Transgenerational Effects and Heritability of Folate Receptor Alpha Autoantibodies in Autism Spectrum Disorder
Abstract
1. Introduction
- •
- Whether FRAA titers are higher in multiplex vs. simplex families, as higher FRAA titers in multiplex families could explain the recurrent risk.
- •
- Whether FRAAs are associated with increased parental age as such a relationship could explain the association of ASD with advanced parental age, especially advanced paternal age.
- •
- Whether FRAAs are transmitted across generations and whether this transmission demonstrates anticipation (increase severity across generations), as anticipation could explain the increase in ASD prevalence in successive generations.
- •
- Whether FRAAs in parents influence the cognitive and behavioral development of their offspring, as such a finding would highlight the importance of the role of parental FRAAs in neurodevelopment.
2. Results
2.1. Participant Characteristics
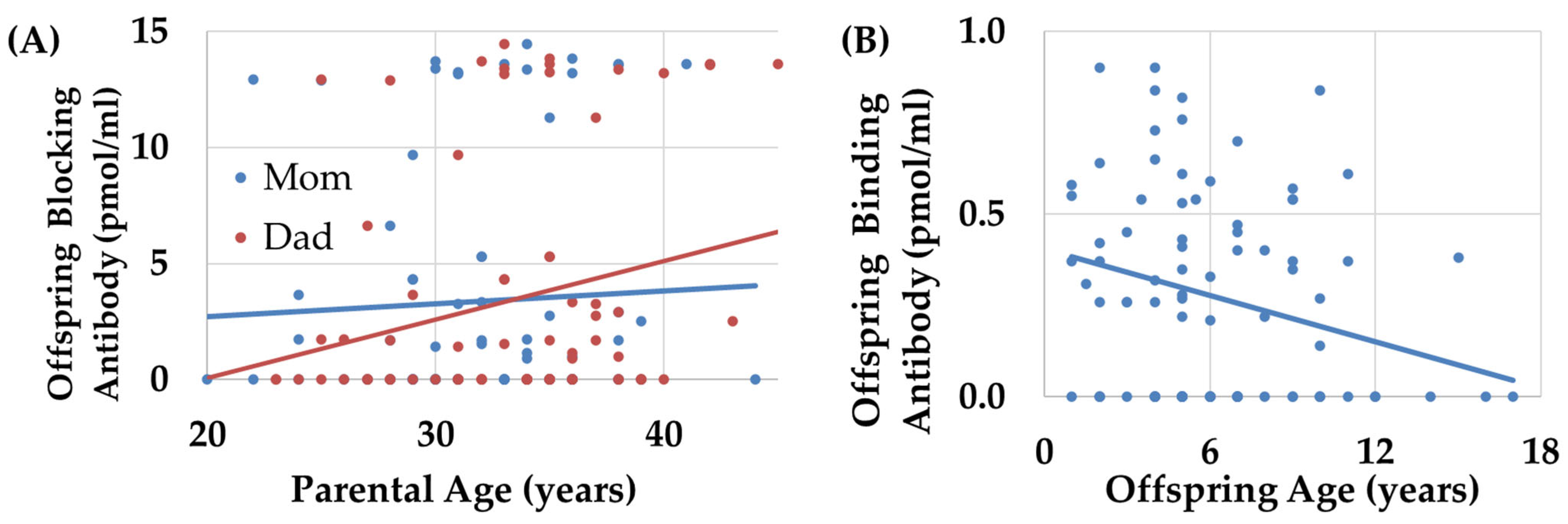
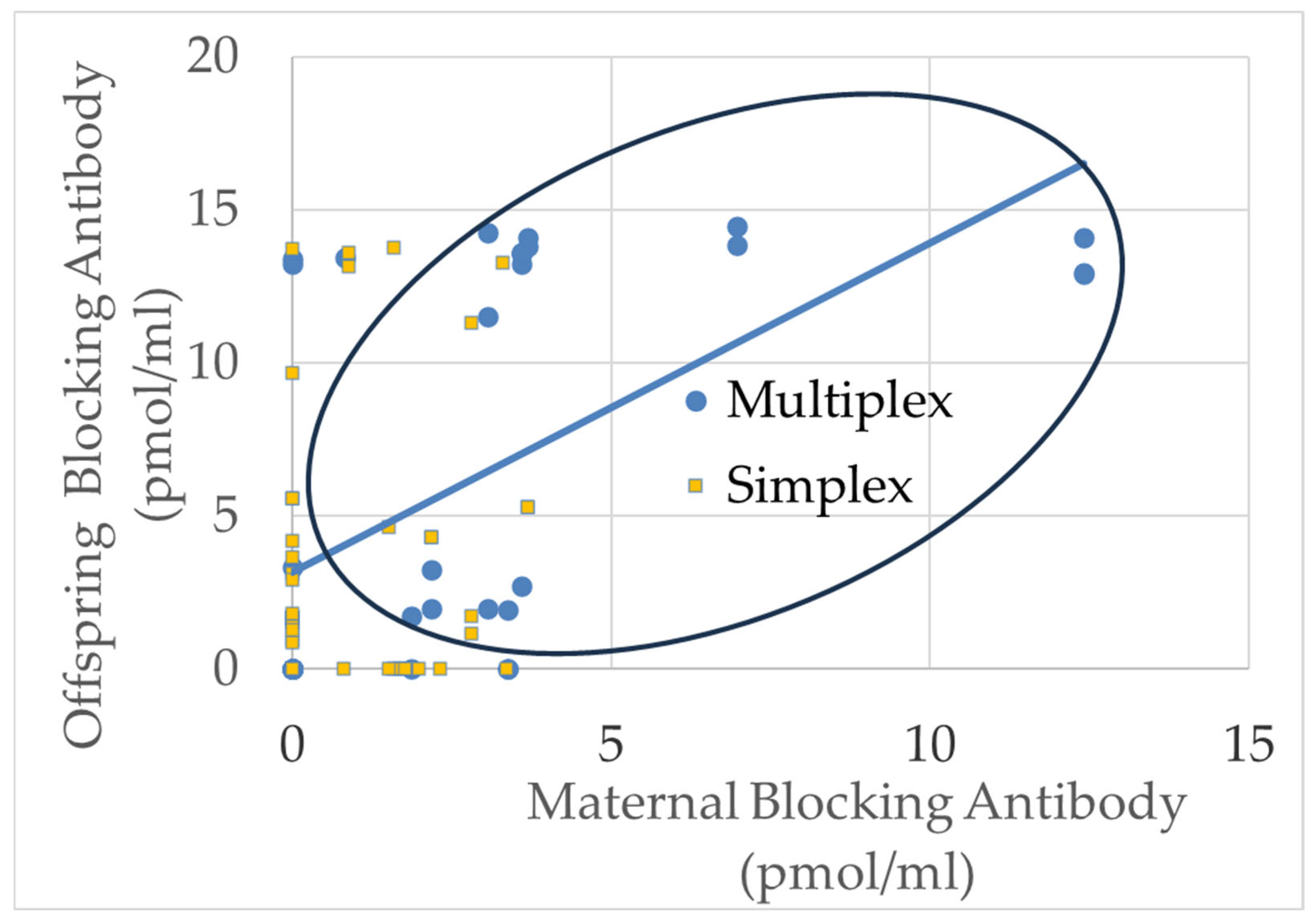
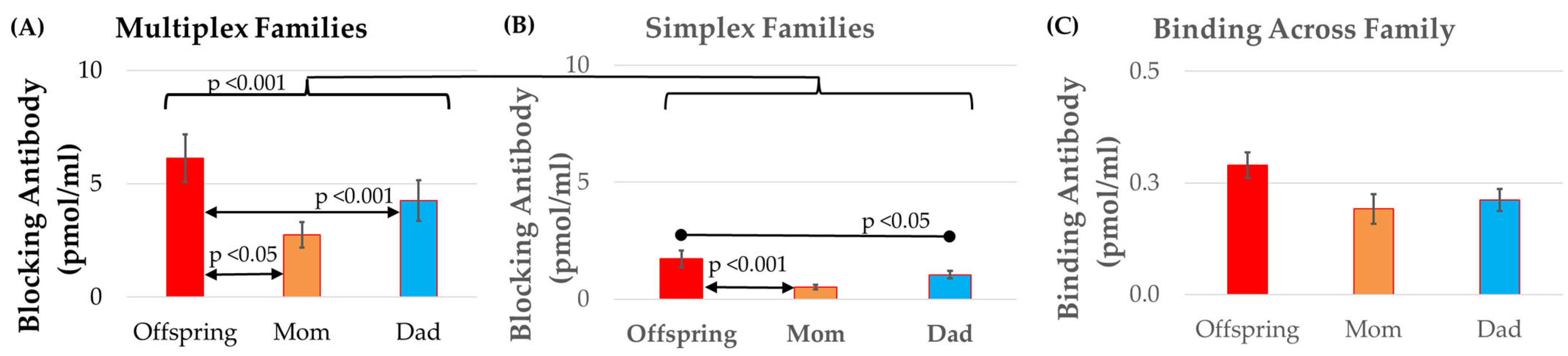
2.2. Effect of Pregnancy and ASD Family Type on FRAA Titers
2.3. Transgenerational Changes in FRAA Titers
2.4. Effect of ASD on Family Titers
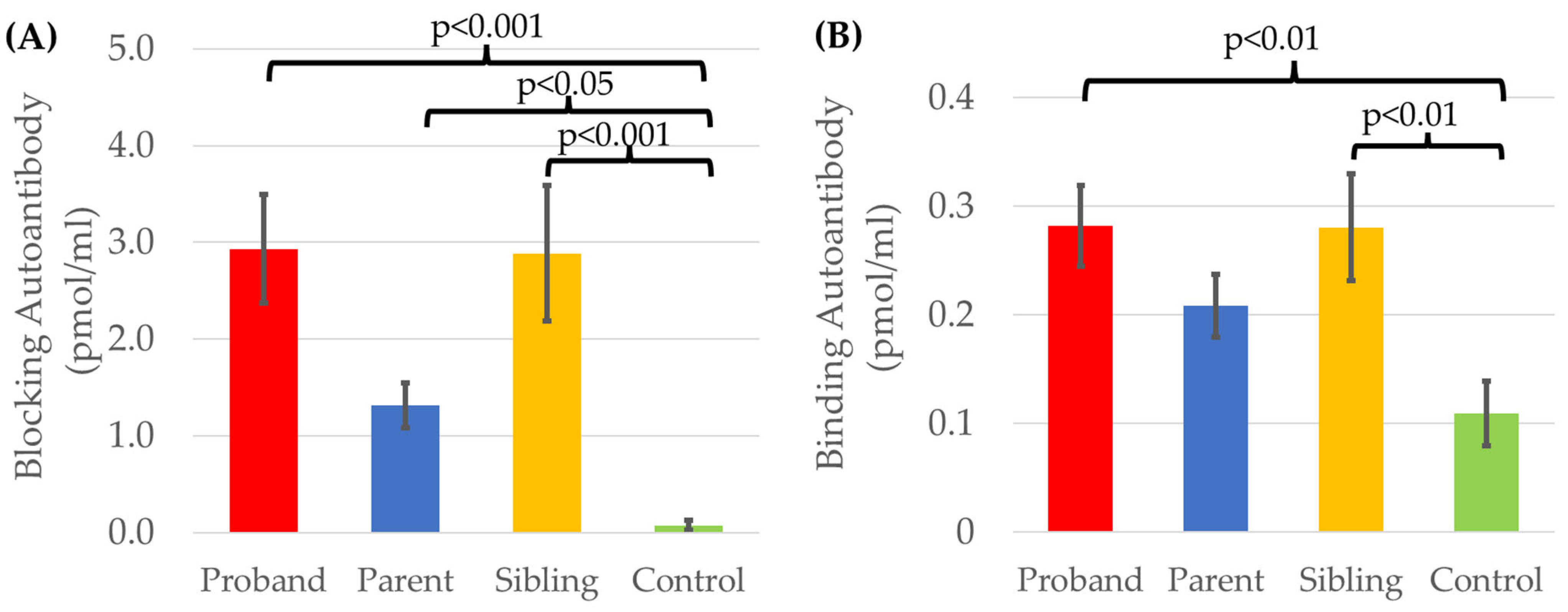
2.5. Familial FRAA Titers Compared to Controls
2.6. Behavior and Development
3. Discussion
4. Materials and Methods
4.1. Titer Measurement
4.2. Developmental Assessments
4.3. Statistical Analysis
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 5-MTHF/CH3-THF | 5-methyltetrahydrofolate |
| ASD | Autism Spectrum Disorder |
| B12 | Vit B12 (cobalamin) |
| BH4 | Tetrahydrobiopterin |
| BHMT | Betaine-homocysteine methyltransferase |
| CBS | Cystathionine beta-synthase |
| CFD | Cerebral folate deficiency |
| CH2-THF | 5,10-methylenetetrahydrofolate |
| COMT | Catechol-O-methyltransferase |
| CTH | Cystathionine gamma-lyase |
| DHFR | Dihydrofolate reductase |
| DNA | Deoxyribonucleic acid |
| DNMT1 | DNA methyltransferase 1 |
| DNMT3A | DNA methyltransferase 3A |
| ELISA | Enzyme-linked immunosorbent assay |
| F | Folate |
| FOLR1 | Folate receptor alpha |
| FRAAs | Folate Receptor Alpha Autoantibodies |
| FRα | Folate Receptor Alpha |
| GABA | γ-Aminobutyric acid (GABA) |
| GCH1 | GTP cyclohydrolase I |
| GS | Glutathione synthetase |
| GTP | Guanosine 5′-triphosphate |
| IBR | Institute for Basic Research in Developmental Disabilities |
| MAT | Methionine adenosyltransferase |
| MBD4 | Methyl-CpG-binding domain protein 4 |
| Me | Methyl group |
| MTHFD1 | Methylenetetrahydrofolate dehydrogenase 1 |
| MTHFD2 | Methylenetetrahydrofolate dehydrogenase 2 |
| MTHFR | Methylenetetrahydrofolate reductase |
| MTR | Methionine synthase |
| MTRR | Methionine synthase reductase |
| PCFC | Protein-coupled folate carrier |
| PDDBI | Pervasive Developmental Disorder Behavior Inventory |
| RFC | Reduced folate carrier |
| RNA | Ribonucleic acid |
| SAH | S-adenosyl-L-homocysteine |
| SAHH | S-adenosylhomocysteine hydrolase |
| SAM | S-adenosyl-L-methionine |
| SHMT | Serine hydroxymethyltransferase |
| SNP | Single nucleotide polymorphism |
| TCN2 | Transcobalamin II |
| THF | Tetrahydrofolate |
| VABSs | Vineland Adaptive Behavior Scales |
| SD | Standard Deviation |
| USA | United States of America |
| SUNY | State University of New York |
References
- Association, A.P. Diagnostic and Statistical Manual of Mental Disorders (DSM-5®), 5th ed.; American Psychiatric Association Publishing: Washington, DC, USA, 2013. [Google Scholar]
- Shaw, K.A.; Williams, S.; Patrick, M.E.; Valencia-Prado, M.; Durkin, M.S.; Howerton, E.M.; Ladd-Acosta, C.M.; Pas, E.T.; Bakian, A.V.; Bartholomew, P.; et al. Prevalence and Early Identification of Autism Spectrum Disorder Among Children Aged 4 and 8 Years—Autism and Developmental Disabilities Monitoring Network, 16 Sites, United States, 2022. MMWR Surveill. Summ. 2025, 74, 1–22. [Google Scholar] [CrossRef]
- Maenner, M.J.; Warren, Z.; Williams, A.R.; Amoakohene, E.; Bakian, A.V.; Bilder, D.A.; Durkin, M.S.; Fitzgerald, R.T.; Furnier, S.M.; Hughes, M.M.; et al. Prevalence and Characteristics of Autism Spectrum Disorder Among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2020. MMWR Surveill. Summ. 2023, 72, 1–14. [Google Scholar] [CrossRef]
- Sandin, S.; Lichtenstein, P.; Kuja-Halkola, R.; Larsson, H.; Hultman, C.M.; Reichenberg, A. The familial risk of autism. JAMA 2014, 311, 1770–1777. [Google Scholar] [CrossRef]
- Schaefer, G.B.; Mendelsohn, N.J.; Professional, P.; Guidelines, C. Clinical genetics evaluation in identifying the etiology of autism spectrum disorders: 2013 guideline revisions. Genet. Med. 2013, 15, 399–407. [Google Scholar] [CrossRef]
- O’Roak, B.J.; Stessman, H.A.; Boyle, E.A.; Witherspoon, K.T.; Martin, B.; Lee, C.; Vives, L.; Baker, C.; Hiatt, J.B.; Nickerson, D.A.; et al. Recurrent de novo mutations implicate novel genes underlying simplex autism risk. Nat. Commun. 2014, 5, 5595. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Li, K.; Wang, Z.; Wang, X.; Li, B.; Zhao, G.; Fang, Z.; Ling, Z.; Luo, T.; et al. Functional relationships between recessive inherited genes and genes with de novo variants in autism spectrum disorder. Mol. Autism 2020, 11, 75. [Google Scholar] [CrossRef]
- Bar, O.; Vahey, E.; Mintz, M.; Frye, R.E.; Boles, R.G. Reanalysis of Trio Whole-Genome Sequencing Data Doubles the Yield in Autism Spectrum Disorder: De Novo Variants Present in Half. Int. J. Mol. Sci. 2024, 25, 1192. [Google Scholar] [CrossRef]
- Frye, R.E.; McCarty, P.J.; Werner, B.A.; Rose, S.; Scheck, A.C. Bioenergetic signatures of neurodevelopmental regression. Front. Physiol. 2024, 15, 1306038. [Google Scholar] [CrossRef]
- Frye, R.E.; Rincon, N.; McCarty, P.J.; Brister, D.; Scheck, A.C.; Rossignol, D.A. Biomarkers of mitochondrial dysfunction in autism spectrum disorder: A systematic review and meta-analysis. Neurobiol. Dis. 2024, 197, 106520. [Google Scholar] [CrossRef]
- Rose, S.; Niyazov, D.M.; Rossignol, D.A.; Goldenthal, M.; Kahler, S.G.; Frye, R.E. Clinical and Molecular Characteristics of Mitochondrial Dysfunction in Autism Spectrum Disorder. Mol. Diagn. Ther. 2018, 22, 571–593. [Google Scholar] [CrossRef]
- Hollowood, K.; Melnyk, S.; Pavliv, O.; Evans, T.; Sides, A.; Schmidt, R.J.; Hertz-Picciotto, I.; Elms, W.; Guerrero, E.; Kruger, U.; et al. Maternal metabolic profile predicts high or low risk of an autism pregnancy outcome. Res. Autism Spectr. Disord. 2018, 56, 72–82. [Google Scholar] [CrossRef]
- Zhu, Y.; Mordaunt, C.E.; Durbin-Johnson, B.P.; Caudill, M.A.; Malysheva, O.V.; Miller, J.W.; Green, R.; James, S.J.; Melnyk, S.B.; Fallin, M.D.; et al. Expression Changes in Epigenetic Gene Pathways Associated With One-Carbon Nutritional Metabolites in Maternal Blood From Pregnancies Resulting in Autism and Non-Typical Neurodevelopment. Autism Res. 2021, 14, 11–28. [Google Scholar] [CrossRef]
- Howsmon, D.P.; Kruger, U.; Melnyk, S.; James, S.J.; Hahn, J. Classification and adaptive behavior prediction of children with autism spectrum disorder based upon multivariate data analysis of markers of oxidative stress and DNA methylation. PLoS Comput. Biol. 2017, 13, e1005385. [Google Scholar] [CrossRef]
- Gardner, R.M.; Brynge, M.; Sjöqvist, H.; Dalman, C.; Karlsson, H. Maternal immune activation and autism in the offspring-what is the evidence for causation? Biol. Psychiatry 2024, 97, 1127–1138. [Google Scholar] [CrossRef]
- Hughes, H.K.; Moreno, R.J.; Ashwood, P. Innate immune dysfunction and neuroinflammation in autism spectrum disorder (ASD). Brain Behav. Immun. 2023, 108, 245–254. [Google Scholar] [CrossRef]
- Hallmayer, J.; Cleveland, S.; Torres, A.; Phillips, J.; Cohen, B.; Torigoe, T.; Miller, J.; Fedele, A.; Collins, J.; Smith, K.; et al. Genetic heritability and shared environmental factors among twin pairs with autism. Arch. Gen. Psychiatry 2011, 68, 1095–1102. [Google Scholar] [CrossRef]
- Frye, R.E.; Cakir, J.; Rose, S.; Palmer, R.F.; Austin, C.; Curtin, P. Physiological mediators of prenatal environmental influences in autism spectrum disorder. Bioessays 2021, 43, e2000307. [Google Scholar] [CrossRef]
- Frye, R.E.; Cakir, J.; Rose, S.; Palmer, R.F.; Austin, C.; Curtin, P.; Arora, M. Mitochondria May Mediate Prenatal Environmental Influences in Autism Spectrum Disorder. J. Pers. Med. 2021, 11, 218. [Google Scholar] [CrossRef]
- Rossignol, D.A.; Genuis, S.J.; Frye, R.E. Environmental toxicants and autism spectrum disorders: A systematic review. Transl. Psychiatry 2014, 4, e360. [Google Scholar] [CrossRef]
- Greenblatt, J.M.; Huffman, L.C.; Reiss, A.L. Folic acid in neurodevelopment and child psychiatry. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 1994, 18, 647–660. [Google Scholar] [CrossRef]
- Black, M.M. Effects of vitamin B12 and folate deficiency on brain development in children. Food Nutr. Bull. 2008, 29, S126–S131. [Google Scholar] [CrossRef]
- Nagarajan, R.P.; Hogart, A.R.; Gwye, Y.; Martin, M.R.; LaSalle, J.M. Reduced MeCP2 expression is frequent in autism frontal cortex and correlates with aberrant MECP2 promoter methylation. Epigenetics. Off. J. DNA Methylation Soc. 2006, 1, e1–e11. [Google Scholar] [CrossRef]
- Lintas, C.; Sacco, R.; Persico, A.M. Differential methylation at the RELN gene promoter in temporal cortex from autistic and typically developing post-puberal subjects. J. Neurodev. Disord. 2016, 8, 18. [Google Scholar] [CrossRef]
- Nardone, S.; Sams, D.S.; Zito, A.; Reuveni, E.; Elliott, E. Dysregulation of Cortical Neuron DNA Methylation Profile in Autism Spectrum Disorder. Cereb. Cortex 2017, 27, 5739–5754. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E.; Slattery, J.C.; Quadros, E.V. Folate metabolism abnormalities in autism: Potential biomarkers. Biomark. Med. 2017, 11, 687–699. [Google Scholar] [CrossRef]
- Devlin, B.; Scherer, S.W. Genetic architecture in autism spectrum disorder. Curr. Opin. Genet. Dev. 2012, 22, 229–237. [Google Scholar] [CrossRef]
- Neale, B.M.; Kou, Y.; Liu, L.; Ma’ayan, A.; Samocha, K.E.; Sabo, A.; Lin, C.F.; Stevens, C.; Wang, L.S.; Makarov, V.; et al. Patterns and rates of exonic de novo mutations in autism spectrum disorders. Nature 2012, 485, 242–245. [Google Scholar] [CrossRef]
- Sanders, S.J.; Murtha, M.T.; Gupta, A.R.; Murdoch, J.D.; Raubeson, M.J.; Willsey, A.J.; Ercan-Sencicek, A.G.; DiLullo, N.M.; Parikshak, N.N.; Stein, J.L.; et al. De novo mutations revealed by whole-exome sequencing are strongly associated with autism. Nature 2012, 485, 237–241. [Google Scholar] [CrossRef]
- Frye, R.E. Central tetrahydrobiopterin concentration in neurodevelopmental disorders. Front. Neurosci. 2010, 4, 52. [Google Scholar] [CrossRef][Green Version]
- Frye, R.E.; Huffman, L.C.; Elliott, G.R. Tetrahydrobiopterin as a novel therapeutic intervention for autism. Neurotherapeutics 2010, 7, 241–249. [Google Scholar] [CrossRef]
- Frye, R.E.; DeLatorre, R.; Taylor, H.B.; Slattery, J.; Melnyk, S.; Chowdhury, N.; James, S.J. Metabolic effects of sapropterin treatment in autism spectrum disorder: A preliminary study. Transl. Psychiatry 2013, 3, e237. [Google Scholar] [CrossRef]
- James, S.J.; Melnyk, S.; Jernigan, S.; Pavliv, O.; Trusty, T.; Lehman, S.; Seidel, L.; Gaylor, D.W.; Cleves, M.A. A functional polymorphism in the reduced folate carrier gene and DNA hypomethylation in mothers of children with autism. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2010, 153B, 1209–1220. [Google Scholar] [CrossRef]
- Orenbuch, A.; Fortis, K.; Taesuwan, S.; Yaffe, R.; Caudill, M.A.; Golan, H.M. Prenatal Nutritional Intervention Reduces Autistic-Like Behavior Rates Among Mthfr-Deficient Mice. Front. Neurosci. 2019, 13, 383. [Google Scholar] [CrossRef]
- Ramaekers, V.T.; Rothenberg, S.P.; Sequeira, J.M.; Opladen, T.; Blau, N.; Quadros, E.V.; Selhub, J. Autoantibodies to folate receptors in the cerebral folate deficiency syndrome. N. Engl. J. Med. 2005, 352, 1985–1991. [Google Scholar] [CrossRef]
- Ramaekers, V.T.; Blau, N.; Sequeira, J.M.; Nassogne, M.C.; Quadros, E.V. Folate receptor autoimmunity and cerebral folate deficiency in low-functioning autism with neurological deficits. Neuropediatrics 2007, 38, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Moretti, P.; Peters, S.U.; Del Gaudio, D.; Sahoo, T.; Hyland, K.; Bottiglieri, T.; Hopkin, R.J.; Peach, E.; Min, S.H.; Goldman, D.; et al. Brief report: Autistic symptoms, developmental regression, mental retardation, epilepsy, and dyskinesias in CNS folate deficiency. J. Autism Dev. Disord. 2008, 38, 1170–1177. [Google Scholar] [CrossRef]
- Frye, R.E.; Sequeira, J.M.; Quadros, E.V.; James, S.J.; Rossignol, D.A. Cerebral folate receptor autoantibodies in autism spectrum disorder. Mol. Psychiatry 2013, 18, 369–381. [Google Scholar] [CrossRef]
- Rossignol, D.A.; Frye, R.E. Cerebral Folate Deficiency, Folate Receptor Alpha Autoantibodies and Leucovorin (Folinic Acid) Treatment in Autism Spectrum Disorders: A Systematic Review and Meta-Analysis. J. Pers. Med. 2021, 11, 1141. [Google Scholar] [CrossRef]
- Batebi, N.; Moghaddam, H.S.; Hasanzadeh, A.; Fakour, Y.; Mohammadi, M.R.; Akhondzadeh, S. Folinic Acid as Adjunctive Therapy in Treatment of Inappropriate Speech in Children with Autism: A Double-Blind and Placebo-Controlled Randomized Trial. Child. Psychiatry Hum. Dev. 2021, 52, 928–938. [Google Scholar] [CrossRef]
- Panda, P.K.; Sharawat, I.K.; Saha, S.; Gupta, D.; Palayullakandi, A.; Meena, K. Efficacy of oral folinic acid supplementation in children with autism spectrum disorder: A randomized double-blind, placebo-controlled trial. Eur. J. Pediatr. 2024, 183, 4827–4835. [Google Scholar] [CrossRef]
- Renard, E.; Leheup, B.; Guéant-Rodriguez, R.M.; Oussalah, A.; Quadros, E.V.; Guéant, J.L. Folinic acid improves the score of Autism in the EFFET placebo-controlled randomized trial. Biochimie 2020, 173, 57–61. [Google Scholar] [CrossRef]
- Zhang, C.; Chen, Y.; Hou, F.; Li, Y.; Wang, W.; Guo, L.; Zhang, C.; Li, L.; Lu, C. Safety and Efficacy of High-Dose Folinic Acid in Children with Autism: The Impact of Folate Metabolism Gene Polymorphisms. Nutrients 2025, 17, 1602. [Google Scholar] [CrossRef]
- Frye, R.E. The Folate Fix: Exploring the Role of Folate in Autism and Neurodevelopmental Disorders; Metabolic Learning Resource, LLC: Phoenix, AZ, USA, 2025. [Google Scholar]
- Quadros, E.V.; Sequeira, J.M.; Brown, W.T.; Mevs, C.; Marchi, E.; Flory, M.; Jenkins, E.C.; Velinov, M.T.; Cohen, I.L. Folate receptor autoantibodies are prevalent in children diagnosed with autism spectrum disorder, their normal siblings and parents. Autism Res. 2018, 11, 707–712. [Google Scholar] [CrossRef]
- Sebat, J.; Lakshmi, B.; Malhotra, D.; Troge, J.; Lese-Martin, C.; Walsh, T.; Yamrom, B.; Yoon, S.; Krasnitz, A.; Kendall, J.; et al. Strong association of de novo copy number mutations with autism. Science 2007, 316, 445–449. [Google Scholar] [CrossRef]
- Yuen, R.K.; Thiruvahindrapuram, B.; Merico, D.; Walker, S.; Tammimies, K.; Hoang, N.; Chrysler, C.; Nalpathamkalam, T.; Pellecchia, G.; Liu, Y.; et al. Whole-genome sequencing of quartet families with autism spectrum disorder. Nat. Med. 2015, 21, 185–191. [Google Scholar] [CrossRef]
- Gardener, H.; Spiegelman, D.; Buka, S.L. Perinatal and neonatal risk factors for autism: A comprehensive meta-analysis. Pediatrics 2011, 128, 344–355. [Google Scholar] [CrossRef]
- Karmel, B.Z.; Gardner, J.M.; Meade, L.S.; Cohen, I.L.; London, E.; Flory, M.J.; Lennon, E.M.; Miroshnichenko, I.; Rabinowitz, S.; Parab, S.; et al. Early medical and behavioral characteristics of NICU infants later classified with ASD. Pediatrics 2010, 126, 457–467. [Google Scholar] [CrossRef]
- Cohen, I.L.; Gardner, J.M.; Karmel, B.Z.; Phan, H.T.; Kittler, P.; Gomez, T.R.; Gonzalez, M.G.; Lennon, E.M.; Parab, S.; Barone, A. Neonatal brainstem function and 4-month arousal-modulated attention are jointly associated with autism. Autism Res. 2013, 6, 11–22. [Google Scholar] [CrossRef]
- Ozonoff, S.; Young, G.S.; Bradshaw, J.; Charman, T.; Chawarska, K.; Iverson, J.M.; Klaiman, C.; Landa, R.J.; McDonald, N.; Messinger, D.; et al. Familial Recurrence of Autism: Updates From the Baby Siblings Research Consortium. Pediatrics 2024, 154, e2023065297. [Google Scholar] [CrossRef]
- McDonald, N.M.; Senturk, D.; Scheffler, A.; Brian, J.A.; Carver, L.J.; Charman, T.; Chawarska, K.; Curtin, S.; Hertz-Piccioto, I.; Jones, E.J.H.; et al. Developmental Trajectories of Infants With Multiplex Family Risk for Autism: A Baby Siblings Research Consortium Study. JAMA Neurol. 2020, 77, 73–81. [Google Scholar] [CrossRef]
- Virkud, Y.V.; Todd, R.D.; Abbacchi, A.M.; Zhang, Y.; Constantino, J.N. Familial aggregation of quantitative autistic traits in multiplex versus simplex autism. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2009, 150B, 328–334. [Google Scholar] [CrossRef]
- Gerdts, J.A.; Bernier, R.; Dawson, G.; Estes, A. The broader autism phenotype in simplex and multiplex families. J. Autism Dev. Disord. 2013, 43, 1597–1605. [Google Scholar] [CrossRef]
- Oerlemans, A.M.; van der Meer, J.M.; van Steijn, D.J.; de Ruiter, S.W.; de Bruijn, Y.G.; de Sonneville, L.M.; Buitelaar, J.K.; Rommelse, N.N. Recognition of facial emotion and affective prosody in children with ASD (+ADHD) and their unaffected siblings. Eur. Child Adolesc. Psychiatry 2014, 23, 257–271. [Google Scholar] [CrossRef]
- Oerlemans, A.M.; Hartman, C.A.; Franke, B.; Buitelaar, J.K.; Rommelse, N.N. Does the cognitive architecture of simplex and multiplex ASD families differ? J. Autism Dev. Disord. 2016, 46, 489–501. [Google Scholar] [CrossRef]
- Qin, X.Y.; Ha, S.Y.; Chen, L.; Zhang, T.; Li, M.Q. Recent Advances in Folates and Autoantibodies against Folate Receptors in Early Pregnancy and Miscarriage. Nutrients 2023, 15, 4882. [Google Scholar] [CrossRef]
- Rothenberg, S.P.; da Costa, M.P.; Sequeira, J.M.; Cracco, J.; Roberts, J.L.; Weedon, J.; Quadros, E.V. Autoantibodies against folate receptors in women with a pregnancy complicated by a neural-tube defect. N. Engl. J. Med. 2004, 350, 134–142. [Google Scholar] [CrossRef]
- Cabrera, R.M.; Shaw, G.M.; Ballard, J.L.; Carmichael, S.L.; Yang, W.; Lammer, E.J.; Finnell, R.H. Autoantibodies to folate receptor during pregnancy and neural tube defect risk. J. Reprod. Immunol. 2008, 79, 85–92. [Google Scholar] [CrossRef]
- Boyles, A.L.; Ballard, J.L.; Gorman, E.B.; McConnaughey, D.R.; Cabrera, R.M.; Wilcox, A.J.; Lie, R.T.; Finnell, R.H. Association between inhibited binding of folic acid to folate receptor alpha in maternal serum and folate-related birth defects in Norway. Hum. Reprod. 2011, 26, 2232–2238. [Google Scholar] [CrossRef]
- Vo, H.D.; Sequeira, J.M.; Quadros, E.V.; Schwarz, S.M.; Perenyi, A.R. The role of folate receptor autoantibodies in preterm birth. Nutrition 2015, 31, 1224–1227. [Google Scholar] [CrossRef]
- Berrocal-Zaragoza, M.I.; Fernandez-Ballart, J.D.; Murphy, M.M.; Cavallé-Busquets, P.; Sequeira, J.M.; Quadros, E.V. Association between blocking folate receptor autoantibodies and subfertility. Fertil. Steril. 2009, 91, 1518–1521. [Google Scholar] [CrossRef][Green Version]
- Malm, J.; Birn, H.; Frohm, B.; Hansen, S.I.; Høier-Madsen, M.; Holm, J. A minor fraction of a high-affinity folate binding protein from the epididymis is associated with membranous vesicles and spermatozoa in human semen. Int. J. Androl. 2005, 28, 267–274. [Google Scholar] [CrossRef]
- Holm, J.; Hansen, S.I. Characterization of soluble folate receptors (folate binding proteins) in humans. Biological roles and clinical potentials in infection and malignancy. Biochim. Biophys. Acta Proteins Proteom. 2020, 1868, 140466. [Google Scholar] [CrossRef]
- Díaz-Fuster, L.; Sáez-Espinosa, P.; Moya, I.; Peinado, I.; Gómez-Torres, M.J. Updating the Role of JUNO and Factors Involved in Its Function during Fertilization. Cells Tissues Organs 2025, 1, 1–16. [Google Scholar] [CrossRef]
- Gurugubelli, K.R.; Ballambattu, V.B. Perspectives on folate with special reference to epigenetics and neural tube defects. Reprod. Toxicol. 2024, 125, 108576. [Google Scholar] [CrossRef]
- Caffrey, A.; Lamers, Y.; Murphy, M.M.; Letourneau, N.; Irwin, R.E.; Pentieva, K.; Ward, M.; Tan, A.; Rojas-Gómez, A.; Santos-Calderón, L.A.; et al. Epigenetic effects of folate and related B vitamins on brain health throughout life: Scientific substantiation and translation of the evidence for health improvement strategies. Nutr. Bull. 2023, 48, 267–277. [Google Scholar] [CrossRef]
- Lintas, C. Linking genetics to epigenetics: The role of folate and folate-related pathways in neurodevelopmental disorders. Clin. Genet. 2019, 95, 241–252. [Google Scholar] [CrossRef]
- Wang, S.; Zeng, Y.; Pei, P.; He, X.; Liu, F.; Zhang, T. Abnormal transcriptome-wide DNA demethylation induced by folate deficiency causes neural tube defects. Front. Genet. 2022, 13, 987210. [Google Scholar] [CrossRef]
- Khaledi, F.; Ghasemi, S. A Review on Epigenetic Effects of Environmental Factors Causing and Inhibiting Cancer. Curr. Mol. Med. 2022, 22, 8–24. [Google Scholar] [CrossRef] [PubMed]
- Tando, Y.; Matsui, Y. Inheritance of environment-induced phenotypic changes through epigenetic mechanisms. Environ. Epigenetics. 2023, 9, dvad008. [Google Scholar] [CrossRef] [PubMed]
- Gaskins, A.J.; Mínguez-Alarcón, L.; Fong, K.C.; Abu Awad, Y.; Di, Q.; Chavarro, J.E.; Ford, J.B.; Coull, B.A.; Schwartz, J.; Kloog, I.; et al. Supplemental Folate and the Relationship Between Traffic-Related Air Pollution and Livebirth Among Women Undergoing Assisted Reproduction. Am. J. Epidemiol. 2019, 188, 1595–1604. [Google Scholar] [CrossRef]
- Dickerson, A.S.; Schmidt, R.J. Invited Perspective: Protect and Serve—The Potential Role of Folate in Lead Risk Reduction. Environ. Health Perspect. 2024, 132, 101302. [Google Scholar] [CrossRef]
- Greenberg, J.A.; Bell, S.J.; Guan, Y.; Yu, Y.H. Folic Acid supplementation and pregnancy: More than just neural tube defect prevention. Rev. Obstet. Gynecol. 2011, 4, 52–59. [Google Scholar]
- Liu, X.; Zou, M.; Sun, C.; Wu, L.; Chen, W.X. Prenatal Folic Acid Supplements and Offspring’s Autism Spectrum Disorder: A Meta-analysis and Meta-regression. J. Autism Dev. Disord. 2022, 52, 522–539. [Google Scholar] [CrossRef]
- Schmidt, R.J.; Kogan, V.; Shelton, J.F.; Delwiche, L.; Hansen, R.L.; Ozonoff, S.; Ma, C.C.; McCanlies, E.C.; Bennett, D.H.; Hertz-Picciotto, I.; et al. Combined Prenatal Pesticide Exposure and Folic Acid Intake in Relation to Autism Spectrum Disorder. Environ. Health Perspect. 2017, 125, 097007. [Google Scholar] [CrossRef]
- Hoxha, B.; Hoxha, M.; Domi, E.; Gervasoni, J.; Persichilli, S.; Malaj, V.; Zappacosta, B. Folic Acid and Autism: A Systematic Review of the Current State of Knowledge. Cells 2021, 10, 1976. [Google Scholar] [CrossRef] [PubMed]
- Naninck, E.F.G.; Stijger, P.C.; Brouwer-Brolsma, E.M. The Importance of Maternal Folate Status for Brain Development and Function of Offspring. Adv. Nutr. 2019, 10, 502–519. [Google Scholar] [CrossRef]
- Virdi, S.; Jadavji, N.M. The Impact of Maternal Folates on Brain Development and Function after Birth. Metabolites 2022, 12, 876. [Google Scholar] [CrossRef] [PubMed]
- Sequeira, J.M.; Desai, A.; Berrocal-Zaragoza, M.I.; Murphy, M.M.; Fernandez-Ballart, J.D.; Quadros, E.V. Exposure to Folate Receptor Alpha Antibodies during Gestation and Weaning Leads to Severe Behavioral Deficits in Rats: A Pilot Study. PLoS ONE 2016, 11, e0152249. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.; Sequeira, J.M.; Quadros, E.V. Prevention of behavioral deficits in rats exposed to folate receptor antibodies: Implication in autism. Mol. Psychiatry 2017, 22, 1291–1297. [Google Scholar] [CrossRef]
- Bobrowski-Khoury, N.; Sequeira, J.M.; Arning, E.; Bottiglieri, T.; Quadros, E.V. Absorption and Tissue Distribution of Folate Forms in Rats: Indications for Specific Folate Form Supplementation during Pregnancy. Nutrients 2022, 14, 2397. [Google Scholar] [CrossRef]
- Ramaekers, V.T.; Sequeira, J.M.; DiDuca, M.; Vrancken, G.; Thomas, A.; Philippe, C.; Peters, M.; Jadot, A.; Quadros, E.V. Improving Outcome in Infantile Autism with Folate Receptor Autoimmunity and Nutritional Derangements: A Self-Controlled Trial. Autism Res. Treat. 2019, 2019, 7486431. [Google Scholar] [CrossRef]
- Shapira, I.; Sequeira, J.M.; Quadros, E.V. Folate receptor autoantibodies in pregnancy related complications. Birth Defects Res. A Clin. Mol. Teratol. 2015, 103, 1028–1030. [Google Scholar] [CrossRef]
- Patanwala, I.; King, M.J.; Barrett, D.A.; Rose, J.; Jackson, R.; Hudson, M.; Philo, M.; Dainty, J.R.; Wright, A.J.; Finglas, P.M.; et al. Folic acid handling by the human gut: Implications for food fortification and supplementation. Am. J. Clin. Nutr. 2014, 100, 593–599. [Google Scholar] [CrossRef]
- Wright, A.J.A.; Dainty, J.R.; Finglas, P.M. Folic acid metabolism in human subjects revisited: Potential implications for proposed mandatory folic acid fortification in the UK. Br. J. Nutr. 2007, 98, 667–675. [Google Scholar] [CrossRef]
- Chen, P.; Tang, L.; Song, Y.; Wang, B.; Qin, X.; Zhang, N.; Wei, Y.; Xu, X.; Zhou, Z.; He, Q.; et al. Association of folic acid dosage with circulating unmetabolized folic acid in Chinese adults with H-type hypertension: A multicenter, double-blind, randomized controlled trial. Front. Nutr. 2023, 10, 1191610. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.S.; Cait, A.; White, R.M.; Arabshahi, H.J.; O’Sullivan, D.; Gasser, O. MR1-dependence of unmetabolized folic acid side-effects. Front. Immunol. 2022, 13, 946713. [Google Scholar] [CrossRef] [PubMed]
- Tam, C.; O’Connor, D.; Koren, G. Circulating unmetabolized folic Acid: Relationship to folate status and effect of supplementation. Obstet. Gynecol. Int. 2012, 2012, 485179. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Pervasive Developmental Disorders. In Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association: Washington, DC, USA, 1994; pp. 65–78. [Google Scholar]
- American Psychiatric Association. Pervasive Developmental Disorders. In Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; Text revised; American Psychiatric Association: Washington, DC, USA, 2000; pp. 69–84. [Google Scholar]
- American Psychiatric Association. Neurodevelopmental Disorders. In Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013; pp. 31–86. [Google Scholar]
- Sequeira, J.M.; Ramaekers, V.T.; Quadros, E.V. The diagnostic utility of folate receptor autoantibodies in blood. Clin. Chem. Lab. Med. 2013, 51, 545–554. [Google Scholar] [CrossRef]
- Griffiths, R. The Abilities of Young Children: A Comprehensive System of Mental Measurement for the First Eight Years of Life (Revised edition); ARICD. The Test Agency Limited.: Bucks, UK, 1984. [Google Scholar]
- Cohen, I.L.; Sudhalter, V. The PDD Behavior Inventory; Psychological Assessment Resources, Inc.: Lutz, FL, USA, 2005. [Google Scholar]
- Pine, E.; Luby, J.; Abbacchi, A.; Constantino, J.N. Quantitative assessment of autistic symptomatology in preschoolers. Autism Int. J. Res. Pract. 2006, 10, 344–352. [Google Scholar] [CrossRef]


| N | Sex [% Male (N)] | Age Range (Years) | Average Age (SD) | |
|---|---|---|---|---|
| ASD | 82 | 79% (65) | 1.6 to 15 | 5.18 (2.6) |
| Typically Developing Siblings | 53 | 42% (22) | 2 to 17 | 6.7 (4.2) |
| Parents of Children with ASD | 135 | 48% (65) | 15 to 54 | 32.4 (5.8) |
| Unrelated, non-ASD controls | 52 | 46% (24) | 3 to 17 | 11.8 (4.0) |
| Mom | Dad | |||
|---|---|---|---|---|
| Griffth’s Mental Development Scales | ||||
| Neg N = 18 | Pos N = 29 | Neg N = 10 | Pos N = 32 | |
| Total Scale (GQ) | 69.1 (5.2) | 75.4 (3.9) | 85.2 (8.5) * | 70.4 (3.4) |
| Locomotor Development | 83.9 (3.6) | 86.6 (2.5) | 93.5 (6.8) * | 83.4 (1.9) |
| Personal Social Development | 64.6 (5.2) | 65.3 (3.9) | 74.0 (8.3) | 63.0 (3.2) |
| Hearing and Speech | 54.8 (6.9) | 61.3 (6.9) | 80.8 (15.0) ** | 51.9 (27.7) |
| Hand and Eye Coordination | 68.3 (5.7) | 76.9 (4.1) | 85.1 (7.8) * | 71.2 (3.8) |
| Performance | 80.9 (6.4) | 88.2 (4.0) | 96.5 (7.4) | 83.9 (4.2) |
| Practical Reasoning | 63.2 (6.3) | 69.0 (5.2) | 81.2 (11.0) * | 63.3 (4.2) |
| PDD (Pervasive Developmental Disorder) Behavior Inventory | ||||
| Neg N = 41 | Pos N = 63 | Neg N = 25 | Pos N = 72 | |
| Autism Composite | 49.1 (1.7) | 51.0 (1.3) | 45.9 (1.8) * | 51.3 (1.2) |
| Sensory/Perceptual Approach Behaviors | 51.4 (1.5) | 48.7 (1.2) | 46.7 (1.7) | 50.4 (1.2) |
| Ritualisms/Resistance to Change | 48.3 (1.3) | 50.3 (1.3) | 45.9 (1.6) | 49.6 (1.2) |
| Social Pragmatic Problems | 47.0 (1.6) ** | 53.1 (1.1) | 48.9 (1.7) | 51.1 (1.2) |
| Semantic/Pragmatic Problems | 51.5 (2.1) | 48.8 (1.1) | 49.7 (2.2) | 49.2 (1.1) |
| Arousal Regulation Problems | 52.3 (1.5) | 50.3 (1.3) | 49.9 (1.8) | 51.1 (1.2) |
| Specific Fears | 48.8 (1.5) | 50.7 (1.2) | 47.7 (2.1) | 50.7 (1.1) |
| Aggressiveness | 48.8 (1.3) | 50.9 (1.6) | 48.5 (2.5) | 49.5 (1.1) |
| Repetitive, Ritualistic, and Pragmatic Prob Comp | 50.7 (1.8) | 50.5 (1.2) | 48.4 (1.9) | 50.5 (1.2) |
| Approach/Withdrawal Problems Composite | 50.6 (1.9) | 51.3 (1.3) | 49.4 (2.3) | 50.7 (1.3) |
| Social Approach Behaviors | 50.0 (1.8) | 48.4 (1.2) | 51.7 (2.0) | 47.3 (1.2) |
| Expressive Language | 50.1 (1.6) | 49.1 (1.2) | 53.4 (1.9) * | 47.8 (1.1) |
| Learning, Memory, and Receptive Language | 50.1 (1.6) | 49.6 (1.1) | 53.0 (2.0) * | 48.2 (1.0) |
| Expressive Social Comm Abilities Composite | 50.0 (1.8) | 49.6 (1.2) | 52.7 (1.9) * | 48.0 (1.2) |
| Receptive/Expressive Social Comm Composite | 50.0 (1.8) | 49.7 (1.2) | 52.7 (1.9) * | 48.0 (1.2) |
| Vineland Adaptive Behavior Scales | ||||
| Neg N = 29 | Pos N = 44 | Neg N = 19 | Pos N = 49 | |
| Communication | 71.0 (4.7) | 63.7 (3.3) | 77.5 (5.8) * | 63.4 (3.3) |
| Daily Living Skills | 62.0 (3.2) | 58.0 (2.2) | 69.5 (4.7) ** | 56.5 (1.7) |
| Socialization | 66.1 (3.3) | 60.3 (1.9) | 72.1 (4.5) ** | 59.3 (1.8) |
| Motor Skills | 70.2 (4.0) | 70.8 (2.4) | 79.6 (5.2) * | 67.7 (2.4) |
| Developmental Gross Motor Milestones | ||||
| Neg N = 15 | Pos N = 21 | Neg N = 10 | Pos N = 26 | |
| Age when sat without support | 6.7 (0.3) | 6.9 (0.3) | 6.5 (0.2) | 7.1 (0.3) |
| Age when walking without support | 13.4 (3.1) | 14.0 (0.7) | 12.8 (0.7) | 14.4 (0.7) |
| Factor | Impact on Folate Receptor Alpha Autoantibodies |
|---|---|
| Multiple Birth Pregnancy | Higher Binding in the Offspring |
| Multiplex ASD Families | Higher Blocking in the Offspring, Mother, and Father |
| Offspring Age | Lower Binding in the Offspring |
| Offspring | Higher Blocking and Binding than Parents Higher Binding than Typically Development Children from Non-ASD Families |
| ASD | Higher Blocking than Parents and Typically Developing Sibling |
| ASD Families | Higher Blocking than Typically Development Children from Non-ASD Families |
| Increased Maternal Age | Higher Blocking in the Offspring |
| Higher Maternal Blocking | Higher Offspring Blocking |
| Maternal FRAA Positive | Greater Pragmatic Problems |
| Increased Paternal Age | Higher Blocking in the Offspring |
| Paternal FRAA Positive | Lower general intelligence, locomotor activity, hearing and speech, hand and eye coordination, practical reasoning skills, learning, memory, receptive and expressive language and social communication, and daily living, socialization, and motor skills. Greater ASD symptoms |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frye, R.E.; Cohen, I.L.; Sequeira, J.M.; Hill, Z.; Espinoza, A.; Brown, W.T.; Mevs, C.; Marchi, E.; Flory, M.; Jenkins, E.C.; et al. Transgenerational Effects and Heritability of Folate Receptor Alpha Autoantibodies in Autism Spectrum Disorder. Int. J. Mol. Sci. 2025, 26, 8293. https://doi.org/10.3390/ijms26178293
Frye RE, Cohen IL, Sequeira JM, Hill Z, Espinoza A, Brown WT, Mevs C, Marchi E, Flory M, Jenkins EC, et al. Transgenerational Effects and Heritability of Folate Receptor Alpha Autoantibodies in Autism Spectrum Disorder. International Journal of Molecular Sciences. 2025; 26(17):8293. https://doi.org/10.3390/ijms26178293
Chicago/Turabian StyleFrye, Richard E., Ira L. Cohen, Jeffrey M. Sequeira, Zoe Hill, Alina Espinoza, W. Ted Brown, Clifford Mevs, Elaine Marchi, Michael Flory, Edmund C. Jenkins, and et al. 2025. "Transgenerational Effects and Heritability of Folate Receptor Alpha Autoantibodies in Autism Spectrum Disorder" International Journal of Molecular Sciences 26, no. 17: 8293. https://doi.org/10.3390/ijms26178293
APA StyleFrye, R. E., Cohen, I. L., Sequeira, J. M., Hill, Z., Espinoza, A., Brown, W. T., Mevs, C., Marchi, E., Flory, M., Jenkins, E. C., Velinov, M. T., & Quadros, E. V. (2025). Transgenerational Effects and Heritability of Folate Receptor Alpha Autoantibodies in Autism Spectrum Disorder. International Journal of Molecular Sciences, 26(17), 8293. https://doi.org/10.3390/ijms26178293






