Photodynamic Activation of a Novel Chlorophyll-Enriched Green Propolis Compound Triggers Apoptosis in Renal Cell Carcinoma
Abstract
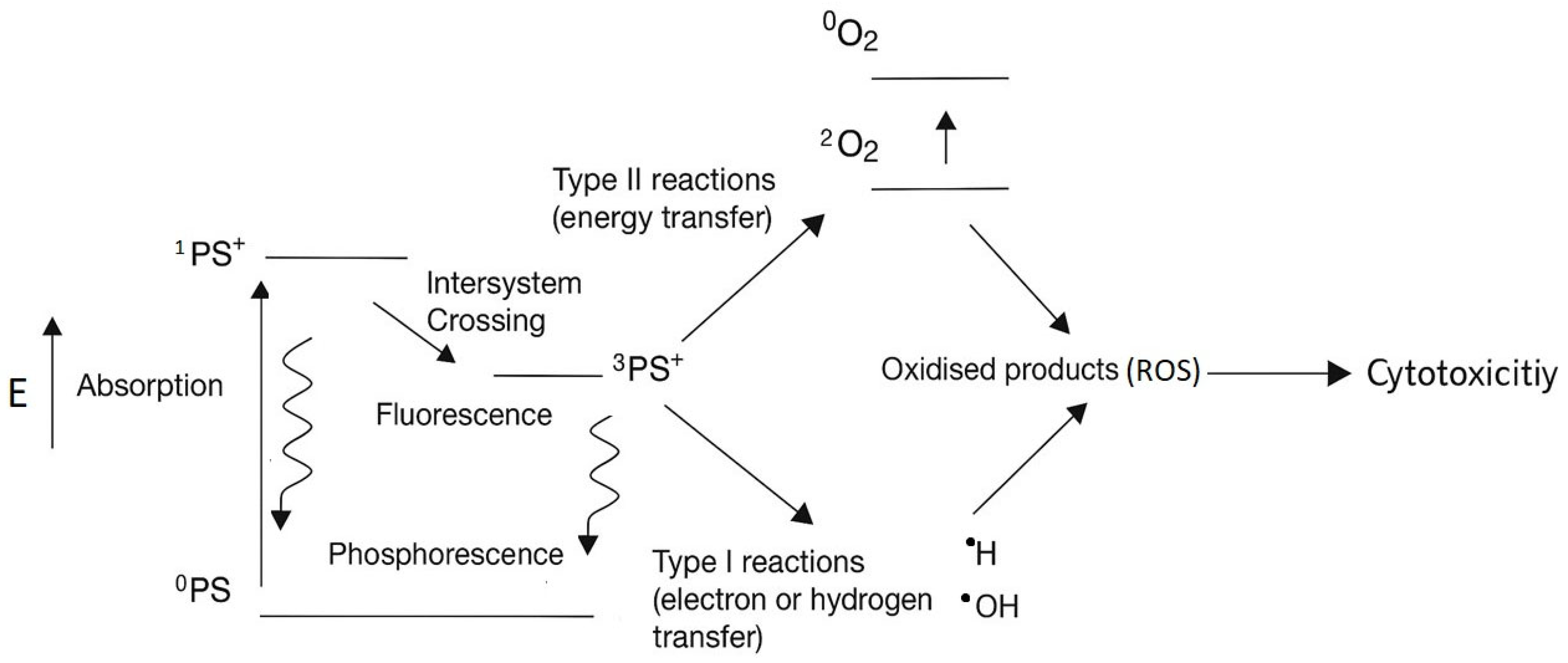
1. Introduction
2. Results
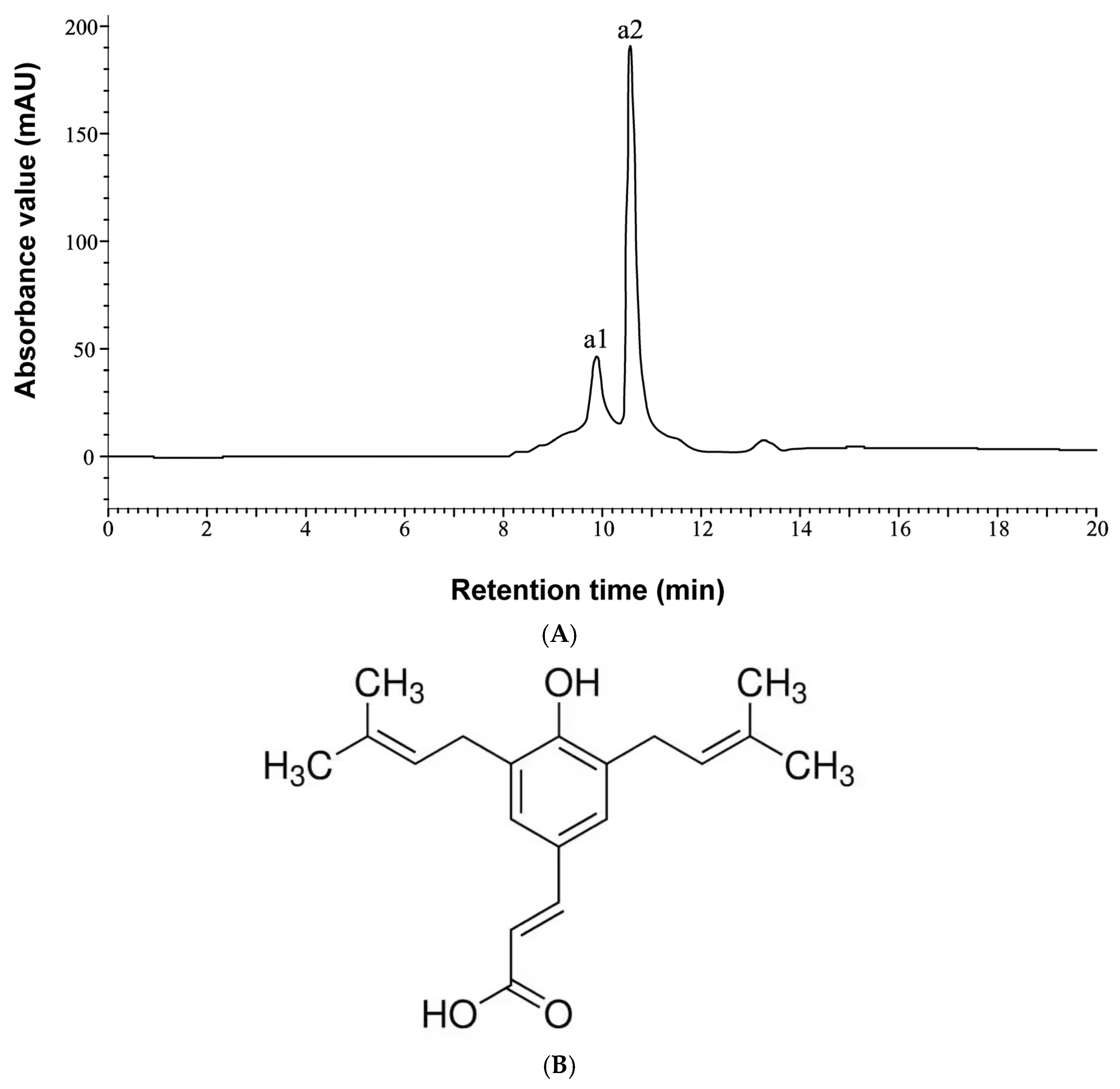
2.1. Phytochemical Analysis of the Extract Compound by HPLC
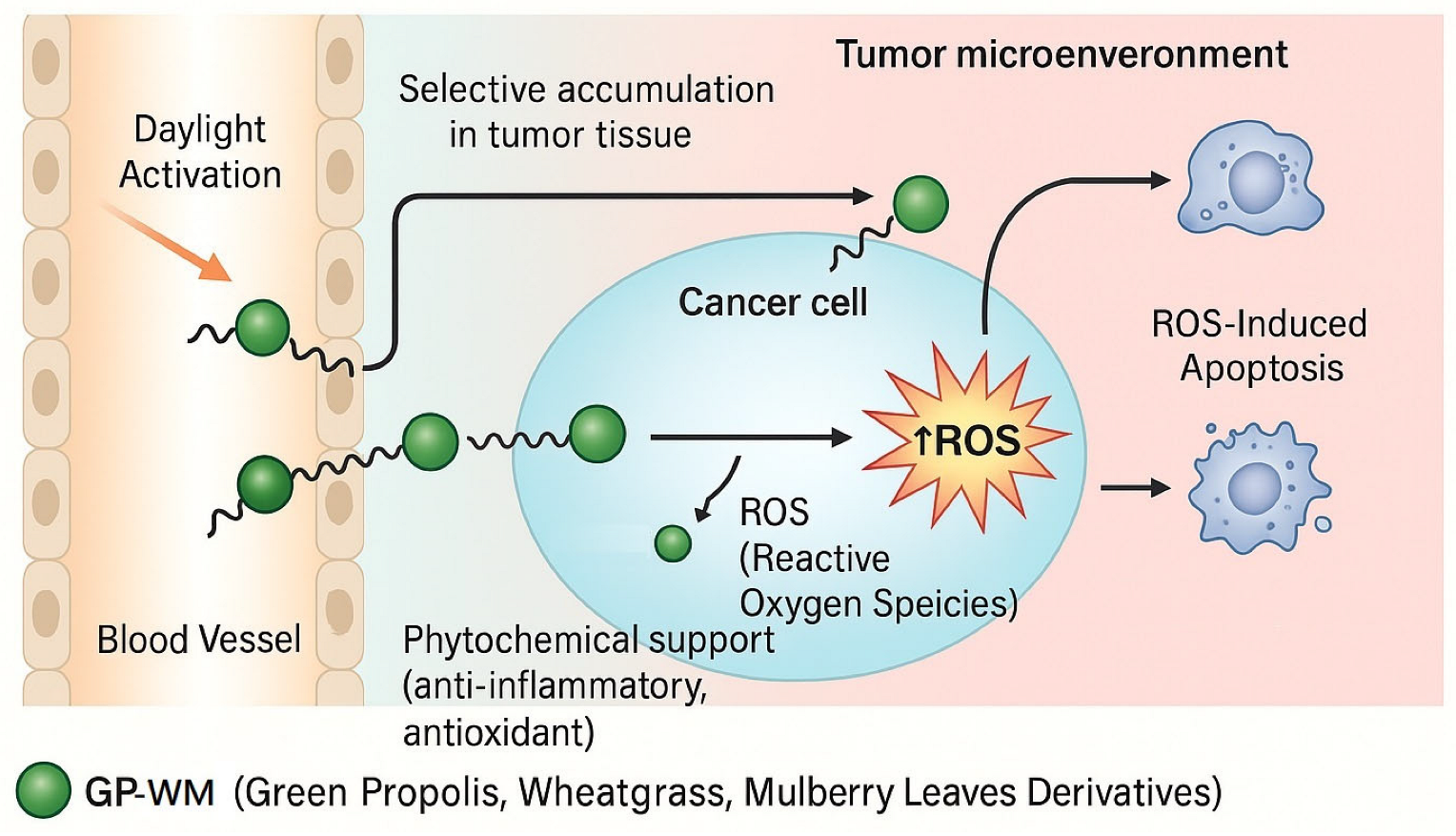
2.2. Evaluation of the Effects of the GP-WM Extract on 786-O Cell Apoptosis with and Without Daylight PDT
3. Discussion
4. Materials and Methods
4.1. Plant and Bee Product Sourcing
4.2. Cell Lines and Culture Conditions
4.3. Preparation of the Extract Compound
4.3.1. Green Propolis (GP) Extraction
4.3.2. Wheatgrass and Mulberry Leaf (WM) Extraction
4.3.3. Composite GP-WM Extract Preparation
4.4. Analyses of the Extract and Cell Viability
4.4.1. High-Performance Liquid Chromatography (HPLC) Analysis
4.4.2. Cell Viability Assay
4.4.3. Statistical Analysis
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.; Ma, J.; Zou, Z.; Jemal, A. Cancer statistics, 2014. CA Cancer J. Clin. 2014, 64, 9–29. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Jonasch, E.; Agarwal, N.; Alva, A.; Baine, M.; Beckermann, K.; Motter, A. Kidney Cancer, Version 3.2022, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc Netw. 2022, 20, 71–90. [Google Scholar] [CrossRef] [PubMed]
- Patel, S. Emerging Adjuvant Therapy for Cancer: Propolis and its Constituents. J. Diet. Suppl. 2016, 13, 245–268. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Zhang, C.-P.; Wang, K.; Li, G.Q.; Hu, F.-L. Recent Advances in the Chemical Composition of Propolis. Molecules 2014, 19, 19610–19632. [Google Scholar] [CrossRef] [PubMed]
- Ghallab, D.; Shawky, E.; Mohyeldin, M.; Metwally, A.M.; Ibrahim, R.S. Propolis: An update on its chemical diversity, botanical origin and biological activities. J. Adv. Pharm. Sci. 2025, 2, 76–99. [Google Scholar] [CrossRef]
- Khalil, M.L. Biological activity of bee propolis in health and disease. Asian Pac. J. Cancer Prev. 2006, 7, 22–31. [Google Scholar] [PubMed]
- Bankova, V.S.; Castro, S.L.d.; Marcucci, M.C. Propolis: Recent advances in chemistry and plant origin. Apidologie 2000, 31, 3–15. [Google Scholar] [CrossRef]
- Huang, W.-D.; Peng, S.-F.; Tsao, N.-W.; Wang, S.-Y.; Tzeng, S.-L.; Hu, N.-J. Taiwan Green Propolis Nanoparticles Induce Antiproliferation and Apoptosis in Oral Cancer Cells. Biomedicines 2025, 13, 921. [Google Scholar] [CrossRef] [PubMed]
- Sawicka, D.; Car, H.; Borawska, M.H.; Nikliński, J. The anticancer activity of propolis. Folia Histochem. Cytobiol. 2012, 50, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Oršolić, N.; Bašić, I. Antitumor, hematostimulative and radioprotective action of water-soluble derivative of propolis (WSDP). Biomed. Pharmacother. 2005, 59, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Ferraz, A.P.C.R.; Figueiredo, P.d.O.; Yoshida, N.C. Black Mulberry (Morus nigra L.): A Review of Attributes as an Anticancer Agent to Encourage Pharmaceutical Development. Adv. Pharmacol. Pharm. Sci. 2024, 2024, 3784092. [Google Scholar] [CrossRef] [PubMed]
- Tamraz, M.; Al Ghossaini, N.; Temraz, S. The Role of Wheatgrass in Colorectal Cancer: A Review of the Current Evidence. Int. J. Mol. Sci. 2024, 25, 5166. [Google Scholar] [CrossRef] [PubMed]
- Mansoori, B.; Mohammadi, A.; Amin Doustvandi, M.; Mohammadnejad, F.; Kamari, F.; Gjerstorff, M.F.; Baradaran, B.; Hamblin, M.R. Photodynamic therapy for cancer: Role of natural products. Photodiagn. Photodyn. Ther. 2019, 26, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Bar-Sela, G.; Cohen, M.; Ben-Arye, E.; Epelbaum, R. The Medical Use of Wheatgrass: Review of the Gap Between Basic and Clinical Applications. Mini Rev. Med. Chem. 2015, 15, 1002–1010. [Google Scholar] [CrossRef] [PubMed]
- Gore, R.D.; Palaskar, S.J.; Bartake, A.R. Wheatgrass: Green Blood can Help to Fight Cancer. J. Clin. Diagn. Res. 2017, 11, ZC40–ZC42. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.H.; Chang, Y.T.; Tseng, T.H.; Wang, C.-J. Mulberry leaf extract inhibit hepatocellular carcinoma cell proliferation via depressing IL-6 and TNF-α derived from adipocyte. J. Food Drug Anal. 2018, 26, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Lan, M.; Zhao, S.; Liu, W.; Lee, C.S.; Zhang, W.; Wang, P. Photosensitizers for Photodynamic Therapy. Adv. Healthc. Mater. 2019, 8, 1900132. [Google Scholar] [CrossRef] [PubMed]
- Sulaiman, C.; George, B.P.; Balachandran, I.; Abrahamse, H. Photoactive Herbal Compounds: A Green Approach to Photodynamic Therapy. Molecules 2022, 27, 5084. [Google Scholar] [CrossRef] [PubMed]
- Wiegell, S.R.; Wulf, H.C.; Szeimies, R.M.; Basset-Seguin, N.; Bissonnette, R.; Gerritsen, M.J.; Braathen, L.R. Daylight photodynamic therapy for actinic keratosis: An international consensus: International Society for Photodynamic Therapy in Dermatology. J. Eur. Acad. Dermatol. Venereol. 2012, 26, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Kubrak, T.P.; Kołodziej, P.; Sawicki, J.; Mazur, A.; Koziorowska, K.; Aebisher, D. Some Natural Photosensitizers and Their Medicinal Properties for Use in Photodynamic Therapy. Molecules 2022, 27, 1192. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.T.-H.; Huang, S.Y.; Liu, T.C.; Chen, Y.K. Novel compounds of Taiwanese green propolis induce apoptosis of human glioblastoma cells by daylight photodynamic action. Future Sci. OA 2025, 11, 2464491. [Google Scholar] [CrossRef] [PubMed]
- Forma, E.; Bryś, M. Anticancer Activity of Propolis and Its Compounds. Nutrients 2021, 13, 2594. [Google Scholar] [CrossRef] [PubMed]
- Souza, R.P.; Bonfim-Mendonça, P.S.; Damke, G.; Carvalho, A.R.D.-A.; Ratti, B.A.; Dembogurski, D.S.O.; Da-Silva, V.R.; Silva, S.O.; Da-Silva, D.B.; Bruschi, M.L.; et al. Artepillin C Induces Selective Oxidative Stress and Inhibits Migration and Invasion in a Comprehensive Panel of Human Cervical Cancer Cell Lines. Anticancer Agents Med. Chem. 2018, 18, 1750–1760. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Cardile, V.; Sanchez, F.; Troncoso, N.; Vanella, A.; Garbarino, J. Chilean propolis: Antioxidant activity and antiproliferative action in human tumor cell lines. Life Sci. 2004, 76, 545–558. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Yañez, N.; Ruiz-Hurtado, P.A.; Rivera-Yañez, C.R.; Arciniega-Martínez, I.M.; Yepez-Ortega, M.; Mendoza-Arroyo, B.; Rebollar-Ruíz, X.A.; Méndez-Cruz, A.R.; Reséndiz-Albor, A.A.; Nieto-Yañez, O. The Role of Propolis as a Natural Product with Potential Gastric Cancer Treatment Properties: A Systematic Review. Foods 2023, 12, 415. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-H.; Zhang, L.-J.; Sun, J.-J.; Yan, Y.-J.; Zhang, L.-X.; Chen, N.; Chen, Z.-L. Photodynamic efficiency of a chlorophyll-a derivative in vitro and in vivo. Biomed. Pharmacother. 2016, 81, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.-Y.; Lee, Y.; Kim, T.-H.; Heo, S.-J.; Shin, H.; Lee, J.; Yi, M.; Kang, H.W.; Jung, W.-K. Anti-Cancer Effect of Chlorophyllin-Assisted Photodynamic Therapy to Induce Apoptosis through Oxidative Stress on Human Cervical Cancer. Int. J. Mol. Sci. 2023, 24, 11565. [Google Scholar] [CrossRef] [PubMed]
- Ji, B.; Wei, M.; Yang, B. Recent advances in nanomedicines for photodynamic therapy (PDT)-driven cancer immunotherapy. Theranostics 2022, 12, 434–458. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.N.; Wu, C.L.; Lin, J.K. Apoptosis of human melanoma cells induced by the novel compounds propolin A and propolin B from Taiwenese propolis. Cancer Lett. 2007, 245, 218–231. [Google Scholar] [CrossRef] [PubMed]
- van Meerloo, J.; Kaspers, G.J.; Cloos, J. Cell sensitivity assays: The MTT assay. Methods Mol. Biol. 2011, 731, 237–245. [Google Scholar] [PubMed]

| HPLC Instrument | Chromaster HPLC System, Equipped with a Pump (CM 5110) and a Diode Array Detector (CM 5430) (Hitachi High-Tech Corporation, Tokyo, Japan) |
|---|---|
| Type of chromatography column | C18 Column (Cosmosil® 5C18-MS-II, Nacalai Tesque Inc., Kyoto, Japan) |
| Size of chromatography column | length: 250 mm inner diameter: 4.6 mm |
| Detection wavelength | 320 nm |
| Mobile phase | Methanol (A)/0.4% phosphoric acid (B)(in methanol) (80:20, v/v) |
| Gradient elution | The mobile phase was conducted for 25 min as follows: A:B was 80:20 (v/v) for 0.1–9 min, A:B was 70:30 (v/v) for 9.1–12 min, A:B was 95:5 (v/v) for 12.1–15 min, A:B was 80:20 (v/v) for 15.1–25 min. |
| Flow rate of test sample | 0.3 mL/min |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.-K.; Chiu, H.-M.; Huang, S.-Y.; Liu, T.-C.; Chen, D.T.-H. Photodynamic Activation of a Novel Chlorophyll-Enriched Green Propolis Compound Triggers Apoptosis in Renal Cell Carcinoma. Int. J. Mol. Sci. 2025, 26, 6897. https://doi.org/10.3390/ijms26146897
Chen Y-K, Chiu H-M, Huang S-Y, Liu T-C, Chen DT-H. Photodynamic Activation of a Novel Chlorophyll-Enriched Green Propolis Compound Triggers Apoptosis in Renal Cell Carcinoma. International Journal of Molecular Sciences. 2025; 26(14):6897. https://doi.org/10.3390/ijms26146897
Chicago/Turabian StyleChen, Yao-Kuan, Hui-Min Chiu, Shin-Yi Huang, Ta-Chun Liu, and Daniel Tzu-Hsuan Chen. 2025. "Photodynamic Activation of a Novel Chlorophyll-Enriched Green Propolis Compound Triggers Apoptosis in Renal Cell Carcinoma" International Journal of Molecular Sciences 26, no. 14: 6897. https://doi.org/10.3390/ijms26146897
APA StyleChen, Y.-K., Chiu, H.-M., Huang, S.-Y., Liu, T.-C., & Chen, D. T.-H. (2025). Photodynamic Activation of a Novel Chlorophyll-Enriched Green Propolis Compound Triggers Apoptosis in Renal Cell Carcinoma. International Journal of Molecular Sciences, 26(14), 6897. https://doi.org/10.3390/ijms26146897





