Characteristics of Intestinal Barrier State and Immunoglobulin-Bound Fraction of Stool Microbiota in Advanced Melanoma Patients Undergoing Anti-PD-1 Therapy
Abstract
1. Introduction
2. Results
2.1. Intestinal Barrier State Biomarkers
2.1.1. A High Baseline Concentration of Fecal SIgA Is Associated with Response and Clinical Benefit from the Anti-PD-1 Therapy
2.1.2. Elevated Levels of Intestinal Barrier State Biomarkers in Advanced Melanoma Patients
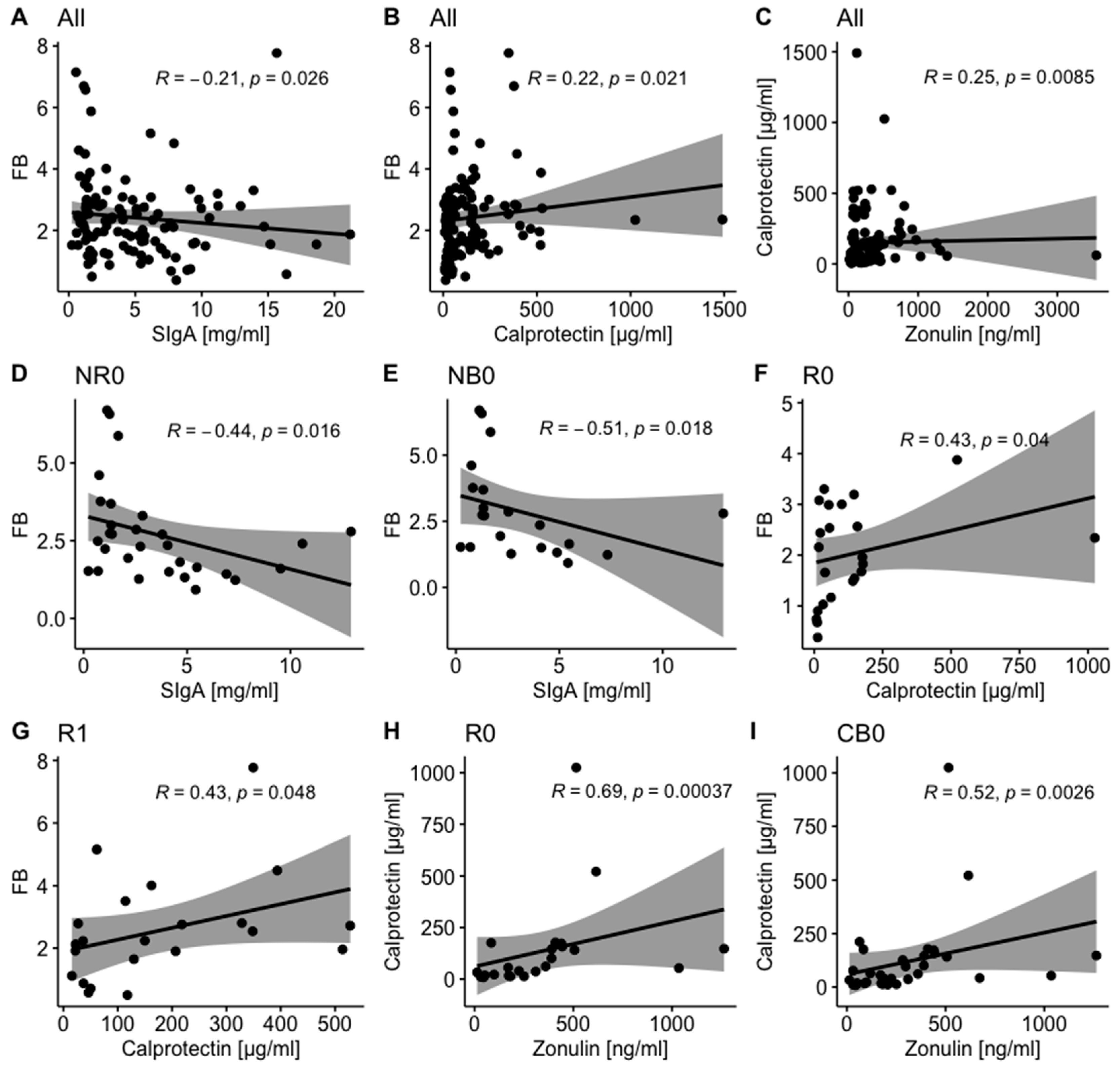
2.1.3. Mutual Correlations Between Intestinal Barrier State Biomarkers and Total Stool Microbiota Composition
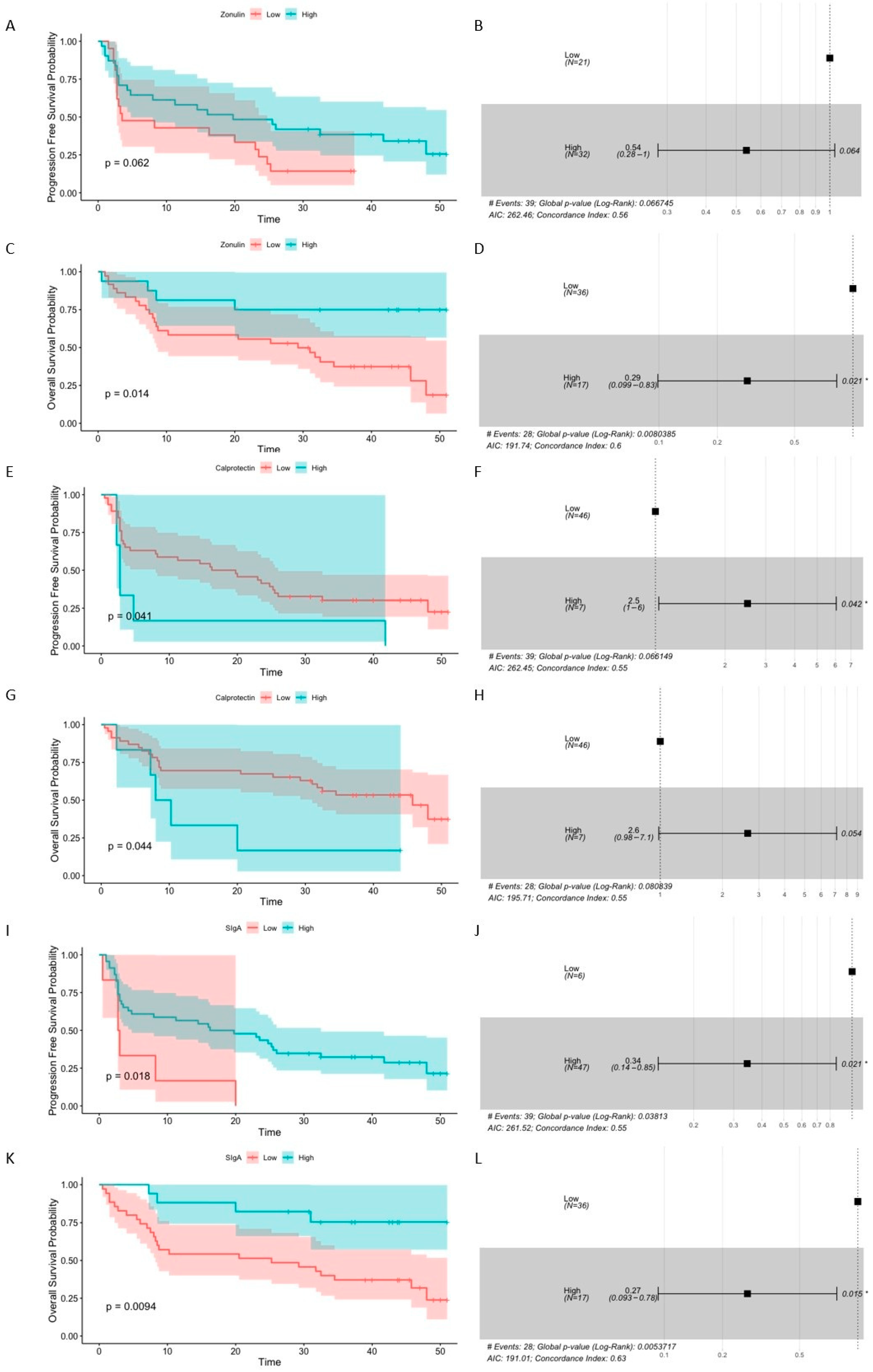
2.1.4. Baseline Levels of Intestinal Barrier State Biomarkers Are Associated with the Survival Outcomes
2.2. Ig-Bound Stool Microbiota Fraction
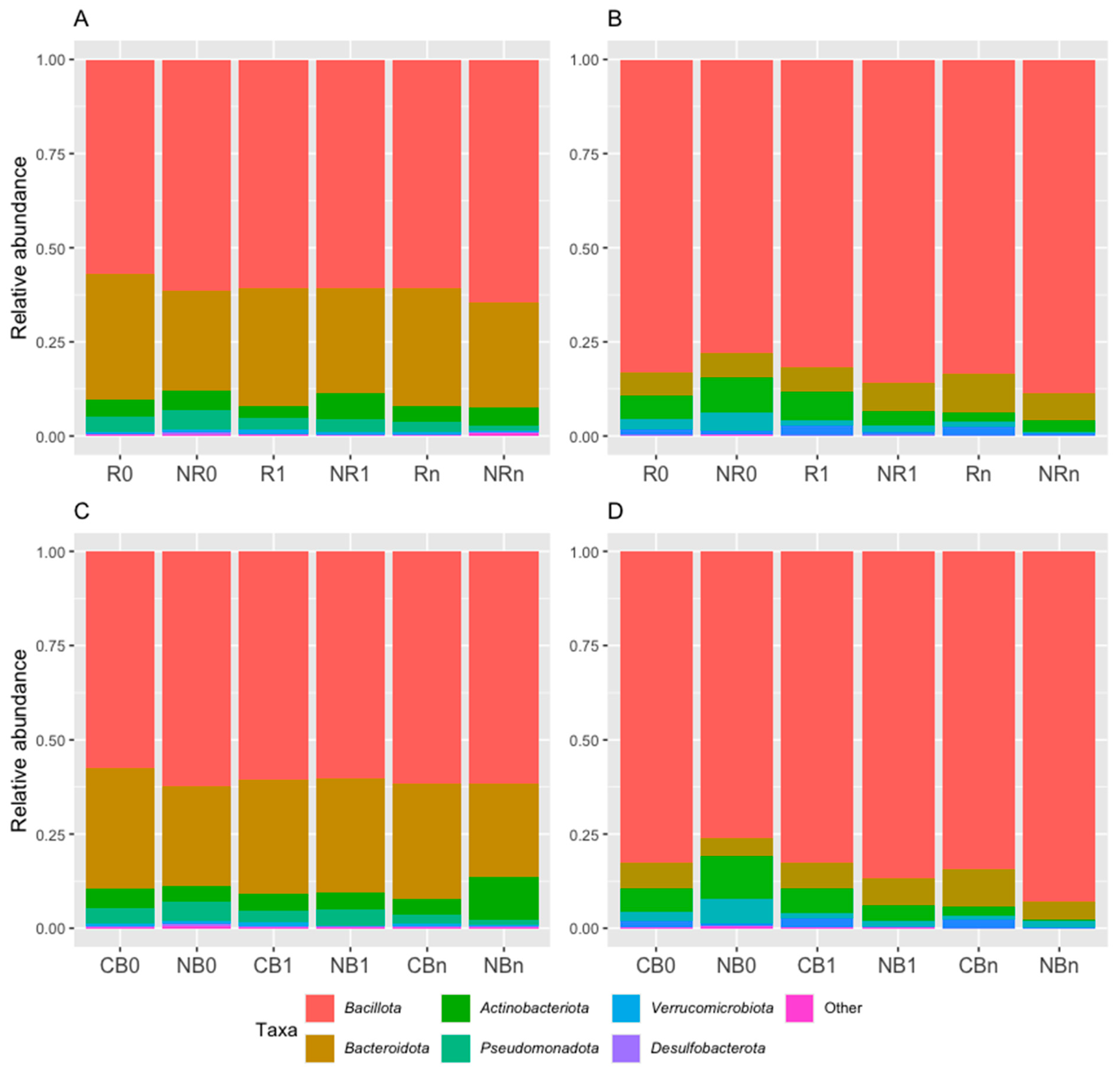
2.2.1. The Dominance of Bacillota Phylum in the Ig-Bound Stool Microbiota and Changes in the Taxonomic Profile at the Phylum Level over the Study Period
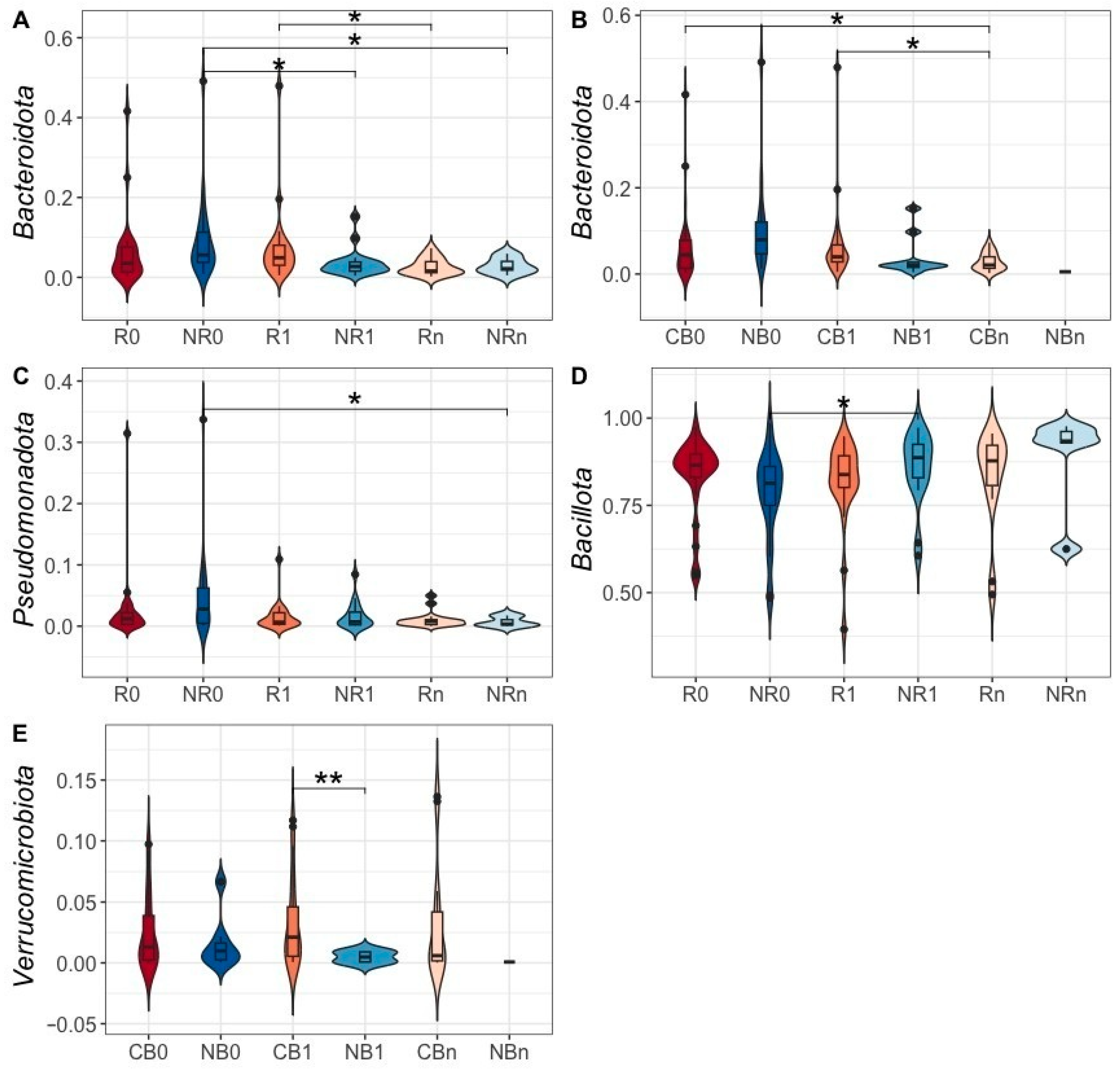
2.2.2. Differences in the Ig-Bound Stool Microbiota Signatures Between Patients with Favorable and Unfavorable Clinical Outcomes at Baseline (at T0) and During the Anti-PD-1 Therapy (at T1 and Tn)
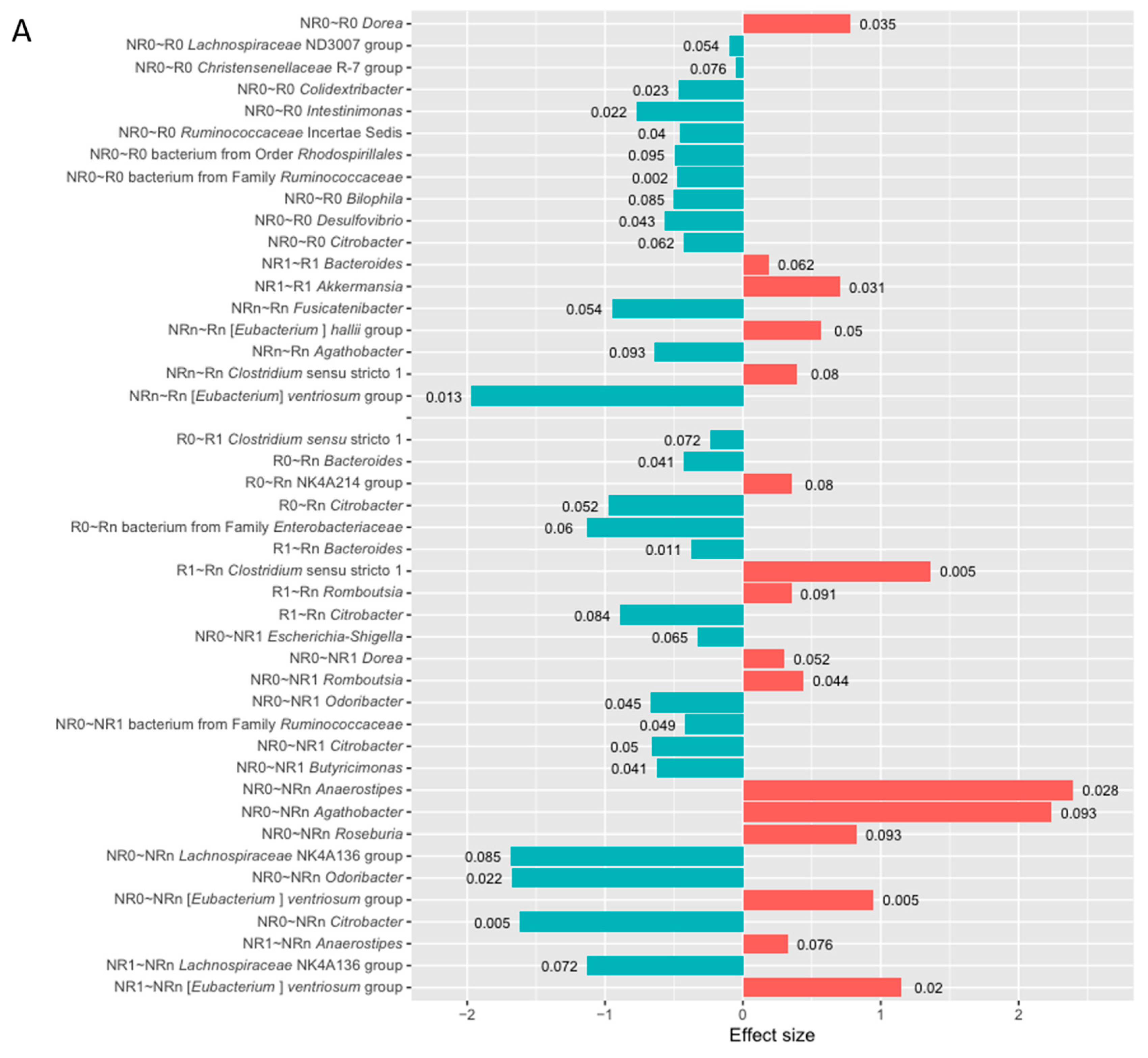
2.2.3. Changes in the Ig-Bound Stool Microbiota Signatures During the Anti-PD-1 Therapy in Patients with Favorable and Unfavorable Clinical Outcomes (T0 vs. T1, T0 vs. Tn, and T1 vs. Tn)
2.2.4. Comparison Between the Ig-Bound and Total Stool Microbiota Composition
3. Discussion
4. Materials and Methods
4.1. Study Cohort Description and Collection of Clinical Data and Stool Samples
4.2. Measurement of the Intestinal Barrier State Biomarker Concentration
4.3. Purification of Ig-Bound Stool Microbiota Fraction
4.4. Metagenomic DNA Extraction and Sequencing of the V3–V4 Regions in the 16S rRNA Gene
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Liu, X.; Chen, Y.; Zhang, S.; Dong, L. Gut Microbiota-Mediated Immunomodulation in Tumor. J. Exp. Clin. Cancer Res. 2021, 40, 221. [Google Scholar] [CrossRef]
- McCulloch, J.A.; Davar, D.; Rodrigues, R.R.; Badger, J.H.; Fang, J.R.; Cole, A.M.; Balaji, A.K.; Vetizou, M.; Prescott, S.M.; Fernandes, M.R.; et al. Intestinal Microbiota Signatures of Clinical Response and Immune-Related Adverse Events in Melanoma Patients Treated with Anti-PD-1. Nat. Med. 2022, 28, 545–556. [Google Scholar] [CrossRef]
- Pietrzak, B.; Tomela, K.; Olejnik-Schmidt, A.; Galus, Ł.; Mackiewicz, J.; Kaczmarek, M.; Mackiewicz, A.; Schmidt, M. A Clinical Outcome of the Anti-PD-1 Therapy of Melanoma in Polish Patients Is Mediated by Population-Specific Gut Microbiome Composition. Cancers 2022, 14, 5369. [Google Scholar] [CrossRef]
- Björk, J.R.; Bolte, L.A.; Maltez Thomas, A.; Lee, K.A.; Rossi, N.; Wind, T.T.; Smit, L.M.; Armanini, F.; Asnicar, F.; Blanco-Miguez, A.; et al. Longitudinal Gut Microbiome Changes in Immune Checkpoint Blockade-Treated Advanced Melanoma. Nat. Med. 2024, 30, 785–796. [Google Scholar] [CrossRef] [PubMed]
- Gunjur, A.; Shao, Y.; Rozday, T.; Klein, O.; Mu, A.; Haak, B.W.; Markman, B.; Kee, D.; Carlino, M.S.; Underhill, C.; et al. A Gut Microbial Signature for Combination Immune Checkpoint Blockade across Cancer Types. Nat. Med. 2024, 30, 797–809. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Deng, Y.; Chu, Q.; Zhang, P. Gut Microbiome and Cancer Immunotherapy. Cancer Lett. 2019, 447, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Thursby, E.; Juge, N. Introduction to the Human Gut Microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef]
- Ouaknine Krief, J.; Helly de Tauriers, P.; Dumenil, C.; Neveux, N.; Dumoulin, J.; Giraud, V.; Labrune, S.; Tisserand, J.; Julie, C.; Emile, J.-F.; et al. Role of Antibiotic Use, Plasma Citrulline and Blood Microbiome in Advanced Non-Small Cell Lung Cancer Patients Treated with Nivolumab. J. Immunother. Cancer 2019, 7, 176. [Google Scholar] [CrossRef]
- Jendraszak, M.; Gałęcka, M.; Kotwicka, M.; Schwiertz, A.; Regdos, A.; Pazgrat-Patan, M.; Andrusiewicz, M. Impact of Biometric Patient Data, Probiotic Supplementation, and Selected Gut Microorganisms on Calprotectin, Zonulin, and sIgA Concentrations in the Stool of Adults Aged 18–74 Years. Biomolecules 2022, 12, 1781. [Google Scholar] [CrossRef]
- Fasano, A. All Disease Begins in the (Leaky) Gut: Role of Zonulin-Mediated Gut Permeability in the Pathogenesis of Some Chronic Inflammatory Diseases. F1000Research 2020, 9, 69. [Google Scholar] [CrossRef] [PubMed]
- Sturgeon, C.; Fasano, A. Zonulin, a Regulator of Epithelial and Endothelial Barrier Functions, and Its Involvement in Chronic Inflammatory Diseases. Tissue Barriers 2016, 4, e1251384. [Google Scholar] [CrossRef] [PubMed]
- Jukic, A.; Bakiri, L.; Wagner, E.F.; Tilg, H.; Adolph, T.E. Calprotectin: From Biomarker to Biological Function. Gut 2021, 70, 1978–1988. [Google Scholar] [CrossRef]
- León, E.D.; Francino, M.P. Roles of Secretory Immunoglobulin A in Host-Microbiota Interactions in the Gut Ecosystem. Front. Microbiol. 2022, 13, 880484. [Google Scholar] [CrossRef]
- Pietrzak, B.; Tomela, K.; Olejnik-Schmidt, A.; Mackiewicz, A.; Schmidt, M. Secretory IgA in Intestinal Mucosal Secretions as an Adaptive Barrier against Microbial Cells. Int. J. Mol. Sci. 2020, 21, 9254. [Google Scholar] [CrossRef]
- Smeekens, J.M.; Johnson-Weaver, B.T.; Hinton, A.L.; Azcarate-Peril, M.A.; Moran, T.P.; Immormino, R.M.; Kesselring, J.R.; Steinbach, E.C.; Orgel, K.A.; Staats, H.F.; et al. Fecal IgA, Antigen Absorption, and Gut Microbiome Composition Are Associated with Food Antigen Sensitization in Genetically Susceptible Mice. Front. Immunol. 2021, 11, 599637. [Google Scholar] [CrossRef] [PubMed]
- Immundiagnostik AG. IDK® sIgA ELISA Manual; Immundiagnostik AG: Bensheim, Germany, 2020. [Google Scholar]
- Immundiagnostik AG. IDK® Calprotectin ELISA Manual; Immundiagnostik AG: Bensheim, Germany, 2019. [Google Scholar]
- Immundiagnostik AG. IDK® Zonulin ELISA Manual; Immundiagnostik AG: Bensheim, Germany, 2019. [Google Scholar]
- Kusnadi, Y.; Saleh, M.I.; Ali, Z.; Hermansyah, H.; Murti, K.; Hafy, Z.; Yuristo, N.S.E. Firmicutes/Bacteroidetes Ratio of Gut Microbiota and Its Relationships with Clinical Parameters of Type 2 Diabetes Mellitus: A Systematic Review. Open Access Maced. J. Med. Sci. 2023, 11, 67–72. [Google Scholar] [CrossRef]
- Stojanov, S.; Berlec, A.; Štrukelj, B. The Influence of Probiotics on the Firmicutes/Bacteroidetes Ratio in the Treatment of Obesity and Inflammatory Bowel Disease. Microorganisms 2020, 8, 1715. [Google Scholar] [CrossRef]
- Drymel, B.; Tomela, K.; Galus, Ł.; Olejnik-Schmidt, A.; Mackiewicz, J.; Kaczmarek, M.; Mackiewicz, A.; Schmidt, M. Circulating Cell-Free Microbial DNA Signatures and Plasma Soluble CD14 Level Are Associated with Clinical Outcomes of Anti-PD-1 Therapy in Advanced Melanoma Patients. Int. J. Mol. Sci. 2024, 25, 12982. [Google Scholar] [CrossRef]
- Yi, M.; Yu, S.; Qin, S.; Liu, Q.; Xu, H.; Zhao, W.; Chu, Q.; Wu, K. Gut Microbiome Modulates Efficacy of Immune Checkpoint Inhibitors. J. Hematol. Oncol. 2018, 11, 47. [Google Scholar] [CrossRef] [PubMed]
- Nomura, M.; Nagatomo, R.; Doi, K.; Shimizu, J.; Baba, K.; Saito, T.; Matsumoto, S.; Inoue, K.; Muto, M. Association of Short-Chain Fatty Acids in the Gut Microbiome with Clinical Response to Treatment with Nivolumab or Pembrolizumab in Patients with Solid Cancer Tumors. JAMA Netw. Open 2020, 3, e202895. [Google Scholar] [CrossRef] [PubMed]
- Ponziani, F.R.; De Luca, A.; Picca, A.; Marzetti, E.; Petito, V.; Del Chierico, F.; Reddel, S.; Paroni Sterbini, F.; Sanguinetti, M.; Putignani, L.; et al. Gut Dysbiosis and Fecal Calprotectin Predict Response to Immune Checkpoint Inhibitors in Patients with Hepatocellular Carcinoma. Hepatol. Commun. 2022, 6, 1492–1501. [Google Scholar] [CrossRef]
- Coutzac, C.; Jouniaux, J.-M.; Paci, A.; Schmidt, J.; Mallardo, D.; Seck, A.; Asvatourian, V.; Cassard, L.; Saulnier, P.; Lacroix, L.; et al. Systemic Short Chain Fatty Acids Limit Antitumor Effect of CTLA-4 Blockade in Hosts with Cancer. Nat. Commun. 2020, 11, 2168. [Google Scholar] [CrossRef]
- Verdam, F.J.; Fuentes, S.; de Jonge, C.; Zoetendal, E.G.; Erbil, R.; Greve, J.W.; Buurman, W.A.; de Vos, W.M.; Rensen, S.S. Human Intestinal Microbiota Composition Is Associated with Local and Systemic Inflammation in Obesity. Obesity 2013, 21, E607–E615. [Google Scholar] [CrossRef] [PubMed]
- Hiippala, K.; Kainulainen, V.; Suutarinen, M.; Heini, T.; Bowers, J.R.; Jasso-Selles, D.; Lemmer, D.; Valentine, M.; Barnes, R.; Engelthaler, D.M.; et al. Isolation of Anti-Inflammatory and Epithelium Reinforcing Bacteroides and Parabacteroides Spp. from A Healthy Fecal Donor. Nutrients 2020, 12, 935. [Google Scholar] [CrossRef]
- Rajput, M.; Momin, T.; Singh, A.; Banerjee, S.; Villasenor, A.; Sheldon, J.; Paudel, P.; Rajput, R. Determining the Association between Gut Microbiota and Its Metabolites with Higher Intestinal Immunoglobulin A Response. Vet. Anim. Sci. 2023, 19, 100279. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Mogno, I.; Contijoch, E.J.; Borgerding, J.N.; Aggarwala, V.; Li, Z.; Siu, S.; Grasset, E.K.; Helmus, D.S.; Dubinsky, M.C.; et al. Fecal IgA Levels Are Determined by Strain-Level Differences in Bacteroides Ovatus and Are Modifiable by Gut Microbiota Manipulation. Cell Host Microbe 2020, 27, 467–475.e6. [Google Scholar] [CrossRef] [PubMed]
- Passarelli, A.; Mannavola, F.; Stucci, L.S.; Tucci, M.; Silvestris, F. Immune System and Melanoma Biology: A Balance between Immunosurveillance and Immune Escape. Oncotarget 2017, 8, 106132–106142. [Google Scholar] [CrossRef]
- Tomela, K.; Pietrzak, B.; Schmidt, M.; Mackiewicz, A. The Tumor and Host Immune Signature, and the Gut Microbiota as Predictive Biomarkers for Immune Checkpoint Inhibitor Response in Melanoma Patients. Life 2020, 10, 219. [Google Scholar] [CrossRef]
- Wu, C.; Qiu, Y.; Zhang, R.; Li, X.; Liang, H.; Wang, M.; Li, F.; Zhu, M.; Ye, G.; Liu, H.; et al. Association of Peripheral Basophils with Tumor M2 Macrophage Infiltration and Outcomes of the Anti-PD-1 Inhibitor plus Chemotherapy Combination in Advanced Gastric Cancer. J. Transl. Med. 2022, 20, 386. [Google Scholar] [CrossRef]
- Rosner, S.; Kwong, E.; Shoushtari, A.N.; Friedman, C.F.; Betof, A.S.; Brady, M.S.; Coit, D.G.; Callahan, M.K.; Wolchok, J.D.; Chapman, P.B.; et al. Peripheral Blood Clinical Laboratory Variables Associated with Outcomes Following Combination Nivolumab and Ipilimumab Immunotherapy in Melanoma. Cancer Med. 2018, 7, 690–697. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, B.; Li, D.; Yang, Y.; Lin, S.; Zhao, R.; Li, Y.; Peng, L. Peripheral Blood Cell Counts as Predictors of Immune-Related Adverse Events in Cancer Patients Receiving Immune Checkpoint Inhibitors: A Systematic Review and Meta-Analysis. Front. Immunol. 2025, 16, 1528084. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, C.; Peng, Z.; Qi, C.; Gong, J.; Zhang, X.; Li, J.; Shen, L. Association of Lymphocyte-to-Monocyte Ratio with Survival in Advanced Gastric Cancer Patients Treated with Immune Checkpoint Inhibitor. Front. Oncol. 2021, 11, 589022. [Google Scholar] [CrossRef]
- Nakamura, Y.; Kitano, S.; Takahashi, A.; Tsutsumida, A.; Namikawa, K.; Tanese, K.; Abe, T.; Funakoshi, T.; Yamamoto, N.; Amagai, M.; et al. Nivolumab for Advanced Melanoma: Pretreatment Prognostic Factors and Early Outcome Markers during Therapy. Oncotarget 2016, 7, 77404–77415. [Google Scholar] [CrossRef]
- Kuribayashi, T.; Kinoshita, R.; Ninomiya, K.; Makimoto, G.; Kubo, T.; Rai, K.; Ichihara, E.; Hotta, K.; Tabata, M.; Maeda, Y.; et al. Plasma S100A8/A9 Level Predicts Response to Immune Checkpoint Inhibitors in Patients with Advanced Non-Small Cell Lung Cancer. Sci. Rep. 2025, 15, 2577. [Google Scholar] [CrossRef]
- Chalkias, A.; Nikotian, G.; Koutsovasilis, A.; Bramis, J.; Manouras, A.; Mystrioti, D.; Katergiannakis, V. Patients with Colorectal Cancer Are Characterized by Increased Concentration of Fecal Hb-Hp Complex, Myeloperoxidase, and Secretory IgA. Am. J. Clin. Oncol. 2011, 34, 561. [Google Scholar] [CrossRef]
- Joshi, S.; Lewis, S.J.; Creanor, S.; Ayling, R.M. Age-Related Faecal Calprotectin, Lactoferrin and Tumour M2-PK Concentrations in Healthy Volunteers. Ann. Clin. Biochem. 2010, 47, 259–263. [Google Scholar] [CrossRef]
- Ohlsson, B.; Roth, B.; Larsson, E.; Höglund, P. Calprotectin in Serum and Zonulin in Serum and Feces Are Elevated after Introduction of a Diet with Lower Carbohydrate Content and Higher Fiber, Fat and Protein Contents. Biomed. Rep. 2017, 6, 411–422. [Google Scholar] [CrossRef]
- Marino, M.; Del Bo’, C.; Martini, D.; Perna, S.; Porrini, M.; Cherubini, A.; Gargari, G.; Meroño, T.; Hidalgo-Liberona, N.; Andres-Lacueva, C.; et al. A (Poly)Phenol-Rich Diet Reduces Serum and Faecal Calprotectin in Older Adults with Increased Intestinal Permeability: The MaPLE Randomised Controlled Trial. BMC Geriatr. 2024, 24, 707. [Google Scholar] [CrossRef] [PubMed]
- Lopez, S.H.; Jalving, M.; Fehrmann, R.S.N.; Nagengast, W.B.; de Vries, E.G.E.; de Haan, J.J. The Gut Wall’s Potential as a Partner for Precision Oncology in Immune Checkpoint Treatment. Cancer Treat. Rev. 2022, 107, 102406. [Google Scholar] [CrossRef] [PubMed]
- Renga, G.; Nunzi, E.; Pariano, M.; Puccetti, M.; Bellet, M.M.; Pieraccini, G.; D’Onofrio, F.; Santarelli, I.; Stincardini, C.; Aversa, F.; et al. Optimizing Therapeutic Outcomes of Immune Checkpoint Blockade by a Microbial Tryptophan Metabolite. J. Immunother. Cancer 2022, 10, e003725. [Google Scholar] [CrossRef]
- Chaput, N.; Lepage, P.; Coutzac, C.; Soularue, E.; Le Roux, K.; Monot, C.; Boselli, L.; Routier, E.; Cassard, L.; Collins, M.; et al. Baseline Gut Microbiota Predicts Clinical Response and Colitis in Metastatic Melanoma Patients Treated with Ipilimumab. Ann. Oncol. 2017, 28, 1368–1379, Erratum in Ann. Oncol. 2019, 30, 2012. [Google Scholar] [CrossRef] [PubMed]
- Nearing, J.T.; Douglas, G.M.; Hayes, M.G.; MacDonald, J.; Desai, D.K.; Allward, N.; Jones, C.M.A.; Wright, R.J.; Dhanani, A.S.; Comeau, A.M.; et al. Microbiome Differential Abundance Methods Produce Different Results across 38 Datasets. Nat. Commun. 2022, 13, 342. [Google Scholar] [CrossRef]
- de la Cuesta-Zuluaga, J.; Escobar, J.S. Considerations for Optimizing Microbiome Analysis Using a Marker Gene. Front. Nutr. 2016, 3, 26. [Google Scholar] [CrossRef]
- Kawamoto, S.; Tran, T.H.; Maruya, M.; Suzuki, K.; Doi, Y.; Tsutsui, Y.; Kato, L.M.; Fagarasan, S. The Inhibitory Receptor PD-1 Regulates IgA Selection and Bacterial Composition in the Gut. Science 2012, 336, 485–489. [Google Scholar] [CrossRef]
- Maruya, M.; Kawamoto, S.; Kato, L.M.; Fagarasan, S. Impaired Selection of IgA and Intestinal Dysbiosis Associated with PD-1-Deficiency. Gut Microbes 2013, 4, 165. [Google Scholar] [CrossRef]
- Zhang, X.-Y.; Liu, Z.-M.; Zhang, H.; Li, Y.-S.; Wen, S.-H.; Shen, J.-T.; Liu, K.-X. Decreased PD-1/PD-L1 Expression Is Associated with the Reduction in Mucosal Immunoglobulin A in Mice with Intestinal Ischemia Reperfusion. Dig. Dis. Sci. 2015, 60, 2662–2669. [Google Scholar] [CrossRef]
- Rollenske, T.; Burkhalter, S.; Muerner, L.; Von Gunten, S.; Lukasiewicz, J.; Wardemann, H.; Macpherson, A.J. Parallelism of Intestinal Secretory IgA Shapes Functional Microbial Fitness. Nature 2021, 598, 657–661. [Google Scholar] [CrossRef]
- Frankel, A.E.; Coughlin, L.A.; Kim, J.; Froehlich, T.W.; Xie, Y.; Frenkel, E.P.; Koh, A.Y. Metagenomic Shotgun Sequencing and Unbiased Metabolomic Profiling Identify Specific Human Gut Microbiota and Metabolites Associated with Immune Checkpoint Therapy Efficacy in Melanoma Patients. Neoplasia 2017, 19, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Matson, V.; Fessler, J.; Bao, R.; Chongsuwat, T.; Zha, Y.; Alegre, M.-L.; Luke, J.J.; Gajewski, T.F. The Commensal Microbiome Is Associated with Anti-PD-1 Efficacy in Metastatic Melanoma Patients. Science 2018, 359, 104–108. [Google Scholar] [CrossRef]
- Routy, B.; Le Chatelier, E.; Derosa, L.; Duong, C.P.M.; Alou, M.T.; Daillère, R.; Fluckiger, A.; Messaoudene, M.; Rauber, C.; Roberti, M.P.; et al. Gut Microbiome Influences Efficacy of PD-1–Based Immunotherapy against Epithelial Tumors. Science 2018, 359, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Peters, B.A.; Wilson, M.; Moran, U.; Pavlick, A.; Izsak, A.; Wechter, T.; Weber, J.S.; Osman, I.; Ahn, J. Relating the Gut Metagenome and Metatranscriptome to Immunotherapy Responses in Melanoma Patients. Genome Med. 2019, 11, 61. [Google Scholar] [CrossRef]
- Derosa, L.; Routy, B.; Fidelle, M.; Iebba, V.; Alla, L.; Pasolli, E.; Segata, N.; Desnoyer, A.; Pietrantonio, F.; Ferrere, G.; et al. Gut Bacteria Composition Drives Primary Resistance to Cancer Immunotherapy in Renal Cell Carcinoma Patients. Eur. Urol. 2020, 78, 195–206. [Google Scholar] [CrossRef]
- Richards, A.F.; Baranova, D.E.; Pizzuto, M.S.; Jaconi, S.; Willsey, G.G.; Torres-Velez, F.J.; Doering, J.E.; Benigni, F.; Corti, D.; Mantis, N.J. Recombinant Human Secretory IgA Induces Salmonella Typhimurium Agglutination and Limits Bacterial Invasion into Gut-Associated Lymphoid Tissues. ACS Infect. Dis. 2021, 7, 1221–1235. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.; Roy, N.; Ali, H.; Naeem, M. Fecal Microbiota Transplants for Inflammatory Bowel Disease Treatment: Synthetic- and Engineered Communities-Based Microbiota Transplants Are the Future. Gastroenterol. Res. Pract. 2022, 2022, 9999925. [Google Scholar] [CrossRef]
- Szmulski, Ł. Obwieszczenie Nr 65 Ministra Zdrowia z Dnia 30 Sierpnia 2019 r. w Sprawie Wykazu Refundowanych Leków, Środków Spożywczych Specjalnego Przeznaczenia Żywieniowego Oraz Wyrobów Medycznych. J. Minist. Health-Pol 2019, 65, 906–913. [Google Scholar]
- Dore, J.; Ehrlich, S.D.; Levenez, F.; Pelletier, E.; Alberti, A.; Bertand, L. IHMS_SOP 05 V1; Standard Operating Procedure for Fecal Samples Preserved in Stabilizing Solution Self-Collection, Laboratory Analysis Handled Within 24 Hours to 7 Days (24 Hours < X ≤ 7 Days); International Human Microbiome Standards (IHMS): Jouy-en-Josas, France, 2015. [Google Scholar]
- Madhwani, T.; McBain, A.J. The Application of Magnetic Bead Selection to Investigate Interactions between the Oral Microbiota and Salivary Immunoglobulins. PLoS ONE 2016, 11, e0158288. [Google Scholar] [CrossRef]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of General 16S Ribosomal RNA Gene PCR Primers for Classical and Next-Generation Sequencing-Based Diversity Studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef]
- Schmidt, M. DADA2 Formatted Silva SSU Taxonomic Training Data (Silva Version 138.1) with Emended Description of the Genus Lactobacillus Beijerinck 1901. Zenodo 2023. [Google Scholar] [CrossRef]
- Houtman, T.A.; Eckermann, H.A.; Smidt, H.; de Weerth, C. Gut Microbiota and BMI throughout Childhood: The Role of Firmicutes, Bacteroidetes, and Short-Chain Fatty Acid Producers. Sci. Rep. 2022, 12, 3140. [Google Scholar] [CrossRef]
- Lozupone, C.A.; Hamady, M.; Kelley, S.T.; Knight, R. Quantitative and Qualitative β Diversity Measures Lead to Different Insights into Factors That Structure Microbial Communities. Appl. Environ. Microbiol. 2007, 73, 1576–1585. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, A.T.; Katsyv, I.; Song, W.-M.; Wang, M.; Zhang, B. DGCA: A Comprehensive R Package for Differential Gene Correlation Analysis. BMC Syst. Biol. 2016, 10, 106. [Google Scholar] [CrossRef] [PubMed]
- Legendre, P.; Gallagher, E.D. Ecologically Meaningful Transformations for Ordination of Species Data. Oecologia 2001, 129, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Yerke, A.; Fry Brumit, D.; Fodor, A.A. Proportion-Based Normalizations Outperform Compositional Data Transformations in Machine Learning Applications. Microbiome 2024, 12, 45. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Drymel, B.; Tomela, K.; Galus, Ł.; Olejnik-Schmidt, A.; Mackiewicz, J.; Kaczmarek, M.; Mackiewicz, A.; Schmidt, M. Characteristics of Intestinal Barrier State and Immunoglobulin-Bound Fraction of Stool Microbiota in Advanced Melanoma Patients Undergoing Anti-PD-1 Therapy. Int. J. Mol. Sci. 2025, 26, 8063. https://doi.org/10.3390/ijms26168063
Drymel B, Tomela K, Galus Ł, Olejnik-Schmidt A, Mackiewicz J, Kaczmarek M, Mackiewicz A, Schmidt M. Characteristics of Intestinal Barrier State and Immunoglobulin-Bound Fraction of Stool Microbiota in Advanced Melanoma Patients Undergoing Anti-PD-1 Therapy. International Journal of Molecular Sciences. 2025; 26(16):8063. https://doi.org/10.3390/ijms26168063
Chicago/Turabian StyleDrymel, Bernadeta, Katarzyna Tomela, Łukasz Galus, Agnieszka Olejnik-Schmidt, Jacek Mackiewicz, Mariusz Kaczmarek, Andrzej Mackiewicz, and Marcin Schmidt. 2025. "Characteristics of Intestinal Barrier State and Immunoglobulin-Bound Fraction of Stool Microbiota in Advanced Melanoma Patients Undergoing Anti-PD-1 Therapy" International Journal of Molecular Sciences 26, no. 16: 8063. https://doi.org/10.3390/ijms26168063
APA StyleDrymel, B., Tomela, K., Galus, Ł., Olejnik-Schmidt, A., Mackiewicz, J., Kaczmarek, M., Mackiewicz, A., & Schmidt, M. (2025). Characteristics of Intestinal Barrier State and Immunoglobulin-Bound Fraction of Stool Microbiota in Advanced Melanoma Patients Undergoing Anti-PD-1 Therapy. International Journal of Molecular Sciences, 26(16), 8063. https://doi.org/10.3390/ijms26168063








