Triglycerides as Determinants of Global Lipoprotein Derangement: Implications for Cardiovascular Prevention
Abstract
1. Introduction
2. Results
2.1. Demographic, Clinical, and Biochemical Characteristics
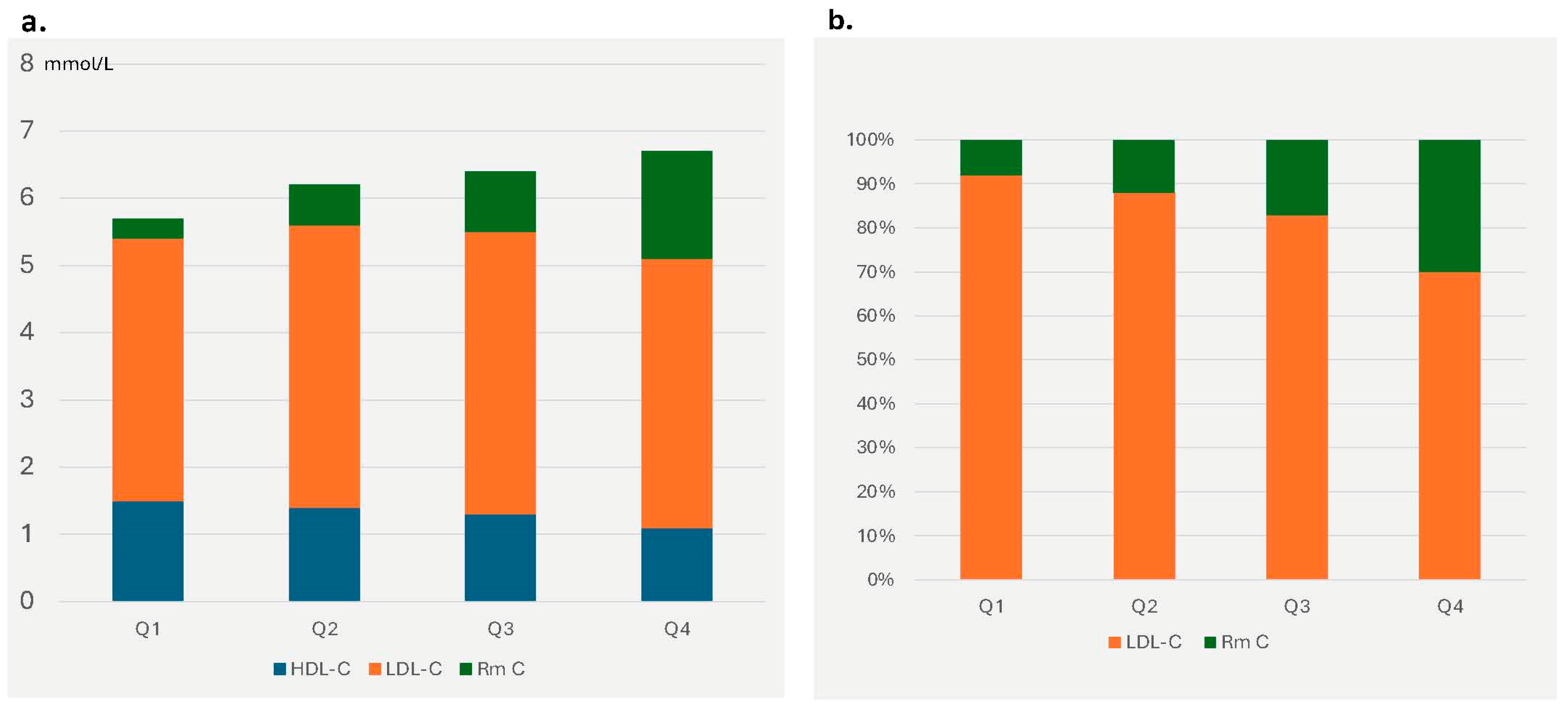
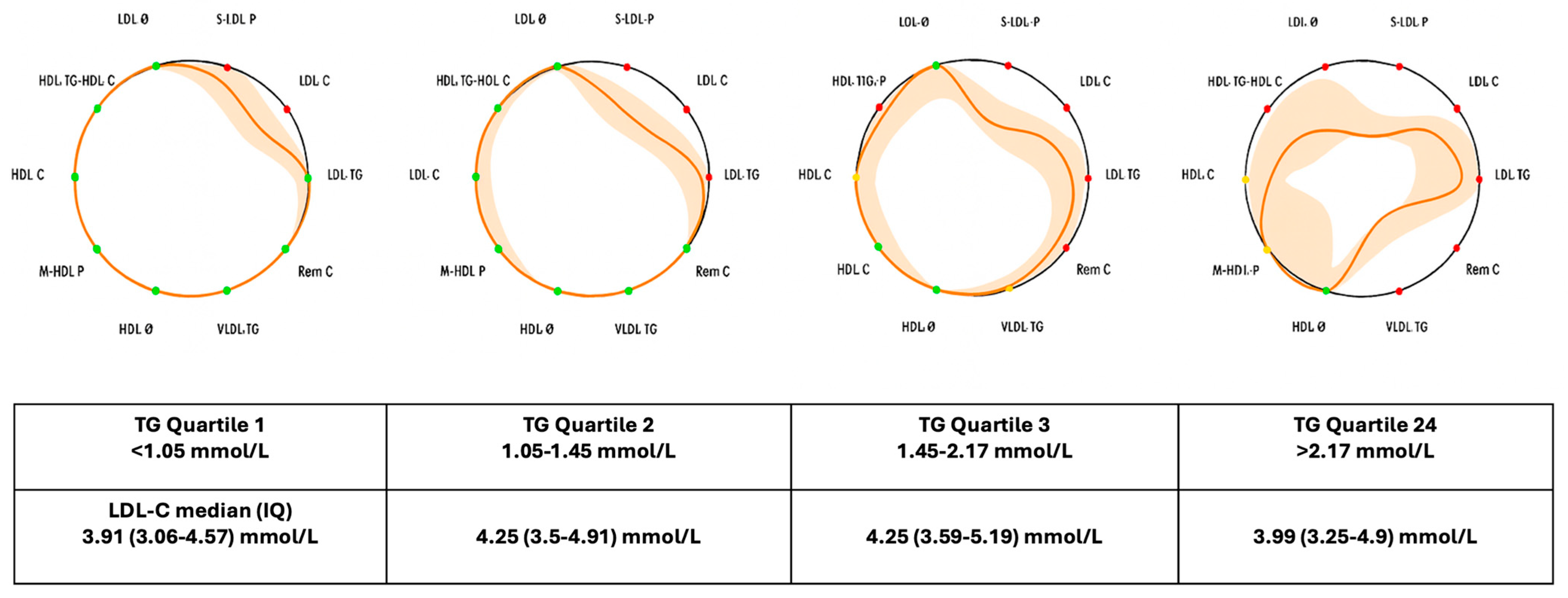
2.2. 1H–NMR Lipoprotein Data
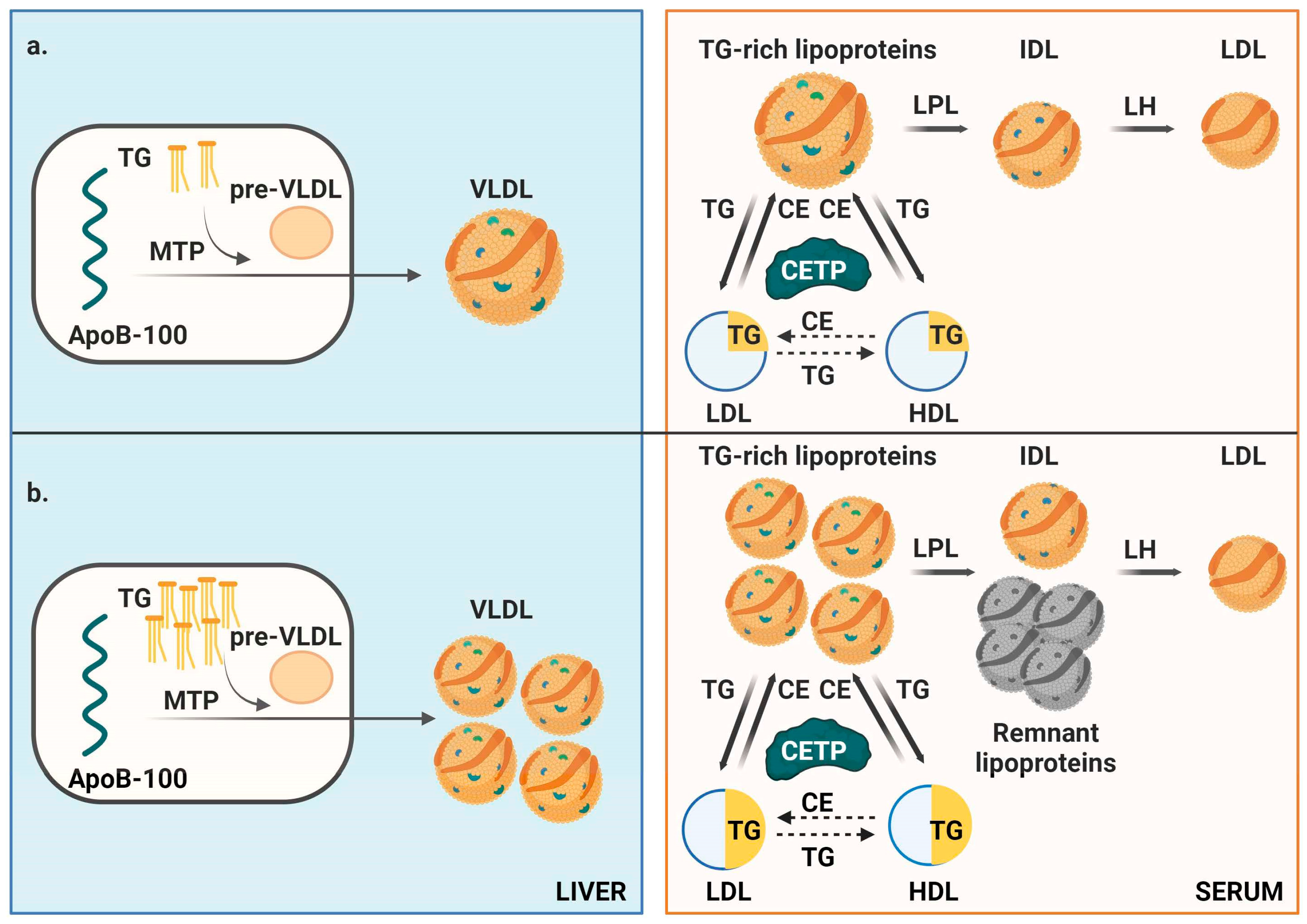
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. 1H–NMR Lipoprotein Profiling
4.3. 1H–NMR Data Reporting
4.4. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
Abbreviations
| apoB | Apolipoprotein B |
| ASCVD | Atherosclerotic Cardiovascular Disease |
| CETP | Cholesteryl Ester Transfer Protein |
| CV | Cardiovascular |
| D/O | Diabetes and/or Obesity |
| FA | Fatty Acid |
| FCS | Familial Chylomicronemia Syndrome |
| HDL | High-Density Lipoprotein |
| HDL–C | HDL Cholesterol |
| HDL–TG | HDL Triglycerides |
| HDL Ø | HDL particle diameter |
| M–HDL–P | Medium-size HDL particle number |
| HDL–TG/HDL–Ch | HDL Triglyceride-to-Cholesterol Ratio |
| IDL | Intermediate-Density Lipoprotein |
| IQR | Interquartile Range |
| LED | Longitudinal Eddy Current Delay |
| LLT | Lipid-Lowering Therapy |
| Lp(a) | Lipoprotein(a) |
| LS | Lipid Silhouette |
| LDL | Low-Density Lipoprotein |
| LDL–C | LDL Cholesterol |
| LDL–TG | LDL Triglycerides |
| LDL Ø | LDL particle diameter |
| M–HDL–P | Medium HDL Particles |
| NMR | Nuclear Magnetic Resonance |
| NOESY | Nuclear Overhauser Effect Spectroscopy |
| PPAR–a/g | peroxisome proliferator–activated receptor–α/γ agonist |
| Q1–Q4 | Quartiles 1 through 4 |
| Rm–Chol | Remnant Cholesterol |
| S–LDL–P | Smaller LDL Particles |
| TG | Triglycerides |
| TRL | Triglyceride-Rich Lipoprotein |
| VLDL | Very Low-Density Lipoprotein |
| VLDL–TG | VLDL Triglycerides |
| VLDL Ø | VLDL particle diameter |
References
- Borén, J.; Chapman, M.J.; Krauss, R.M.; Packard, C.J.; Bentzon, J.F.; Binder, C.J.; Daemen, M.J.; Demer, L.L.; Hegele, R.A.; Nicholls, S.J.; et al. Low–density lipoproteins cause atherosclerotic cardiovascular disease: Pathophysiological, genetic, and therapeutic insights: A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur. Heart J. 2020, 41, 2313–2330. [Google Scholar] [CrossRef]
- Gaba, P.; O’Donoghue, M.L.; Park, J.G.; Wiviott, S.D.; Atar, D.; Kuder, J.F.; Furtado, R.H.M.; Bohula, E.A.; Sever, P.S.; Keech, A.C.; et al. Association between achieved low–density lipoprotein cholesterol levels and long–term cardiovascular and safety outcomes: An analysis of FOURIER–OLE. Circulation 2023, 147, 1192–1203. [Google Scholar] [CrossRef]
- Hoogeveen, R.C.; Ballantyne, C.M. Residual cardiovascular risk at low LDL: Remnants, lipoprotein(a), and inflammation. Clin. Chem. 2021, 67, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Ference, B.A.; Braunwald, E.; Catapano, A.L. The LDL cumulative exposure hypothesis: Evidence and practical applications. Nat. Rev. Cardiol. 2024, 21, 701–716. [Google Scholar] [CrossRef] [PubMed]
- Nordestgaard, B.G.; Langsted, A. Lipoprotein(a) and cardiovascular disease. Lancet 2024, 404, 1255–1264. [Google Scholar] [CrossRef] [PubMed]
- Toth, P.P.; Banach, M. High triglycerides and low HDL cholesterol as markers of cardiovascular risk. J. Am. Coll. Cardiol. 2013, 62 (Suppl. 10), S92–S99. [Google Scholar]
- Lorenzatti, A.J.; Toth, P.P. New Perspectives on Atherogenic Dyslipidaemia and Cardiovascular Disease. Eur. Cardiol. 2020, 15, 1–9. [Google Scholar] [CrossRef]
- Nordestgaard, B.G.; Wootton, R.; Lewis, B. Selective retention of VLDL, IDL, and LDL in the arterial intima of genetically hyperlipidemic rabbits: Molecular size as a determinant of arterial entry. Arterioscler. Thromb. Vasc. Biol. 1995, 15, 534–542. [Google Scholar] [CrossRef]
- Castañer, O.; Pintó, X.; Subirana, I.; Amor, A.J.; Ros, E.; Hernáez, Á.; Martínez–González, M.Á.; Corella, D.; Salas–Salvadó, J.; Estruch, R.; et al. Remnant cholesterol, not LDL cholesterol, is associated with incident cardiovascular disease. J. Am. Coll. Cardiol. 2020, 76, 2712–2724. [Google Scholar] [CrossRef] [PubMed]
- Balling, M.; Afzal, S.; Varbo, A.; Langsted, A.; Davey Smith, G.; Nordestgaard, B.G. VLDL cholesterol accounts for one–half of the risk of myocardial infarction associated with apoB–containing lipoproteins. J. Am. Coll. Cardiol. 2020, 76, 2725–2735. [Google Scholar] [CrossRef] [PubMed]
- Johannesen, C.D.L.; Langsted, A.; Nordestgaard, B.G.; Mortensen, M.B. Excess apolipoprotein B and cardiovascular risk in women and men. J. Am. Coll. Cardiol. 2024, 83, 2262–2273. [Google Scholar] [CrossRef]
- ACCORD Study Group; Ginsberg, H.N.; Elam, M.B.; Lovato, L.C.; Crouse, J.R.; Leiter, L.A.; Linz, P.; Friedewald, W.T.; Buse, J.B.; Gerstein, H.C.; et al. Effects of combination lipid therapy in type 2 diabetes mellitus. N. Engl. J. Med. 2010, 362, 1563–1574, https://doi.org/10.1056/NEJMoa1001282; Erratum in N. Engl. J. Med. 2010, 362, 1748. [Google Scholar] [CrossRef]
- Landray, M.J.; Haynes, R.; Hopewell, J.C.; Parish, S.; Aung, T.; Tomson, J.; Wallendszus, K.; Craig, M.; Jiang, L.; Collins, R.; et al. HPS2–THRIVE Collaborative Group Effects of extended–release niacin with laropiprant in high–risk patients. N. Engl. J. Med. 2014, 371, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Barter, P.J.; Caulfield, M.; Eriksson, M.; Grundy, S.M.; Kastelein, J.J.P.; Komajda, M.; Lopez-Sendon, J.; Mosca, L.; Tardif, J.-C.; Waters, D.D.; et al. Effects of torcetrapib in patients at high risk for coronary events. N. Engl. J. Med. 2007, 357, 2109–2122. [Google Scholar] [CrossRef]
- Pradhan, A.D.; Glynn, R.J.; Fruchart, J.C.; MacFadyen, J.G.; Zaharris, E.S.; Everett, B.M.; Campbell, S.E.; Oshima, R.; Amarenco, P.; Blom, D.J.; et al. Triglyceride lowering with pemafibrate to reduce cardiovascular risk. N. Engl. J. Med. 2022, 387, 1923–1934. [Google Scholar] [CrossRef]
- Bhatt, D.L.; Steg, P.G.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Ketchum, S.B.; Doyle, R.T., Jr.; Juliano, R.A.; Jiao, L.; Granowitz, C.; et al. REDUCE–IT Investigators Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N. Engl. J. Med. 2019, 380, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Mallol, R.; Amigó, N.; Rodríguez, M.A.; Heras, M.; Vinaixa, M.; Plana, N.; Rock, E.; Ribalta, J.; Yanes, O.; Masana, L.; et al. Liposcale: A novel advanced lipoprotein test based on 2D diffusion–ordered 1H NMR spectroscopy. J. Lipid Res. 2015, 56, 737–746. [Google Scholar] [CrossRef]
- Lin, K.-H.; Amigó, N.; Ortiz, P.; Mallol, R.; Heras, M.; Correig, X.; Chalasani, N.P.; Gawrieh, S. The Athero–Contour: A novel tool for global and rapid assessment of atherogenic parameters. Clin. Investig. Arterioscler. 2024, 34, 192–200. [Google Scholar]
- Wadström, B.N.; Wulff, A.B.; Pedersen, K.M.; Nordestgaard, B.G. Small remnants versus large triglyceride–lipoproteins in risk of atherosclerotic cardiovascular disease. Clin. Chem. 2025, 71, 463–473. [Google Scholar] [CrossRef]
- Sniderman, A.D.; Thanassoulis, G.; Glavinovic, T.; Navar, A.M.; Pencina, M.; Catapano, A.L.; Ference, B.A. Apolipoprotein B particles and cardiovascular disease: A narrative review. JAMA Cardiol. 2019, 4, 1287–1295. [Google Scholar] [CrossRef]
- Girona, J.; Amigó, N.; Ibarretxe, D.; Plana, N.; Rodríguez, M.A.; Heras, M.; Masana, L.; Pintó, X.; Subirana, I. HDL triglycerides: A new marker of metabolic and cardiovascular risk. Int. J. Mol. Sci. 2019, 20, 3151. [Google Scholar] [CrossRef]
- Asztalos, B.F.; Tani, M.; Schaefer, E.J. Metabolic and functional relevance of HDL subspecies. Curr. Opin. Lipidol. 2011, 22, 176–185. [Google Scholar] [CrossRef]
- Amigó, N.; Mallol, R.; Heras, M.; Plana, N.; Rodríguez, M.A.; Correig, X.; Blanco-Vaca, F.; Escola-Gil, J.C.; Yanes, O.; Masana, L. Lipoprotein hydrophobic core lipids are partially extruded to the surface in smaller HDL: “Herniated” HDL, a common feature in diabetes. Sci. Rep. 2016, 6, 19249. [Google Scholar] [CrossRef]
- Laufs, U.; Parhofer, K.G.; Ginsberg, H.N.; Hegele, R.A. Clinical review on triglycerides. Eur. Heart J. 2020, 41, 99–109c. [Google Scholar] [CrossRef]
- Chen, J.; Fang, Z.; Luo, Q.; Wang, X.; Warda, M.; Das, A.; Luo, F. Unlocking the mysteries of VLDL: Exploring its production, intracellular trafficking, and metabolism as therapeutic targets. Lipids Health Dis. 2024, 23, 14. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, H.N.; Packard, C.J.; Chapman, M.J.; Borén, J.; Aguilar-Salinas, C.A.; Averna, M.; Ference, B.A.; Gaudet, D.; Hegele, R.A.; Kersten, S.; et al. Triglyceride–rich lipoproteins and their remnants: Metabolic insights, role in atherosclerotic cardiovascular disease, and emerging therapeutic strategies—A consensus statement from the European Atherosclerosis Society. Eur. Heart J. 2021, 42, 4791–4806. [Google Scholar] [CrossRef]
- Borén, J.; Taskinen, M.-R.; Packard, C.J. Biosynthesis and metabolism of ApoB–containing lipoproteins. Annu. Rev. Nutr. 2024, 44, 179–204. [Google Scholar] [CrossRef] [PubMed]
- Ipsen, D.H.; Lykkesfeldt, J.; Tveden-Nyborg, P. Molecular mechanisms of hepatic lipid accumulation in non-alcoholic fatty liver disease. Cell. Mol. Life Sci. 2018, 75, 3313–3327. [Google Scholar] [CrossRef]
- Amigó, N.; Akinkuolie, A.O.; Chiuve, S.E.; Correig, X.; Cook, N.R.; Mora, S. Habitual fish consumption, n-3 fatty acids, and nuclear magnetic resonance lipoprotein subfractions in women. J. Am. Heart Assoc. 2020, 9, e014963. [Google Scholar] [CrossRef] [PubMed]
- Rajaram, S.; Cofán, M.; Sala-Vila, A.; Haddad, E.; Serra-Mir, M.; Bitok, E.; Roth, I.; Freitas-Simoes, T.M.; Kaur, A.; Valls-Pedret, C.; et al. Effects of walnut consumption for 2 years on lipoprotein subclasses among healthy elders: Findings from the WAHA randomized controlled trial. Circulation 2021, 144, 1083–1085. [Google Scholar] [CrossRef]
- Lincoff, A.M.; Brown-Frandsen, K.; Colhoun, H.M.; Deanfield, J.; Emerson, S.S.; Esbjerg, S.; Hardt-Lindberg, S.; Hovingh, G.K.; Kahn, S.E.; Kushner, R.F.; et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N. Engl. J. Med. 2023, 389, 2221–2232. [Google Scholar] [CrossRef] [PubMed]
- Parhofer, K.G.; Chapman, M.J.; Nordestgaard, B.G. Efficacy and safety of icosapent ethyl in hypertriglyceridaemia: A recap. Eur. Heart J. Suppl. 2020, 22 (Suppl. J), J21–J33. [Google Scholar] [CrossRef]
- Pintó, X.; Masana, L.; Civeira, F.; Real, J.; Ibarretxe, D.; Candas, B.; Puzo, J.; Díaz, J.L.; Amigó, N.; Esteban, M.; et al. Consensus document of an expert group from the Spanish Society of Arteriosclerosis (SEA) on the clinical use of nuclear magnetic resonance to assess lipoprotein metabolism (Liposcale®). Clin. Investig. Arterioscler. 2020, 32, 219–229. [Google Scholar] [CrossRef]
- Johnson, C.S., Jr. Diffusion ordered nuclear magnetic resonance spectroscopy: Principles and applications. Prog. Nucl. Magn. Reson. Spectrosc. 1999, 34, 203–256. [Google Scholar] [CrossRef]
- Soriguer, F.; Goday, A.; Bosch-Comas, A.; Bordiú, E.; Calle-Pascual, A.; Carmena, R.; Casamitjana, R.; Castaño, L.; Delgado, E.; Gaztambide, S.; et al. Prevalence of diabetes mellitus and impaired glucose regulation in Spain: The Di@bet.es Study. Diabetologia 2012, 55, 88–93. [Google Scholar] [CrossRef]
- Valdés, S.; Doulatram-Gamgaram, V.; Maldonado-Araque, C.; García-Escobar, E.; García-Serrano, S.; Oualla-Bachiri, W.; Segura-Cuevas, E.; Acosta-Camargo, M.; Botella-Carretero, J.I.; Rojo-Martínez, G.; et al. Association between exposure to air pollution and blood lipids in the general population of Spain. Eur. J. Clin. Investig. 2024, 54, e14101, https://doi.org/10.1111/eci.14101; Erratum in Eur. J. Clin. Investig. 2025, 55, e70047. [Google Scholar] [CrossRef]
| All Population | ||||||
|---|---|---|---|---|---|---|
| All Patients | Quartile 1 <1.05 mmol/L | Quartile 2 1.051–1.45 mmol/L | Quartile 3 1.46–2.17 mmol/L | Quartile 4 >2.17 mmol/L | p Value | |
| Number of participants | 822 | 206 | 205 | 205 | 206 | |
| Age (years) | 48.3 ± 13.4 | 44.2 ± 15.1 | 48.7 ± 12.5 | 49.6 ± 12.4 | 50.6 ± 12.7 | <0.001 |
| Women (%) | 367 (44.8%) | 117 (56.8%) | 102 (50%) | 81 (39.5%) | 67 (32.8%) | <0.001 |
| Obesity (%) | 259 (31.7%) | 29 (14.1%) | 50 (24.5%) | 68 (33.2%) | 112 (54.9%) | <0.001 |
| BMI (kg/m2) | 27.9 ± 5.14 | 25.1 ± 4.27 | 27.3 ± 5 | 28.3 ± 4.58 | 30.8 ± 5.01 | <0.001 |
| Diabetes (%) | 95 (11.6%) | 10 (4.85%) | 13 (6.34%) | 24 (11.7%) | 48 (23.3%) | <0.001 |
| Hypertension (%) | 164 (20%) | 23 (11.2%) | 25 (12.2%) | 44 (21.5%) | 72 (35%) | <0.001 |
| Cardiovascular disease (%) | 71 (8.64%) | 19 (9.22%) | 19 (9.27%) | 15 (7.32%) | 18 (8.74%) | 0.88 |
| Plaque (%) | 59 (7.18%) | 7 (3.4%) | 18 (8.78%) | 19 (9.27%) | 15 (7.28%) | 0.087 |
| Standard Lipid Profile 1 | ||||||
| Total cholesterol (mmol/L) | 6.18 (5.17–7.06) | 6.03 (4.81–6.88) | 6.17 (5.24–6.9) | 6.23 (5.43–7.14) | 6.36 (5.31–7.31) | 0.003 |
| Triglycerides (mmol/L) | 1.57 (1.04–2.45) | 0.86 (0.69–0.97) | 1.28 (1.17–1.45) | 1.93 (1.73–2.15) | 3.8 (2.83–5.48) | <0.001 |
| LDL–C (mmol/L) | 3.92 (2.92–4.68) | 3.87 (2.71–4.54) | 4.11 (3.2–4.82) | 4.03 (3.23–4.86) | 3.52 (2.46–4.4) | <0.001 |
| HDL–C (mmol/L) | 1.27 (1.03–1.58) | 1.55 (1.29–1.93) | 1.38 (1.16–1.66) | 1.24 (1.06–1.47) | 0.98 (0.81–1.16) | <0.001 |
| Apo B–100 (mg/dL) | 133 (108–154) | 122 (92.8–142) | 134 (109–153) | 139 (117–162) | 137 (114–161) | <0.001 |
| Apo A–I (mg/dL) | 144 (127–165) | 154 (134–175) | 149 (133–166) | 143 (129–165) | 133 (115–150) | <0.001 |
| Lp(a) (nmol/L) | 36.9 (10–138) | 37 (11–134) | 47 (16–182) | 39 (11–138) | 23 (7.5–88.5) | <0.001 |
| All Population | ||||||
|---|---|---|---|---|---|---|
| Quartile 1 <1.05 mmol/L | Quartile 2 1.05–1.45 mmol/L | Quartile 3 1.46–2.17 mmol/L | Quartile 4 >2.17 mmol/L | p-Trend | p-Trend adj. | |
| VLDL–C (mmol/L) | 0.12 (0.08–0.19) | 0.29 (0.23–0.36) | 0.52 (0.44–0.6) | 1.12 (0.86–1.62) | <0.001 | <0.001 |
| IDL–C (mmol/L) | 0.21 (0.16–0.27) | 0.3 (0.24–0.35) | 0.36 (0.29–0.47) | 0.46 (0.34–0.6) | <0.001 | <0.001 |
| LDL–C (mmol/L) | 3.91 (3.06–4.57) | 4.24 (3.49–4.92) | 4.25 (3.59–5.19) | 3.99 (3.25–4.9) | 0.022 | 0.003 |
| HDL–C (mmol/L) | 1.52 (1.28–1.72) | 1.39 (1.16–1.57) | 1.29 (1.13–1.47) | 1.08 (0.94–1.22) | <0.001 | <0.001 |
| Remnant C (mmol/L) | 0.34 (0.28–0.44) | 0.59 (0.51–0.68) | 0.89 (0.78–1.02) | 1.63 (1.27–2.14) | <0.001 | <0.001 |
| Total Cholesterol (mmol/L) | 5.89 (4.79–6.58) | 6.3 (5.42–7.04) | 6.5 (5.68–7.47) | 6.82 (5.99–7.99) | <0.001 | <0.001 |
| VLDL–TG (mmol/L) | 0.45 (0.37–0.52) | 0.75 (0.63–0.85) | 1.18 (1.02–1.34) | 2.54 (1.9–3.85) | <0.001 | <0.001 |
| IDL–TG (mmol/L) | 0.1 (0.08–0.12) | 0.13 (0.11–0.15) | 0.16 (0.13–0.19) | 0.19 (0.14–0.24) | <0.001 | <0.001 |
| LDL–TG (mmol/L) | 0.19 (0.14–0.24) | 0.23 (0.18–0.27) | 0.25 (0.19–0.3) | 0.25 (0.18–0.34) | <0.001 | <0.001 |
| HDL–TG (mmol/L) | 0.12 (0.09–0.15) | 0.14 (0.11–0.17) | 0.17 (0.13–0.21) | 0.2 (0.13–0.26) | <0.001 | <0.001 |
| Total Triglycerides (mmol/L) | 0.89 (0.78–0.97) | 1.24 (1.15–1.33) | 1.74 (1.59–1.9) | 3.25 (2.53–4.5) | <0.001 | <0.001 |
| VLDL–P (nmol/L) | 26.2 (22.5–31.9) | 45.7 (38.8–52.2) | 74.1 (64.8–85.6) | 160 (124–234) | <0.001 | <0.001 |
| Large VLDL–P (nmol/L) | 0.71 (0.56–0.89) | 1.09 (0.89–1.31) | 1.67 (1.42–1.96) | 3.42 (2.68–4.92) | <0.001 | <0.001 |
| Medium VLDL–P (nmol/L) | 2.79 (2.11–3.38) | 4.5 (3.81–5.27) | 6.56 (5.17–7.88) | 14.9 (9.97–26.7) | <0.001 | <0.001 |
| Small VLDL–P (nmol/L) | 22.7 (19.3–27.7) | 39.8 (33.7–46) | 65.7 (56.4–76.9) | 138 (111–205) | <0.001 | <0.001 |
| LDL–P (nmol/L) | 1470 (1179–1688) | 1640 (1396–1861) | 1700 (1476–2034) | 1721 (1452–2100) | <0.001 | <0.001 |
| Large LDL–P (nmol/L) | 217 (170–256) | 235 (193–270) | 230 (198–262) | 205 (153–248) | 0.93 | 0.14 |
| Medium LDL–P (nmol/L) | 521 (335–657) | 571 (411–704) | 528 (389–689) | 394 (252–570) | 0.003 | 0.26 |
| Small LDL–P (nmol/L) | 721 (630–807) | 832 (743–930) | 935 (820–1079) | 1104 (889–1280) | <0.001 | <0.001 |
| HDL–P (μmol/L) | 27.5 (23.6–31.8) | 27 (22.9–31) | 26 (23.6–29.8) | 24.6 (20.8–28.3) | <0.001 | 0.49 |
| Large HDL–P (μmol/L) | 0.28 (0.24–0.32) | 0.3 (0.27–0.33) | 0.31 (0.27–0.35) | 0.29 (0.24–0.34) | 0.11 | <0.001 |
| Medium HDL–P (μmol/L) | 9.76 (8.36–11.4) | 9.33 (8.01–10.6) | 8.96 (7.86–10.1) | 8.2 (6.75–9.61) | <0.001 | 0.003 |
| Small HDL–P (μmol/L) | 17.8 (14.9–20.5) | 17.5 (14.6–20) | 17.1 (14.6–19.8) | 15.8 (13.4–19) | 0.052 | 0.71 |
| VLDL–Z (nm) | 42.2 (42–42.3) | 42 (41.9–42.2) | 41.9 (41.7–42.1) | 42 (41.7–42.3) | 0.012 | 0.019 |
| LDL–Z (nm) | 21.2 (21–21.4) | 21.1 (20.9–21.4) | 20.9 (20.8–21.2) | 20.6 (20.3–20.8) | <0.001 | <0.001 |
| HDL–Z (nm) | 8.28 (8.23–8.33) | 8.26 (8.22–8.31) | 8.26 (8.21–8.31) | 8.25 (8.19–8.31) | 0.19 | 0.3 |
| HDL–TG/HDL–C | 0.18 (0.14–0.22) | 0.23 (0.19–0.28) | 0.29 (0.23–0.36) | 0.41 (0.29–0.59) | <0.001 | <0.001 |
| No DM and BMI < 30 (n = 527) | DM or BMI > 30 (n = 287) | |||||||
|---|---|---|---|---|---|---|---|---|
| Variable | Tertile 1 <1.1 mmol/L | Tertile 2 1.1–1.6 mmol/L | Tertile 3 >1.6 mmol/L | p Value | Tertile 1 <1.5 mmol/L | Tertile 2 1.5–2.5 mmol/L | Tertile 3 >2.5 mmol/L | p Value |
| Number of participants | 176 | 175 | 176 | 96 | 95 | 96 | ||
| Age (years) | 42.9 ± 14.5 | 46.8 ± 12.8 | 48.6 ± 12.5 | <0.001 | 50.9 ± 14.2 | 52.4 ± 11 | 52.3 ± 12.6 | 0.81 |
| Women (%) | 104 (59.1%) | 86 (49.1%) | 66 (37.5%) | <0.001 | 49 (51%) | 32 (33.7%) | 28 (29.2%) | 0.004 |
| BMI (kg/m2) | 23.9 ± 3.1 | 25 ± 2.8 | 26.3 ± 2.41 | <0.001 | 32.1 ± 4.8 | 32.8 ± 3.82 | 34.1 ± 4.17 | 0.004 |
| Hypertension (%) | 11 (6.25%) | 15 (8.57%) | 27 (15.3%) | 0.013 | 24 (25%) | 41 (43.2%) | 46 (47.9%) | 0.003 |
| Cardiovascular disease (%) | 14 (7.95%) | 13 (7.43%) | 7 (3.98%) | 0.26 | 12 (12.5%) | 12 (12.6%) | 11 (11.5%) | 0.96 |
| Plaque (%) | 6 (3.41%) | 16 (9.14%) | 12 (6.82%) | 0.089 | 7 (7.29%) | 7 (7.37%) | 9 (9.38%) | 0.83 |
| Standard Lipid Profile 1 | ||||||||
| Total cholesterol (mmol/L) | 6.1 (4.81–6.91) | 6.36 (5.59–6.98) | 6.44 (5.6–7.29) | 0.002 | 5.53 (4.72–6.59) | 5.87 (4.69–6.93) | 6.54 (5.42–7.68) | <0.001 |
| Triglycerides (mmol/L) | 0.85 (0.68–0.95) | 1.35 (1.21–1.52) | 2.37 (2.01–3.07) | <0.001 | 1.2 (0.99–1.38) | 2.03 (1.82–2.39) | 4.74 (3.56–6.35) | <0.001 |
| LDL–C (mmol/L) | 3.9 (2.77–4.56) | 4.22 (3.47–4.93) | 4.03 (3.13–4.87) | 0.012 | 3.7 (2.77–4.48) | 3.85 (2.66–4.5) | 3.54 (2.54–4.16) | 0.12 |
| HDL–C (mmol/L) | 1.59 (1.37–2) | 1.42 (1.22–1.71) | 1.14 (0.96–1.34) | <0.001 | 1.32 (1.11–1.58) | 1.16 (0.93–1.36) | 0.93 (0.76–1.1) | <0.001 |
| Apo B–100 (mg/dL) | 122 (91–141) | 137 (116–157) | 139 (119–165) | <0.001 | 122 (98–143) | 138 (106–154) | 140 (118–160) | <0.001 |
| Apo A–I (mg/dL) | 156 (140–180) | 152 (134–172) | 139 (126–160) | <0.001 | 141 (131–160) | 140 (120–162) | 130 (113–145) | 0.001 |
| Lp(a) (nmol/L) | 45 (16.9–155) | 47 (14.2–170) | 21 (8.45–89.2) | <0.001 | 34 (10.1–127) | 44 (13.5–142) | 27 (9–81) | 0.25 |
| No DM and BMI < 30 (n = 527) | DM or BMI > 30 (n = 287) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Variable | Tertile 1 | Tertile 2 | Tertile 3 | p-Trend | p-Trend adj. | Tertile 1 | Tertile 2 | Tertile 3 | p-Trend | p-Trend adj. |
| VLDL–C (mmol/L) | 0.11 (0.08–0.18) | 0.32 (0.25–0.39) | 0.7 (0.54–1.05) | <0.001 | <0.001 | 0.28 (0.18–0.35) | 0.53 (0.46–0.72) | 1.3 (1.01–1.76) | <0.001 | <0.001 |
| IDL–C (mmol/L) | 0.21 (0.16–0.26) | 0.31 (0.24–0.39) | 0.44 (0.32–0.55) | <0.001 | <0.001 | 0.26 (0.2–0.32) | 0.34 (0.28–0.44) | 0.48 (0.35–0.68) | <0.001 | <0.001 |
| LDL–C (mmol/L) | 3.99 (3.04–4.59) | 4.3 (3.81–5.01) | 4.26 (3.57–5.16) | 0.002 | <0.001 | 3.75 (3.28–4.54) | 4.04 (3.39–4.85) | 3.98 (3.38–4.94) | 0.048 | 0.035 |
| HDL–C (mmol/L) | 1.54 (1.31–1.75) | 1.43 (1.2–1.61) | 1.19 (1.07–1.39) | <0.001 | <0.001 | 1.34 (1.1–1.55) | 1.22 (1.08–1.37) | 1.05 (0.85–1.18) | 0.001 | 0.01 |
| Remnant C (mmol/L) | 0.33 (0.27–0.44) | 0.62 (0.55–0.74) | 1.14 (0.93–1.47) | <0.001 | <0.001 | 0.52 (0.4–0.67) | 0.92 (0.82–1.04) | 1.87 (1.55–2.46) | <0.001 | <0.001 |
| Cholesterol (mmol/L) | 6.09 (4.79–6.61) | 6.43 (5.81–7.26) | 6.84 (5.91–7.76) | <0.001 | <0.001 | 5.61 (4.98–6.62) | 6.14 (5.46–7.14) | 6.91 (6.21–8.24) | <0.001 | <0.001 |
| VLDL–TG (mmol/L) | 0.43 (0.36–0.51) | 0.77 (0.64–0.87) | 1.48 (1.23–2.1) | <0.001 | <0.001 | 0.66 (0.55–0.82) | 1.28 (1.12–1.64) | 3.35 (2.43–4.64) | <0.001 | <0.001 |
| IDL–TG (mmol/L) | 0.1 (0.09–0.11) | 0.13 (0.11–0.16) | 0.18 (0.14–0.21) | <0.001 | <0.001 | 0.12 (0.1–0.13) | 0.16 (0.13–0.19) | 0.19 (0.15–0.24) | <0.001 | <0.001 |
| LDL–TG (mmol/L) | 0.2 (0.14–0.24) | 0.24 (0.2–0.29) | 0.27 (0.19–0.34) | <0.001 | <0.001 | 0.19 (0.16–0.25) | 0.23 (0.19–0.27) | 0.25 (0.16–0.32) | <0.001 | <0.001 |
| HDL–TG (mmol/L) | 0.12 (0.1–0.15) | 0.15 (0.12–0.18) | 0.18 (0.14–0.24) | <0.001 | <0.001 | 0.13 (0.1–0.16) | 0.17 (0.13–0.2) | 0.18 (0.08–0.26) | <0.001 | <0.001 |
| Triglycerides (mmol/L) | 0.88 (0.78–0.97) | 1.29 (1.17–1.43) | 2.16 (1.82–2.72) | <0.001 | <0.001 | 1.14 (0.96–1.33) | 1.84 (1.66–2.13) | 3.99 (3.14–5.33) | <0.001 | <0.001 |
| VLDL–P (nmol/L) | 25.5 (22.2–31.3) | 47.3 (39.1–55.6) | 96.4 (78.8–138) | <0.001 | <0.001 | 41.7 (33.3–51.6) | 82 (69.7–101) | 205 (152–289) | <0.001 | <0.001 |
| Large VLDL–P (nmol/L) | 0.68 (0.56–0.83) | 1.08 (0.91–1.31) | 2.15 (1.75–2.97) | <0.001 | <0.001 | 1.1 (0.84–1.32) | 1.91 (1.58–2.35) | 4.32 (3.29–6.2) | <0.001 | <0.001 |
| Medium VLDL–P (nmol/L) | 2.8 (2.06–3.4) | 4.56 (3.85–5.41) | 8.4 (6.37–12.6) | <0.001 | <0.001 | 4.13 (3.1–5.15) | 7.02 (5.71–8.67) | 21.8 (14.1–33.1) | <0.001 | <0.001 |
| Small VLDL–P (nmol/L) | 21.9 (19–27) | 41.3 (34.1–49.3) | 85.7 (70.4–123) | <0.001 | <0.001 | 36.7 (28.9–45.4) | 71.8 (61.4–90.8) | 175 (136–249) | <0.001 | <0.001 |
| LDL–P (nmol/L) | 1482 (1168–1702) | 1684 (1482–1894) | 1746 (1513–2067) | <0.001 | <0.001 | 1488 (1282–1708) | 1652 (1418–1990) | 1739 (1479–2178) | <0.001 | <0.001 |
| Large LDL–P (nmol/L) | 228 (170–261) | 238 (205–272) | 230 (190–264) | 0.15 | 0.01 | 208 (170–245) | 213 (169–244) | 197 (150–245) | 0.61 | 0.66 |
| Medium LDL–P (nmol/L) | 533 (330–672) | 589 (460–711) | 489 (354–674) | 0.87 | 0.32 | 473 (382–635) | 473 (335–630) | 360 (240–534) | 0.06 | 0.12 |
| Small LDL–P (nmol/L) | 719 (627–803) | 842 (741–957) | 996 (879–1177) | <0.001 | <0.001 | 785 (686–897) | 959 (828–1083) | 1148 (892–1346) | <0.001 | <0.001 |
| HDL–P (μmol/L) | 28.4 (24.6–32.2) | 27.7 (23.8–31.3) | 25.5 (23.1–29.5) | 0.007 | 0.21 | 25.7 (21.9–29.3) | 25.2 (22.8–27.6) | 22.8 (18.9–27) | 0.19 | 0.6 |
| Large HDL–P (μmol/L) | 0.29 (0.25–0.33) | 0.31 (0.27–0.34) | 0.31 (0.27–0.35) | 0.002 | <0.001 | 0.28 (0.25–0.31) | 0.29 (0.26–0.32) | 0.27 (0.21–0.33) | 0.7 | 0.89 |
| Medium HDL–P (μmol/L) | 10 (8.52–11.6) | 9.58 (8.18–10.9) | 8.87 (7.56–10.2) | <0.001 | 0.0063 | 9.15 (7.81–10.3) | 8.47 (7.48–9.49) | 7.86 (6.19–9.04) | 0.009 | 0.048 |
| Small HDL–P (μmol/L) | 18.2 (15.5–20.8) | 17.9 (15.2–21.2) | 16.7 (14.4–19.5) | 0.23 | 0.66 | 16.4 (13.6–19.3) | 16.9 (14.2–18.7) | 15.6 (11.9–18.3) | 0.57 | 0.87 |
| VLDL–Z (nm) | 42.2 (42–42.3) | 42 (41.9–42.2) | 41.9 (41.7–42.2) | <0.001 | <0.001 | 42 (41.9–42.2) | 41.9 (41.7–42.1) | 42.1 (41.9–42.4) | 0.12 | 0.23 |
| LDL–Z (nm) | 21.2 (21–21.5) | 21.2 (20.9–21.4) | 20.8 (20.6–21.1) | <0.001 | <0.001 | 21.1 (20.9–21.3) | 20.8 (20.6–21.1) | 20.4 (20.1–20.8) | <0.001 | <0.001 |
| HDL–Z (nm) | 8.27 (8.23–8.32) | 8.27 (8.22–8.31) | 8.25 (8.21–8.3) | 0.048 | 0.66 | 8.28 (8.23–8.33) | 8.26 (8.21–8.3) | 8.24 (8.18–8.31) | 0.4 | 0.24 |
| HDL–TG/HDL–C | 0.18 (0.14–0.22) | 0.24 (0.19–0.29) | 0.35 (0.26–0.47) | <0.001 | <0.001 | 0.22 (0.17–0.29) | 0.32 (0.23–0.39) | 0.37 (0.21–0.62) | <0.001 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amigó, N.; Torné, P.; Nordestgaard, L.T.; Di Giacomo-Barbagallo, F.; Merino, C.; Magni, P.; González-Lleó, A.; Andreychuk, N.; Catapano, A.L.; Masana, L.; et al. Triglycerides as Determinants of Global Lipoprotein Derangement: Implications for Cardiovascular Prevention. Int. J. Mol. Sci. 2025, 26, 8284. https://doi.org/10.3390/ijms26178284
Amigó N, Torné P, Nordestgaard LT, Di Giacomo-Barbagallo F, Merino C, Magni P, González-Lleó A, Andreychuk N, Catapano AL, Masana L, et al. Triglycerides as Determinants of Global Lipoprotein Derangement: Implications for Cardiovascular Prevention. International Journal of Molecular Sciences. 2025; 26(17):8284. https://doi.org/10.3390/ijms26178284
Chicago/Turabian StyleAmigó, Núria, Pol Torné, Liv T. Nordestgaard, Francesco Di Giacomo-Barbagallo, Carla Merino, Paolo Magni, Ana González-Lleó, Natalia Andreychuk, Alberico L. Catapano, Lluís Masana, and et al. 2025. "Triglycerides as Determinants of Global Lipoprotein Derangement: Implications for Cardiovascular Prevention" International Journal of Molecular Sciences 26, no. 17: 8284. https://doi.org/10.3390/ijms26178284
APA StyleAmigó, N., Torné, P., Nordestgaard, L. T., Di Giacomo-Barbagallo, F., Merino, C., Magni, P., González-Lleó, A., Andreychuk, N., Catapano, A. L., Masana, L., & Ibarretxe, D. (2025). Triglycerides as Determinants of Global Lipoprotein Derangement: Implications for Cardiovascular Prevention. International Journal of Molecular Sciences, 26(17), 8284. https://doi.org/10.3390/ijms26178284









