Biphasic Electrical Stimulation of Schwann Cells on Conducting Polymer-Coated Carbon Microfibers
Abstract
1. Introduction
2. Results
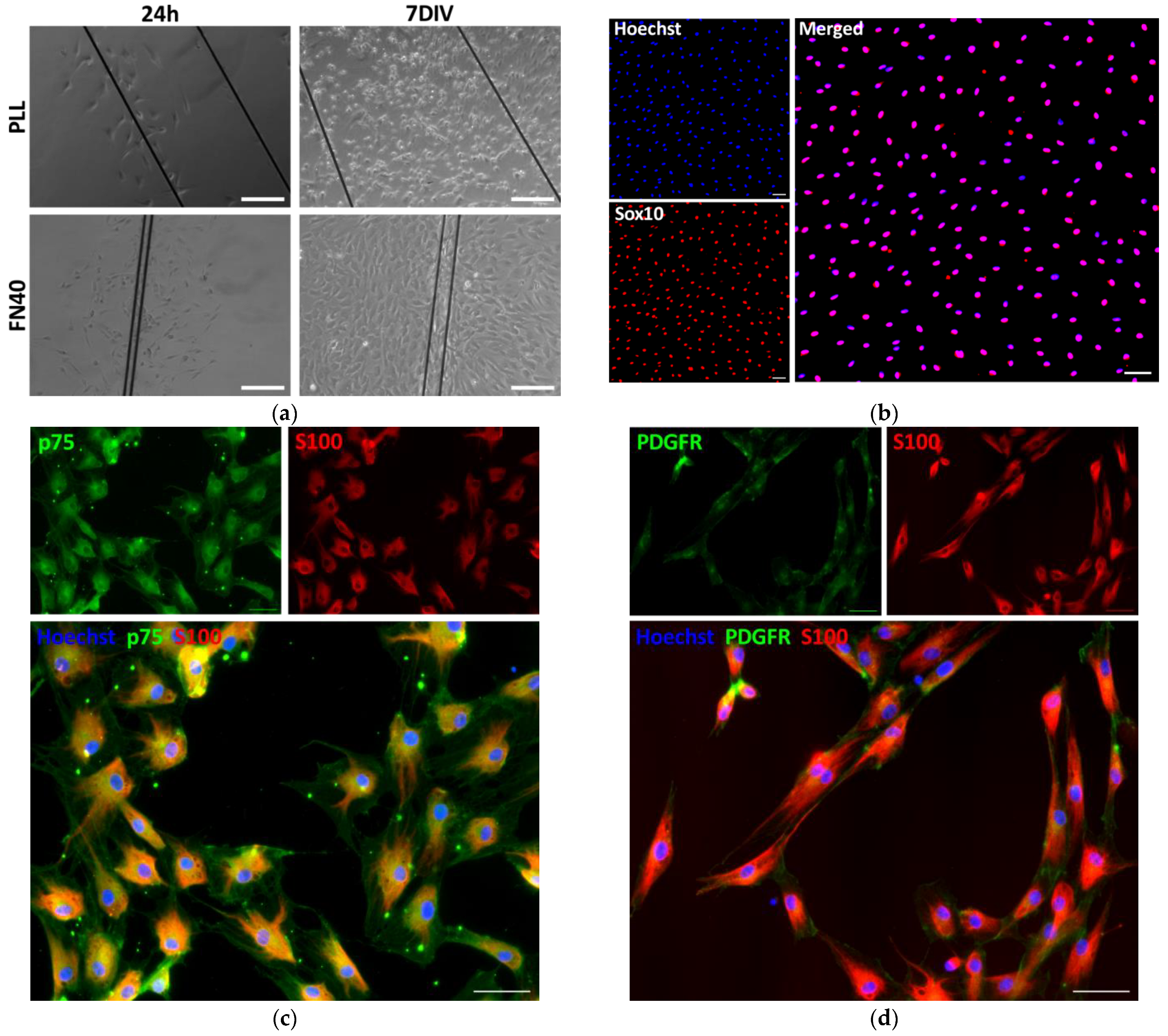
2.1. Optimal SC Substrate Coating
2.2. Essential Components of the SC Culture Medium
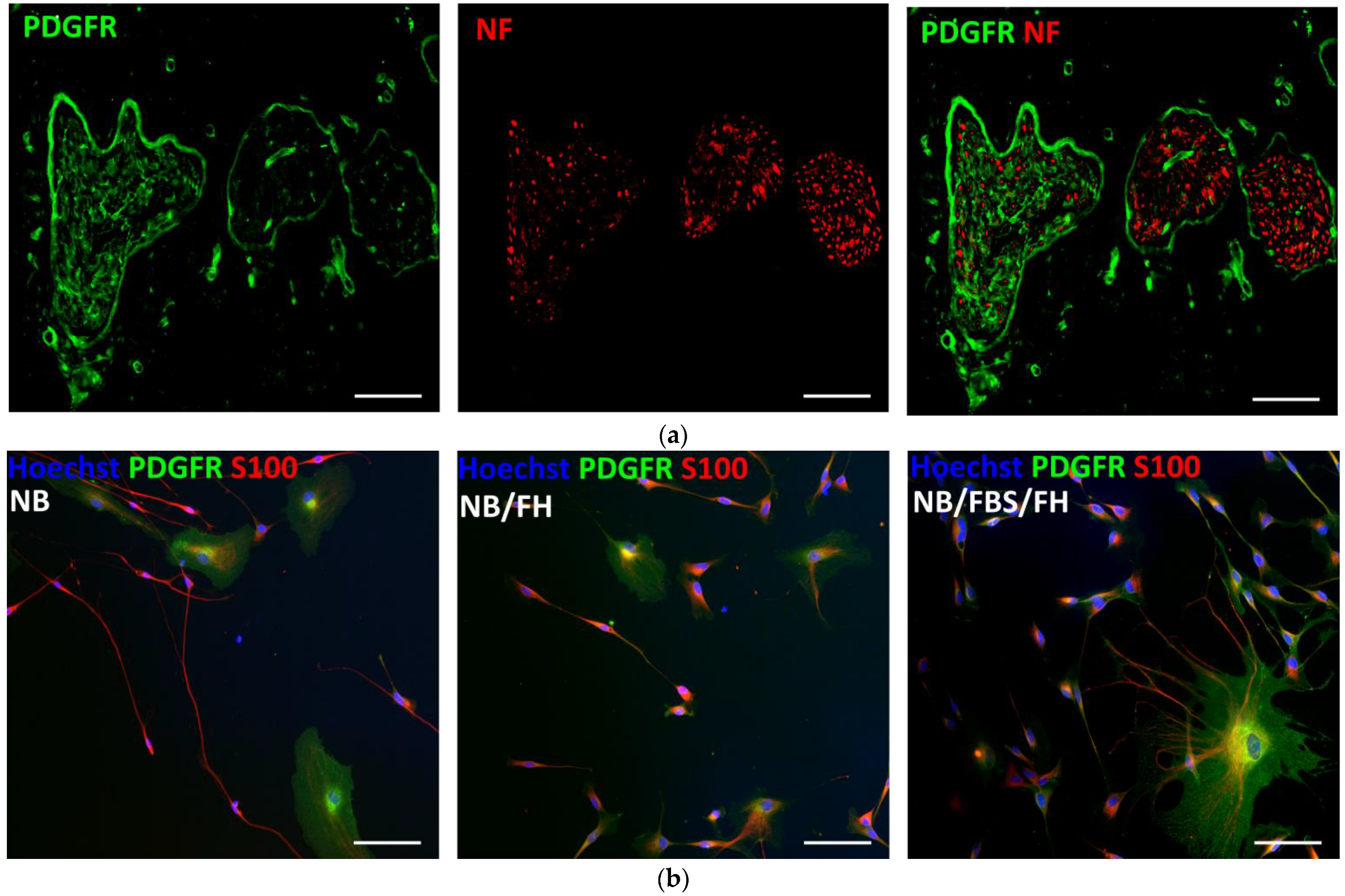
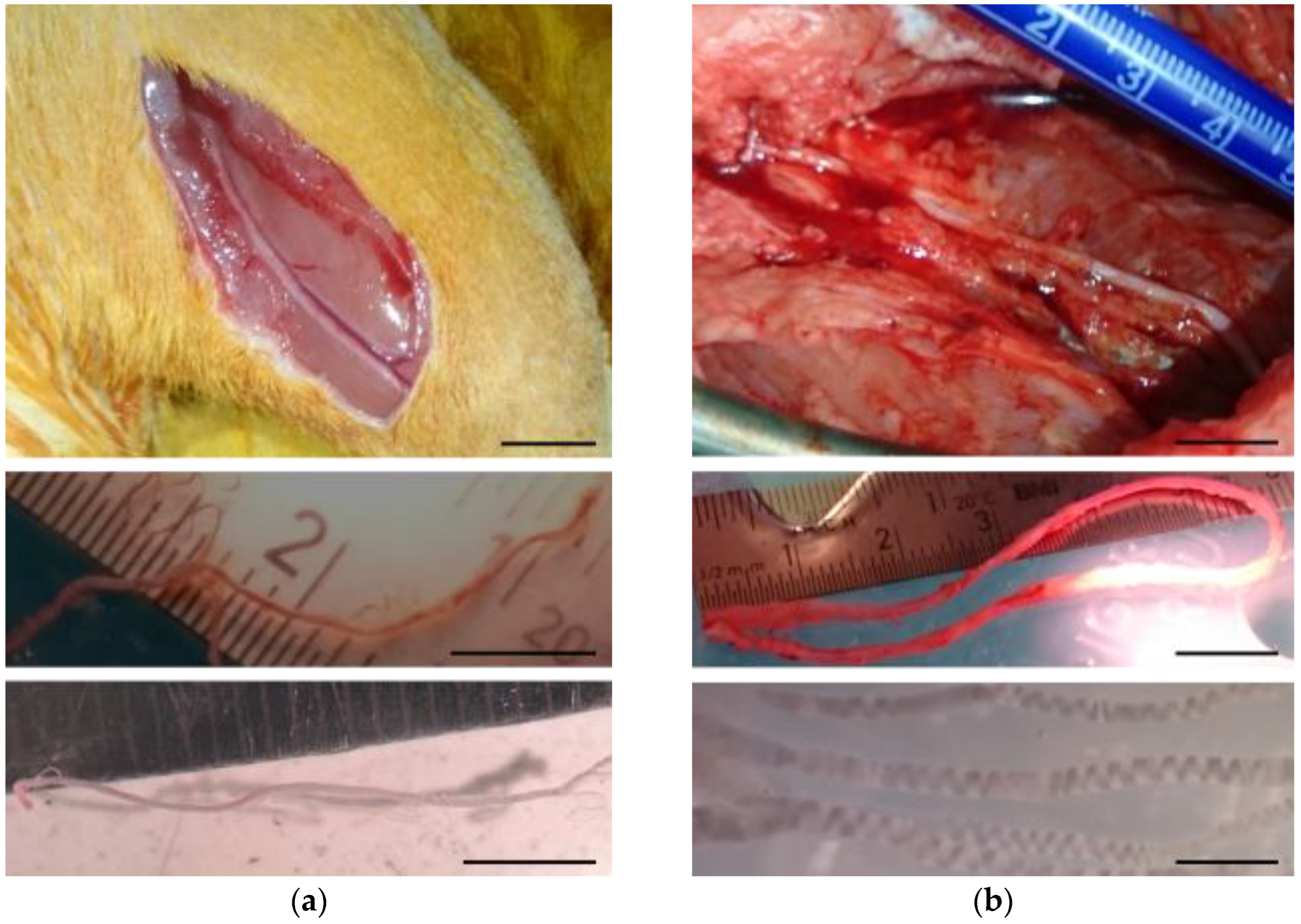
2.3. Cell Growth from Peripheral Nerves on PEDOT-Coated Carbon Microfibers
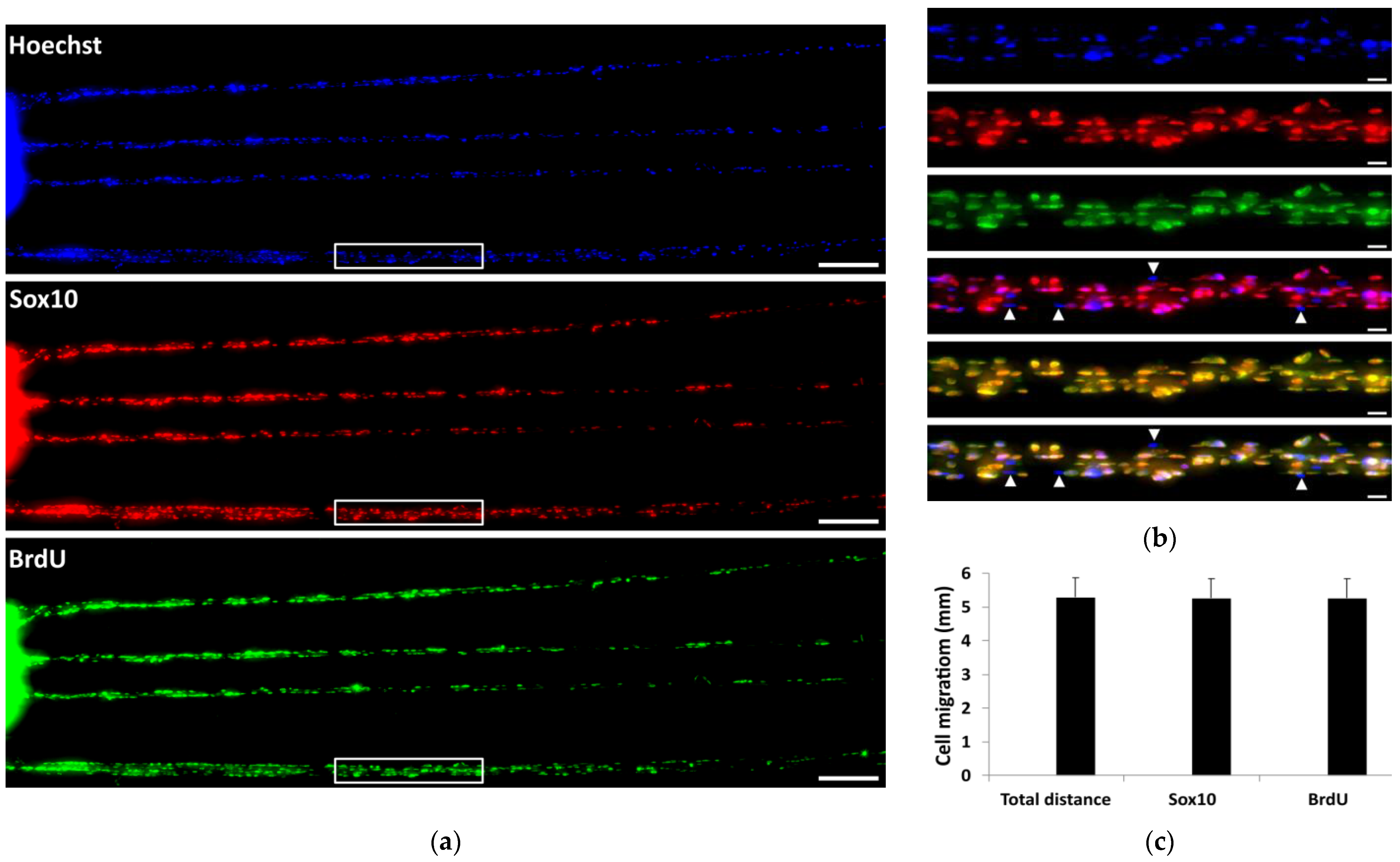
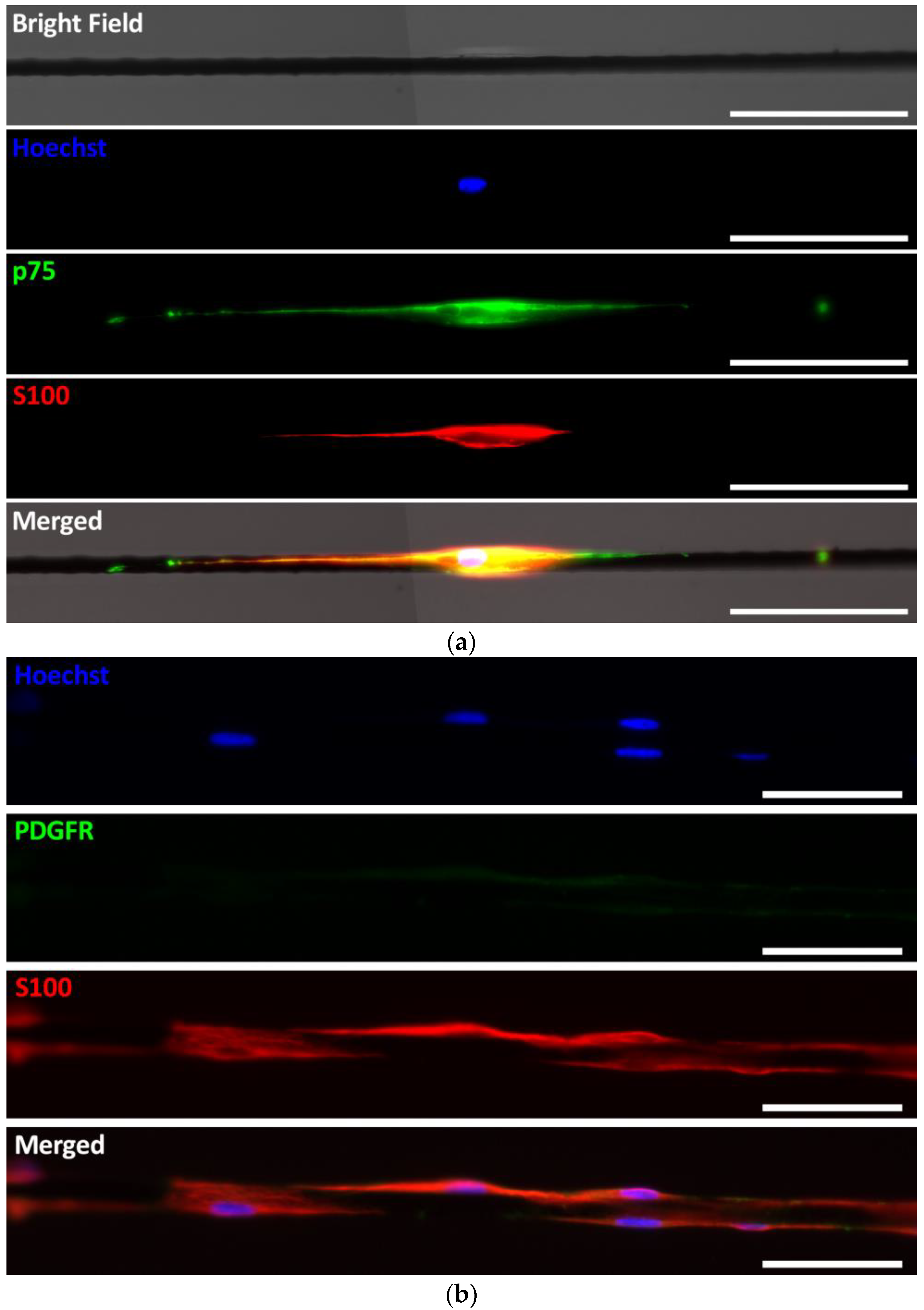
2.4. Sub-Culturing Cells That Migrate Along PCMFs
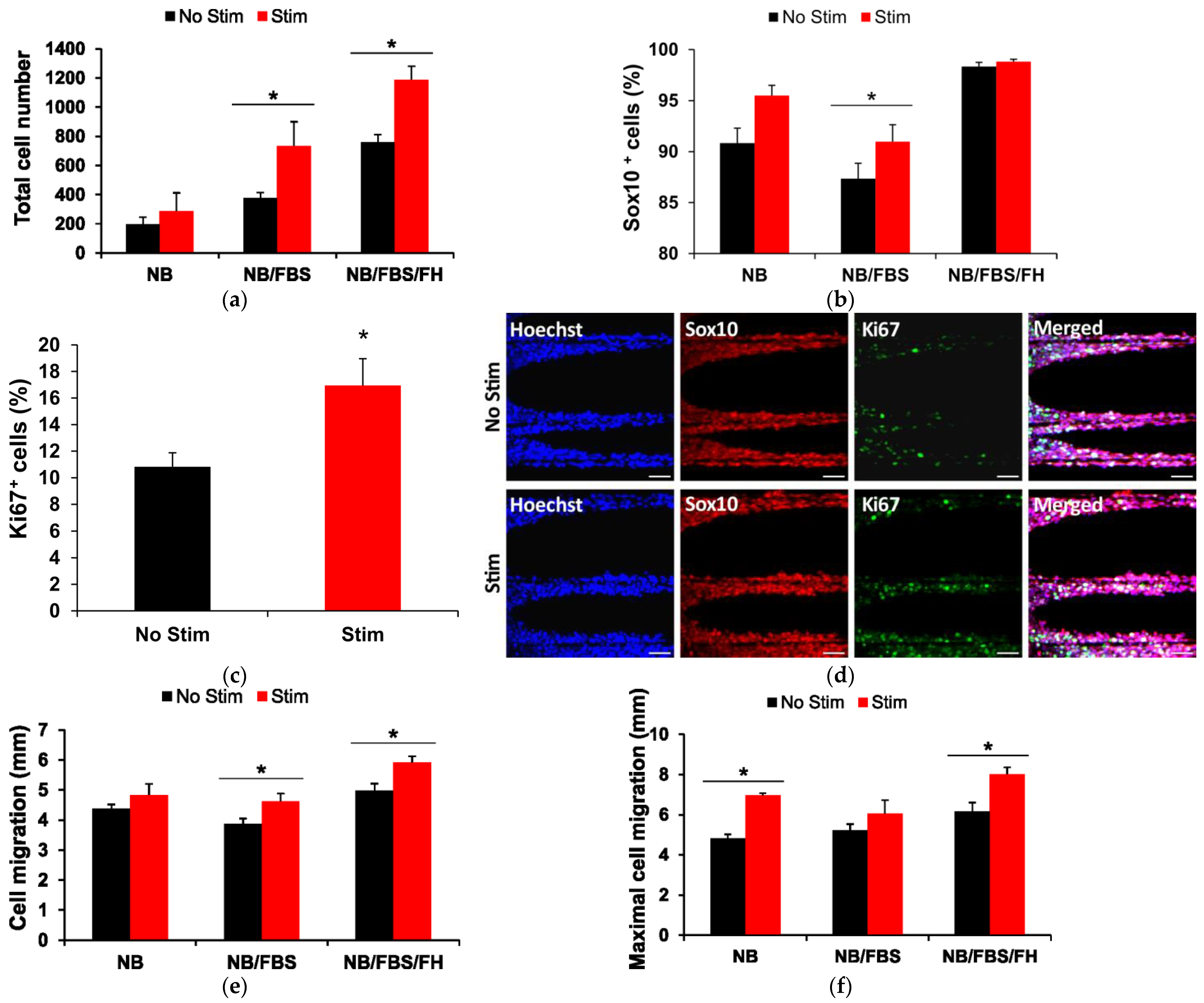
2.5. Cellular Effects of ES Through PCMFs and Synergies with Soluble Factors
3. Discussion
4. Materials and Methods
4.1. Selection of Molecules for Substrate Coating
4.2. Preparation of Cell Culture Chambers Containing Biofunctionalized PCMFs
4.3. Peripheral Nerve Sources
4.4. Nerve Processing and Cell Culture Media
4.4.1. Dissociated SC Culture
4.4.2. Nerves Cultured on PCMFs
4.4.3. Secondary Culture of the Cells Migrating on PCMFs
4.5. Electrical Stimulation
4.6. Immunocytochemistry and Cell Quantification
4.7. Statistics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| bFGF | Basic fibroblast growth factor |
| BrdU | 5-Bromo-2′-desoxiuridine |
| DIV | Days in vitro |
| EDC | N-(3-Dimethylaminopropyl)-N′-ethylcarbodiimide |
| ES | Electrical stimulation |
| FBS | Fetal bovine serum |
| FH | Forskolin and heregulin β-1 |
| HBSS | Hank’s balanced salts solution |
| NB | Neurobasal cell culture medium with L-glutamine, Penicillin-Streptomycin, Gentamicin, and B27 |
| NHS | N-Hydroxysuccinimide |
| PBS | Phosphate buffered saline |
| PCMFs | Carbon microfibers coated with PEDOT doped with PSS-co-MA |
| PDGFR | Platelet derived growth factor receptor |
| PEDOT | Poly(3,4-ethylenedioxythiophene) |
| PLL | Poly-L-lysine |
| PNI | Peripheral nerve injury |
| PSS-co-MA | Poly [(4-styrenesulfonic acid)-co-(maleic acid)] |
| RT | Room temperature |
| SCs | Schwann cells |
References
- Murphy, R.N.A.; de Schoulepnikoff, C.; Chen, J.H.C.; Columb, M.O.; Bedford, J.; Wong, J.K.; Reid, A.J. The incidence and management of peripheral nerve injury in England (2005–2020). J. Plast. Reconstr. Aesthet. Surg. 2023, 80, 75–85. [Google Scholar] [CrossRef]
- Ciaramitaro, P.; Mondelli, M.; Logullo, F.; Grimaldi, S.; Battiston, B.; Sard, A.; Scarinzi, C.; Migliaretti, G.; Faccani, G.; Cocito, D. Italian Network for Traumatic Neuropathies. Traumatic peripheral nerve injuries: Epidemiological findings, neuropathic pain and quality of life in 158 patients. J. Peripher. Nerv. Syst. 2010, 15, 120–127. [Google Scholar] [CrossRef]
- Asplund, M.; Nilsson, M.; Jacobsson, A.; von Holst, H. Incidence of traumatic peripheral nerve injuries and amputations in Sweden between 1998 and 2006. Neuroepidemiology 2009, 32, 217–228. [Google Scholar] [CrossRef]
- Bekelis, K.; Missios, S.; Spinner, R.J. Falls and peripheral nerve injuries: An age-dependent relationship. J. Neurosurg. 2015, 123, 1223–1229. [Google Scholar] [CrossRef]
- Stonner, M.M.; Mackinnon, S.E.; Kaskutas, V. Predictors of functional outcome after peripheral nerve injury and compression. J. Hand Ther. 2021, 34, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Grosu-Bularda, A.; Vancea, C.V.; Hodea, F.V.; Cretu, A.; Bordeanu-Diaconescu, E.M.; Dumitru, C.S.; Ratoiu, V.A.; Teodoreanu, R.N.; Lascar, I.; Hariga, C.S. Optimizing peripheral nerve regeneration: Surgical techniques, biomolecular and regenerative strategies—A narrative review. Int. J. Mol. Sci. 2025, 26, 3895. [Google Scholar] [CrossRef]
- Gordon, T. Brief electrical stimulation promotes recovery after surgical repair of injured peripheral nerves. Int. J. Mol. Sci. 2024, 25, 665. [Google Scholar] [CrossRef]
- Min, Q.; Parkinson, D.B.; Dun, X.P. Migrating Schwann cells direct axon regeneration within the peripheral nerve bridge. Glia 2021, 69, 235–254. [Google Scholar] [CrossRef]
- Bosch-Queralt, M.; Fledrich, R.; Stassart, R.M. Schwann cell functions in peripheral nerve development and repair. Neurobiol. Dis. 2023, 176, 105952. [Google Scholar] [CrossRef]
- Bolívar, S.; Navarro, X.; Udina, E. Schwann cell role in selectivity of nerve regeneration. Cells 2020, 9, 2131. [Google Scholar] [CrossRef] [PubMed]
- Stocco, E.; Barbon, S.; Emmi, A.; Tiengo, C.; Macchi, V.; De Caro, R.; Porzionato, A. Bridging gaps in peripheral nerves: From current strategies to future perspectives in conduit design. Int. J. Mol. Sci. 2023, 24, 9170. [Google Scholar] [CrossRef]
- Redolfi Riva, E.; Özkan, M.; Contreras, E.; Pawar, S.; Zinno, C.; Escarda-Castro, E.; Kim, J.; Wieringa, P.; Stellacci, F.; Micera, S.; et al. Beyond the limiting gap length: Peripheral nerve regeneration through implantable nerve guidance conduits. Biomater. Sci. 2024, 12, 1371–1404. [Google Scholar] [CrossRef] [PubMed]
- Ronchi, G.; Ackva, C.; Fregnan, F.; Zen, F.; Muratori, L.; Crosio, A.; Metzen, J.; Haastert-Talini, K. Chitosan-based materials for peripheral nerve repair—New pre-clinical data on degradation behavior at the nerve repair site and critical opinion on their translational impact. Int. J. Mol. Sci. 2025, 26, 1214. [Google Scholar] [CrossRef] [PubMed]
- Kuffler, D.P.; Foy, C. Restoration of Neurological Function Following Peripheral Nerve Trauma. Int. J. Mol. Sci. 2020, 21, 1808. [Google Scholar] [CrossRef] [PubMed]
- Vijayavenkataraman, S. Nerve guide conduits for peripheral nerve injury repair: A review on design, materials and fabrication methods. Acta Biomater. 2020, 106, 54–69. [Google Scholar] [CrossRef]
- Ribeiro-Resende, V.T.; Koenig, B.; Nichterwitz, S.; Oberhoffner, S.; Schlosshauer, B. Strategies for inducing the formation of bands of Büngner in peripheral nerve regeneration. Biomaterials 2009, 30, 5251–5259. [Google Scholar] [CrossRef]
- Gonzalez-Perez, F.; Hernández, J.; Heimann, C.; Phillips, J.B.; Udina, E.; Navarro, X. Schwann cells and mesenchymal stem cells in laminin- or fibronectin-aligned matrices and regeneration across a critical size defect of 15 mm in the rat sciatic nerve. J. Neurosurg. Spine 2018, 28, 109–118. [Google Scholar] [CrossRef]
- Fang, Y.; Wang, C.; Liu, Z.; Ko, J.; Chen, L.; Zhang, T.; Xiong, Z.; Zhang, L.; Sun, W. 3D printed conductive multiscale nerve guidance conduit with hierarchical fibers for peripheral nerve regeneration. Adv. Sci. 2023, 10, e2205744. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, X.; Wang, C.; Li, F.; Qiao, Z.; Zeng, L.; Wang, Z.; Liu, H.; Ding, J.; Yang, H. Conductive composite fiber with optimized alignment guides neural regeneration under electrical stimulation. Adv. Healthc. Mater. 2021, 10, e2000604. [Google Scholar] [CrossRef]
- Al-Majed, A.A.; Brushart, T.M.; Gordon, T. Electrical stimulation accelerates and increases expression of BDNF and trkB mRNA in regenerating rat femoral motoneurons. Eur. J. Neurosci. 2000, 12, 4381–4390. [Google Scholar] [CrossRef]
- Al-Majed, A.A.; Siu, L.T.; Gordon, T. Electrical stimulation accelerates and enhances expression of regeneration-associated genes in regenerating rat femoral motoneurons. Cell. Mol. Neurobiol. 2004, 24, 379–402. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Ye, Z.; Hu, X.; Lu, L.; Luo, Z. Electrical stimulation induces calcium-dependent release of NGF from cultured Schwann cells. Glia 2010, 58, 622–631. [Google Scholar] [CrossRef]
- Luo, B.; Huang, J.; Lu, L.; Hu, X.; Luo, Z.; Li, M. Electrically induced brain-derived neurotrophic factor release from Schwann cells. J. Neurosci. Res. 2014, 92, 893–903. [Google Scholar] [CrossRef]
- Kim, I.S.; Song, Y.M.; Cho, T.H.; Pan, H.; Lee, T.H.; Kim, S.J.; Hwang, S.J. Biphasic electrical targeting plays a significant role in Schwann cell activation. Tissue Eng. Part A 2011, 17, 1327–1340. [Google Scholar] [CrossRef]
- Huang, J.; Lu, L.; Zhang, J.; Hu, X.; Zhang, Y.; Liang, W.; Wu, S.; Luo, Z. Electrical stimulation to conductive scaffold promotes axonal regeneration and remyelination in a rat model of large nerve defect. PLoS ONE 2012, 7, e39526. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Liang, Y.; Ding, S.; Zhang, K.; Mao, H.Q.; Yang, Y. Application of conductive PPy/SF composite scaffold and electrical stimulation for neural tissue engineering. Biomaterials 2020, 255, 120164. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Liu, Y.; Yushan, M.; Yusufu, A. Enhanced nerve regeneration by bionic conductive nerve scaffold under electrical stimulation. Front. Neurosci. 2022, 16, 810676. [Google Scholar] [CrossRef]
- Borah, R.; Diez Clarke, D.; Upadhyay, J.; Monaghan, M.G. From innovation to clinic: Emerging strategies harnessing electrically conductive polymers to enhance electrically stimulated peripheral nerve repair. Mater. Today Bio 2024, 30, 101415. [Google Scholar] [CrossRef]
- Wang, J.; Fang, J.; Weng, Z.; Nan, L.; Chen, Y.; Shan, J.; Chen, F.; Liu, J. Advanced development of conductive biomaterials for enhanced peripheral nerve regeneration: A review. RSC Adv. 2025, 15, 12997–13009. [Google Scholar] [CrossRef]
- Thakkar, V.; Mehdipour, M.; Chang, S. Unlocking nerve regeneration: Electrical stimulation and bioscaffolds to enhance peripheral nerve regeneration. Front. Neurosci. 2025, 19, 1594435. [Google Scholar] [CrossRef]
- Cui, X.; Martin, D.C. Electrochemical deposition and characterization of poly(3,4-ethylenedioxythiophene) on neural microelectrode arrays. Sens. Actuators B Chem. 2003, 89, 92–102. [Google Scholar] [CrossRef]
- Collazos-Castro, J.E.; Polo, J.L.; Hernández-Labrado, G.R.; Padial-Cañete, V.; García-Rama, C. Bioelectrochemical control of neural cell development on conducting polymers. Biomaterials 2010, 31, 9244–9255. [Google Scholar] [CrossRef]
- Collazos-Castro, J.E.; Hernández-Labrado, G.R.; Polo, J.L.; García-Rama, C. N-Cadherin- and L1-functionalised conducting polymers for synergistic stimulation and guidance of neural cell growth. Biomaterials 2013, 34, 3603–3617. [Google Scholar] [CrossRef]
- Vallejo-Giraldo, C.; Kelly, A.; Biggs, M.J. Biofunctionalisation of electrically conducting polymers. Drug Discov. Today 2014, 19, 88–94. [Google Scholar] [CrossRef]
- Collazos-Castro, J.E.; García-Rama, C.; Alves-Sampaio, A. Glial progenitor cell migration promotes CNS axon growth on functionalized electroconducting microfibers. Acta Biomater. 2016, 35, 42–56. [Google Scholar] [CrossRef]
- Wu, B.; Cao, B.; Taylor, I.M.; Woeppel, K.; Cui, X.T. Facile synthesis of a 3,4-ethylene-dioxythiophene (EDOT) derivative for ease of bio-functionalization of the conducting polymer PEDOT. Front. Chem. 2019, 7, 178. [Google Scholar] [CrossRef]
- Baker, C.; Wagner, K.; Wagner, P.; Officer, D.L.; Mawad, D. Biofunctional conducting polymers: Synthetic advances, challenges, and perspectives towards their use in implantable bioelectronic devices. Adv. Phys. X 2021, 6, 1899850. [Google Scholar] [CrossRef]
- Alves-Sampaio, A.; García-Rama, C.; Collazos-Castro, J.E. Biofunctionalized PEDOT-coated microfibers for the treatment of spinal cord injury. Biomaterials 2016, 89, 98–113. [Google Scholar] [CrossRef]
- El Waly, B.; Escarrat, V.; Perez-Sanchez, J.; Kaur, J.; Pelletier, F.; Collazos-Castro, J.E.; Debarbieux, F. Intravital assessment of cells responses to conducting polymer-coated carbon microfibres for bridging spinal cord injury. Cells 2021, 10, 73. [Google Scholar] [CrossRef]
- Escarrat, V.; Perez-Sanchez, J.; El-Waly, B.; Collazos-Castro, J.E.; Debarbieux, F. Composite fibrin and carbon microfibre implant to modulate postraumatic inflammation after spinal cord injury. Cells 2023, 12, 839. [Google Scholar] [CrossRef]
- Alves-Sampaio, A.; Del-Cerro, P.; Collazos-Castro, J.E. Composite fibrin/carbon microfiber implants for bridging spinal cord injury: A translational approach in pigs. Int. J. Mol. Sci. 2023, 24, 11102. [Google Scholar] [CrossRef]
- Brushart, T.M.; Jari, R.; Verge, V.; Rohde, C.; Gordon, T. Electrical stimulation restores the specificity of sensory axon regeneration. Exp. Neurol. 2005, 194, 221–229. [Google Scholar] [CrossRef]
- Ma, C.H.E.; Omura, T.; Cobos, E.J.; Latrémolière, A.; Ghasemlou, N.; Brenner, G.J.; van Veen, E.; Barrett, L.; Sawada, T.; Gao, F.; et al. Accelerating axonal growth promotes motor recovery after peripheral nerve injury in mice. J. Clin. Investig. 2011, 121, 4332–4347. [Google Scholar] [CrossRef]
- Brooks, A.E.; Athauda, G.; Bunge, M.B.; Khan, A. Culture and expansion of rodent and porcine Schwann cells for preclinical animal studies. Methods Mol. Biol. 2018, 1739, 111–126. [Google Scholar] [CrossRef]
- Morrissey, T.K.; Kleitman, N.; Bunge, R.P. Isolation and functional characterization of Schwann cells derived from adult peripheral nerve. J. Neurosci. 1991, 11, 2433–2442. [Google Scholar] [CrossRef]
- Finzsch, M.; Schreiner, S.; Kichko, T.; Reeh, P.; Tamm, E.R.; Bösl, M.R.; Meijer, D.; Wegner, M. Sox10 is required for Schwann cell identity and progression beyond the immature Schwann cell stage. J Cell Biol. 2010, 189, 701–712. [Google Scholar] [CrossRef]
- Aberle, T.; Walter, A.; Piefke, S.; Hilgärtner, S.; Wüst, H.M.; Wegner, M.; Küspert, M. Sox10 Activity and the Timing of Schwann Cell Differentiation Are Controlled by a Tle4-Dependent Negative Feedback Loop. Int. J. Mol. Sci. 2024, 25, 5234. [Google Scholar] [CrossRef]
- Levi, A.D.; Bunge, R.P.; Lofgren, J.A.; Meima, L.; Hefti, F.; Nikolics, K.; Sliwkowski, M.X. The influence of heregulins on human Schwann cell proliferation. J. Neurosci. 1995, 15, 1329–1340. [Google Scholar] [CrossRef]
- Syroid, D.E.; Maycox, P.R.; Burrola, P.G.; Liu, N.; Wen, D.; Lee, K.F.; Lemke, G.; Kilpatrick, T.J. Cell death in the Schwann cell lineage and its regulation by neuregulin. Proc. Natl. Acad. Sci. USA 1996, 93, 9229–9234. [Google Scholar] [CrossRef]
- Sugimura, K.; Haimoto, H.; Nagura, H.; Kato, K.; Takahashi, A. Immunohistochemical differential distribution of S-100 alpha and S-100 beta in the peripheral nervous system of the rat. Muscle Nerve 1989, 12, 929–935. [Google Scholar] [CrossRef]
- Schnell, E.; Klinkhammer, K.; Balzer, S.; Brook, G.; Klee, D.; Dalton, P.; Mey, J. Guidance of glial cell migration and axonal growth on electrospun nanofibers of poly-epsilon-caprolactone and a collagen/poly-epsilon-caprolactone blend. Biomaterials 2007, 28, 3012–3025. [Google Scholar] [CrossRef]
- Gisbert Roca, F.; Serrano Requena, S.; Monleón Pradas, M.; Martínez-Ramos, C. Electrical stimulation increases axonal growth from dorsal root ganglia co-cultured with Schwann cells in highly aligned PLA-PPy-Au microfiber substrates. Int. J. Mol. Sci. 2022, 23, 6362. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Wu, T.; Qiu, J.; Rutledge, S.; Tanes, M.L.; Xia, Y. Promoting cell migration and neurite extension along uniaxially aligned nanofibers with biomacromolecular particles in a density gradient. Adv. Funct. Mater. 2020, 30, 2002031. [Google Scholar] [CrossRef]
- Collazos-Castro, J.E. Biomaterial-based systems as biomimetic agents in the repair of the central nervous system. In Handbook of Innovations in Central Nervous System Regenerative Medicine; Elsevier: Amsterdam, The Netherlands, 2020; pp. 259–289. [Google Scholar]
- Vara, H.; Collazos-Castro, J.E. Biofunctionalized conducting polymer/carbon microfiber electrodes for ultrasensitive neural recordings. ACS Appl. Mater. Interfaces 2015, 7, 27016–27026. [Google Scholar] [CrossRef]
- Vara, H.; Collazos-Castro, J.E. Enhanced spinal cord microstimulation using conducting polymer-coated carbon microfibers. Acta Biomater. 2019, 90, 71–86. [Google Scholar] [CrossRef] [PubMed]
- Vara, H.; Hernández-Labrado, G.R.; Alves-Sampaio, A.; Collazos-Castro, J.E. Stability of conducting polymer-coated carbon microfibers for long-term electrical stimulation of injured neural tissue. Polymers 2024, 16, 2093. [Google Scholar] [CrossRef]
- del Lopez-Buenafe, G.R.; Alonso-Cabrera, J.A.; Marcuello, C.; Ortiz-Perez, M.; Benito-Lopez, F.; Colom, A.; Basabe-Desmonts, L.; Saez, J. Fabrication and Characterization of PEDOT:PSS-Based Microstructured Electrodes for In Vitro Cell Culture. Adv. Mater. Interfaces 2025, 12, 2500097. [Google Scholar] [CrossRef]
- Xu, T.; Ji, W.; Wang, X.; Zhang, Y.; Zeng, H.; Mao, L.; Zhang, M. Support-Free PEDOT:PSS Fibers as Multifunctional Microelectrodes for In Vivo Neural Recording and Modulation. Angew. Chem. Int. Ed. 2022, 61, e202115074. [Google Scholar] [CrossRef]
- Bi, C.X.; Jin, K.Q.; Yan, J.; Qin, Y.; Hong, F.; Huang, W.H.; Liu, Y.L. Nanofiber-based Stretchable Electrodes for Oriented Culture and Mechanotransduction Monitoring of Smooth Muscle Cells. ACS Sens. 2023, 8, 3248–3256. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, X.; Tan, S.; Li, Z.; Sun, J.; Li, Y.; Xie, Z.; Li, Z.; Han, F.; Liu, Y. Design Strategies of PEDOT:PSS-Based Conductive Hydrogels and Their Applications in Health Monitoring. Polymers 2025, 17, 1192. [Google Scholar] [CrossRef]
- Barrejón, M.; Vara, H.; Alves-Sampaio, A.; Uceta, H.; Collazos-Castro, J.E. Electroactive covalent linkers for enhancing conducting polymer adhesion, charge transfer, and biological integration of PEDOT-coated carbon microfibers. Carbon 2025, 232, 119820. [Google Scholar] [CrossRef]
- Mukhatyar, V.J.; Salmerón-Sánchez, M.; Rudra, S.; Mukhopadaya, S.; Barker, T.H.; García, A.J.; Bellamkonda, R.V. Role of fibronectin in topographical guidance of neurite extension on electrospun fibers. Biomaterials 2011, 32, 3958–3968. [Google Scholar] [CrossRef]
- Torres-Mejía, E.; Trümbach, D.; Kleeberger, C.; Dornseifer, U.; Orschmann, T.; Bäcker, T.; Brenke, J.K.; Hadian, K.; Wurst, W.; López-Schier, H.; et al. Sox2 controls Schwann cell self-organization through fibronectin fibrillogenesis. Sci. Rep. 2020, 10, 1984. [Google Scholar] [CrossRef] [PubMed]
- Mosahebi, A.; Wiberg, M.; Terenghi, G. Addition of fibronectin to alginate matrix improves peripheral nerve regeneration in tissue-engineered conduits. Tissue Eng. 2003, 9, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Baron-Van Evercooren, A.; Kleinman, H.K.; Seppä, H.; Rentier, B.; Dubois-Dalcq, M. Fibronectin promotes rat Schwann cell growth and motility. J. Cell Biol. 1982, 93, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro-Resende, V.T.; Carrier-Ruiz, A.; Lemes, R.M.; Reis, R.A.; Mendez-Otero, R. Bone marrow-derived fibroblast growth factor-2 induces glial cell proliferation in the regenerating peripheral nervous system. Mol. Neurodegener. 2012, 7, 34. [Google Scholar] [CrossRef]
- Rodriguez-Sanchez, D.N.; de Carvalho, L.A.M.; Mancilla-Corzo, I.; Cartarozzi, L.P.; Safari, S.; Ermis, M.; d’Ávila, M.A.; Oliveira, A.L.R. Long-gap sciatic nerve regeneration using 3D-printed nerve conduits with controlled FGF-2 release. ACS Appl. Mater. Interfaces 2025, 17, 40237–40257. [Google Scholar] [CrossRef]
- Monje, P.V.; Sant, D.; Wang, G. Phenotypic and functional characteristics of human Schwann cells as revealed by cell-based assays and RNA-seq. Mol. Neurobiol. 2018, 55, 6637–6660. [Google Scholar] [CrossRef]
- Cerro, P.D.; Barriga-Martín, A.; Vara, H.; Romero-Muñoz, L.M.; Rodríguez-De-Lope, Á.; Collazos-Castro, J.E. Neuropathological and motor impairments after incomplete cervical spinal cord injury in pigs. J. Neurotrauma 2021, 38, 2956–2977. [Google Scholar] [CrossRef]
- Burrell, J.C.; Browne, K.D.; Dutton, J.L.; Laimo, F.A.; Das, S.; Brown, D.P.; Sanford, R.; Dmitriy, P.; Zarina, A.; Harry, C.L.; et al. A porcine model of peripheral nerve injury enabling ultra-long regenerative distances: Surgical approach, recovery kinetics, and clinical relevance. Neurosurgery 2020, 87, 833–846. [Google Scholar] [CrossRef]
- Kornfeld, T.; Nessler, J.; Helmer, C.; Hannemann, R.; Waldmann, K.H.; Peck, C.T.; Hoffmann, P.; Brandes, G.; Vogt, P.M.; Radtke, C. Spider silk nerve graft promotes axonal regeneration on long distance nerve defect in a sheep model. Biomaterials 2021, 271, 120692. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.H.; Burrell, J.C.; Browne, K.D.; Katiyar, K.S.; Ezra, M.I.; Dutton, J.L.; Morand, J.P.; Struzyna, L.A.; Laimo, F.A.; Chen, H.I.; et al. Tissue-engineered grafts exploit axon-facilitated axon regeneration and pathway protection to enable recovery after 5-cm nerve defects in pigs. Sci. Adv. 2022, 8, eabm3291. [Google Scholar] [CrossRef] [PubMed]
- Holzer, P.; Chang, E.J.; Rogers, K.; Tarlton, J.; Lu, D.; Gillespie, N.; Adkins, J.; Metea, M.; LaRochelle, A.; Wicks, J.; et al. Large-gap peripheral nerve repair using xenogeneic transplants in rhesus macaques. Xenotransplantation 2023, 30, e12792. [Google Scholar] [CrossRef] [PubMed]
- Contreras, E.; Traserra, S.; Bolívar, S.; Forés, J.; Jose-Cunilleras, E.; Delgado-Martínez, I.; García, F.; Udina, E.; Navarro, X. Repair of long peripheral nerve defects in sheep: A translational model for nerve regeneration. Int. J. Mol. Sci. 2023, 24, 1333. [Google Scholar] [CrossRef]
- Chu, T.H.; Midha, R. Limited Myelination Capacity in Human Schwann Cells in Experimental Models in Comparison to Rodent and Porcine Schwann Cells. Int. J. Mol. Sci. 2025, 26, 6457. [Google Scholar] [CrossRef]
- Shen, N.; Liang, Q.; Liu, Y.; Lai, B.; Li, W.; Wang, Z.; Li, S. Charge-balanced biphasic electrical stimulation inhibits neurite extension of spiral ganglion neurons. Neurosci. Lett. 2016, 624, 92–99. [Google Scholar] [CrossRef]
- Koppes, A.N.; Nordberg, A.L.; Paolillo, G.; Goodsell, N.; Darwish, H.; Zhang, L.; Thompson, D.M. Electrical stimulation of Schwann cells promotes sustained increases in neurite outgrowth. Tissue Eng. Part A 2013, 20, 494–506. [Google Scholar] [CrossRef]
- Aman, M.; Mayrhofer-Schmid, M.; Schwarz, D.; Bendszus, M.; Daeschler, S.C.; Klemm, T.; Kneser, U.; Harhaus, L.; Arne, H. Boecker Avoiding scar tissue formation of peripheral nerves with the help of an acellular collagen matrix. PLoS ONE 2023, 18, e0289677. [Google Scholar] [CrossRef]
- Gant, K.L.; Guest, J.D.; Palermo, A.E.; Vedantam, A.; Jimsheleishvili, G.; Bunge, M.B.; Brooks, A.E.; Anderson, K.D.; Thomas, C.K.; Santamaria, A.J.; et al. Phase 1 Safety Trial of Autologous Human Schwann Cell Transplantation in Chronic Spinal Cord Injury. J. Neurotrauma 2022, 39, 285–299. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alves-Sampaio, A.; Collazos-Castro, J.E. Biphasic Electrical Stimulation of Schwann Cells on Conducting Polymer-Coated Carbon Microfibers. Int. J. Mol. Sci. 2025, 26, 8102. https://doi.org/10.3390/ijms26168102
Alves-Sampaio A, Collazos-Castro JE. Biphasic Electrical Stimulation of Schwann Cells on Conducting Polymer-Coated Carbon Microfibers. International Journal of Molecular Sciences. 2025; 26(16):8102. https://doi.org/10.3390/ijms26168102
Chicago/Turabian StyleAlves-Sampaio, Alexandra, and Jorge E. Collazos-Castro. 2025. "Biphasic Electrical Stimulation of Schwann Cells on Conducting Polymer-Coated Carbon Microfibers" International Journal of Molecular Sciences 26, no. 16: 8102. https://doi.org/10.3390/ijms26168102
APA StyleAlves-Sampaio, A., & Collazos-Castro, J. E. (2025). Biphasic Electrical Stimulation of Schwann Cells on Conducting Polymer-Coated Carbon Microfibers. International Journal of Molecular Sciences, 26(16), 8102. https://doi.org/10.3390/ijms26168102




