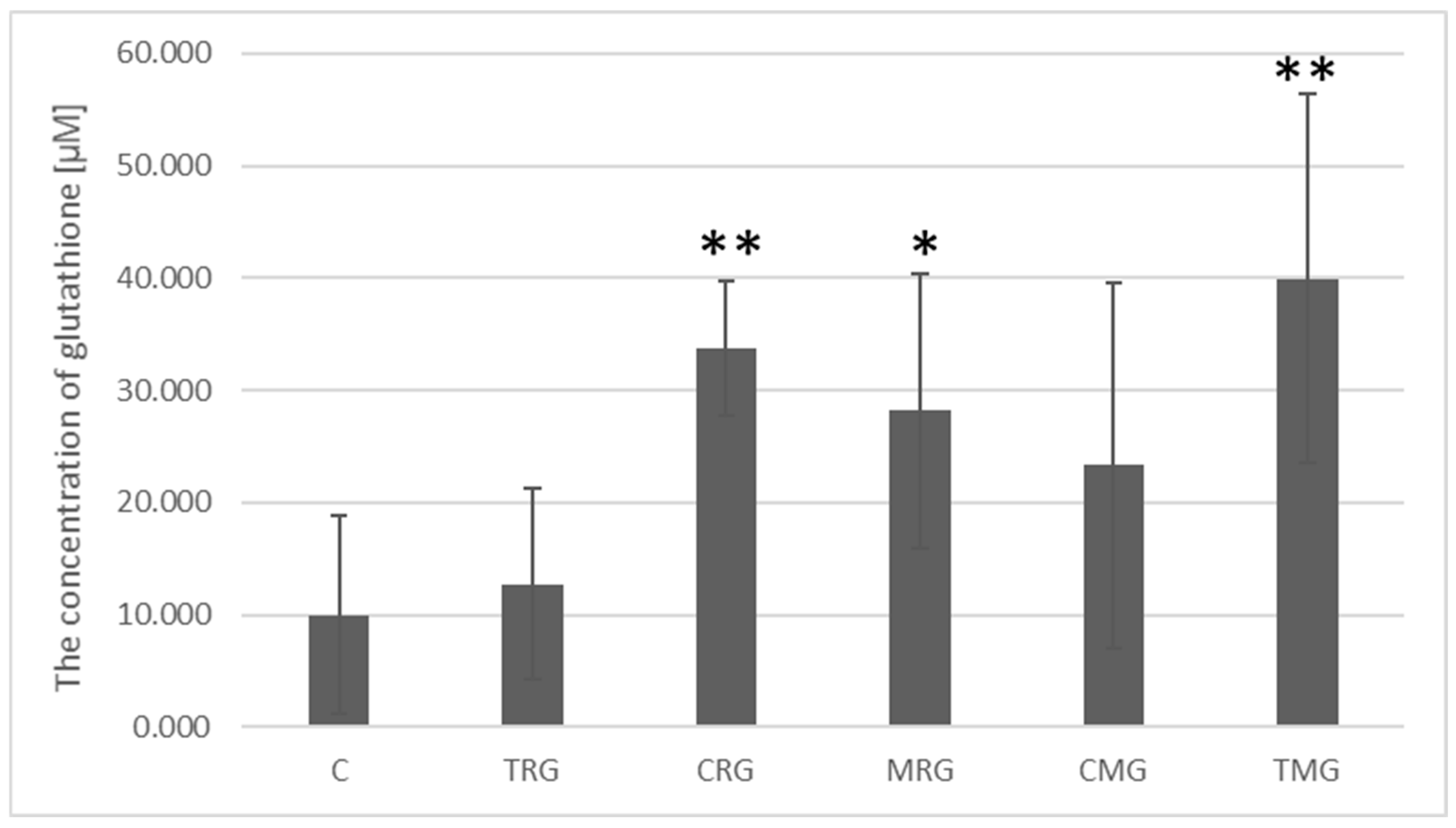
2.7. Quantitative Analysis of Glutathione Concentration
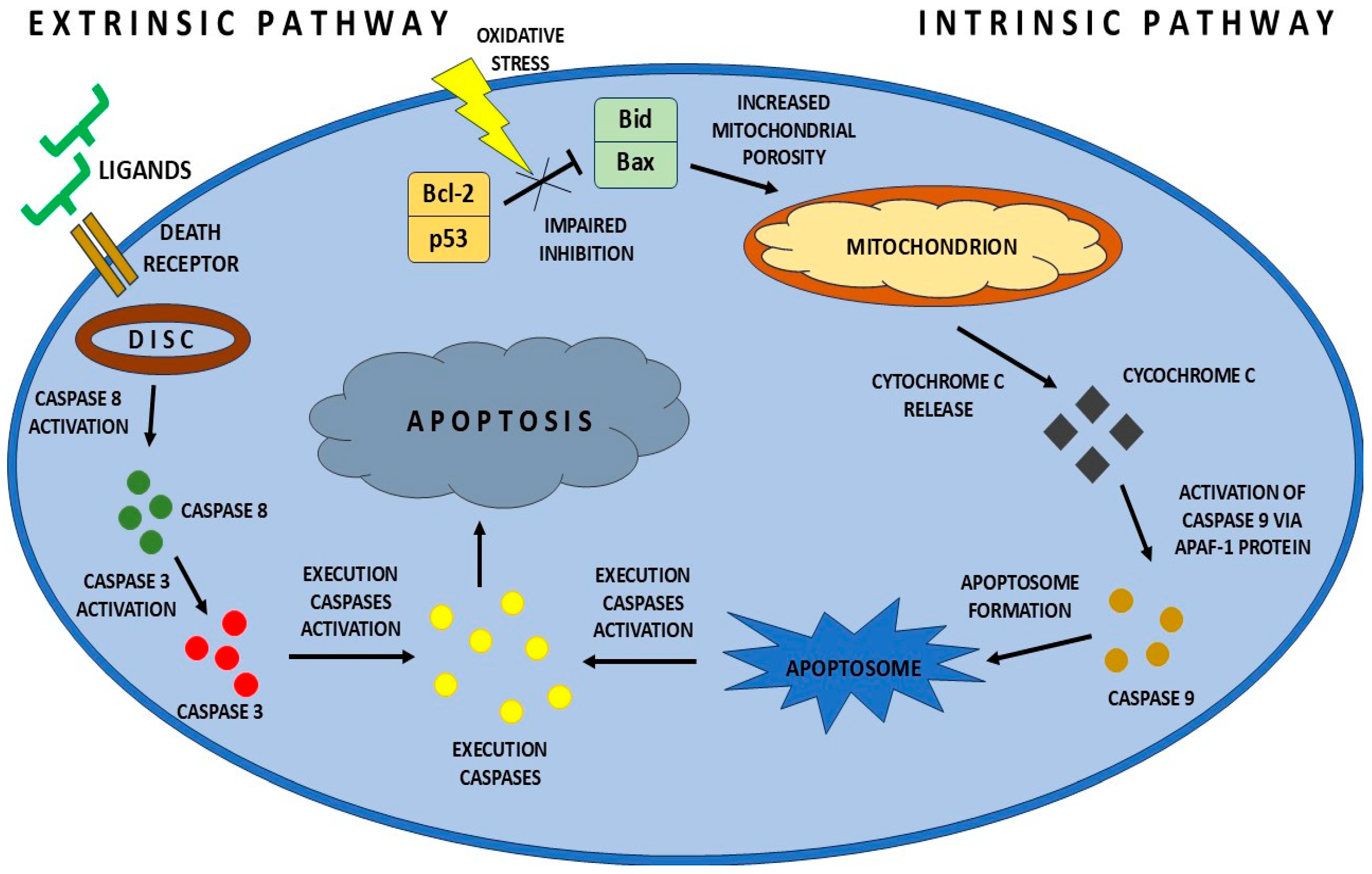
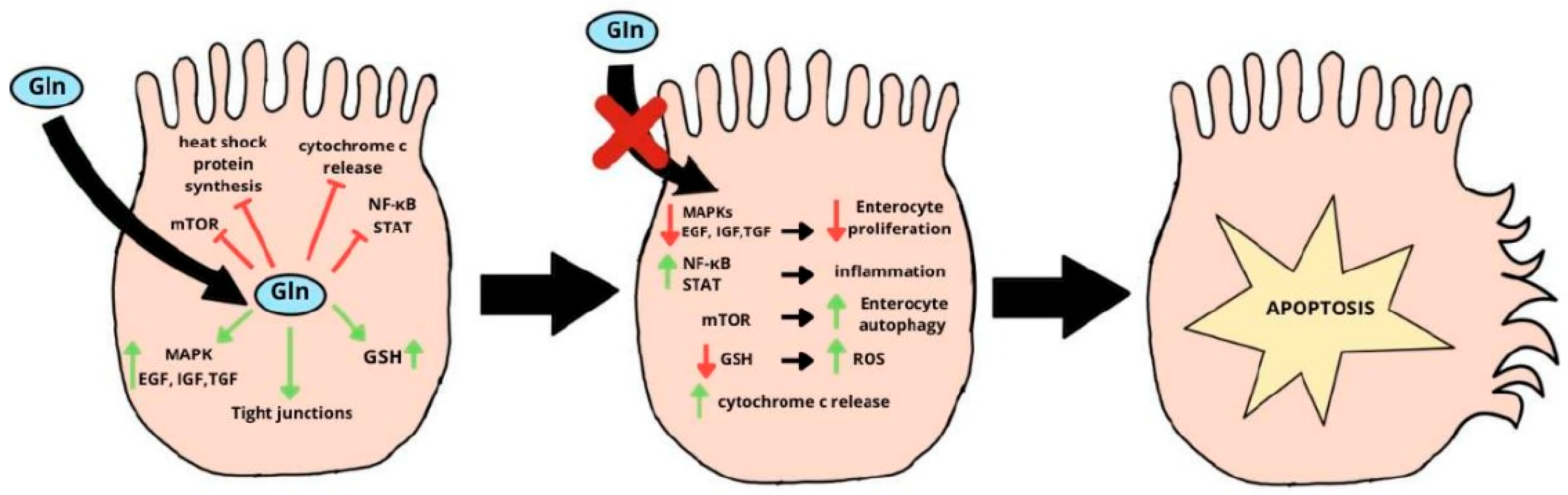
3. Discussion
4. Material and Methods
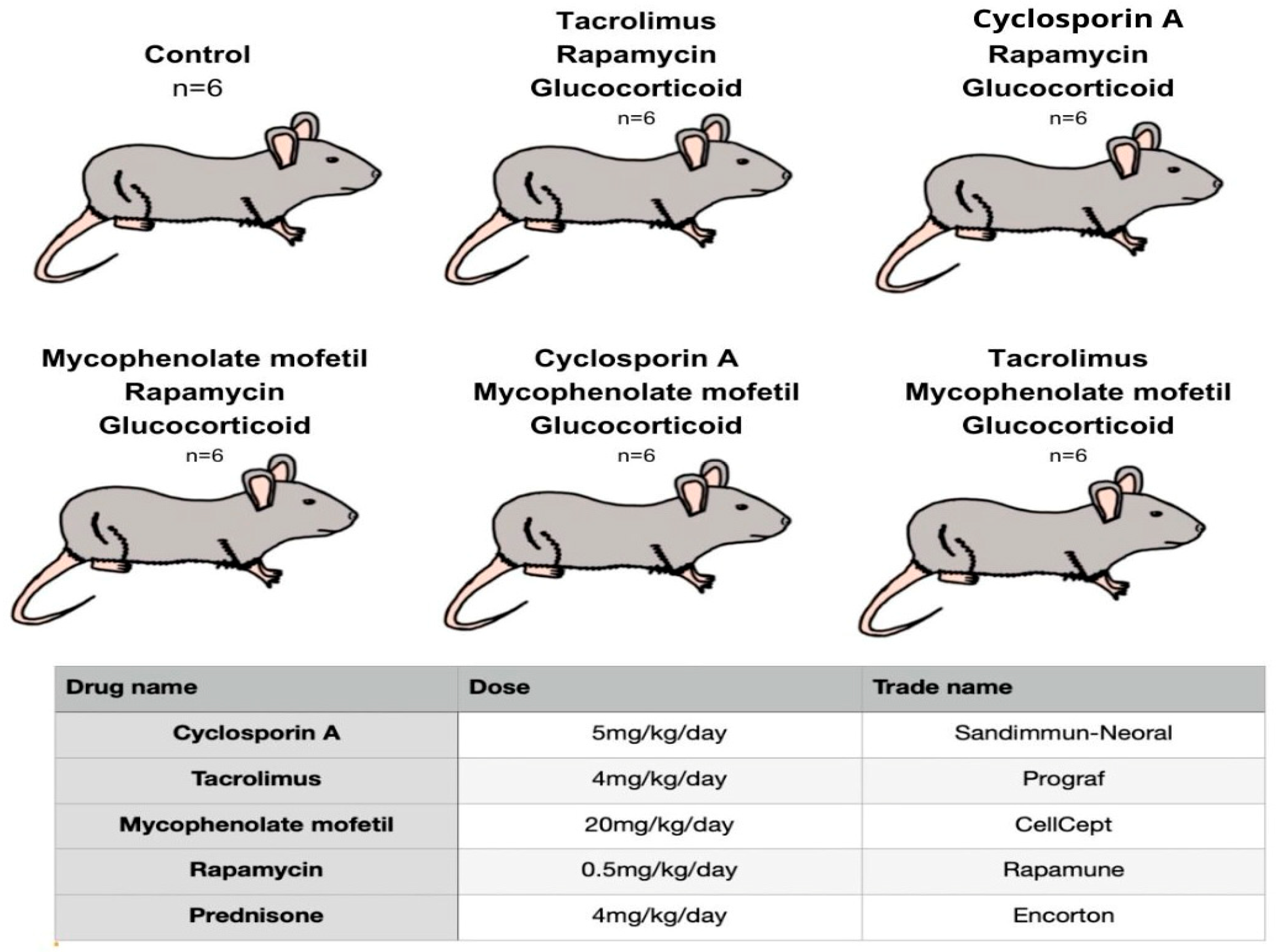
4.1. Material
4.2. Methods
4.2.1. Quantitative Analysis of TUNEL-Positive Cells
4.2.2. Protein Isolation for ELISA and Western Blot Assays
4.2.3. ELISA Immunoenzymatic Test: Determination of Protein Concentrations of Bcl-2, Bax, Caspase 3 and Caspase 9
4.2.4. Western Blot Measurement of SLC5A1 Protein Expression
4.2.5. Determination of Glutathione Concentration by Enzymatic Method
4.2.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data availability Statement
Conflicts of Interest
References
- Grinyó, J.M. Why Is Organ Transplantation Clinically Important? Cold Spring Harb. Perspect. Med. 2013, 3, a014985. [Google Scholar] [CrossRef]
- Newland, D.M.; Nemeth, T.L. Induction and Standard Immunosuppression. In Solid Organ Transplantation in Infants and Children; Dunn, S.P., Horslen, S., Eds.; Organ and Tissue Transplantation; Springer International Publishing: Cham, Switzerland, 2018; pp. 149–182. [Google Scholar] [CrossRef]
- Chang, D.H.; Youn, J.-C.; Dilibero, D.; Patel, J.K.; Kobashigawa, J.A. Heart Transplant Immunosuppression Strategies at Cedars-Sinai Medical Center. Int. J. Heart Fail. 2020, 3, 15–30. [Google Scholar] [CrossRef]
- Arnol, M.; Naumovic, R.; Dimitrov, E.P.; Racki, S.; Bucsa, C.A.; Covic, A.; Mitic, I.; Vavic, N.; Radovanovic, R.M.V.; Zibar, L.; et al. Immunosuppressive regimens following kidney transplantation in five European countries: The observational RECORD study. Transplant. Rep. 2020, 5, 100061. [Google Scholar] [CrossRef]
- Neuwirt, H.; Rudnicki, M.; Schratzberger, P.; Pirklbauer, M.; Kronbichler, A.; Mayer, G. Immunosuppression after renal transplantation. Memo-Mag. Eur. Med. Oncol. 2019, 12, 216–221. [Google Scholar] [CrossRef]
- Opałka, B.; Żołnierczuk, M.; Grabowska, M. Immunosuppressive Agents—Effects on the Cardiovascular System and Selected Metabolic Aspects: A Review. J. Clin. Med. 2023, 12, 6935. [Google Scholar] [CrossRef]
- Tielemans, M.M.; van Boekel, G.A.J.; van Gelder, T.; Tjwa, E.T.; Hilbrands, L.B. Immunosuppressive drugs and the gastrointestinal tract in renal transplant patients. Transplant. Rev. Orlando Fla 2019, 33, 55–63. [Google Scholar] [CrossRef]
- Surówka, A.; Żołnierczuk, M.; Prowans, P.; Grabowska, M.; Kupnicka, P.; Markowska, M.; Szlosser, Z.; Kędzierska-Kapuza, K. The Effect of Chronic Immunosuppressive Regimen Treatment on Apoptosis in the Heart of Rats. Pharmaceuticals 2024, 17, 1188. [Google Scholar] [CrossRef]
- Mustafa, M.; Ahmad, R.; Tantry, I.Q.; Ahmad, W.; Siddiqui, S.; Alam, M.; Abbas, K.; Moinuddin; Hassan, M.I.; Habib, S.; et al. Apoptosis: A Comprehensive Overview of Signaling Pathways, Morphological Changes, and Physiological Significance and Therapeutic Implications. Cells 2024, 13, 1838. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A Review of Programmed Cell Death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Kim, M.-H.; Kim, H. The Roles of Glutamine in the Intestine and Its Implication in Intestinal Diseases. Int. J. Mol. Sci. 2017, 18, 1051. [Google Scholar] [CrossRef]
- Rao, R.; Samak, G. Role of Glutamine in Protection of Intestinal Epithelial Tight Junctions. J. Epithel. Biol. Pharmacol. 2012, 5, 47–54. [Google Scholar] [CrossRef]
- Circu, M.L.; Yee Aw, T. Glutathione and apoptosis. Free Radic. Res. 2008, 42, 689–706. [Google Scholar] [CrossRef]
- Wang, B.; Wu, G.; Zhou, Z.; Dai, Z.; Sun, Y.; Ji, Y.; Li, W.; Wang, W.; Liu, C.; Han, F.; et al. Glutamine and intestinal barrier function. Amino Acids 2015, 47, 2143–2154. [Google Scholar] [CrossRef]
- Souba, W.W.; Smith, R.J.; Wilmore, D.W. Effects of glucocorticoids on glutamine metabolism in visceral organs. Metabolism 1985, 34, 450–456. [Google Scholar] [CrossRef]
- D’Arcy, M.S. Cell death: A review of the major forms of apoptosis, necrosis and autophagy. Cell Biol. Int. 2019, 43, 582–592. [Google Scholar] [CrossRef]
- Jan, R.; Chaudhry, G.-S. Understanding Apoptosis and Apoptotic Pathways Targeted Cancer Therapeutics. Adv. Pharm. Bull. 2019, 9, 205–218. [Google Scholar] [CrossRef]
- Sayers, T.J. Targeting the extrinsic apoptosis signaling pathway for cancer therapy. Cancer Immunol. Immunother. CII 2011, 60, 1173–1180. [Google Scholar] [CrossRef]
- Czabotar, P.E.; Lessene, G.; Strasser, A.; Adams, J.M. Control of apoptosis by the BCL-2 protein family: Implications for physiology and therapy. Nat. Rev. Mol. Cell Biol. 2014, 15, 49–63. [Google Scholar] [CrossRef]
- Martinou, J.-C.; Youle, R.J. Mitochondria in Apoptosis: Bcl-2 family Members and Mitochondrial Dynamics. Dev. Cell 2011, 21, 92–101. [Google Scholar] [CrossRef]
- Häcker, G. The morphology of apoptosis. Cell Tissue Res. 2000, 301, 5–17. [Google Scholar] [CrossRef]
- Faitova, J.; Krekac, D.; Hrstka, R.; Vojtesek, B. Endoplasmic reticulum stress and apoptosis. Cell. Mol. Biol. Lett. 2006, 11, 488–505. [Google Scholar] [CrossRef]
- Beilankouhi, E.A.V.; Sajadi, M.A.; Alipourfard, I.; Hassani, P.; Valilo, M.; Safaralizadeh, R. Role of the ER-induced UPR pathway, apoptosis, and autophagy in colorectal cancer. Pathol.-Res. Pract. 2023, 248, 154706. [Google Scholar] [CrossRef]
- García-Sánchez, A.; Miranda-Díaz, A.G.; Cardona-Muñoz, E.G. The Role of Oxidative Stress in Physiopathology and Pharmacological Treatment with Pro- and Antioxidant Properties in Chronic Diseases. Oxid. Med. Cell. Longev. 2020, 2020, 2082145. [Google Scholar] [CrossRef]
- Gulcin, İ. Antioxidants: A comprehensive review. Arch. Toxicol. 2025, 99, 1893–1997. [Google Scholar] [CrossRef]
- Kwiatkowska, M.; Oldakowska-Jedynak, U.; Wojtaszek, E.; Glogowski, T.; Malyszko, J. Potential Effects of Immunosuppression on Oxidative Stress and Atherosclerosis in Kidney Transplant Recipients. Oxid. Med. Cell. Longev. 2021, 2021, 6660846. [Google Scholar] [CrossRef]
- Longoni, B.; Boschi, E.; Demontis, G.C.; Ratto, G.M.; Mosca, F. Apoptosis and adaptive responses to oxidative stress in human endothelial cells exposed to cyclosporin A correlate with BCL-2 expression levels. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2001, 15, 731–740. [Google Scholar] [CrossRef]
- Trapp, A.; Weis, M. The Impact of Immunosuppression on Endothelial Function. J. Cardiovasc. Pharmacol. 2005, 45, 81. [Google Scholar] [CrossRef]
- Akbasli, A.C.; Keven, K.; Erbay, B.; Nebioglu, S. Changes in oxidative stress in renal graft patients receiving calcineurin inhibitors: Cyclosporine versus tacrolimus. Exp. Clin. Transplant. Off. J. Middle East Soc. Organ Transplant. 2012, 10, 439–445. [Google Scholar] [CrossRef]
- Paquette, J.C.; Guérin, P.J.; Gauthier, E.R. Rapid induction of the intrinsic apoptotic pathway by L-glutamine starvation. J. Cell. Physiol. 2005, 202, 912–921. [Google Scholar] [CrossRef]
- Wischmeyer, P.E. Glutamine and heat shock protein expression. Nutr. Burbank Los Angel. Cty. Calif 2002, 18, 225–228. [Google Scholar] [CrossRef]
- Labarrere, C.A.; Kassab, G.S. Glutathione: A Samsonian life-sustaining small molecule that protects against oxidative stress, ageing and damaging inflammation. Front. Nutr. 2022, 9, 1007816. [Google Scholar] [CrossRef] [PubMed]
- Bröer, S.; Fairweather, S.J. Amino Acid Transport Across the Mammalian Intestine. Compr. Physiol. 2019, 9, 343–373. [Google Scholar] [CrossRef]
- Zuhl, M.; Specht, J.; Beatty, S.; Mermier, C. Glutamine Supplementation: A Possible Strategy to Help Mitigate Health Risks of Heat-Related Illness. J. Nutr. Metab. 2024, 2024, 1638244. [Google Scholar] [CrossRef] [PubMed]
- Kędzierska, K.; Sporniak-Tutak, K.; Sindrewicz, K.; Bober, J.; Domański, L.; Parafiniuk, M.; Urasińska, E.; Ciechanowicz, A.; Domański, M.; Smektała, T.; et al. Effects of immunosuppressive treatment on protein expression in rat kidney. Drug Des. Devel. Ther. 2014, 8, 1695–1708. [Google Scholar] [CrossRef][Green Version]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wawrowski, T.; Surówka, A.; Żołnierczuk, M.; Prowans, P.; Grabowska, M.; Kupnicka, P.; Markowska, M.; Kaczmarkiewicz, M.; Sych, W.; Zagrodnik, E.; et al. The Role of the SLC1A5 Transporter on Glutathione Homeostasis and Enterocyte Apoptosis in Chronic Treatment of Rats with Immunosuppressive Drugs. Int. J. Mol. Sci. 2025, 26, 8330. https://doi.org/10.3390/ijms26178330
Wawrowski T, Surówka A, Żołnierczuk M, Prowans P, Grabowska M, Kupnicka P, Markowska M, Kaczmarkiewicz M, Sych W, Zagrodnik E, et al. The Role of the SLC1A5 Transporter on Glutathione Homeostasis and Enterocyte Apoptosis in Chronic Treatment of Rats with Immunosuppressive Drugs. International Journal of Molecular Sciences. 2025; 26(17):8330. https://doi.org/10.3390/ijms26178330
Chicago/Turabian StyleWawrowski, Tomasz, Anna Surówka, Michał Żołnierczuk, Piotr Prowans, Marta Grabowska, Patrycja Kupnicka, Marta Markowska, Mikołaj Kaczmarkiewicz, Weronika Sych, Edyta Zagrodnik, and et al. 2025. "The Role of the SLC1A5 Transporter on Glutathione Homeostasis and Enterocyte Apoptosis in Chronic Treatment of Rats with Immunosuppressive Drugs" International Journal of Molecular Sciences 26, no. 17: 8330. https://doi.org/10.3390/ijms26178330
APA StyleWawrowski, T., Surówka, A., Żołnierczuk, M., Prowans, P., Grabowska, M., Kupnicka, P., Markowska, M., Kaczmarkiewicz, M., Sych, W., Zagrodnik, E., & Kędzierska-Kapuza, K. (2025). The Role of the SLC1A5 Transporter on Glutathione Homeostasis and Enterocyte Apoptosis in Chronic Treatment of Rats with Immunosuppressive Drugs. International Journal of Molecular Sciences, 26(17), 8330. https://doi.org/10.3390/ijms26178330






